Abstract
Opacity of the lens caused by cataracts could lead to severe visual impairment and even blindness. Oxidative stress caused by exposure of lens epithelial cells to hydrogen peroxide (H2O2) can lead to DNA damage and impair cell function. Therefore, how to prevent lens epithelial cells from being harmed by H2O2 is an urgent problem. The ZNF219 gene belongs to the Kruppel like zinc finger gene family, which is involved in a variety of biological processes. In this study, we found the low expression of ZNF219 in H2O2-induced HLE-B3 cells. We further noticed ZNF219 could improve the survival rate of H2O2-induced HLE-B3 cells, and inhibit the apoptosis and oxidative stress response. Mechanically, ZNF219 protected human lens epithelial cells against H2O2-induced injury via targeting SOX9 through activating AKT/GSK3β pathway. We therefore thought ZNF219 was a key protective protein in the oxidative damage of human lens epithelial cells and the pathogenesis of cataract.
Introduction
Opacity of the lens caused by cataracts could lead to severe visual impairment and even blindness, seriously affecting the quality of life of patients. 1 Although there is no effective way to prevent cataract lens formation, surgical interventions have been effective, nevertheless, surgery can cause cataract recurrence. 2 It is important to explore the molecular mechanism of cataract occurrence. A recent study showed that damage caused by oxidative stress in the lens of the eye is a major cause of cataracts. 3 Oxidative stress caused by exposure of lens epithelial cells to hydrogen peroxide (H2O2) can lead to DNA damage and impair cell and tissue function. 4 H2O2 is a non-free radical member of the reactive oxygen family, which produces hydroxyl radicals that irreversibly damage the lens epithelium, leading to cell death and cataract formation. 5 Therefore, it was urgently needed to prevent lens epithelial cells from being harmed by H2O2.
The ZNF219 gene belongs to the Kruppel like zinc finger gene family, which is involved in a variety of biological processes, such as cell growth, cell differentiation, embryogenesis, and tumorigenesis.6,7 ZSCAN21 or other control transcription factors targeting ZNF219 could be a novel way to treat Parkinson’s disease and other diseases. 8 In addition, through the oligonucleotide microarray hybridization, it was found that ZNF219 was low expression in the lens of cataract patients compared with the normal lens. 9 However, few studies have been conducted on ZNF219 in cataract disease, and its mechanism is not clear. Previous studies have shown that ZNF219 could interact with SOX9 and co-located with SOX9 in the nucleus. 8 Moreover, ZNF219 promoted the differentiation of chondrogenic cells by up-regulating the expression of SOX9. 6
In this study, we detected the expression levels of ZNF219 in H2O2-induced HLE-B3 cells, and investigated whether ZNF219 could improve the survival of H2O2-induced HLE-B3 cells, and detected the effects of ZNF219 on the apoptosis and oxidative stress response of these cells. We further revealed the mechanisms underlying ZNF219 protecting human lens epithelial cells against H2O2-induced injury.
Methods
Clinical samples
Human lens epithelium samples and healthy control samples were collected from 20 ARC patients and 20 healthy post-mortem eyes with no more than 10 h after donor death from Inner Mongolia Chaoju Eye Hospital. The selected samples were recruited from patients without other eye diseases. The samples were stored at −80°C immediately after dissection. The research was performed in accordance with the Declaration of Helsinki related to human subjects. This study was approved by the Ethical Committee of Inner Mongolia Chaoju Eye Hospital.
Cell culture
Human LECs (HLE-B3) cells were accessed from Shanghai Academy of Life Science (Shanghai, China) and cultured in Dulbecco’s modified Eagle’s medium containing 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin in 5% CO2 culture hood. HLE-B3 cells were treated with 50 μM H2O2. ZNF219 plasmid and shRNA were obtained from Addgene and Sigma, respectively. For shRNA and plasmid transfection, Lipo 2000 was used.
Quantitative polymerase chain reaction (qPCR)
Total tissue and cellular RNA were isolated with the RNAiso Plus reagent (Takara Bio, Kusatsu, Japan). Then total RNA was reverse transcribed into cDNA by PrimeScript™ RT reagent Kit (Takara). qPCR assay was carried out with SYBR® Premix Ex Taq™ II (Takara). The primer of ZNF219 was purchased from Santa Cruz. b-actin: FP: 5′-CATTGCTGACAGGATGCAGAAGG-3′, RP: 5′-TGCTGGAAGGTGGACAGTGAGG-3′, SOX9: FP: 5′-GGCAAGCTCTGGAGGCTG-3′, RP: 5′-CCTCCACGAAGGGTCTCTTCT. The PCR reactions were performed on an ABI 7500 Fast system (Life Technologies, Carlsbad, USA).
Western blot
Protein was quantified by bicinchoninic acid (BCA) method, and 30 µg protein was added to each well. The cell samples were isolated to extract the proteins and electrophoresed with 8% SDS-PAGE, sequentially transferred onto the PVDF membranes for 2 h, blocking with 5% fat-free milk in TBST buffer at room temperature for 2 h. PVDF membranes were subsequently cultured with primary antibodies targeting ZNF219 (1:1000, Thermo Scientific, Carlsbad, USA), b-actin (1:10,000; Cell Signaling Technology, Danvers, USA), caspase-3, Bax, Bcl-2, SOX9, p-Akt, Akt, p-GSK3β, and GSK3β (1:1000; Cell Signaling Technology, Danvers, USA) at room temperature for 1 h. Subsequently the membranes were incubated with horseradish peroxidase (HRP) -conjugated secondary antibodies at room temperature for 1 h. Signals were then visualized by the use of an ECL kit.
Cell viability
Cell viability assay was conducted with CCK-8 kit (Bimake, Houston, USA). Cells were suspended and plated in 96-well plates (10 4 cells/well). CCK-8 solution was put into each well every 24 h and the cells were incubated for 1.5 h. The absorbance of each well was read at 450 nm with a microplate reader (Mindray 96-A multifunctional microplate reader, Shenzhen, China).
Cell apoptosis assay
Cells were resuspended and mixed with annexin V-FITC and propidium iodide (PI) at room temperature for 10 min. Subsequently the samples were analyzed by a FACS Calibur flow cytometer, and the apoptosis cells were analyzed and compared among different groups.
Immunofluorescence
The cells were fixed using 4% formaldehyde for 10 min, and permeabilized using 0.1% Triton X-100 for 10 min. Then cells were added with anti-cleaved caspase-3 antibody (1:500 dilution) in 1% goat serum with PBS for 1 h. After washing in PBST, the cells were conjugated with related secondary antibody conjugated with rhodamine for 1 h. The cells were stained with 0.1 μg/ml DAPI and mounted under a coverslip.
Assessment of antioxidant activity
The levels of MDA, SOD, GSH were assessed by the detection kits of Nanjing Jiancheng Bio-engineering Institute (Jiangsu, China) in accordance to the manufacturer’s instructions.
Lactate dehydrogenase (LDH) release activity
For LDH release activity, LDH cytotoxicity detection kit was used (Clontech laboratories Inc., CA, USA), following the manufacturer’s protocol. Briefly, 100 µL of culture supernatant from each well were added with 100 µL of the LDH reaction solution, and then incubated at room temperature for 30 min. Then measure the optical densities of the samples at 490 nm using a microplate reader.
Statistics
GraphPad 7.0 was utilized for statistical analysis in this study. Data were shown as mean ± SEM in this study. Student’s t-test was used for statistical comparisons, and p < 0.05 is thought significant.
Results
ZNF219 is down-regulated in lens epithelium samples of ARC patients and H2O2-induced HLE-B3 cells
The expression level of ZNF219 in lens epithelium samples of ARC patients and healthy donors was measured. As shown in Figure 1(A), ZNF219 was decreased in lens epithelium tissues of ARC patients (Figure 1(A)). Consistently, the expression of ZNF219 was also decreased in lens epithelium tissues of ARC patients as detected by Immunoblot and Immunofluorescence (Figure 1(B) and (C)). In H2O2 stimulated HLE-B3 cells, we also noticed reduced ZNF219 level both in mRNA and protein level (Figure 1(D) and (E)). Therefore, ZNF219 was proved to be downregulated in human lens epithelium samples of ARC patients and H2O2-induced HLE-B3 cells.
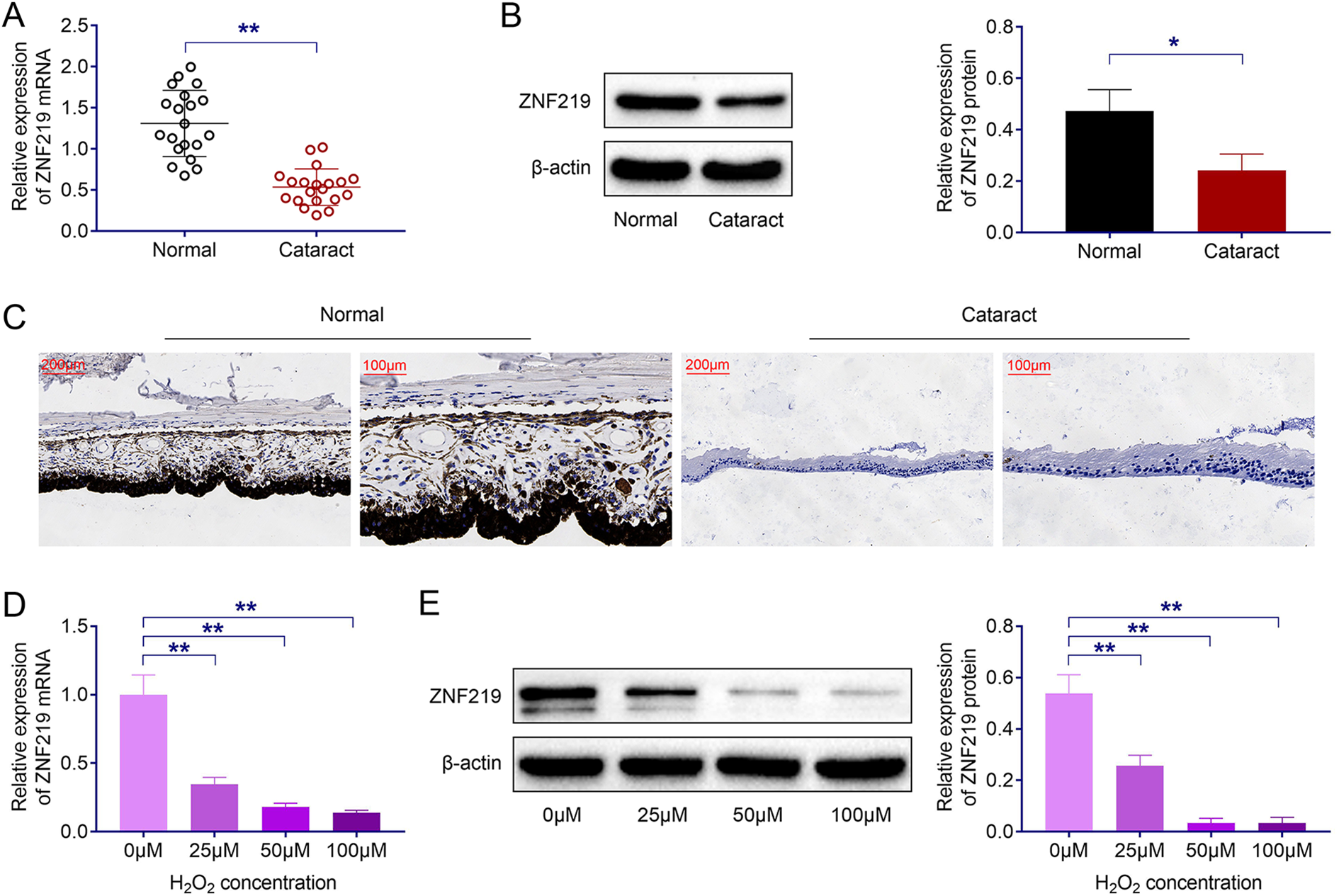
ZNF219 is down-regulated in human lens epithelium samples of ARC patients and H2O2-induced HLE-B3 cells. (A, B, C) The mRNA and protein level of ZNF219 in human len epithelia samples of ARC and healthy people as detected by qPCR, Immunoblot and Immunofluorescence. (D, E) The level of ZNF219 in HLE-B3 cells induced by increasing concentration of H2O2 measured by qPCR and Immunoblot. Results are presented as mean ± SEM, *p < 0.05, **p < 0.01.
ZNF219 promotes cell viability in H2O2-induced HLE-B3 cells
To evaluate the role of ZNF219 in HLE-B3 cells, HLE-B3 cells were transfected with pcDNA 3.0 vector, pcDNA-ZNF219, shNC and shZNF219 to modulate the level of ZNF219. As expected, ZNF219 transfection significantly increased ZNF219 level and shZNF219 transfection reduced ZNF219 level (Figure 2(A)). Subsequently, CCK-8 assay indicated ZNF219 overexpression promoted the viability of HLE-B3 cells and ZNF219 ablation inhibited cell viability (Figure 2(B)). Thus, ZNF219 could modulate cell viability in HLE-B3 cells.

ZNF219 promotes cell viability in H2O2-induced HLE-B3 cells. (A) The expression level of ZNF219 in response to ZNF219 plasmid and shRNA transfection. (B) Cell viability in ZNF219 overexpressed and downregulated HLE-B3 cells. Results are presented as mean ± SEM, *p < 0.05, **p < 0.01.
ZNF219 suppresses cell apoptosis in H2O2 stimulated HLE-B3 cells
To evaluate the function of ZNF219 in cell apoptosis, flow cytometry assay was performed. As shown in Figure 3(A), the apoptosis rate was reduced in HLE-B3 cells after ZNF219 restoration, and cell apoptosis was increased following ZNF219 knockdown (Figure 3(B)). Also, Immunofluorescence indicated decreased or increased accumulation of caspase-3 following ZNF219 overexpression or down expression (Figure 3(C)). Western blot analysis implied that ZNF219 overexpression elevated Bcl-2 level but down-regulated caspase-3 and Bax expression in HLE-B3 cells. However, the level of Bcl-2, Bax and caspase-3 displayed converse change after ZNF219 ablation (Figure 3(D)). These results further implied ZNF219 restoration inhibited the apoptosis of HLE-B3 cells.

ZNF219 suppresses cell apoptosis in H2O2-induced HLE-B3 cells. (A, B) Cell apoptosis in ZNF219 overexpressed and downregulated HLE-B3 cells. (C) Cellular level of caspase3 in ZNF219 overexpressed and downregulated HLE-B3 cells detected by Immunofluorescence. (D) The expression level of Bcl2, Bax and caspase-3 in ZNF219 overexpressed and downregulated HLE-B3 cells. Results are presented as mean ± SEM, *p < 0.05, **p < 0.01.
ZNF219 inhibits oxidative stress in HLE-B3 cells induced by H2O2
We also noticed reduced LDH activity following ZNF219 overexpression and increased LDH activity by ZNF219 knockdown (Figure 4(A)). ZNF219 upregulation increased the level of GSH-Px and SOD levels and reduced MDA level (Figure 4(B)), while ZNF219 downregulation reduced the level of GSH-Px and SOD levels and induced MDA level. Therefore, we demonstrated that ZNF219 inhibited HLE-B3 cells oxidative damage.

ZNF219 inhibits oxidative stress in HLE-B3 cells induced by H2O2. (A) LDH activity in ZNF219 overexpressed and downregulated HLE-B3 cells. (B) The MDA, SOD and GSH-Px level in ZNF219 overexpressed and downregulated HLE-B3 cells. Results are presented as mean ± SEM, *p < 0.05, **p < 0.01.
SOX9 is a target of ZNF219 and modulates the activation of AKT/GSK3β pathway
In human lens epithelium samples of ARC patients and healthy donors samples, SOX9 was downregulated in ARC patients both in mRNA and protein level (Figure 5(A) and (B)). Similarly, SOX9 was downregulated as detected by Immunofluorescence (Figure 5(C)). H2O2 treatment could reduce the protein level of SOX9 (Figure 5(D)). To verify the regulatory effect of ZNF219 on SOX9, we detected SOX9 protein level in response to ZNF219 overexpression or downregulation. ZNF219 overexpression elevated SOX9 level and ZNF219 downregulation reduced SOX9 level (Figure 5(E)). Phosphorylated Akt and GSK3β levels were induced by ZNF219 overexpression and SOX9 ablation totally reversed the activation of AKT/GSK3β pathway (Figure 5(F)). Taken together, SOX9 is a potential target of ZNF219 and could regulate the activation of AKT/GSK3β pathway.

SOX9 is a target of ZNF219 and regulates the activation of AKT/GSK3β pathway. (A, B, C) The mRNA and protein level of SOX9 in human len epithelia samples of ARC and healthy people as detected by qPCR, Immunoblot and Immunofluorescence. (D) The level of SOX9 in HLE-B3 cells induced by H2O2 measured by Immunoblot. (E) The expression level of SOX9 in response to ZNF219 plasmid and shRNA transfection. (F) The expression level of phosphorylated AKT and GSK3β and total protein level of AKT and GSK3β in response to ZNF219 plasmid and shRNA transfection. Results are presented as mean ± SEM, *p < 0.05, **p < 0.01.
SOX9 ablation reverses the effect of ZNF219 in HLE-B3 cells
As SOX9 was a target of ZNF219, we wonder whether SOX9 was involved in ZNF219 mediated cell viability, apoptosis and oxidative stress. ZNF219 overexpression increased cell viability and SOX9 knockdown reversed the effect (Figure 6(A)). As for apoptosis, ZNF219 overexpression reduced cell apoptosis and SOX9 knockdown-induced cell apoptosis induced by ZNF219 (Figure 6(B) and (C)). The reduced LDH activity caused by ZNF219 could be relieved by SOX9 knockdown (Figure 6(D)). Taken together, SOX9 ablation reverses the effect of ZNF219 in cell viability, apoptosis and oxidative stress in HLE-B3 cells.

SOX9 ablation reverses the effect of ZNF219 in HLE-B3 cells. (A) Cell viability in ZNF219 overexpressed and ZNF219 overexpressed plus SOX9 downregulated HLE-B3 cells. (B, C) Cell apoptosis in ZNF219 overexpressed and ZNF219 overexpressed plus SOX9 downregulated HLE-B3 cells. (D) LDH activity in ZNF219 overexpressed and ZNF219 overexpressed plus SOX9 downregulated HLE-B3 cells. Results are presented as mean ± SEM, p < 0.01.
Discussion
Lens opacity is the main cause of cataract, and surgical treatment is the main way to fight against cataract. 10 However, the main cause of the recurrence of cataract is the surgical treatment. For cataract, targeted therapy has the potential advantages of less side effects. 11 In order to improve lens opacity and cataract, we still need to have a deeper understanding of its pathogenesis and find out the key regulatory proteins. In recent years, it is generally believed that the damage of lens epithelial cells caused by oxidative stress is a major cause of cataract. 12 Herein, we noticed that ZNF219 protected human lens epithelial cells from H2O2-induced damage, confirming the important role of this protein in cataract induced by oxidative damage to lens epithelial cells.
Performing CCK-8, FCM, Immunofluorescence, and Immunoblot assays, we found ZNF219 improved the survival, inhibited the apoptosis and oxidative stress response of H2O2-induced HLE-B3 cells. ZNF219 protein, as a transcription factor, is known related to a variety of biological processes, such as cell growth, cell differentiation, embryogenesis, and tumorigenesis.7,8,13 ZNF219 mediated chondrocyte differentiation via assembling a transcription factory SOX9. 8 Similarly, we here noticed that ZNF219 protected lens epithelial cells against H2O2-induced injury via targeting SOX9. It was reported that ZNF219 was related to the regulation of transcription and developmental regulation. 14 Additionally, previous study also revealed that ZNF219 acted as a transcriptional repressor of the HMGN1 promoter. 6 We next should clarify whether ZNF219 protected lens epithelial cells against H2O2-induced injury through repressing HMGN1 promoter. 7 Also, ZNF219 was low expression in the lens of cataract patients compared with the normal lens. Consistently, we confirmed the low expression of ZNF219 in H2O2-induced HLE-B3 cells.
Previous studies have shown that ZNF219 can interact with SOX9 and co-located with SOX9 in the nucleus. 6 Moreover, ZNF219 promoted the differentiation of chondrogenic cells by up-regulating the expression of SOX9. 15 As we know, SOX9 is an important member of the SOX family affecting multiple cellular processes.16–18 SOX9 could activate downstream Akt activity. 19 SOX9 could inhibit apoptosis and inflammatory response by activating Akt /GSK3β pathway, and protect MRC5 of fetal lung fibroblasts from LPS-induced injury. 20 Importantly, we found ZNF219 protected human lens epithelial cells against H2O2-induced injury via targeting SOX9, and the precise regulatory mechanism needs further study.
Akt pathway is involved in a variety of biological processes, including cell proliferation, apoptosis, inflammatory response, and oxidative stress process.21–23 In addition, Akt activation also plays an important role in the protection of cataract lens cells. Resveratrol oxalate inhibited oxidative stress and apoptosis induced by H2O2 by activating the Akt pathway, and protected human lens epithelial cells from damage induced by H2O2. 24 The up-regulation of circhipK3 mediated the proliferation of HLECs and inhibited apoptosis by activating PI3K/Akt, thereby protecting human lens epithelial cells from dysfunction. 25 H2O2 could downregulate the phosphorylation level of Akt in HLECs and aggravate cataract injury. 26 In this study, we confirmed that ZNF219 protected human lens epithelial cells from H2O2-induced damage via Akt pathway, further provided the evidence that Akt pathway regulated the progression of cataracts and lens opacity.
In conclusion, we found the low expression of ZNF219 in H2O2-induced HLE-B3 cells. ZNF219 could improve the survival, inhibit the apoptosis and oxidative stress response of H2O2-induced HLE-B3 cells. We uncovered that ZNF219 protected human lens epithelial cells against H2O2-induced injury via targeting SOX9 via activating Akt pathway.
Footnotes
Author contributions
Qing Guo designed the study, supervised the data collection, Qing Geletu analyzed the data, interpreted the data, Yanlai Zhang prepare the manuscript for publication and reviewed the draft of the manuscript. All authors have read and approved the manuscript.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval was obtained from the Ethics Committee of Inner Mongolia Chaoju Eye Hospital.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
