Abstract
Objective:
In light of the upregulation of p21-activated kinase (PAK7) in a variety of cancers, including hepatocellular carcinoma (HCC), we aimed to investigate the effect of PAK7 on the sensitivity of HCC cells to radiotherapy.
Methods:
PAK7 expression was determined in normal adult liver epithelial THLE-2 and human HCC cell lines. The effect of ionizing radiation (IR) on the HCC cell viability was evaluated by Sulforhodamine B (SRB) assay. HCC cell lines Mahlavu and Huh7 were chosen to assess the effect of PAK7 shRNAs on the viability, clone formation, apoptosis, cycle distribution and γ-H2AX expression after exposure to IR.
Results:
As compared to THLE-2 cells, PAK7 was upregulated in poorly differentiated Mahlavu and SK-Hep-1 cells, but moderately or lowly expressed in well-differentiated Huh7 and HepG2 cells. HCC cells with moderate or low expression of PAK7 presented a decreased viability at 2 Gy IR, which had no significant effect on PAK7high HCC cells. Mahlavu and Huh7 cells transfected with PAK7 shRNAs showed increased inhibitory effect of IR on viability. In addition, PAK7 shRNAs reduced clone formation, facilitated the cell apoptosis, arrested cells at G2/M phase, and increased γ-H2AX expression. Moreover, changes above were more evident in the HCC cells co-treated with IR and PAK7 shRNAs.
Conclusion:
PAK7 downregulation could inhibit the viability, promote the apoptosis, arrest cells in G2/M phase, and induce the DNA damage in HCC cells, thereby enhancing the radiosensitivity in HCC.
Introduction
According to the available statistics, there are 905,677 (4.7%) new cases are diagnosed as liver cancer and nearly 830,180 (8.3%) are dead from liver cancer (https://gco.iarc.fr/today/fact-sheets-cancers). Histologically, liver cancer is subclassified into different types including hepatocellular carcinoma (HCC), 1 which are the most common pathological one in primary HCC, taking up nearly 70–85%. 2 Currently, the prevalence of HCC has been increasing annually, making it a major public health issue.2,3 So far, surgery remains the major method for HCC, 4 but many patients have lost the opportunity of surgical treatment at the time of diagnosis, since HCC is always featured with hidden onset, inconspicuous early symptoms and rapid progression. 5 As for the significant progress in the development of radiotherapy technique, like intensity-modulated radiation therapy (IMRT), helical tomotherapy and stereotactic radiotherapy, radiotherapy has become the necessary method for treatment of HCC.6,7 However, radiotherapy may also result in severe radiation-induced liver diseases, causing an increased mortality rate of patients.8,9 Thus, searching for factors related to the sensitivity of HCC cells to radiotherapy is a major issue to be addressed.
P21-activated kinases (PAKs) constitute a group of highly evolutionarily conservative serine/threonine protein kinases that participate in the regulation of cell survival, apoptosis, angiogenesis, mitosis and other biological functions.10,11 In general, PAKs are divided into PAKs I (PAK1, 2, 3) and PAKs II (PAK4, 5, 6), 12 while PAK5, also known as PAK7, is a newly found but scarcely researched type II PAKs.13,14 Based on the analysis of PAK7 cDNA, PAK7 gene is located in 20p12 in the human genomic DNA with the length of 300 kb, containing 12 exons and encoding 80 kD protein. 15 Recently, some scholars have implicated the role of PAK7 in the pathology of malignant tumors. For instance, PAK7 was upregulated in the human colorectal cancer, showing an association with its tumor progression and metastasis, as suggested by Huang et al. 16 In lung squamous cell carcinoma, PAK7 was also highly expressed and knockout of PAK7 could inhibit the growth and metastasis of its tumor cells. 17 Importantly, PAK7 demonstrated a similar expression pattern as the above in HCC.18,19 More importantly, PAK7 could be used to distinguish the irradiated prostate cancer patients into early and late relapse groups in the study of Seifert et al., 20 suggesting a potential effect of PAK7 on radiotherapy. However, whether PAK7 could affect the sensitivity of HCC cells to radiotherapy is unknown. Thus, we performed a series of experiments to detect PAK7 expression in different HCC cell lines and investigated the effect of varying PAK7 levels on the viability of HCC cells. To avoid a possibility of off-target effects, two shRNA sequences were transfected into the Mahlavu and Huh7 cells and further performed a series of in vitro experiments, including clone formation assay, sulforhodamine B (SRB) assay, Annexin-V-FITC/PT, flow cytometry, qRT-PCR and Western blotting, so as to explore the effect of PAK7 shRNA on the sensitivity of HCC cells to radiotherapy and its underlying mechanism.
Materials and methods
Cell culture
In this study, normal adult liver epithelial THLE-2 cells, poorly differentiated HCC cell lines (Mahlavu and SK-Hep-1 cell) and well-differentiated HCC cell lines (Huh7 and HepG2) provided by American Type Culture Collection (ATCC, Manassas, VA, USA) were sustained in Dulbecco’ s Modified Eagle’ s Medium (DMEM) containing fetal bovine serum (FBS, 10%) and penicillin-streptomycin (1%) at 37°C in 5% humidified CO2 incubator. Cells were subcultured at full confluence or media changed every 2 or 3 days.
Sulforhodamine B (SRB) cytotoxicity assay
A 96-well plate was used to seed the cells at the density of 3 × 103 cells/well for culture. Cells were exposed to the ionizing radiation (IR) at 0.5–2 Gy for 24 h, 21 followed by fixed in 10% trichloroacetic acid (TCA). Cells were washed in ddH2O and stained in 0.4% SRB/acetic acid solution, where the unbound SRB was removed via three washes in 1% acetic acid. Thereafter, cells were treated by Tris, where the bound SRB was dissolved, and cells were subjected to the measurement of optical density (OD) at 570 nm using the Molecular Devices Spectramax M3 multimode microplate reader (Molecular Devices LLC., San Jose, CA, USA). This experiment was conducted in triplicate.
Cell grouping and transfection
Mahlavu and Huh7 cells were divided into Blank group (Cells were not treated), scr-shRNA group (Cells were transfected by scramble shRNA, 5′-TTCTCCGAACGTGTCACGT-3′), PAK7 shRNA#1 group (Cells were transfected by PAK7 shRNA#1, 5′-CCGGGCCTCCATAAATATGATCTATCTCGAGATAGATCATATTTATGGAGGCTTTTT-3′), PAK7 shRNA#2 group (Cells were transfected by PAK7 shRNA#2, 5′-CCGGCGGGATTACCACCATGACAATCTCGAGATTGTCATGGTGGTAATCCCGTTTTT-3′), IR group (Cells were irradiated at 2 Gy for 24 h 21 ), IR + scr-shRNA group (Cells were co-treated by IR at 2 Gy and transfection of 2 µg scr-shRNA for 24 h), IR + PAK7 shRNA#1 group (Cells were co-treated by IR at 2 Gy and transfection of 2 µg PAK7 shRNA#1 for 24 h), IR + PAK7 shRNA#2 group (Cells were co-treated by IR at 2 Gy and transfection of 2 µg PAK7 shRNA#2 for 24 h). Transfection was conducted in accordance with the instruction of Lipofectamine™ 3000 (Sigma, USA). Total RNA and protein extracted 24 h after transfection was used for qRT-PCR and Western blot analyses. Subsequently, cell viability was determined through the Sulforhodamine B Cytotoxicity Assay.
qRT-PCR
The total RNA was extracted by using the TRIzol™ Reagent (Invitrogen™, USA) and used to prepare cDNA via the reverse transcription by using the High-Capacity RNA-to-cDNA™ Kit (Applied Biosystems™, USA), with the following primers: PAK7: Forward: 5′-TATCTGGCCCGTCCAACTTTG-3′, Reverse: 5′-GGGAAGGCCGGTAAACTTCTG-3′; GAPDH: Forward: 5′-GGAGCGAGATCCCTCCAAAAT-3′, Reverse: 5′-GGCTGTTGTCATACTTCTCATGG-3′. Then, PCR reactions were conducted by using Power SYBR™ Green PCR Master Mix (Applied Biosystems™, USA) on Applied Biosystems® QuantStudio® 5 Real-Time PCR System (Applied Biosystems™, USA), with GAPDH (glyceraldehyde-phosphate dehydrogenase) as the endogenous reference. The difference in mRNA expression of targeted genes in experiment groups and control groups was expressed by using the formula of 2−ΔΔCT. This experiment was conducted in triplicate.
Western blotting
Total proteins were extracted from cells and subjected to the measurement of protein concentration by using the BCA Protein Quantification Kit. Proteins were then separated via the 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transfected onto the PVDF membrane, where the unoccupied sites were blocked in 5% non-fat milk at 4°C overnight. Proteins on the membrane were firstly incubated with the primary anti-PAK7 (ab110069; 1: 500, Abcam, USA), anti-γ-H2AX (ab81299) at 1/100,000 dilution, anti-β-actin -loading control (ab8226; 1: 1000, Abcam, USA) antibodies at 4°C overnight. Subsequently, the immunoblots were washed three times in PBS at room temperature, 5 min/wash, and then incubated with the secondary antibodies conjugated with horseradish peroxidase (HRP) for 1 h, followed by three washes in PBS. The final immunoblots were developed by incubation with enhanced chemiluminescence (ECL). With β-actin as the endogenous control, the relative expression of targeted protein was expressed by using the ratio of band intensity of targeted protein to that of β-actin. This experiment was conducted in triplicate.
Clone formation assay
In brief, 1 × 103 Mahlavu or Huh7 cells were exposed to treatment regimen for 24 h. After seeded in 6-well plates in 5% humidified CO2 incubator at 37°C for 2 weeks, cell colonies formed (>50 cells/colony) were stained with crystal violet dye, followed by photographing and counting. This experiment was conducted in triplicate.
Flow cytometry
Cells were collected and placed into 70% cold ethanol for fixation at 4°C overnight and then centrifuged, with the supernatant being abandoned. The sediment was rinsed twice in PBS supplemented with 1% FBS, resuspended in 400 µL binding buffer and incubated with 50 µL RNAase A (Sigma, USA). Then, cells were further incubated with 50 µL PI (50 mg/L) in the dark. Cell cycle was determined by using the flow cytometer.
Cells in each group were digested in 0.25% trypsin and collected into the FCM tubes, followed by centrifugation. With the supernatant discarded, cells in the sediment were rinsed in the cold PBS three times and centrifuged to discard the supernatant. Annexin-V-FITC, PI and HEPES buffer were mixed in proportion of 1:2:50, and in 100 µL of mixture, 1 × 106 cells were resuspended and well mixed for incubation at room temperature for 15 min and then placed in 1 mL HEPES buffer. FITC and PI fluorescent signals were detected at 515 nm and 620 nm to detect cell apoptosis. This experiment was conducted in triplicate.
Statistical analysis
All data were processed by using the SPSS 21.0 software (SPSS, Inc, Chicago, IL, USA). The measurement data were expressed in form of mean ± standard deviation (SD) and compared by using the One-way analysis of variance, followed by the Tukey’s post hoc test. P < 0.05 suggested that the difference had statistical significance.
Results
Effect of IR on the viability of HCC cells with varying levels of PAK7
Results of qRT-PCR and Western blotting (Figure 1(A) to (C)) demonstrated that PAK7 was significantly upregulated in poorly differentiated Mahlavu and SK-Hep-1 (PAK7high cell) (all P < 0.001), but moderately expressed in well-differentiated Huh7 (PAK7moderate cell) (P < 0.05), in comparison with the THLE-2 cells. No significant difference was found between THLE-2 cells and well-differentiated HepG2 (PAK7low cell) (P > 0.05). In addition, the viability of different HCC cells with the increasing IR dosage was evaluated, and as a result, after 24 h of IR at 2 Gy, a significant decrease was found in the viability of PAK7low HepG2 cells and PAK7moderate Huh7 cells (P < 0.05), but for PAK7high Mahlavu cells and PAK7high SK-Hep-1 cells, the increased dose of IR, though decreasing the cell viability, showed no sufficient killing effect on cells (Figure 1(D)), indicating a vital role for PAK7 expression in the radiosensitivity of HCC cells. And the 2 Gy IR was chosen for the subsequent experiment.

Effect of IR at different doses on the viability of HCC cells responsive to varying levels of PAK7. Note: (A–C) qRT-PCR (A) and Western blotting (B and C) are conducted to detect the mRNA and protein expression of PAK7 in normal adult liver epithelial THLE-2 cells, poorly differentiated HCC cell lines (Mahlavu and SK-Hep-1 cell) and well-differentiated HCC cell lines (Huh7 and HepG2). *P < 0.05, ***P < 0.005, ****P < 0.005 vs. the normal adult liver epithelial THLE-2 cells. (D Effect of IR at different doses (0 Gy, 0.5 Gy, 1.5 Gy and 2 Gy) for 24 h on the viability of HCC cells (PAK7low HepG2 cells, PAK7moderate Huh7 cells, PAK7high Mahlavu cells and PAK7high SK-Hep-1 cells) responsive to varying levels of PAK7. Note: *P < 0.05 vs. 0 Gy. This experiment was conducted in triplicate.
Effect of PAK7 shRNAs on the viability of HCC cells after IR
SRB assay was conducted to evaluate the effect of PAK7 downregulation caused by transfection of PAK7 shRNA#1 and PAK7 shRNA#2 on the viability of PAK7moderate Huh7 cells and PAK7high Mahlavu cells, after exposure to the IR (24 h exposure to 2 Gy). As a result, in comparison with the scr-shRNA group, cells in the PAK7 shRNA#1 group and PAK7 shRNA#2 group presented with the significant downregulation of PAK7 (all P < 0.05, Figure 2(A) to (C)). Moreover, the effect of 2 Gy on cell viability was further enhanced in Mahlavu and Huh7 cells following the transfection of PAK7 shRNAs (all P < 0.05, Figure 2(D) and (E)).

Effect of PAK7 shRNAs on the viability of HCC cells after IR. Note: (A–C) Western blotting is conducted to detect the protein expression of PAK7 in HCC Mahlavu and Huh7 cells after transfection of PAK7 shRNA#1 and PAK7 shRNA#2. (D and E) Sulforhodamine B (SRB) assay is conducted to measure the effect of PAK7 shRNAs on the viability of Mahlavu and Huh7 cells after 2 Gy IR for 24 h; *P < 0.05 vs. the Blank group or scr-shRNA group; #P < 0.05 vs. PAK7 shRNA#1 group; &P < 0.05 vs. PAK7 shRNA#2 group; %P < 0.05 vs. IR group; @P < 0.05 vs. the IR + scr-shRNA group. The experiment was repeated three times.
Effect of PAK7 shRNAs on HCC cell clone formation after IR
Results of the clone formation assay (Figure 3) indicated that in comparison with the 21% or 47% loss of clonogenicity elicited by exposure to 2 Gy IR in Mahlavu or Huh7 cells, respectively, PAK7 shRNAs dampened the abilities of Mahlavu or Huh7 cell to form colony by 61% or 76% (PAK7 shRNA#1), and by 62% or 79% (PAK7 shRNA#2). Moreover, Combining PAK7 shRNAs with 2 Gy IR could further reduce the formation of clones in Mahlavu and Huh7 cells (all P < 0.05).
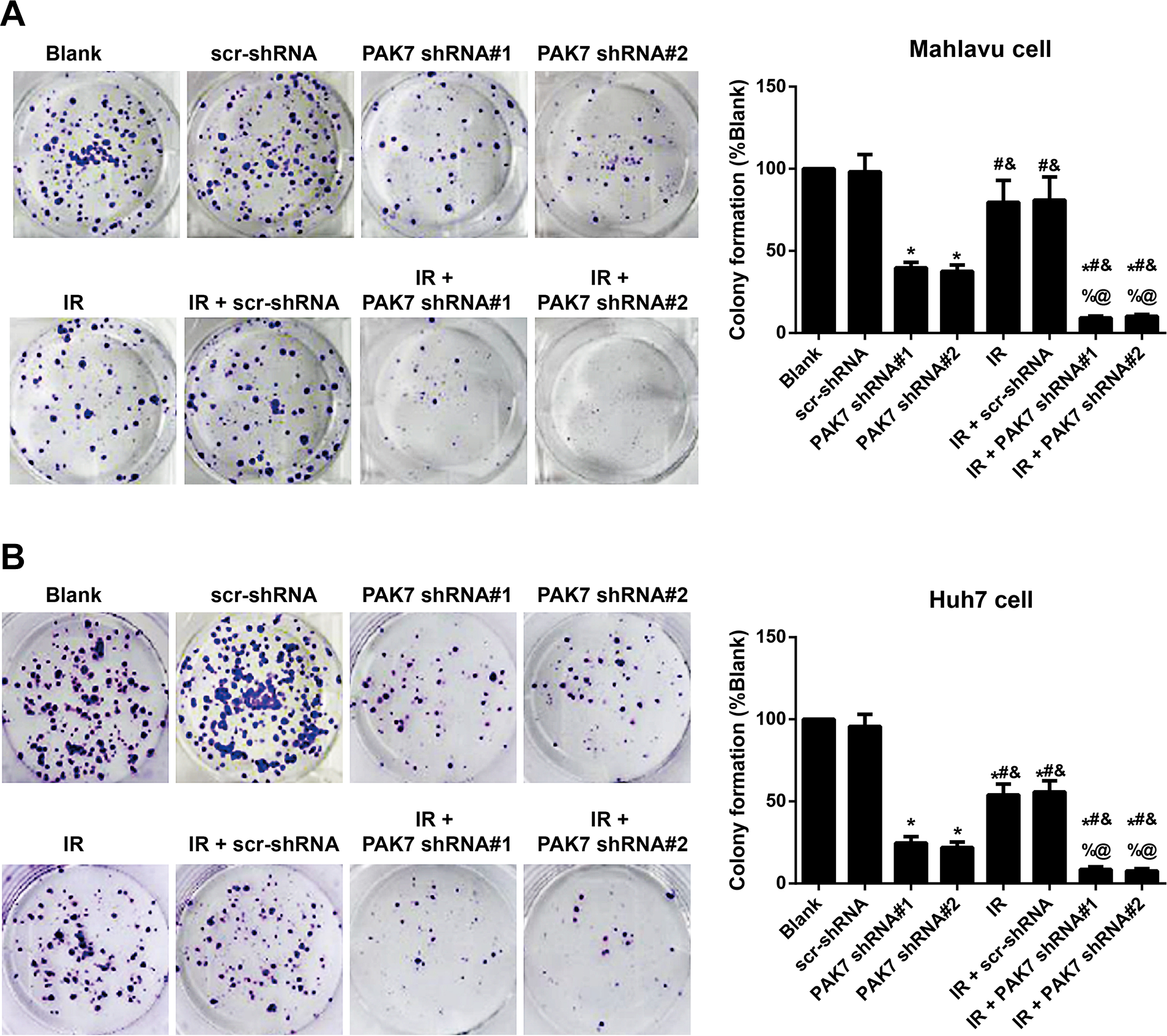
Effect of PAK7 shRNAs on the ability of HCC cells to form the clones after IR. Note: Clone formation assays are conducted to evaluate the effect of 2 Gy IR, PAK7 shRNA#1 or PAK7 shRNA#2 on clonogenicity of Mahlavu cells (A) or Huh7 cells (B); *P < 0.05 vs. the Blank group or scr-shRNA group; #P < 0.05 vs. PAK7 shRNA#1 group; &P < 0.05 vs. PAK7 shRNA#2 group; %P < 0.05 vs. IR group; @P < 0.05 vs. the IR + scr-shRNA group. The experiment was repeated three times.
Effect of PAK7 shRNAs on the apoptosis of HCC cell after IR
Results of Annexin-V-FITC/PI and flow cytometry (Figure 4) showed that PAK7 shRNAs could evidently facilitate the apoptosis of HCC cells, and the apoptotic rates of cell in PAK7 shRNA#1 group (apoptotic rate of Mahlavu cell: 23.71%; apoptotic rate of Huh7 cell: 17.27%) and PAK7 shRNA#2 group (apoptotic rate of Mahlavu cell: 24.91%; apoptotic rate of Huh7 cell: 20.02%) were much higher than those in the scr-shRNA group (apoptotic rate of Mahlavu cell: 3.36%; Apoptotic rate of Huh7 cell: 4.90%) (all P < 0.05). Moreover, as compared to the IR group (apoptotic rate of Mahlavu cells: 8.04%; apoptotic rate of Huh7 cells: 15.95%), the apoptotic rate of cells in IR + PAK7 shRNA#1 group (apoptotic rate of Mahlavu cells: 28.53%; apoptotic rate of Huh7 cells: 35.4%) and IR + PAK7 shRNA#2 group (apoptotic rate of Mahlavu cells: 28.95%; apoptotic rate of Huh7 cells: 36.44%) further increased (all P < 0.05).
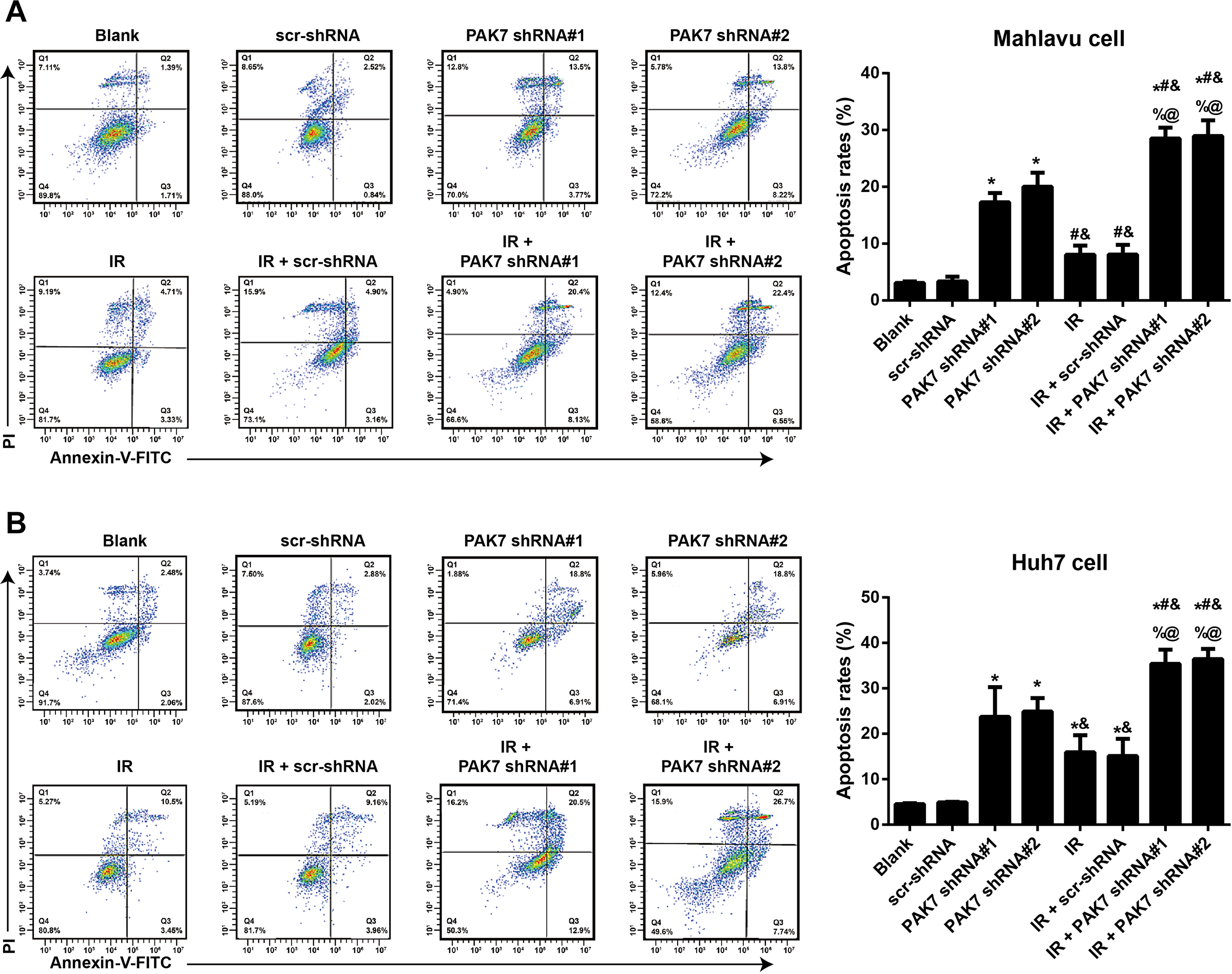
Effect of PAK7 shRNAs on the apoptosis of HCC cells after IR. Note: (A and B) Annexin-V-FITC/PI and flow cytometry are conducted to determine the effect of 2 Gy IR, PAK7 shRNA#1 or PAK7 shRNA#2 on the apoptosis rate of Mahlavu cells (A) or Huh7 cells (B); *P < 0.05 vs. the Blank group or scr-shRNA group; #P < 0.05 vs. PAK7 shRNA#1 group; &P < 0.05 vs. PAK7 shRNA#2 group; %P < 0.05 vs. IR group; @P < 0.05 vs. the IR + scr-shRNA group. The experiment was repeated three times.
Effect of PAK7 shRNAs on the cell cycle and DNA damage in HCC cells after IR
Flow cytometry was carried to determine the cycle distribution of HCC cells in each group (Figure 5), and consequently, in comparison with Mahlavu cell and Huh7 cell in the Blank group or scr-shRNA group, the cell cycle was arrested in G2/M phase in the other groups, especially in the IR + PAK7 shRNA#1 group and IR + PAK7 shRNA#2 group. Moreover, the expression of γ-H2AX, an indicator of DNA damage, was also detected (Figure 6) and PAK7 shRNAs could upregulate the expression of γ-H2AX in HCC cells, which could be further enhanced by the co-treatment with IR and PAK7 shRNAs (all P < 0.05).
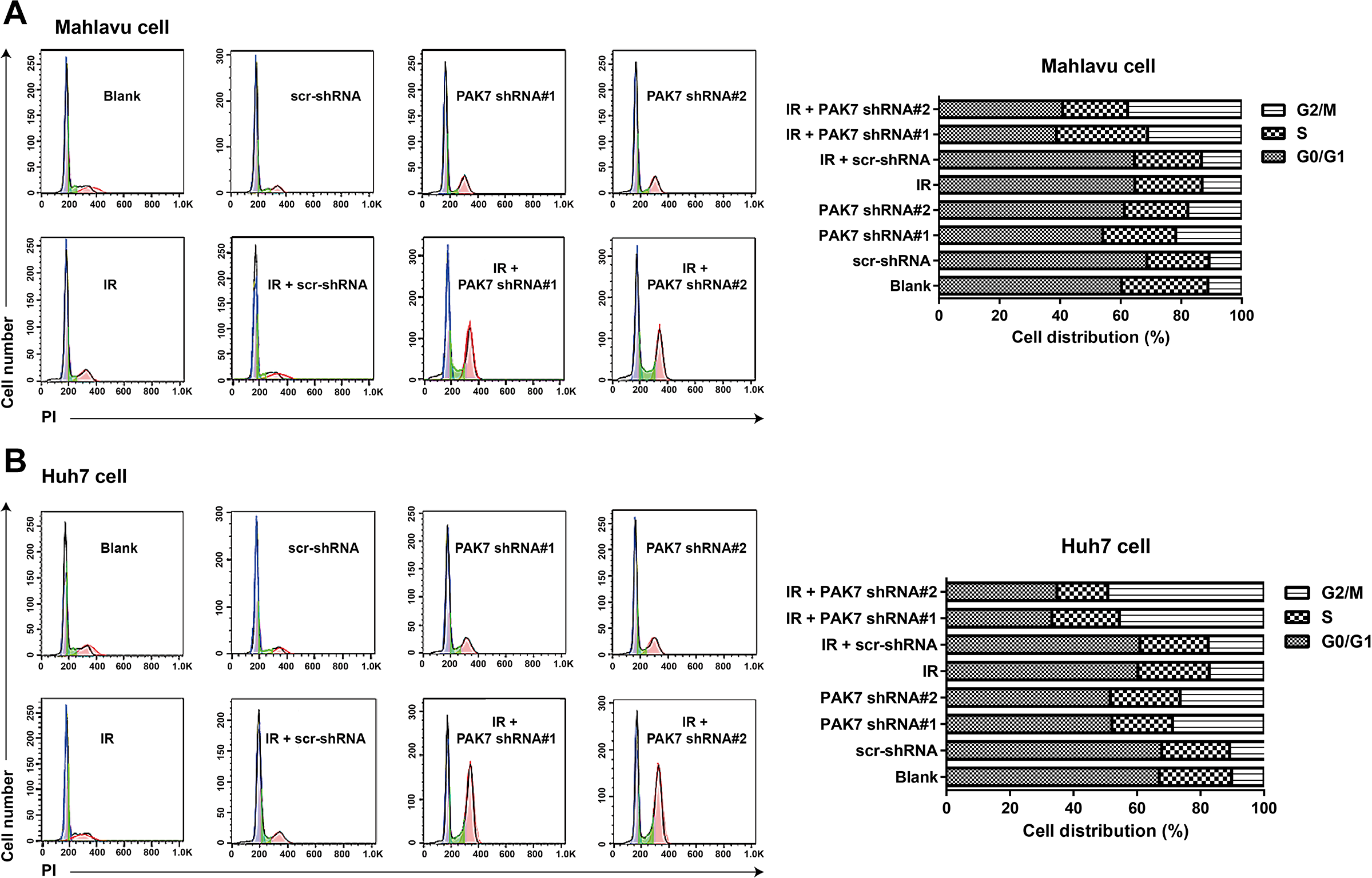
Effect of PAK7 shRNAs on the cell cycle in HCC cells after IR. Note: Flow cytometry is carried out to evaluate the effect of 2 Gy IR, PAK7 shRNA#1 or PAK7 shRNA#2 on cell cycle of Mahlavu cell (A) or Huh7 cell (B); this experiment was repeated three times.
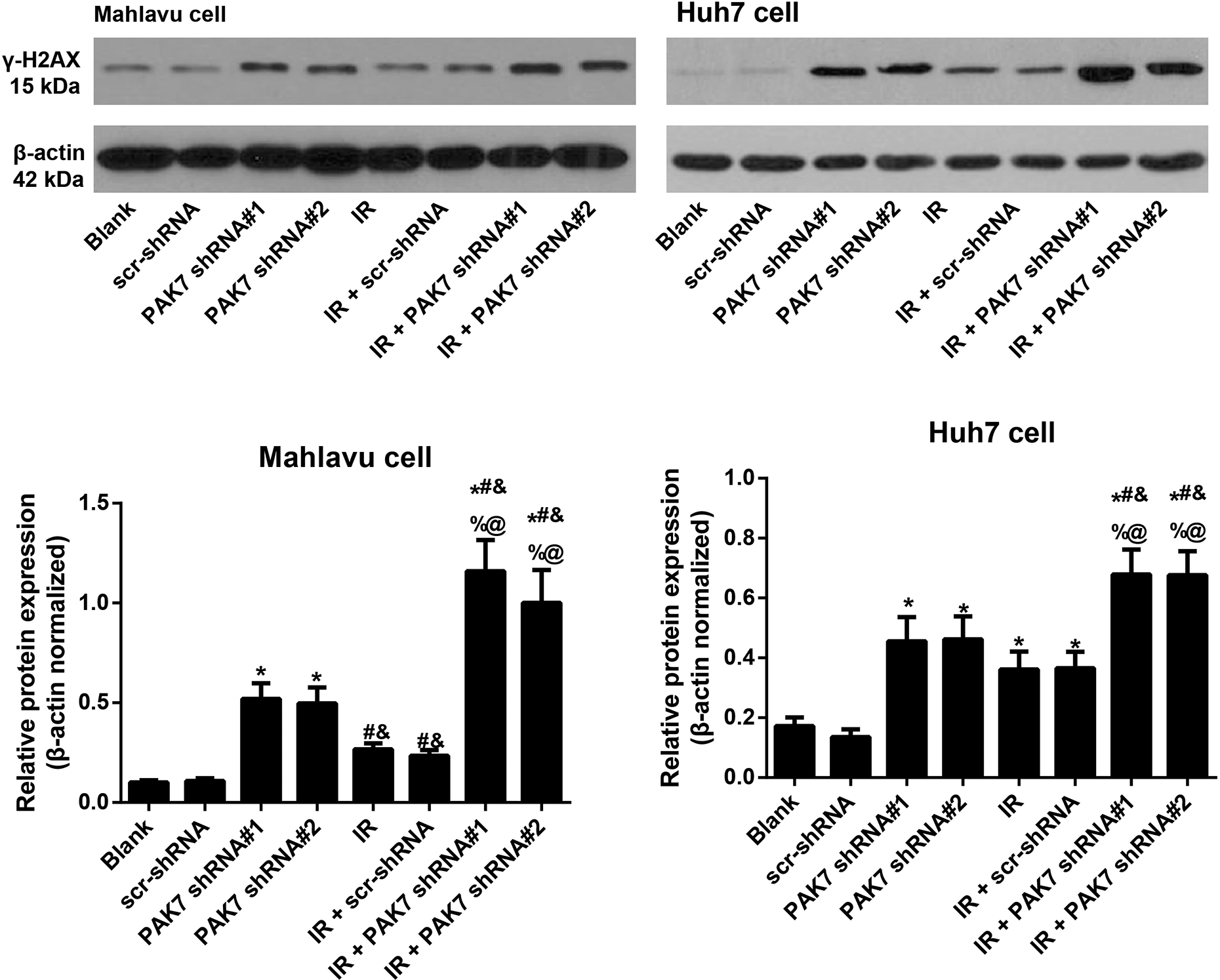
Effect of PAK7 shRNAs on DNA damage in HCC cells after IR. Note: *P < 0.05 vs. the Blank group or scr-shRNA group; #P < 0.05 vs. PAK7 shRNA#1 group; &P < 0.05 vs. PAK7 shRNA#2 group; %P < 0.05 vs. IR group; @P < 0.05 vs. the IR + scr-shRNA group. The experiment was repeated three times.
Discussion
At present, the upregulation of PAK7 has been reported in various cancer.22–24 In this study, PAK7 was upregulated evidently in poorly differentiated HCC cell, and such a result was in line with the previous findings,18,19,25 suggesting a potential role of PAK7 as a cancer addictive oncogene. In recent years, PAK family was shown to be associated to IR. For instance, PAK1 phosphorylation was significantly reduced after IR with the overexpression of transthyretin (TTR). 26 Besides, the cleavage and activation of PAK2 can be induced during the early stages of IR-triggered apoptosis of human epidermal carcinoma A431 cells. 27 Moreover, the combination of PAK6 inhibition with radiotherapy could further decrease the survival rate of prostatic cancer cells. 28 The SRB assay remains one of the most widely used methods for in vitro cytotoxicity screening,29,30 which has also been shown to be effective for in vitro testing of cancer cell sensitivity to radiation, with a sensitivity comparable to that of the standard clonogenic assay. 31 To deepen our understanding on the mechanism of PAK7 on the tumorigenesis and treatment of HCC, we evaluated the effect of IR at different doses on the viability of several HCC cells using SRB assay, and as a result, we found that after 24 h of IR at 2 Gy, significant decreases were shown in the viability of PAK7low HepG2 cells and PAK7moderate Huh7 cells, but we found no significant killing effect on the PAK7high Mahlavu and SK-Hep-1 cells, indicating a vital role of PAK7 expression in the radiosensitivity of HCC cells.
Generally, tumorigenesis results from the abnormal proliferation of cells. Previous research has shown that SOX21-AS1/miR-144-3p/PAK7 axis acts as a carcinogen in the development of glioma through regulating its cell proliferation. 23 Notably, PAK7 overexpression was also correlated with the abnormal proliferation of various tumor cells, such as osteosarcoma 32 and gastric cancer. 33 It should be noted that the ectopic introduction of miR-526a into HCC cells resulted in a significant decrease in cell apoptosis via targeting PAK7 according to the work of Zhan et al. 18 In our work, PAK7 shRNA was able to induce the IR-caused decrease in the viability of HCC cells, while reduce the clone formation ability of HCC cells. Continuous increment in the quantity of cancer cell is correlated to various factors, not only with the proliferation, but also with the pattern of cell death. 34 Cell apoptosis is a dominant pattern for the reduction of tumor cells. 35 At the meantime, radiotherapy, by inducing tumor cell apoptosis, can retard the increased quantity of tumor cells. An increasing body of evidence has shown the correlation between the radiotherapy sensitivity of cells and apoptosis. In addition, endogenous PAK7 shuttles between mitochondria and the nucleus and induces resistance to apoptosis by phosphorylating Bcl2-associated agonist of cell death in the mitochondria, 15 suggesting that PAK7 can alter the tumor cell apoptosis to affect the treatment of cancer. In this study, we noted that transfection of PAK7 shRNAs could enhance the IR-induced apoptosis of HCC cells evidently. Similarly, it had been reported that PAK7 deletion could reduce the proliferation but increase cell apoptosis of breast cancer. 36 By knocking-out of PAK7, the proliferation and colony formation of human osteosarcoma Saos-2 cells were inhibited with enhanced apoptosis. 37 Taken together, reducing PAK7 may enhance the sensitivity of HCC cells to radiotherapy via inhibiting cell viability and proliferation.
Irradiation-caused DNA damage is mainly characterized by DNA double strand breaks (DSBs), while the mechanism affecting the sensitivity of cells to radiotherapy is the recognition of damaged DNA by tumor cells, which can revitalize the damaged DNA to increase the survival rate of tumor cells. 38 Thus, increased DNA repair has become the major cause for the resistance of tumor cells to radiotherapy. 39 In this work, no significant effect of IR on HCC cells was found responsive to the high expression of PAK7, but the viability of HCC cells was suppressed in low PAK7 expression, which provided the possibility that PAK7 in high expression could enhance the activity of DNA repair-related pathway. During the DSB resulted from various factors, H2AX is phosphorylated into γ-H2AX, which, thus, has been taken as an indicator for DNA damage and repair, and its upregulation indicated the exacerbation of DNA damage.40,41 In our study, we found that the knockout of PAK7 in HCC cells could further increase the IR-induced expression of γ-H2AX, which could indirectly implied that PAK7 might reduce the DSB of DNA in HCC cells and accelerate the repair of DNA damage, thereby weakening the sensitivity of cells to radiotherapy. Irradiation-associated DNA damage can lead to G2/M checkpoint activation and consequent G2/M phase arrest, enabling DNA repair to occur prior to cellular entry into mitosis. 42 In this work, we noted that PAK7 knockout could arrest the cycle of cells in G2/M phase, coinciding with the previous report that Ru (II) complex could arrest the cycle of cell in G2/M phase by inhibiting the expression of Cdc42 protein. 43 The loss in cancer cell viability is due to a dual Rac/Cdc42 inhibitor MBQ-167-mediated G2/M cell-cycle arrest and subsequent apoptosis, especially of the detached cells. 44 However, the activation of PAK7 is likely to be independent of Cdc42 binding, which obviously enhanced cellular biologic regulations of PAK7 in tumor development. 45 Overall, the effect of PAK7 on cell cycle may be affected by Cdc42, but the specific effect will be explored in the future work.
In conclusion, in comparison with the normal adult liver epithelial THLE-2 cells, PAK7 was upregulated significantly in poorly differentiated HCC cells, but moderately or lowly expressed in well-differentiated cells. Following 24 h of 2 Gy IR, sharp decreases in viability were noted in the PAK7low HepG2 cells and PAK7moderate Huh7 cells, while PAK7high Mahlavu and SK-Hep-1 cells presented with no significant in viability. Moreover, downregulation of PAK7 may enhance the sensitivity of HCC cells to IR by reducing the clone formation, promoting the apoptosis and inhibiting DNA damage repair in HCC cells, projecting PDK7-targeting as an enhancer of radiosensitivity and a potential new therapeutic approach for patients with HCC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
