Abstract
Objective
To discuss the possible effects of microRNA-141 (miR-141) in sepsis-induced cardiomyopathy (SIC) via targeting death-associated protein kinase 1 (DAPK1).
Methods
An SIC mouse model was constructed by abdominal injection of lipopolysaccharide (LPS) and divided into control, LPS, LPS + pre-miR-141, and LPS + anti-miR-141 groups. Hemodynamic indicators and heart function indexes of mice were detected. ELISA was used to determine the serum levels of inflammatory cytokines, while TUNEL staining to observe the apoptosis of myocardial cells of mice, as well as qRT-PCR and Western blotting to clarify the expression of miR-141 and DAPK1. Lastly, in vitro experiment was also conducted on the primary neonatal rat ventricular cardiomyocytes (NRVCMs) to validate the results.
Results
Mice in the LPS group, as compared to the control group, had lower left ventricular ejection fraction, left ventricular fractional shortening, left ventricular systolic pressure, and ±dp/dt, but a higher left ventricular end-diastolic pressure, while the serum expression of IL-1β, IL-6, TNF-α, and cTn-T was up-regulated evidently with the increased apoptotic index of myocardial tissues. However, miR-141 and Bcl-2/Bax were down-regulated with elevated DAPK1 and cleaved caspase-3. The above changes were ameliorated in mice from the LPS + pre-miR-141 group relative to the LPS group, while those in the LPS + anti-miR-141 group were further deteriorated. In vitro experiment showed that miR-141 overexpression could reduce the apoptosis of LPS-induced NRVCMs and the levels of inflammatory cytokines with the increased cell viability.
Conclusion
MiR-141 could decrease inflammatory response and reduce myocardial cell apoptosis by targeting DAPK1, thereby playing the promising protective role in SIC.
Introduction
Sepsis is widely accepted as the systemic inflammatory response syndrome commonly resulting from the infection. 1 According to the statistical data, there are globally over 19 million sepsis patients worldwide, including six million patients died of sepsis with a mortality rate of over 1/4. 2 In spite of advancements in the medical research over the past decade, sepsis remains the major cause in critically ill patients owing to the rapid progress and difficult treatments. 3 Notably, lipopolysaccharide (LPS), derived from the Gram-negative bacterium, has been reported to be an important virulence factor in pathogenesis of sepsis, where LPS can activate inflammation-related cells to enhance inflammatory responses, eventually inducing the irreversible cardiac dysfunction,4,5 and thus, it has been called as sepsis-induced cardiomyopathy (SIC). 6 Moreover, the cardiac dysfunction can further deteriorate sepsis-related conditions, thereby increasing the mortality rate. 7 A retrospective cohort study also demonstrated a close correlation of SIC with the prognosis of sepsis patients. 8 Hence, searching for an effective myocardia-protective drug to alleviate the condition of SIC is of great clinical significance to improve the prognosis of sepsis patients.
MicroRNA (miRNA) represents a group of non-coding single chain RNA in length of about 22 nt that can bind to the 3’UTR of mRNA of downstream genes, thereby inhibiting or blocking the expression of the targeted gene and regulating the cellular activities. 9 MicroRNA-141 (miR-141), as a member of miR-200 family, can regulate different signal pathways via binding to different targets to modulate a series of biological processes, especially the early embryo implantation, normal pregnancy, angiogenesis, and tumorigenesis.10–12 In the myocardial ischemia/reperfusion model of mouse, caudal injection of miR-141 mimics was found to be able to reduce the infarction area and the inflammatory cell infiltration. 13 However, there remains limited information regarding the role of miR-141 in sepsis. According to the prediction website of target gene, there are potential binding sites between death-associated protein kinase 1 (DAPK1) and miR-141. More importantly, Li et al. found that DAPK1, via inhibiting p38MAPK/NF-κB, can suppress the oxidative stress and alleviate the inflammatory responses, thereby mitigating LPS-induced septic lung injury. 14 Zhai et al. supported that miR-98, through targeting the down-regulation of DAPK1, can relieve oxidative stress and myocardial cell apoptosis to alleviate myocardial ischemia/reperfusion injury. 15 In this respect, miR-141 was suggested to play a regulatory role by targeting DAPK1 in SIC.
Collectively, LPS-induced SIC models were constructed to investigate the role and mechanism of miR-141-targeted DAPK1 in SIC, aiming to provide a new strategy for the treatment of SIC.
Materials and methods
Ethical statement
This study was conducted in accordance with the Guide for the Care and Use of Laboratory Animals published by National Institutes of Health (NIH). 16 All animal-related operations were supervised by the Ethical Board for Medical Laboratory Animal of our hospital.
Construction and grouping of SIC mice
Eight-week-old C57BL/6 male mice, weighing 23–27 g, purchased from Shanghai SLAC Laboratory Animal Co., Ltd, were maintained in a bacteria-free facility at a constant humidity and temperature with free access to the food and water in a 12/12 light–dark cycle. According to the previous literature, SIC models were constructed on mice by abdominal injection of LPS. 17 Prior to the experiment, all mice were fasted for 12 h but could drink freely and then injected abdominally with LPS at dose of 10 mg/kg body weight dissolved in saline, while for the mice in the control group (n = 12), LPS was substituted by the normal saline. The models were divided randomly into the LPS group, LPS + pre-miR-141 group, and LPS + anti-miR-141 group, with 12 mice in each group. Mice in the LPS group, LPS + pre-miR-141 group, and LPS + anti-miR-141 group were injected with 100 μL purified adenovirus solution (1 × 1011 PFU) carrying empty vector, miR-141 precursor, or inhibitor intravenously 4 days prior to LPS injection. 18
Detection of hemodynamic indicators and heart function of mice
Following the anesthesia, mice were subjected to the echocardiogram by using the Mylab30CV (ESAOTE) ultrasound 31 systems with a 15 MHz probe to record the left ventricular ejection fraction (LVEF) and left ventricular fractional shortening (LVFS). Mice were then anesthetized by 1%–1.5% isoflurane for the determination of hemodynamic indicators by using a custom 1.4-F catheter transducer (SPR-853; Millar Instruments, 36 Houston, Texas), including left ventricular systolic pressure (LVSP), left ventricular end-diastolic pressure (LVEDP), and ± dp/dt.
Collection and treatment of samples
Twenty-four hours after model construction, blood samples were collected from the orbit and placed in a clean EP tube for 2 h at room temperature. Then, the samples were centrifuged at 3000 r/min for 20 min to collect the serum in the supernatant to determine the concentrations of IL-1β, IL-6, TNF-α, and cardiac troponin-T (cTn-T) by using the corresponding ELISA kit (Nanjing Jiancheng Biotech Institute). Mice were then fixed in an operation table to open the abdomen and expose the thoracic cavity to take out the heart of mice by removing the bilateral ribs and sterna which was later rinsed in the normal saline and the water on the surface was removed by a filter paper. The heart was then divided into two parts: one part was preserved at −80°C, and the other part was fixed in 4% paraformaldehyde.
Hematoxylin and eosin (HE) staining
The mouse myocardial tissue was fixed in 10% paraformaldehyde and embedded in the paraffin for serial sectioning in thickness of 4 μm. Paraffin of sections was melted and removed at 60°C, and the sections were later immersed in xylene twice (5 min/time) and dehydrated in ethanol of gradient concentrations. The sections were stained in Harris hematoxylin for 10 min and differentiated in 1% HCl–ethanol for 30 s. Later, the sections were blued in the running water for 15 min and stained in 1% eosin, followed by being differentiated in 90% ethanol, washed in 95% ethanol for 1 min and in xylene three times (2 min/time). Lastly, the sections were mounted in the neutral balsam and observed under a light microscope (Olympus BX51, Olympus, Japan). A scoring system was performed according to a previous study. 19 Each category was graded on a 5-point scale: 0 = no lesions, 1 = one or few small lesions, 2 = many small or a few large lesions, 3 = multiple small and large lesions, and 4 = massive lesions.
Transferase (TdT)-mediated D-UTP-biotin nick end labeling (TUNEL) staining
Paraffin sections were de-paraffinized and treated in xylene twice (15 min/time), followed by being hydrated in ethanol of gradient concentrations and digested in protease K at 37°C for 30 min. Then, the sections were placed in 0.3% H2O2–methanol at room temperature for 10 min and incubated with the TUNEL reaction buffer in the water bath of 37°C for 60 min, followed by incubation with the biotinylated anti-digoxin antibody (catalog # MK2362, Wuhan Yide Biotech Co., Ltd) in the 37°C water bath for 60 min. The sections were then counterstained in hematoxylin for 15–30 s, and the residual hematoxylin was removed by being washed in the water. The sections were then dehydrated in ethanol, cleared twice in xylene for 15 min and mounted by using the neutral balsam. Under the high-power lens, five visions were selected randomly without overlap, followed by the image analysis by using the Nikon Image Analysis Software (Nikon Corporation, Tokyo, Japan). Apoptosis index = (Number of TUNEL−positive nuclei/Number of nuclei) × 100%. 20 The apoptosis index could precisely reflect the apoptosis of myocardial cells.
Quantitative reverse transcriptase polymerase chain reaction (qRT-PCR)
The primers of qRT-PCR used in this study.

miR-141: microRNA-141; qRT-PCR: quantitative reverse transcriptase polymerase chain reaction.
Western blotting
Total proteins were extracted from the myocardial tissues of mice in each group and cells and then subjected to the bicinchoninic acid (BCA) method to measure the concentration of protein. Following the separation in 10% SDS-PAGE, proteins in the gel were transferred in the semi-dry transfer (Bio-Rad, USA) onto the Polyvinylidene Fluoride (PVDF) membrane, where the unoccupied sites were blocked in the 5% non-fat milk at room temperature and then rinsed in the phosphate buffer solution with Tween 20 (PBST). Proteins were incubated with the primary antibodies anti-DAPK1 (catalog # MA5-36128, Thermo Fisher Scientific, Inc., USA), anti-Bcl-2 (catalog # ab182858, Abcam, USA), anti-Bax (catalog # ab182733, Abcam, USA), anti-cleaved caspase-3 (catalog # ab214430, Abcam, USA), and anti-β-actin (catalog # ab8226, Abcam, USA) at 4°C overnight, and the incubation was terminated by five washes in PBST (3 min/wash). Thereafter, the immunoblots were further detected by incubation with the horseradish peroxidase–conjugated secondary antibody (catalog #170-6515, Bio-Rad, USA) at room temperature for 1 h. The final immunoblots were visualized by incubation with the reagents in the chemiluminescence (ECL) kit (Amersham, Arlington Heights, IL), and with the band intensity of β-actin as loading control, the expression of targeted proteins was expressed by the ratio of band intensity to that of β-actin.
Isolation, culture, and grouping of the myocardial cells
According to the previous literature, 22 the primary neonatal rat ventricular cardiomyocytes (NRVCMs) were isolated from the neonatal Sprague-Dawley rats, and then cultured in the Dulbecco’s modified Eagle medium/F12 Ham medium (DMEM/F12) (Gibco C11330) supplemented with 15% fetal bovine serum (FBS; HyClone) in an incubator at 37°C and 5% CO2. The medium was refreshed every 48 h. One day prior to the transfection, the cells were seeded into the 12-well plate, and when cell confluence reached about 50%, the cells were transfected according to the instruction of LipofectamineTM 2000 (Invitrogen, USA). The cells were divided into the mock group (the cells were not transfected), LPS group (the cells were cultured in 1 μg/mL LPS 23 for 12 h), LPS + miR-NC group (the cells were transfected with 50 nM miR-NC, followed by treatment of 1 μg/mL LPS for 12 h), LPS + miR-141 mimics group (the cells were transfected with 50 nM miR-141 mimics, followed by the treatment of 1 μg/mL LPS for 12 h), LPS + DAPK1 group (the cells were transfected with 4.0 μg pcDNA3.1-DAPK1, followed by the treatment of 1 μg/mL LPS for 12 h), and LPS + miR-141 mimics + DAPK1 group (the cells were transfected by miR-141 mimics and pcDNA3.1-DAPK1, followed by treatment of 1 μg/mL LPS for 12 h). MiR-141 mimics, miR-NC for negative control, and pcDNA3.1-DAPK1 were provided by Ribo Bio (Guangzhou, China).
Dual luciferase reporter gene assay
In this study, WT-DAPK1 plasmid and MUT-DAPK1 plasmid were constructed and transfected, together with miR-141 mimics and miR-NC, into the NRVCMs by using the LipofectamineTM 2000. Forty-eight hours later, a dual-luciferase reporter assay system (Promega, USA) was used to determine the activity of firefly luciferase and that of renilla, while the relative luciferase activity = firefly luciferase activity/renilla luciferase activity. 24
Cell viability of myocardial cells determined by Cell Count Kit-8 (CCK-8)
Transfected cells were digested and resuspended, where the density of cells was adjusted to 2 × 104 cells/mL, and seeded into the wells of a 96-well plate, with five replicate wells for each group. The plate was then placed in an incubator (37°C, 5% CO2). In each well, 10 μL CCK-8 (Beyotime, Nanjing, China) was added, followed by 1–4 h of incubation. The optical density (OD) of each well was determined by using the microplate reader and the cell viability = (ODexperiment group/ODcontrol group) × 100%. 25
Flow cytometry to determine the apoptosis of cells
The cells were harvested and digested in 0.25% EDTA-free trypsin and washed twice in cold PBS. Then, the cells were resuspended in binding buffer and mixed with 5 μL Annexin V-FITC and 5 μL PI. Binding buffer was added at 5 min prior to the detection, and then the flow cytometry was carried out. Then, cell apoptosis were assessed by flow cytometry (FACScan; BD Biosciences, USA) equipped with CellQuest software (BD Biosciences, USA). Early apoptosis was detected in the lower right quadrant (Q3), late apoptosis was detected in the upper right quadrant (Q2), necrosis cells were detected in the upper left quadrant (Q1), and viable cells were detected in the lower left quadrant (Q4). The apoptosis rate = early apoptosis rate + late apoptosis rate. 26
Statistical analysis
All data were subjected to the statistical analysis in SPSS 21.0 software (SPSS, Inc., Chicago, IL, USA). Measurement data are expressed in form of mean ± standard deviation (SD), while the difference among groups was validated via one-way ANOVA, followed by multiple comparisons by using the Tukey’s post hoc test. p < 0.05 suggested that the difference had statistical significance.
Results
Comparison of the hemodynamic indicators and heart function indexes in mice
The hemodynamic indicators (LVEF and LVFS) and heart function indexes (LVSP, ±dp/dt, and LVEDP) were measured. As shown in Figure 1, mice in the LPS group had lower LVEF, LVFS, LVSP, and ±dp/dt, but a higher LVEDP than the control group (all p < 0.05). In addition, as compared to the LPS group, the above factors were improved in mice from the LPS + pre-miR-141 group but were further aggravated in those from the LPS + anti-miR-141 group (all p < 0.05). The results suggested a protective role of miR-141 in the heart function of SIC mice. Comparison of the hemodynamic indicators and heart function indexes in mice. (a, b) Comparison of the hemodynamic indicators of mice; (c–f) comparison of the heart function indexes of mice; * p < 0.05 versus the control group; # p < 0.05 versus the LPS group; and p < 0.05 versus the LPS + pre-miR-141 group. LPS: lipopolysaccharide.
Serum expression of inflammatory cytokines in mice
ELISA was applied in order to detect the serum levels of inflammatory cytokines in mice. As a result, the expression of IL-1β, IL-6, TNF-α, and cTn-T in serum was increased in mice in the LPS group as compared to the control group (all p < 0.05, Figure 2), while as compared to the LPS group, mice in the LPS + pre-miR-141 group presented with down-regulations of IL-1β, IL-6, TNF-α, and cTn-T, but those factors in the LPS + anti-miR-141 group were increased (all p < 0.05). The above results suggested that miR-141 could ameliorate the inflammation responses in SIC mice. The serum expression of inflammatory cytokines in mice. (a–d) ELISA was used to detect the expression of IL-1β (a), IL-6 (b), TNF-α (c), and cTn-T (d) in serum of mice; * p < 0.05 versus the control group; # p < 0.05 versus the LPS group; and p < 0.05 versus the LPS + pre-miR-141 group. LPS: lipopolysaccharide.
Pathological changes and apoptosis of myocardial cells in mice
The pathological changes and apoptosis of myocardial cells in mice were detected by HE staining and TUNEL staining, respectively. According to the results of HE staining in Figure 3, myocardial tissues were clear and normally arranged, with no evident infiltration of inflammatory cells in control mice, while those in the LPS group presented with the poorly arranged myocardial fibers, edema, denaturation, and widened interspace of myocardial cells and massive infiltration of inflammatory cells, which, however, was found to be alleviated in the mice from the LPS + pre-miR-141 group but deteriorated in those from the LPS + anti-miR-141 group. Besides, the quantitative results indicated that in comparison with the control group, mice in the LPS group had higher histopathological score and apoptotic index in the myocardial tissues (all p < 0.05), while compared to the LPS group, the histopathological score, and apoptotic index were reduced in the LPS + pre-miR-141 group and enhanced in the LPS + anti-miR-141 group (all p < 0.05). These data suggested miR-141 could protect against myocardial apoptosis in SIC mice. Pathological changes and apoptosis of myocardial cells in mice. (a) The pathological changes and apoptosis of myocardial tissues in mice determined by HE staining and TUNEL staining (arrow indicated the apoptotic nucleus); (b, c)comparison of the histopathological score and apoptotic indexes of myocardial tissues of mice; * p < 0.05 versus the control group; # p < 0.05 versus the LPS group; and p < 0.05 versus the LPS + pre-miR-141 group. LPS: lipopolysaccharide; TUNEL: transferase (TdT)-mediated D-UTP-biotin nick end labeling.
Expression of miR-141 and DAPK1 in myocardial tissues of mice
The expression of miR-141 and DAPK1 in the myocardial tissues of mice was measured by qRT-PCR and Western blotting (Figure 4(a)–(c)). As a result, in comparison with the control group, the expression of miR-141 and Bcl-2/Bax was down-regulated while DAPK1 and cleaved caspase-3 were up-regulated in the mice of LPS group (all p < 0.05). When compared to the LPS group, miR-141 and Bcl-2/Bax were up-regulated, but DAPK1 and cleaved caspase-3 were down-regulated in the LPS + pre-miR-141 group (all p < 0.05). However, mice in the LPS + anti-miR-141 group presented with the opposite changes (all p < 0.05). According to the prediction website of TargetScan.org, the binding site between miR-141 and DAPK1 was found (Figure 4(d)), and the results of luciferase reporter gene assay (Figure 4(e)) demonstrated that in comparison with the miR-NC group, co-transfection of miR-141 mimics, and WT-DAPK1 could decrease the activity of luciferase (p < 0.05), while co-transfection with MUT-DAPK1 resulted in no evident changes in the luciferase activity (p > 0.05). The results of this experiment implied that DAPK1 was inferred to be a target gene of miR-141 and DAPK1 was inhibited by miR-141 in the myocardial tissues of SIC mice. Expression of miR-141 and DAPK1 in myocardial tissues of mice. (a) Expression of miR-141 in the myocardial tissue of mice detected by qRT-PCR; (b, c) expression of DAPK1, Bcl-2/Bax, and cleaved caspase-3 in the myocardial tissue of mice detected by Western blotting; * p < 0.05 versus the control group; # p < 0.05 versus the LPS group; and p < 0.05 versus the LPS + pre-miR-141 group; (d) online prediction indicated the binding site between miR-141 and DAPK1; (e) results of luciferase reporter gene assay; * p < 0.05 versus the miR-NC group. LPS: lipopolysaccharide; qRT-PCR: quantitative reverse transcriptase polymerase chain reaction.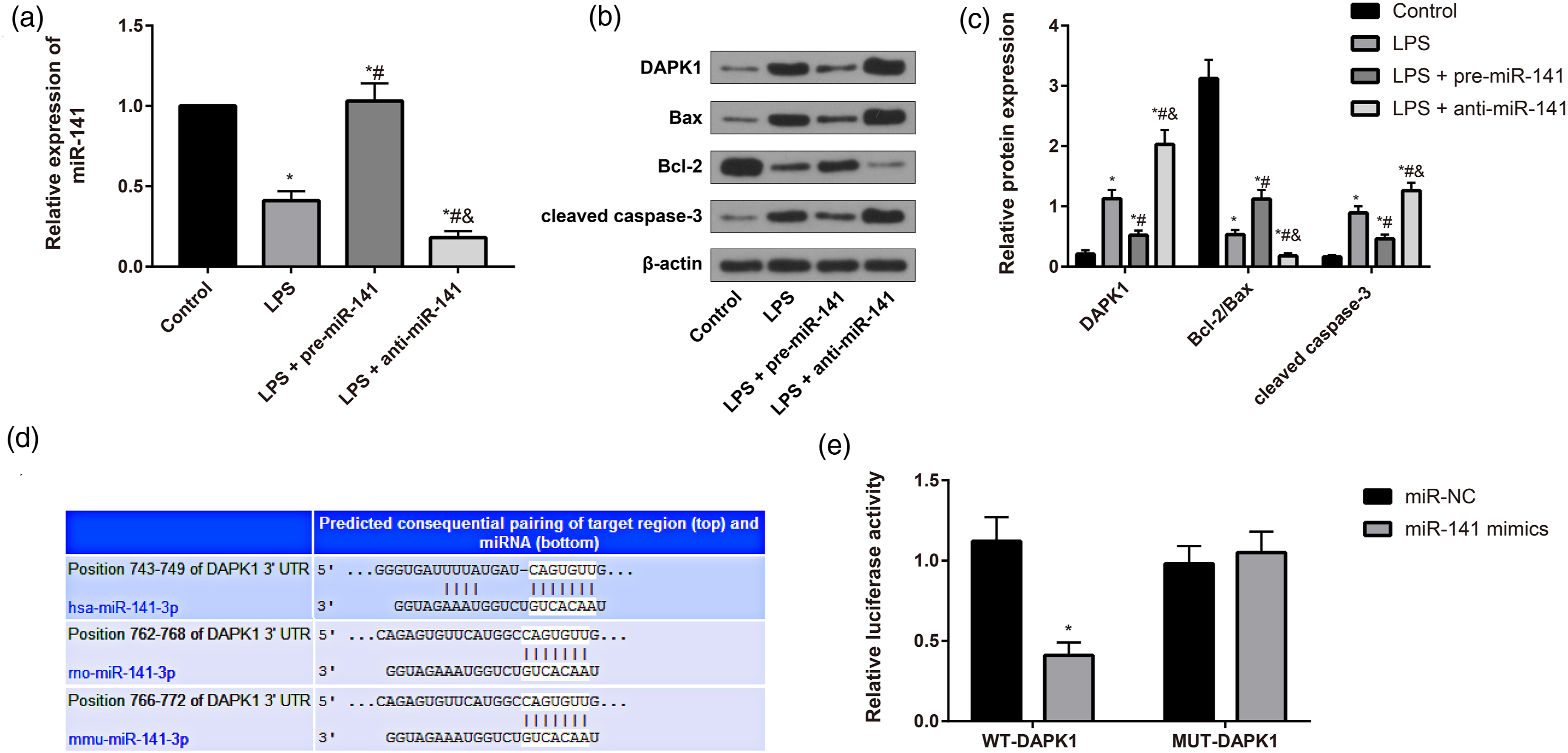
Cell viability and apoptosis of NRVCMs after treatment
To further confirm the potential effects of miR-141 on cardiomyocyte apoptosis induced by LPS, the cell viability and apoptosis of NRVCMs were detected by CCK-8 assay and flow cytometry, respectively. Consequently, by comparison with the mock group, NRVCMs in the LPS group had a sharp decrease in the cell viability but an increase in the apoptosis rate, while compared to the LPS group, NRVCMs in the LPS + miR-141 mimics group had a sharp increase in the cell viability but a decrease in apoptosis rate, while NRVCMs demonstrated the opposite changes (all p < 0.05, Figure 5). Moreover, in comparison with the LPS + mimics group, NRVCMs in the LPS + miR-141 mimics + DAPK1 group had a decrease in cell viability and an increase in the apoptosis rate (all p < 0.05). These data further indicated the protective effect of miR-141 against apoptosis of myocardial cells induced by LPS in vitro. Cell viability and apoptosis of NRVCMs after treatment. (a, b) The apoptosis of NRVCMs was detected by flow cytometry; Q1(upper left quadrant): necrosis cells; Q2 (upper right quadrant): late apoptosis; Q3 (lower right quadrant): early apoptosis; Q4 (lower left quadrant): viable cells; (c) the cell viability of NRVCMs was determined by CCK-8; * p < 0.05 versus the Mock group; # p < 0.05 versus the LPS group; and p < 0.05 versus the LPS + miR-141 mimics group; % p < 0.05 versus the LPS + DAPK1 group. LPS: lipopolysaccharide; NRVCM: neonatal rat ventricular cardiomyocytes; CCK-8: cell count kit-8.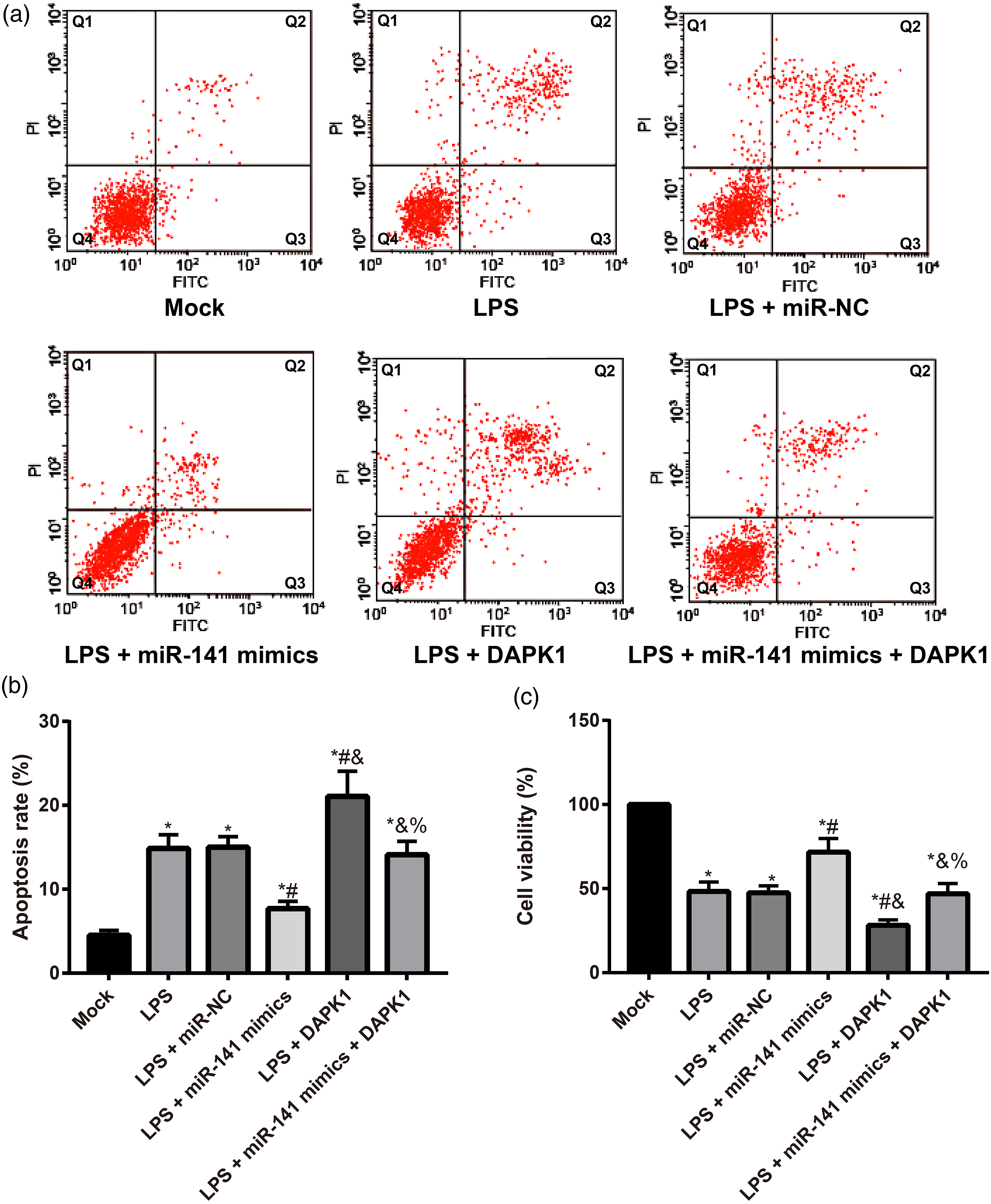
Comparison of the expression of inflammatory cytokines in NRVCMs among groups
Further, the expression of inflammatory cytokines (IL-1β, IL-6, and TNF-α) in NRVCMs was measured using qRT-PCR and ELISA. As shown in Figure 6, we found that in comparison with the mock group, NRVCMs had evident up-regulations of IL-1β, IL-6, and TNF-α in the LPS group (all p < 0.05), while compared to the LPS group, NRVCMs presented with the down-regulations of IL-1β, IL-6, and TNF-α in the LPS + miR-141 mimics group, but NRVCMs in the LPS + DAPK1 group showed the opposite changes (all p < 0.05). In addition, in comparison with the LPS + mimics group, NRVCMs in the LPS + miR-141 mimics + DAPK1 group manifested the up-regulations of IL-1β, IL-6, and TNF-α (all p < 0.05). These results supported the protective effect of miR-141 against cardiomyocyte inflammatory response induced by LPS in vitro. Comparison of the expression of inflammatory cytokines in NRVCMs among groups. (a, b) The expression of IL-1β, IL-6, and TNF-α in NRVCMs was detected by qRT-PCR and ELISA; * p < 0.05 versus the Mock group; # p < 0.05 versus the LPS group; and p < 0.05 versus the LPS + miR-141 mimics group; % p < 0.05 versus the LPS + DAPK1 group. LPS: lipopolysaccharide; NRVCM: neonatal rat ventricular cardiomyocytes; qRT-PCR: quantitative reverse transcriptase polymerase chain reaction. The molecular mechanism diagram shows that miR-141 could decrease the inflammatory response and reduce the apoptosis of myocardial cells through down-regulating the expression of DAPK1 in sepsis-induced cardiomyopathy.

Discussion
In the present article, SIC models on mice were established via the abdominal injection of LPS on the basis of the previous literature,27,28 and consequently, mice in the LPS group had decreased LVEF, LVFS, LVSP, and ±dp/dt but a significant increased LVEDP, which was in agreement with the previous findings,29,30 exhibiting the evident defect in myocardial function of sepsis mice, with the manifestation of left ventricular dilation and decreased systolic function. However, these changes were rescued in LPS mice treated with pre-miR-141 but the inhibition of miR-141 could deteriorate the damage to the heart function of these mice. Similar to the study of Liu et al., miR-141 overexpression could mitigate the inflammatory infiltration and reduce the infarction area in myocardial cells, so as to alleviate the myocardial ischemia/reperfusion injury, 13 suggesting that miR-141 may play a protective role in the heart function of SIC mice.
Sepsis, as reported, usually has the histological manifestation of necrosis and apoptosis of myocardial cells. 31 Cardiac markers in serum, such as troponin t, elevate rapidly, and at the same time, the pathological staining showed the disordered arrangement and enlargement of myocardial cells. In addition to its necrosis or apoptosis, the infiltration of inflammatory cells and then the release of inflammatory cytokines, including TNF-α, IL-1β, and IL-6, could further damage the myocardial cells.32–34 TNF-α is the earliest released inflammatory cytokines after sepsis attack, which can activate NF-κB to induce the cytokine cascade reaction to trigger the massive generation of TNF-α, IL-1, and IL-6, while the interaction among these cytokines and the underlying positive feedback mechanism can further aggravate the inflammation to activate the inflammatory cascades.35–37 Also, TNF-α can activate the apoptosis and damage the myocardial cells by activating the caspase. 34 The disordered myocardial cells are thought to have a direct association with the development and progression of sepsis, and the myocardial cell apoptosis can cause anomaly in the myocardial structure and function, thereby resulting in the cardiac dysfunction. 38 In this study, LPS mouse was observed to present with the significant increased expression of IL-1β, IL-6, TNF-α, and cTn-T, as well as the elevated apoptotic index in myocardial tissues. By contrast, miR-141 overexpression could decrease the levels of the above inflammatory cytokines and inhibit the apoptosis of myocardial cells. Consistently, miR-146a, another miRNA, could also protect mouse from the LPS-induced SIC by suppressing inflammatory cytokines and decreasing myocardial cell apoptosis. 39 Quan et al. also demonstrated that miR-141, by inhibiting p38 MAPK and NF-κB signaling pathway, could mitigate LPS-induced inflammatory injury to WI-38 fibroblasts. 40 The work of Tang et al. showed that knockout of CRNDE, via up-regulating miR-141, could down-regulate the expression of IL-1β, IL-6, and TNF-α to promote LPS-induced proliferation and inhibit the apoptosis in MRC-5 cells. 41 As such, miR-141 was considered to mitigate LPS-induced inflammation in SIC mice to reduce the apoptosis and protect the cardiac function of myocardial cells.
Furthermore, the myocardial tissues of LPS mice were presented with the down-regulations of miR-141 and Bcl-2/Bax and the up-regulations of DAPK1 and cleaved caspase-3, which were reversed by the overexpression of miR-141. Recent evidence indeed pointed out that DAPK1 was involved in a series of inflammatory regulation positively or negatively, 42 which may correlate with the effect of DAPK1 on NF-κB activities and various cell types. 43 IL-1β, as a pro-inflammatory factor, plays a key role in the septic infection and inflammation. 44 DAPK1 is critical to the generation of IL-1β, 45 where inflammation could activate NF-κB to promote the synthesis of IL-1β precursor which is divided into the mature p17 IL-1β under the catalytic effect of caspase-1 to induce the synthesis of NLRP3.46,47 DAPK1 deletion, possibly via altering the synthesis of NLRP3 inflammasome, could affect the synthesis of NLRP3 inflammasome to inhibit the synthesis of IL-1β and curb the activity of caspase-1. 48 Hu et al. also reported that in the mouse with sepsis-induced acute renal injury, DAPK1 knockout showed the protective role by ameliorating the inflammation. 49 Besides, inhibition of DAPK1, as reported by Cui et al., can reduce the generation of inflammatory cytokines and the apoptosis of neutrophils, thereby relieving lung injury in LPS-induced ARDS. 50 The luciferase reporter gene assay in this study confirmed that DAPK1 was a target gene of miR-141, while the in vitro experiment showed that overexpression of DAPK1 in LPS-induced myocardial cells could decrease the cell viability, promote the apoptosis, increase the levels of IL-1β, IL-6, and TNF-α and reverse the protective effect of miR-141 mimics on LPS-induced myocardial cells. Findings of Zhu et al. also found that miR-141-3p overexpression, through targeting HMGB1, can inhibit the inflammation and apoptotic injuries in LPS-induced nasal epithelial cells. 51 Zhai et al. demonstrated that miR-93, possibly by down-regulating the expression of DAPK1, can reduce the apoptosis and improve the cardiac function in myocardial ischemia/reperfusion rats. 15 Thus, miR-141 could mitigate the inflammation and ameliorate the cardiac function in LPS-induced SIC via down-regulating the expression of DAPK1(Figure 7).
Taken together, we found that miR-141 could decrease the inflammatory response and reduce the apoptosis of myocardial cells through down-regulating the expression of DAPK1 in SIC. The findings of this study provide novel insights into the treatment of SIC.
Supplemental Material
sj-jpg-1-het-10.1177_09603271211033768 – Supplemental Material for MiR-141 attenuates sepsis-induced cardiomyopathy by targeting DAPK1
Supplemental Material, sj-jpg-1-het-10.1177_09603271211033768 for MiR-141 attenuates sepsis-induced cardiomyopathy by targeting DAPK1 by Bo Song, Xin-Xiang Wang, Hai-Yan Yang, Ling-Ting Kong and Hong-Yan Sun in Human & Experimental Toxicology
Supplemental Material
sj-jpg-2-het-10.1177_09603271211033768 – Supplemental Material for MiR-141 attenuates sepsis-induced cardiomyopathy by targeting DAPK1
Supplemental Material, sj-jpg-2-het-10.1177_09603271211033768 for MiR-141 attenuates sepsis-induced cardiomyopathy by targeting DAPK1 by Bo Song, Xin-Xiang Wang, Hai-Yan Yang, Ling-Ting Kong and Hong-Yan Sun in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
All relevant data are within the paper.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
