Abstract
Glutathione S-transferase placental form-positive (GST-P+) foci are markers of preneoplastic lesions in rat hepatocarcinogenesis. Our previous studies using reporter gene transgenic rats showed that furan, a hepatocarcinogen in rodents, rapidly induces the formation of GST-P+ foci after short exposure without reporter gene mutation. We hypothesized that GST-P+ foci induced by furan may have biological characteristics different from those induced by diethylnitrosamine (DEN), a genotoxic hepatocarcinogen. Accordingly, we compared the cell kinetics of GST-P+ foci after cessation of DEN treatment and performed comprehensive gene expression in DEN- or furan-induced GST-P+ foci. The number and area of DEN-induced GST-P+ foci were increased after cessation of treatment, whereas furan decreased these parameters. Size distribution analysis showed that large furan-induced GST-P+ foci disappeared after cessation of treatment. Hierarchical cluster analysis showed that all samples from GST-P+ foci induced by furan were separated from those induced by DEN. SOX9 expression was upregulated in furan-induced GST-P+ foci and was detected by immunohistochemistry in large furan-induced GST-P+ foci. Our results indicated that large furan-induced GST-P+ foci were quite different from DEN-induced GST-P+ foci at the molecular and cellular levels. And one of the properties of disappearing large GST-P+ foci were characterized by inclusion of hepatocytes expressing SOX9.
Keywords
Introduction
Glutathione S-transferase placental form-positive (GST-P+) foci are preneoplastic lesions in rat hepatocarcinogenesis and can be used as an end point marker in medium-term rat liver bioassays (eg, Ito’s model). 1,2 The good correlation between outcomes in this model and those in carcinogenic bioassays suggests that rat hepatocytes express GST-P during the process of neoplastic conversion. However, rat hepatocytes exhibiting GST-P expression do not always convert into neoplastic cells because the incidences and multiplicities of GST-P+ foci induced by various hepatocarcinogens are much higher than those observed in subsequent hepatocellular adenomas and carcinomas. 3 -7 Thus, the mechanisms through which GST-P+ hepatocytes transform into neoplastic cells are still unclear, and other key molecules are also expected to be involved in the conversion of GST-P+ cells into neoplastic cells.
Glutathione S-transferase placental form-positive foci are spontaneously observed in aged rats, consistent with the increased incidence of hepatocyte-derived tumors in untreated rats. In addition, even tumor-promoting agents that do not induce DNA damage can increase the number of GST-P+ foci. According to our previous studies using gpt delta rats, genotoxic hepatocarcinogens rapidly induce large numbers of GST-P+ foci and increase reporter gene mutations. 4,8 -10 In contrast, nongenotoxic hepatocarcinogens require long-term exposure to induce the formation of GST-P+ foci. 9,11 -13
Furan, which is an intermediate in the chemical manufacturing industry and a contaminant in various foods, is a hepatocarcinogen in rodents. 5 Because of inconsistent results in conventional genotoxic studies, 5,14 -16 it is unclear how furan induces hepatocarcinogenicity. However, in vivo reporter gene assays have shown that furan yields clearly negative results. Previously, we conducted in vivo mutation assays and quantitative analysis of GST-P+ foci using gpt delta rats treated with furan at carcinogenic doses. As a result, furan induced striking elevation of GST-P+ foci within a very short duration of exposure, despite not inducing significant changes in reporter gene mutation frequency in the liver. 3 These results allowed us to hypothesize that GST-P+ foci induced by furan may have different biological characteristics and hepatocarcinogenic process from those induced by genotoxic hepatocarcinogens.
Accordingly, in this study, we compared the cell kinetics of GST-P+ foci induced by a potent genotoxic carcinogen, diethylnitrosamine (DEN) or furan after the cessation of their treatment. In addition, comprehensive gene expression analysis was performed using messenger RNA (mRNA) extracted from DEN- or furan-induced GST-P+ foci isolated by laser microdissection. SOX9 was selected as an upregulated gene and evaluated by immunohistochemical staining in GST-P+ foci.
Materials and Methods
Reagents
Diethylnitrosamine was purchased from Tokyo Chemical Industry. Furan was obtained from FUJIFILM Wako Pure Chemical.
Experimental Animals
Specific pathogen-free male F344 rats (5 weeks old) were purchased from Japan SLC. All animals were housed in a room with a barrier system and maintained under the following conditions: temperature of 23 °C ± 2 °C, relative humidity of 55% ± 15%, ventilation frequency of 12 times/h, and a 12-hour light/dark cycle. Animals were given free access to a CRF-1 basal diet (Oriental Yeast Co) and tap water. At the end of each experiment, all animals were euthanized by exsanguination via transection of the abdominal aorta under deep anesthesia. The protocols for the current study were approved by the Animal Care and Utilization Committee of the National Institute of Health Sciences (no. 385; August 1, 2012, no. 545; July 17, 2015).
Animal Treatment
Experiment I
Experiment I was conducted to investigate the kinetics of GST-P+ foci induced by DEN or furan after 13 weeks of carcinogen treatment (Figure 1A). Following 1 week of acclimatization, groups of 30 rats were given 10 ppm DEN in the drinking water or 8 mg/kg/d (5 d/wk) furan by gavage for 13 weeks. The control group was given tap water. After 13 weeks of treatment, 20 rats in each group were euthanized under deep anesthesia. The remaining rats were maintained on a basal diet and tap water for 7 weeks. During necropsy, the livers were immediately removed and fixed in 10% buffered formaldehyde and routinely processed by preparing hematoxylin and eosin specimen.
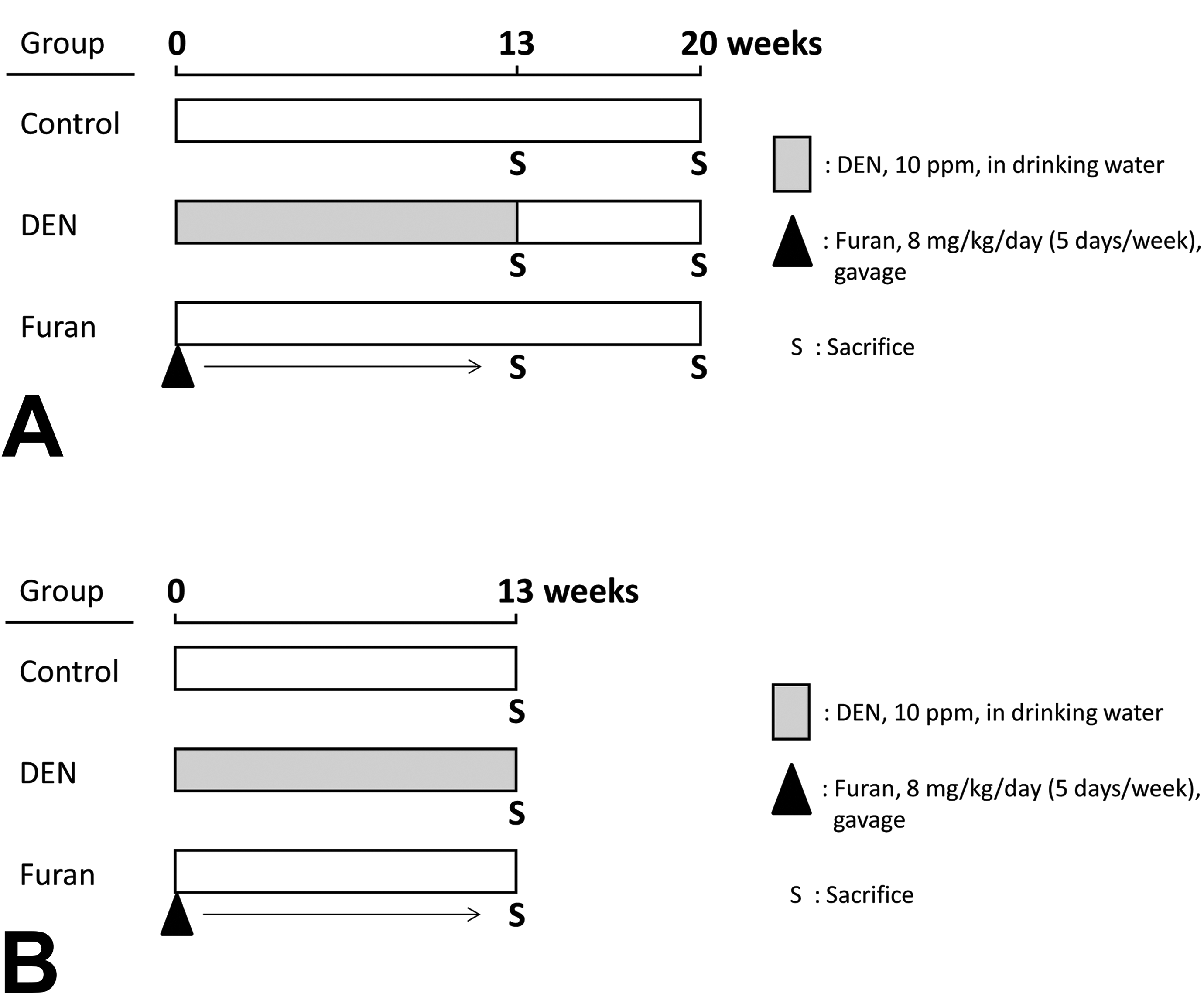
Graphical schematic of the experimental protocol. In experiment I, groups of 30 rats were given 10 ppm DEN in the drinking water or 8 mg/kg/d (5 d/wk) furan by gavage for 13 weeks. The control group was given tap water. After 13 weeks of treatment, 20 rats in each group were killed. The remaining rats were maintained on a basal diet and tap water for 7 weeks. At the end of the experiments, all rats were killed, and quantification of GST-P+ foci was conducted (A). In experimental II, 10 or 30 rats were given 10 ppm DEN in the drinking water or 8 mg/kg/d (5 d/wk) furan by gavage for 13 weeks. The control group was given tap water. After treatment, all rats were killed and global gene expression in GST-P+ foci was analyzed (B). DEN indicates diethylnitrosamine; GST-P+, glutathione S-transferase placental form-positive.
Experiment II
Experiment II was conducted to examine gene expression by complementary DNA (cDNA) microarray specifically in GST-P+ foci (Figure 1B). Following 1 week of acclimatization, 10 or 30 rats were given 10 ppm DEN in the drinking water or 8 mg/kg/d (5 d/wk) furan by gavage for 13 weeks. Numbers of rats were designed based on the number of GST-P+ foci induced by DEN or furan per animal in the experiment I. The control group was given tap water. After treatment, the livers were immediately removed and frozen after embedding in OCT compound (Sakura Finetek Japan).
Immunohistochemical Staining for GST-P, SOX9, and CK19
Formalin-fixed, paraffin-embedded, and frozen liver sections were stained using polyclonal antibodies against GST-P (MBL), SOX9, or cytokeratin (CK) 19 (Abcam). Visualization was performed using 3,3′-diaminobenzidine tetrahydrochloride or TSA Plus Fluorescein and Cyanine 3 System (PerkinElmer). In formalin-fixed, paraffin-embedded liver sections, the numbers (number/cm2) and areas (mm2/cm2) of GST-P+ foci consisting of 5 or more hepatocytes were evaluated using an IPAP image analyzer (Sumika Technos).
Total RNA Collection
Frozen livers embedded in OCT compound were sectioned using cryostats and stained with 0.05% toluidine blue solution. We selected 3 rats from each group and collected GST-P+ foci, including large GST-P+ foci in furan-treated group, from serial frozen sections with a mirror section stained by GST-P immunohistochemical staining as a guide for laser microdissection (Leica Microsystems; Figure 2A). The GST-P-negative area was collected from liver tissues surrounding GST-P+ foci. Total RNA was isolated from microdissected samples using an RNeasy Plus Micro Kit (Qiagen GmbH). The concentration and quality of the total RNA were analyzed using an Agilent 2100 Bioanalyzer (Agilent Technologies). The average RNA integrity number was 8.0 (range 7.5-9.2) and considered adequate for microarray analysis. Extracted total RNA was amplified and synthesized into SPIA cDNA using an Ovation PicoSL WTA System V2 (NuGEN) for cDNA microarray.
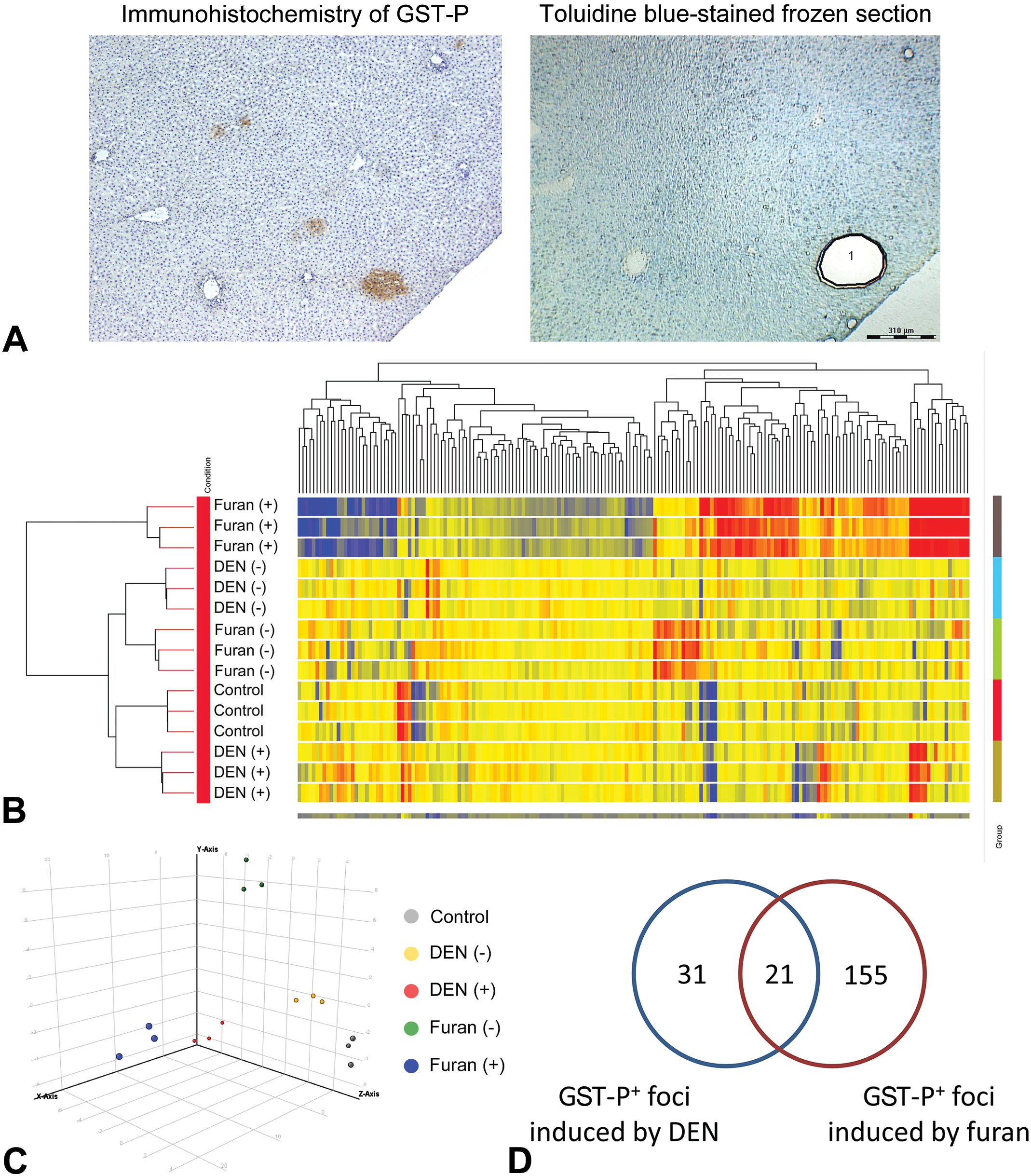
Gene expression profiles of GST-P+ foci induced by DEN or furan. A, Sampling of GST-P+ foci by laser microdissection. Area 1 in toluidine blue-stained frozen section indicated a focus captured using laser microdissection as GST-P+ focus. B, Hierarchical cluster analysis illustrated that gene expression patterns in GST-P+ foci induced by furan were separated from those induced by DEN and from the GST-P-negative area. C, Principal component analysis results of gene expressions that differentiate between control, GST-P+ foci induced by DEN or furan, and GST-P-negative area. D, Venn diagram of significantly altered genes furan (+), furan-induced GST-P+ foci; furan (−), GST-P-negative area of livers treated with furan; DEN (+), DEN-induced GST-P+ foci; DEN (−), GST-P-negative area of the liver treated with DEN. DEN indicates diethylnitrosamine; GST-P+, glutathione S-transferase placental form-positive.
Global Gene Expression Analysis by cDNA Microarray
SPIA cDNA was labeled with cyanine 3 dye using a SureTag Complete DNA labeling kit (Agilent Technologies). Complementary DNA concentration, dye incorporation, and quality were analyzed using an ultraviolet–visible spectrophotometer and Agilent 2100 Bioanalyzer. Fluorescently labeled complementary RNA was hybridized to Agilent 4 × 44 K whole rat genome expression microarrays following the manufacturer’s protocol (Agilent Technologies). Hybridized microarrays were then scanned using an Agilent Microarray Scanner (model G2565BA; Agilent Technologies). Feature Extraction Software (Agilent Technologies) was employed for image analysis and data extraction processes. Normalization of gene expression data and filtering of probe sets by expression levels, flags, and errors were performed using GeneSpring software (Agilent Technologies). Significant differences in gene expression between DEN- or furan-induced GST-P+ foci and corresponding GST-P-negative areas were analyzed by analysis of variance (P < .05; multiple testing correction: Benjamini-Hochberg false discovery rate) and fold change (>2.0 or <−2.0). The identified genes were analyzed by hierarchical clustering and principal component analysis using GeneSpring software.
Statistics
The data for number and area of GST-P+ foci in DEN- or furan-treated groups and GST-P+ foci with Sox9 expression were checked for homogeneity with the F test. Student t tests and Welch t test were applied for homogeneous or heterogeneous data, respectively. Differences with P values of <.05 were considered significant. Incidences of GST-P+ foci were compared with Fisher exact probability test.
Results
Kinetics of the Formation of GST-P+ Foci After Cessation of Carcinogen Treatment
The results of quantitative analysis of GST-P+ foci are summarized in Figure 3. The number and area of GST-P+ foci in DEN- or furan-treated groups were significantly increased compared with those in the control just after carcinogen treatment. At 7 weeks after cessation, the number and area of GST-P+ foci induced by DEN were significantly increased (Figures 3A and 3B). In contrast, those induced by furan were significantly decreased or tended to be decreased (Figures 3A and 3C). Notably, large GST-P+ foci (>0.3 mm2), which were composed of hepatocytes stained more weakly by anti-GST-P antibodies than those induced by DEN, were significantly increased only in the liver just after 13-week furan treatment and were not observed at 7 weeks after cessation (Table 1, Figure 3A). In contrast, small GST-P+ foci (<0.05 mm2) were significantly observed in 13-week DEN treatment group, and total number of small GST-P+ foci (<0.05 mm2) was significantly increased after cessation of DEN treatment (Table 1, Figure 3A)
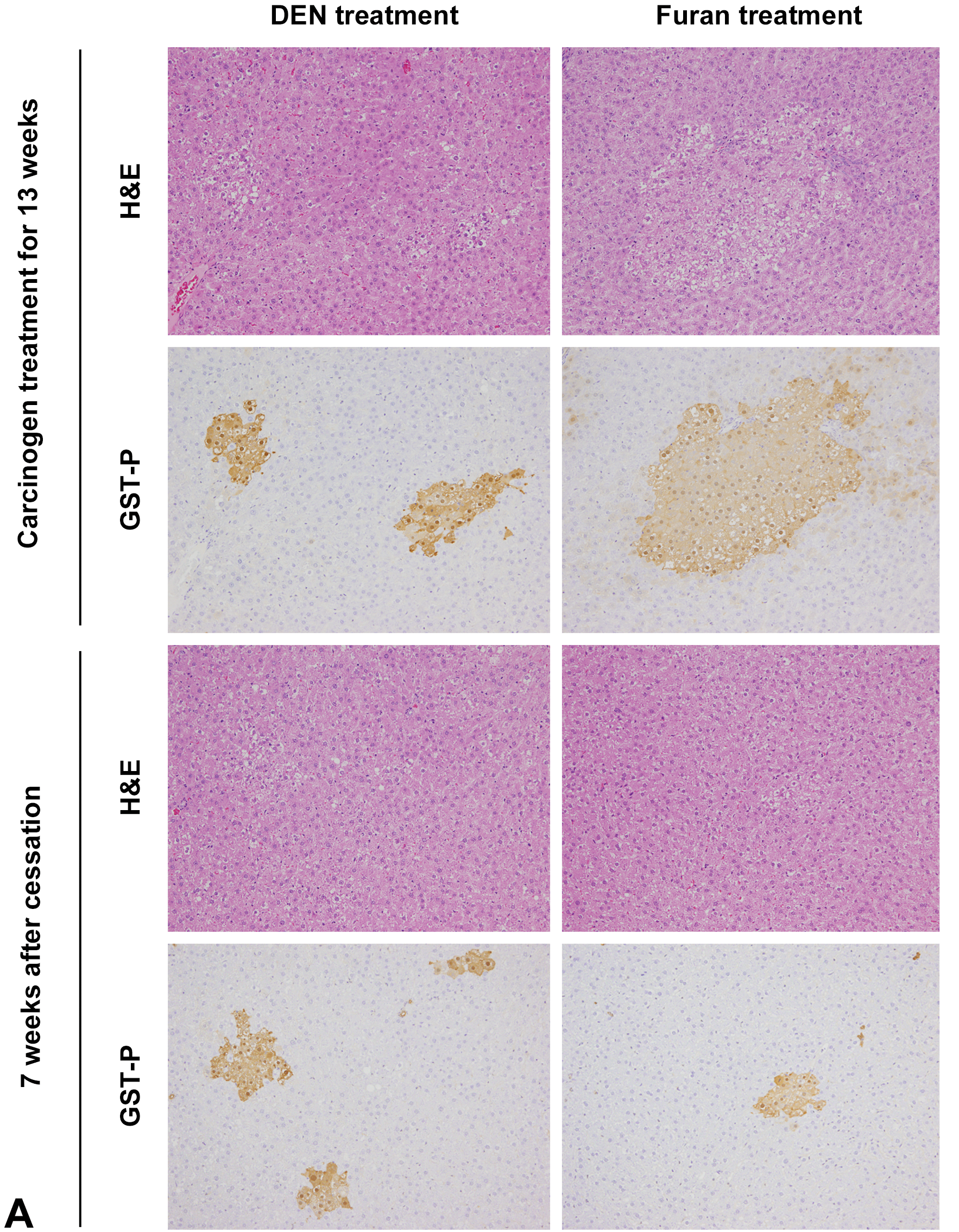
Representative images of hematoxylin and eosin and GST-P immunohistochemistry in experiment I (A). The number and area of GST-P+ foci induced by DEN (B) or furan (C). * Significantly different from the 13-week treatment group at P < .05. DEN indicates diethylnitrosamine; GST-P+, glutathione S-transferase placental form-positive.
Size Distribution of GST-P+ Foci Induced by DEN or Furan.
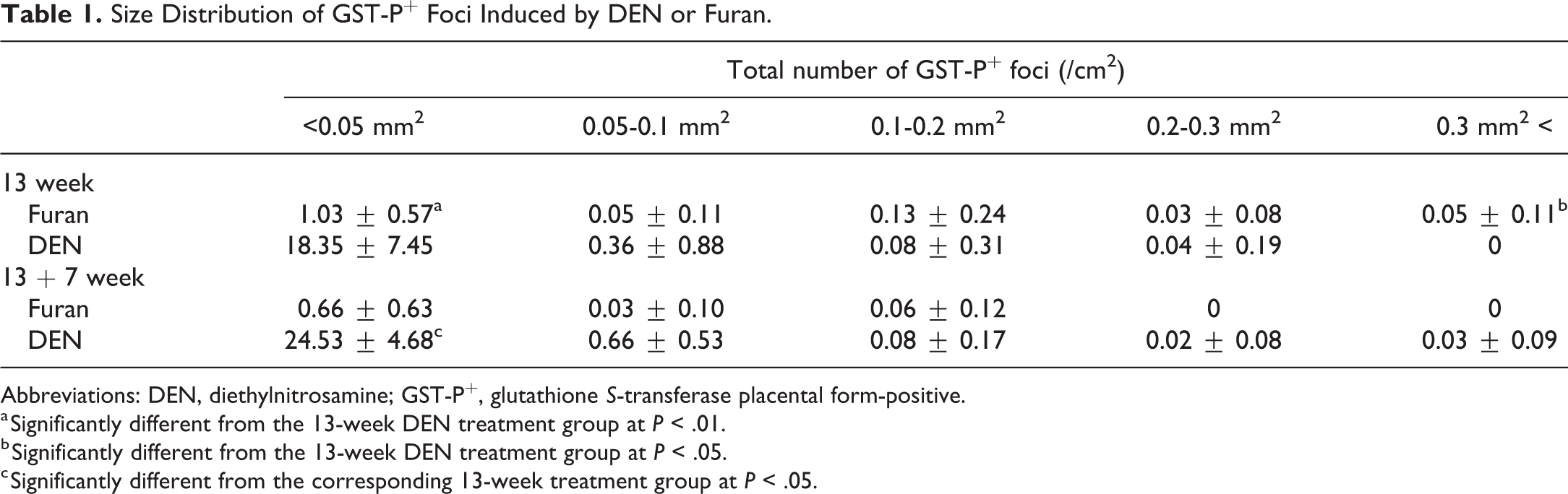
Abbreviations: DEN, diethylnitrosamine; GST-P+, glutathione S-transferase placental form-positive.
a Significantly different from the 13-week DEN treatment group at P < .01.
b Significantly different from the 13-week DEN treatment group at P < .05.
c Significantly different from the corresponding 13-week treatment group at P < .05.
Global Gene Expression Analysis Using cDNA Microarrays
Filtering, which was performed to minimize errors, generated 10,575 probe sets. The expression of 267 genes was significantly altered among all groups. The genes that were significantly altered among all groups were subjected to hierarchical cluster analysis. The results showed that all 3 samples in each group were clustered together. The principal bifurcation in the clustering occurred between the samples of GST-P+ foci induced by furan and other groups (Figure 2B). Principal component analysis was applied on genes significantly altered among groups to identify those genes that could distinguish between control, GST-P+ foci induced by DEN or furan, and GST-P-negative area (Figure 2C). When comparing GST-P+ foci with its negative area in DEN- or furan-treated groups, 21 (14 upregulated, 7 downregulated) genes were commonly altered in both GST-P+ foci types (Figure 2D, Table 2). The expression levels of 31 (11 upregulated, 20 downregulated) or 155 (57 upregulated, 98 downregulated) genes were significantly altered by at least 2.0-fold only in GST-P+ foci induced by DEN or furan, respectively (Figure 2D, Tables 3 and 4).
A List of the 10 Genes Most Highly Upregulated Both in GST-P-Positive Foci Induced by DEN and Furan.
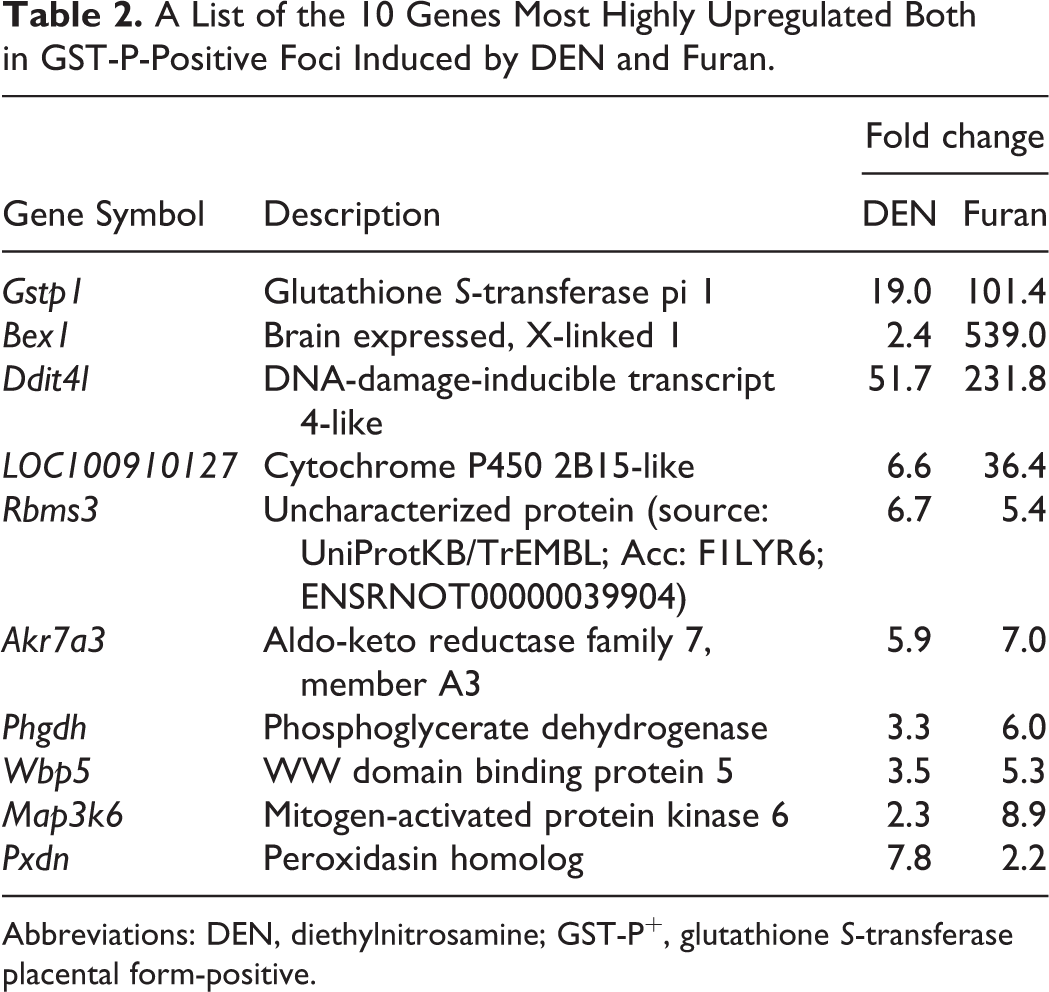
Abbreviations: DEN, diethylnitrosamine; GST-P+, glutathione S-transferase placental form-positive.
A List of Upregulated or Downregulated Genes Only in GST-P-Positive Foci Induced by DEN.
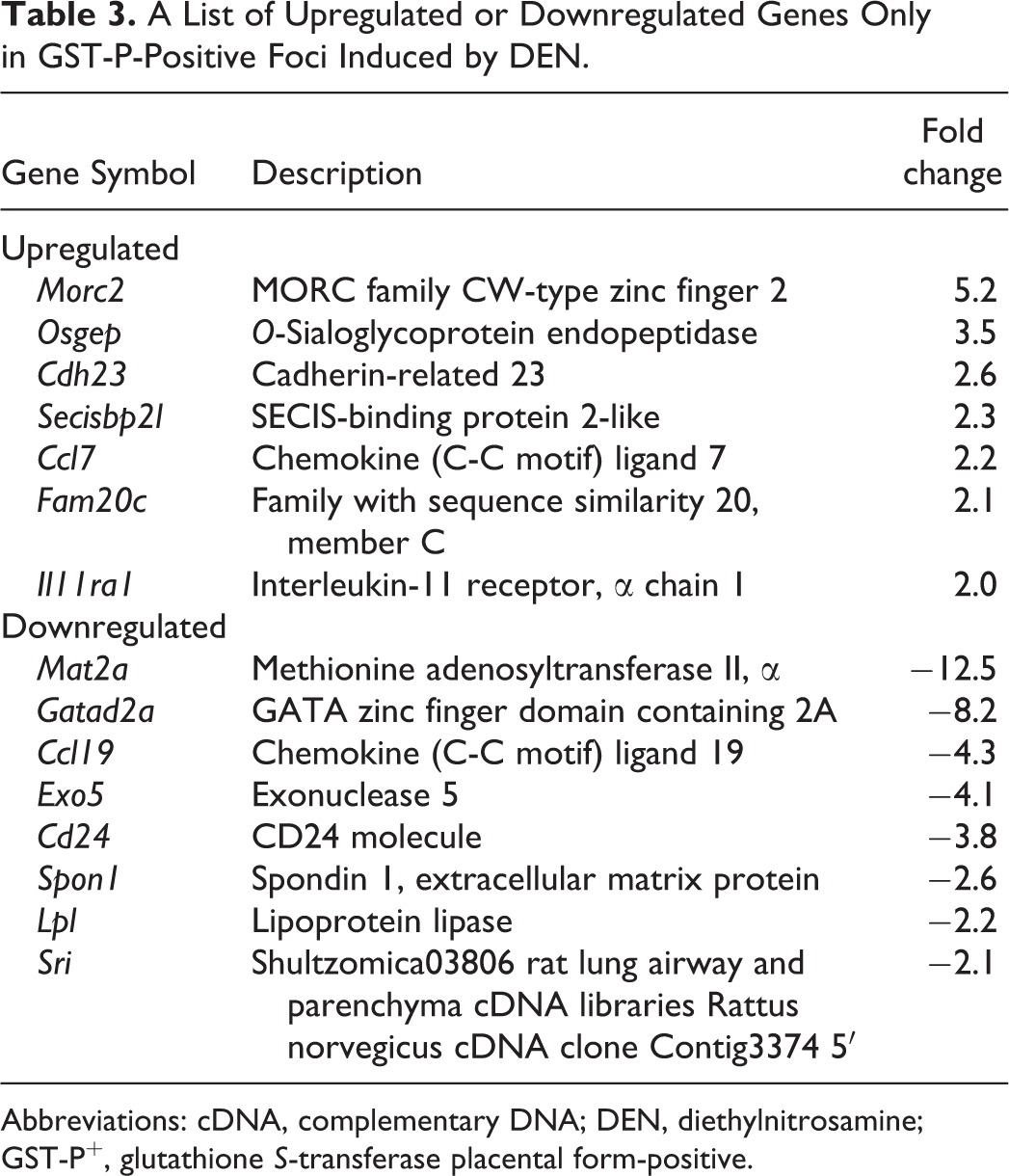
Abbreviations: cDNA, complementary DNA; DEN, diethylnitrosamine; GST-P+, glutathione S-transferase placental form-positive.
A List of 10 Genes Most Highly Upregulated or Downregulated Only in GST-P-Positive Foci Induced by Furan.
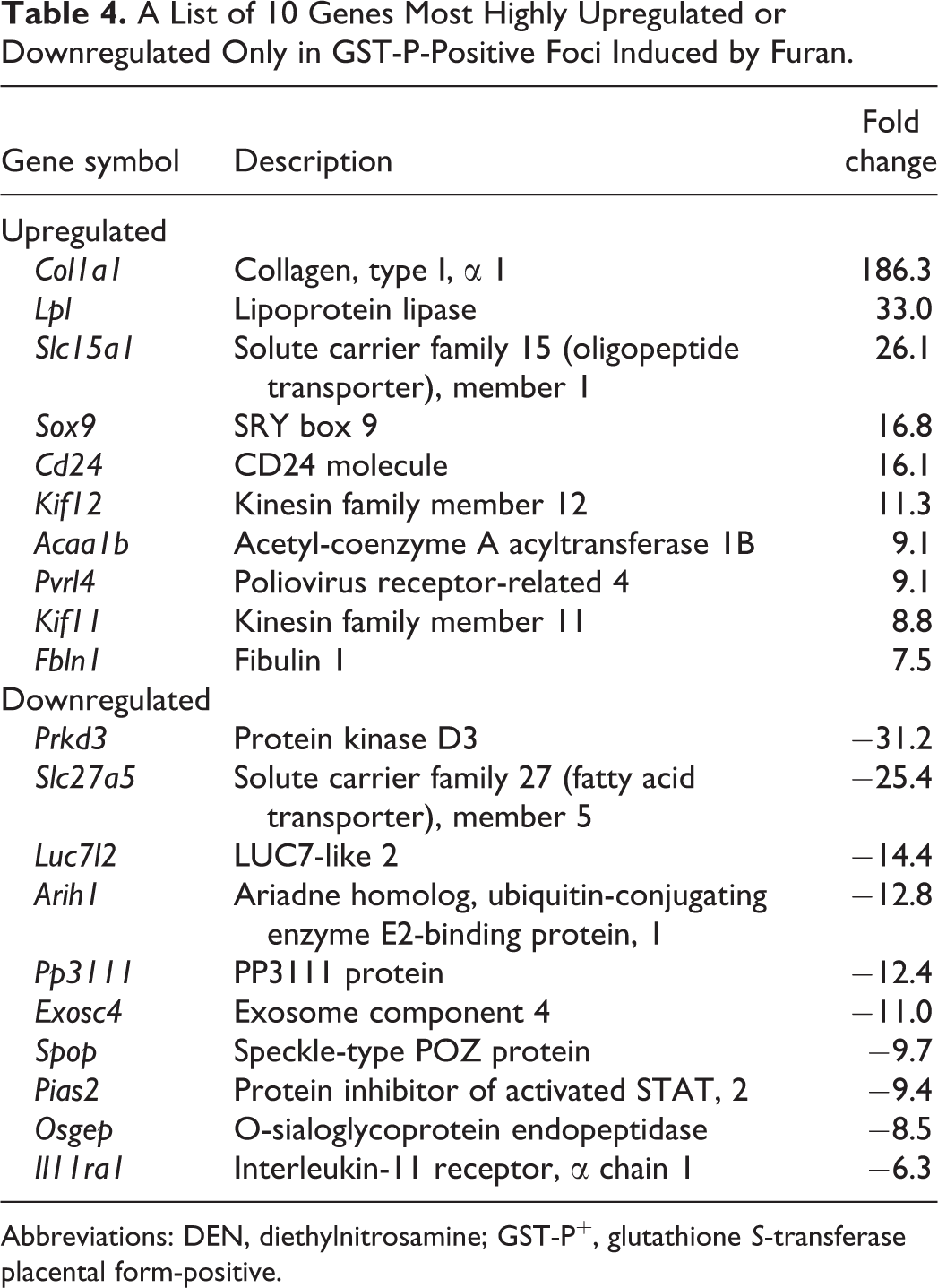
Abbreviations: DEN, diethylnitrosamine; GST-P+, glutathione S-transferase placental form-positive.
Expression of SOX9 in GST-P+ Foci
SOX9 expression levels in GST-P+ foci are summarized in Table 5. Immunofluorescence staining showed that SOX9 expression was detected in bile duct epithelial cells (Figure 4A), which is consistent with the previous report that SOX9 is a biliary marker. 17 Foci composed of hepatocytes expressing both GST-P and SOX9 were observed in furan-treated groups. However, GST-P+ hepatocyte expressing SOX9 was not detected in DEN-treated groups (Figure 4A). SOX9 expression was immunohistochemically detected in furan-induced GST-P+ foci (6/30, 20%), and no SOX9 expression was observed in DEN-induced GST-P+ foci (0/50, 0%). At 7 weeks after cessation, there were no SOX9+ cells in furan- and DEN-induced GST-P+ foci. Importantly, GST-P+ foci with SOX9 expression were larger than those without SOX9 expression (Table 5). Immunofluorescence staining for CK19, one of the marker for oval cell/bile duct cell, 18 in representative GST-P+ foci in the present study, which were presented in Figure 4A, showed that CK19+ cells were scattered in SOX9+ GST-P+ foci, whereas CK19+ cells were not detected in Sox9-negative GST-P+ foci induced by furan or DEN (Figure 4B).
Expression of Sox9 in GST-P-Positive Foci Induced by DEN or Furan.
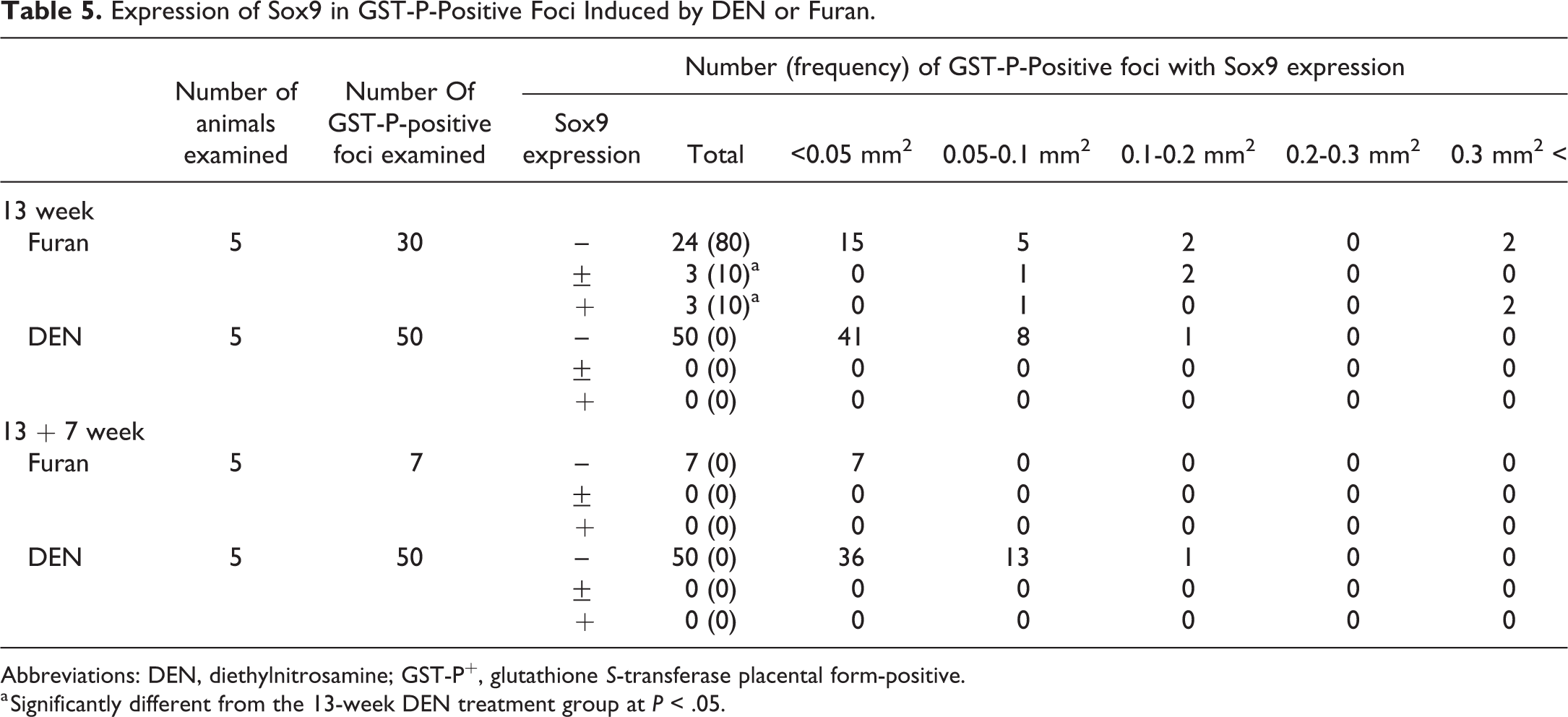
Abbreviations: DEN, diethylnitrosamine; GST-P+, glutathione S-transferase placental form-positive.
a Significantly different from the 13-week DEN treatment group at P < .05.
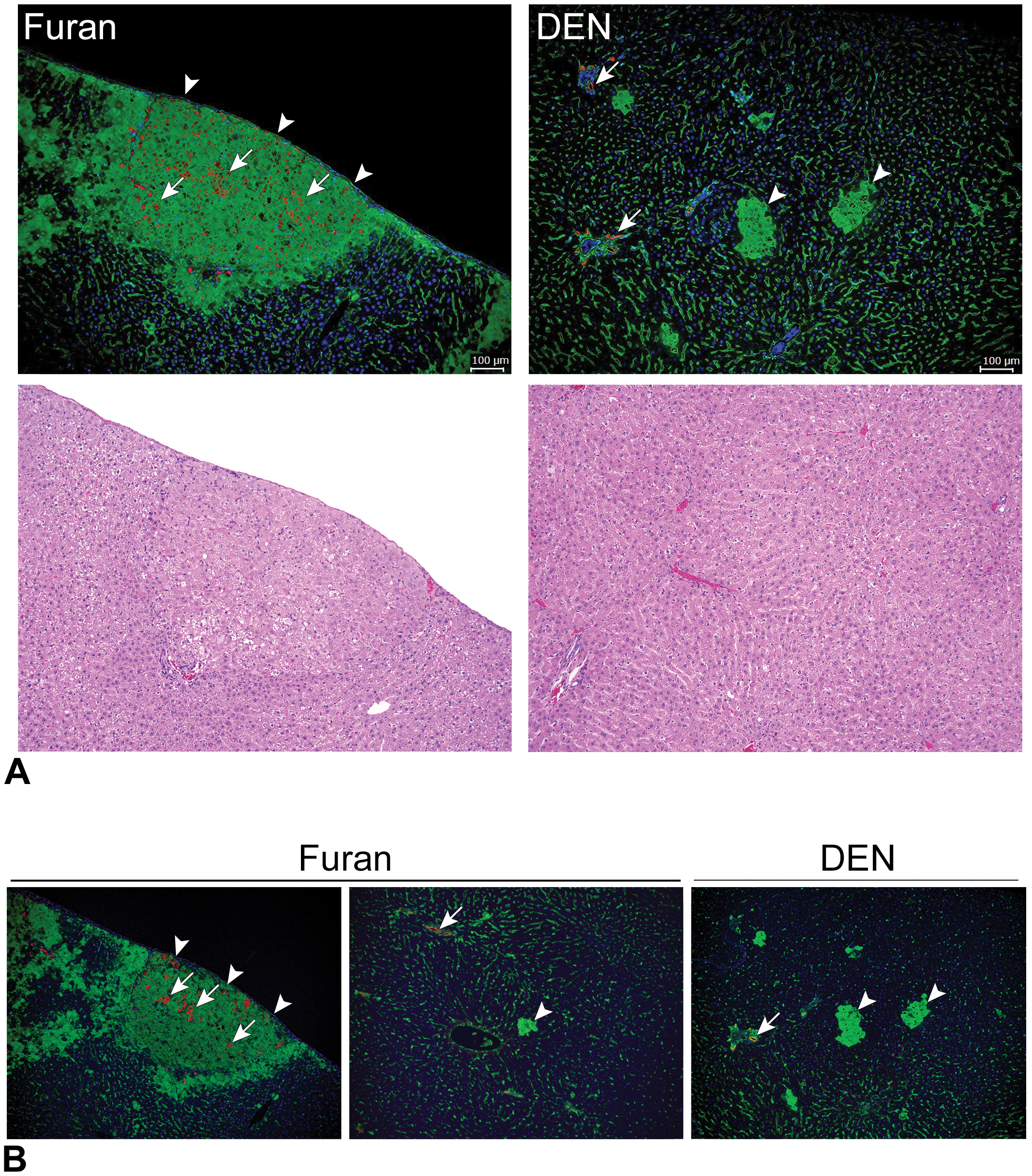
SOX9 expression in GST-P+ foci. A, Representative photographs of hematoxylin and eosin and glutathione S-transferase placental form (green) and SOX9 (red) immunofluorescence staining in DEN- or furan-induced GST-P+ foci. Arrowheads indicates GST-P+ foci. Arrow indicates Sox9-positive cells. B, Representative photographs of glutathione S-transferase placental form (green) and CK19 (red) immunofluorescence staining in DEN- or furan-induced GST-P+ foci. Arrowhead indicates GST-P+ foci. Arrows indicates CK19-positive cells. DEN indicates diethylnitrosamine; GST-P+, glutathione S-transferase placental form-positive.
Discussion
In the current study, our comparison of the cell kinetics of GST-P+ foci revealed that GST-P+ foci induced by furan were decreased after cessation of treatment, whereas those induced by DEN were increased after cessation of treatment. In particular, large GST-P+ foci, which were observed only in furan treatment group, disappeared after the cessation of furan treatment. Although GST-P+ foci are considered good markers of preneoplastic lesions in rat hepatocarcinogenesis, GST-P+ hepatocytes do not always progress to neoplastic cells. Additionally, the expression of GST-P mRNA in some foci declines after release from 2-acetylaminofluorene (2-AAF) treatment. 19 However, in a previous study, the size of disappearing GST-P+ foci induced by DEN and 2-AAF was relatively smaller, 19 inconsistent with our current results. According to detailed observations of GST-P immunochemistry, large GST-P+ foci induced by furan were composed of hepatocytes weakly stained by anti-GST-P antibodies, whereas foci induced by DEN or small foci induced by furan showed opposite results. These differences strongly supported our current hypothesis that large GST-P+ foci induced by furan showed different characteristics regarding cell kinetics and morphology from typical GST-P+ foci.
In addition to cell kinetics, global gene expression property also supported the biological difference of large GST-P+ foci induced by furan from GST-P+ foci induced by DEN. Our global gene expression analysis using mRNA extracted from GST-P+ foci collected by laser microdissection revealed common genes upregulated in furan- and DEN-induced foci, including Ddit4l, Akr7a3, Wbp5, and Gstp1, which were previously reported to be upregulated in foci induced by DEN, 2-AAF, and two-thirds partial hepatectomy. 20 -22 However, hierarchical clustering analysis clearly indicated different groups of fluctuation genes in furan-induced foci from those in DEN-induced foci. Thus, large GST-P+ foci induced by furan were quite different from those induced by DEN at the molecular and cellular levels.
The mRNA expression levels of various genes fluctuated only in furan-induced foci. In the list of upregulated or downregulated genes only in furan-induced foci, genes encoding factors related with collagen component (Col1a1), lipid metabolism (Lpl, Slc27a5), and peptide transporter (Slc15a1) were included. 23 -26 Among them, we selected Sox9 because of several reports showing relationships between Sox9 expression and liver cell differentiation. SOX9 regulates the development of intrahepatic bile ducts through tubulogenesis. 17 SOX9+ ductal epithelial cells serve as endogenous hepatocyte progenitor cells, contributing to hepatocytes during liver homeostasis and after injuries. 27 Recent studies have shown that Sox9 is expressed in a small subpopulation of periportal hepatocytes in the normal liver; SOX9+ hepatocytes give rise to both hepatocytes and ductal cells after liver injury. 28,29 Immunohistochemical analysis of SOX9 showed that hepatocytes expressing SOX9 were located only in furan-induced foci and that these cells disappeared after the cessation of furan treatment. Size distribution analysis of foci, including hepatocytes expressing SOX9, showed that small GST-P+ foci (<0.05 mm2) were negative and large GST-P+ foci (>0.3 mm2) were positive for SOX9. Therefore, it is highly probable that disappearing large GST-P+ foci induced by furan were characterized by inclusion of hepatocytes expressing SOX9.
Disappearing GST-P+ foci were commonly observed after treatment with furan derivatives (unpublished data). To clarify the biological property of GST-P+ cells composing large SOX9+ GST-P+ foci, expression of CK19 in a representative GST-P+ focus was examined. Immunofluorescence analysis showed that CK19+ cells were scattered in a representative SOX9+ GST-P+ focus, whereas they were not detected in a SOX9-negative GST-P+ focus induced by furan or DEN. Previously, it has been reported that CK19+ hepatocytes were located in hepatocyte foci and nodules. 30,31 However, biological significances of CK19+ cells in hepatic lesion are not fully understood. Based on the role of SOX9 in the development of liver cells, as described above, 17,27 -29 and CK19 expression, status of cell differentiation in SOX9+ GST-P+ foci induced by furan might be different from those of DEN-induced GST-P+ foci. However, further examination to elucidate the relationship between SOX9+ GST-P+ foci and CK19+ GST-P+ foci appears to be needed. In addition, the cell characteristics including cell proliferation, cell marker expression, and gene expression of SOX9+ GST-P+ foci should be clarified in further study, which will lead to understanding of mechanisms of furan-induced hepatocarcinogenesis.
Footnotes
Acknowledgments
The authors thank Ms Ayako Saikawa and Ms Yoshimi Komatsu for their expert technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid from the Ministry of Health, Labour, and Welfare, Japan (grant no. H27-kagaku-wakate-010) and the grants from the Japan Food Chemical Research Foundation (H29-008 and H30-006).
