Abstract
This study aimed to investigate the anti-cancer effect of lobetyolin on breast cancer cells. Lobetyolin was incubated with MDA-MB-231 and MDA-MB-468 breast cancer cells for 24 h. Glucose uptake and the mRNA expression of GLUT4 (SLC2A4), HK2 and PKM2 were detected to assess the effect of lobetyolin on glucose metabolism. Glutamine uptake and the mRNA expression of ASCT2 (SLC1A5), GLS1, GDH and GLUL were measured to assess the effect of lobetyolin on glutamine metabolism. Annexin V/PI double staining and Hoechst 33342 staining were used to investigate the effect of lobetyolin on cell apoptosis. Immunoblot was employed to estimate the effect of lobetyolin on the expression of proliferation-related markers and apoptosis-related markers. SLC1A5 knockdown with specific siRNA was performed to study the role of ASCT2 played in the anti-cancer effect of lobetyolin on MDA-MB-231 and MDA-MB-468 breast cancer cells. C-MYC knockdown with specific siRNA was performed to study the role of c-Myc played in lobetyolin-induced ASCT2 down-regulation. Myr-AKT overexpression was performed to investigate the role of AKT/GSK3β signaling played in lobetyolin-induced down-regulation of c-Myc and ASCT2. The results showed that lobetyolin inhibited the proliferation of both MDA-MB-231 and MDA-MB-468 breast cancer cells. Lobetyolin disrupted glutamine uptake via down-regulating ASCT2. SLC1A5 knockdown attenuated the anti-cancer effect of lobetyolin. C-MYC knockdown attenuated lobetyolin-caused down-regulation of ASCT2 and Myr-AKT overexpression reversed lobetyolin-caused down-regulation of both c-Myc and ASCT2. In conclusion, the present work suggested that lobetyolin exerted anti-cancer effect via ASCT2 down-regulation-induced apoptosis in breast cancer cells.
Introduction
Breast cancer has become one of the most common malignant tumors in women owing to its increasing incidence every year. 1 Despite several therapeutic strategies including surgery, chemotherapy and radiotherapy are used in clinic, the prognosis of breast cancer remains unsatisfactory. Thus, novel and more efficient anti-tumor drugs are still urgently needed.
Lobetyolin is a bioactive ingredient extracted from Codonopsis pilosula (Franch.) Nannf., a famous traditional Chinese medicine which has been used to enhance the immune system, suppress blood pressure, attenuate gastrointestinal function, improve appetite and treat gastric ulcer. 2 Increasing evidence suggested that lobetyolin possessed anti-inflammatory, anti-oxidative3,4 and xanthine oxidase inhibiting properties. 5 Importantly, the lobetyolin containing Chinese medicine Bu-Fei decoction showed anti-epithelial-mesenchymal transition (EMT) effect on non-small-cell lung cancer cells 6 and the steamed Codonopsis lanceolate, which consisted of lobetyolin, performed beneficial effect on H22 tumor-bearing mice. 7 Recently, lobetyolin was reported to inhibit the proliferation of colon cancer cells via disrupting glutamine metabolism. 8 However, whether and how lobetyolin possesses an anti-cancer effect on breast cancer remain unclear.
Increased need for nutrients including glucose and glutamine is a hallmark of rapidly growing cancer cells. Glucose is the most important substance of energy source which provides ATP for bioactivity and carbon for biosynthesis. 9 Recent years, a larger number of studies indicated that cancer cells reprogramed their metabolic pathways to increase glutamine uptake.10,11 Glutamine can provide nitrogen for the synthesis of purines and pyrimidines, as well as non-essential amino acids (NEAAs). At the same time, glutamine is also an important carbon source. 11 In addition to being nitrogen source and carbon source for synthesizing biomolecules and participating in the production of ATP by tricarboxylic acid cycle (TCA), glutamine is also closely related to the oxidation-reduction homeostasis of cells. 12 After glutamine being metabolized to α-KG and entering TCA cycle, part of malate can be catalyzed to generate pyruvate and NADPH under the catalysis of NADP+ dependent malic enzyme (ME-1). 13 NADPH is an important antioxidant, which is able to reduce oxidized glutathione (GSSG) to reduced glutathione (GSH). In addition, glutamate, which can be converted from glutamine, is directly involved in the synthesis of glutathione (GSH), the most important antioxidant inside of cells. 14
In present study, we aimed to study the anti-cancer effect of lobetyolin on breast cancer cells and its underlying mechanism. Our data revealed that lobetyolin suppressed the growth of MDA-MB-231 and MDA-MB-468 breast cancer cells. Further studies indicated that lobetyolin inhibited ASCT2-mediated glutamine uptake without influencing glucose metabolism and induced oxidative stress-mediated apoptosis. SLC1A5 knockdown attenuated the anti-cancer effect and apoptosis-inducing effect of lobetyolin. In addition, lobetyolin suppressed ASCT2 expression via AKT/GSK3β/c-Myc axis. Taken together, our results demonstrate that lobetyolin inhibits the proliferation of breast cancer cells via ASCT2 down-regulation-induced apoptosis.
Materials and methods
Materials
Lobetyolin (purity > 98%) was purchased from Yuanye Bio-Technology (Shanghai, China). L-15 medium, dimethyl sulfoxide (DMSO), fetal bovine serum (FBS) and trypsin-EDTA were purchased from Gibco (Grand Island, NY). Culture plates, antibiotics, and chemicals used for cell culture were purchased from KeyGEN BioTECH (Jiangsu, China). Annexin V/PI double staining kit was purchased from Vazyme Biotech (Jiangsu, China). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and Hoechst 33342 were purchased from Beyotime Biotechnology (Jiangsu, China).
Cell culture
MDA-MB-231 and MDA-MB-468 human breast cancer cell lines obtained from American Type Culture Collection (ATCC, Rockville, MD, USA) were cultured in L-15 supplemented with 10% FBS, 100 U/ml penicillin, and 100 U/ml streptomycin under 95% air/5% CO2 at 37°C. The culture medium was changed every 2 days, and cells were subcultured twice a week.
MTT assay
MTT assay was performed to examine cell viability. Briefly, MDA-MB-231 and MDA-MB-468 breast cancer cells were incubated with 0.25 mg MTT/ml for 4 h at 37°C. The amount of MTT formazan product was determined by measuring absorbance at 570 nm (630 nm as a reference) using a microplate reader (Bio-Rad, Hercules, CA, USA). Cell viability was expressed as a percentage of control groups.
Proliferation curve test
Proliferation curve test was performed to examine cell proliferation. Briefly, around 5 × 104 cells were seed into 12 well plates, the cell number was counted as Day 0. Then lobetyolin (10 μM) group, lobetyolin (20 μM) group, lobetyolin (40 μM) group were treated with lobetyolin (10 μM), lobetyolin (20 μM), lobetyolin (40 μM) respectively. The cells of every group were digested and the cell number of every group was counted using Countstar Automated Cell Counter (Shanghai, China).
Immunoblot
Total protein was obtained using radio-immunoprecipitation assay (RIPA) buffer (Beyotime, Shanghai, China) supplemented with the protease inhibitor cocktail (Sigma-Aldrich). In certain experiments, cytoplasmic and mitochondrial proteins were extracted using Cell Mitochondria Isolation Kit according to the manufacturer’s instructions (Sigma, USA). Protein concentrations were measured using BCA protein assay kit (Beyotime). The protein samples were separated by SDS-PAGE and blotted onto polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA, USA). Next, the membranes were incubated at 4°C overnight with primary antibodies against p-4EBP1(Ser65), 4EBP1, p-p70S6k (Ser371), p70S6k, ASCT2, Cleaved Caspase 3, Cleaved Caspase 9, Cleaved PARP, Cytochrome C, p-GSK3β(Ser9), GSK3β, c-Myc, p53, RNF5, p-c-Myc (Thr58), p-AKT (Ser473) (Cell Signaling Technology, Beverly, MA, USA), Bcl-2, Bax, GAPDH, COX IV, Caspase 3, Caspase 9, PARP, AKT (Proteintech, Wuhan, China), followed by horseradish peroxidase-conjugated secondary antibodies. The immunoblot signals were visualized by enhanced chemiluminescence detection kit (Amersham Pharmacia Biotec, Buckinghamshire, UK). The gray-scale values were quantified by Image-J software (NIH, Bethesda, MD, USA).
Glucose uptake
Glucose uptake was measured using the Glucose Uptake Colorimetric Assay Kit (K676, Biovision, USA) according to manufacturer’s instruction.
Glutamine uptake
Glutamine uptake was measured using the Glutamine Assay Kit (ab197011, Abcam, UK) according to manufacturer’s instruction.
RNA extraction and real-time reverse transcriptase polymerase chain reaction (RT-qPCR)
Total RNA was isolated from MDA-MB-231 and MDA-MB-468 breast cancer cells using RNA isolater Total RNA Extraction Reagent (Vazyme Biotech Co., Ltd) and reversely transcribed into cDNA using HiScript II Q Select RT SuperMix for qPCR (Vazyme Biotech Co., Ltd) according to the supplier’s protocol. Real-time PCR was performed with gene specific primers (Table 1, Thermo Fisher Scientific Inc.) with 18s as internal control. The reactions were performed in an ABI Prism® 7900 High-Throughput RealTime PCR System (Applied Biosystems, Foster City, CA, USA). The comparative threshold cycle (CT) method was used for relative quantification of target gene expression, which was plotted as fold of control.
Primer sequences in quantitative RT-PCR.

Measurement of intracellular ATP content
Intracellular ATP content was measured using ATP Assay Kit (S0026, Beyotime Biotechnology, China) according to manufacturer’s instruction.
Detection of GSH and ROS
Cells were washed twice with PBS (pH 7.2), then were scraped from the plates and followed by homogenizing in PBS containing 0.5 mM butylated hydroxytoluene to prevent further oxidation of sample. The homogenate was centrifuged at 3000 g for 20 min at 4°C, and the supernatant was used for these assays according to the manufacturer’ s instructions. GSH or GSSG concentration (ng/mg protein) was quantified by a commercial colorimetric assay kit (OxisResearch, Portland, OR, USA). ROS level was determined using a ROS Assay Kit (Beyotime Biotechnology, China). Cells were incubated with 10 μM DCFH-DA dye for 20 min in 37°C and washed with PBS. Fluorescence value was measured by using a fluorescence microplate reader (Molecular Devices, USA) at excitation and emission wavelengths of 488 and 525 nm, respectively. Relative fluorescence unit (RFU) was the difference in fluorescence values obtained at time 0 and 5 min.
Annexin V/PI double staining
After treatment with 10 μM, 20 μM, 40 μM lobetyolin for 24 h, MDA-MB-231 and MDA-MB-468 breast cancer cells were digested with 0.25% trypsin without EDTA and were collected via centrifugation at a rotate speed of 500 rpm/min. Apoptotic cells were identified by the Annexin V-FITC Apoptosis Detection kit (Vazyme, Jiangsu, China) in accordance with the manufacturer’s instructions. Cells were washed with fresh PBS buffer for three times. Take out around 5 × 105 cells into another tube, centrifugate it at a rotate speed of 500 rpm/min for 5 min. Then remove the supernatant carefully. Add 100 µl 1 × Binding Buffer and gently blow into the single-cell suspension. Then add 5 µl Annexin V-FITC solution and 5 µl PI Staining Solution for staining for 10 min in dark. Flow cytometric analysis was performed immediately after supravital staining. Data acquisition and analysis were performed in a Becton Dickinson FACS-Calibur flow cytometer using the Cell Quest software (Franklin Lakes). For blocking reactive oxygen species (ROS), cells were treated with 5 mM N-acetylcysteine (Beyotime, Jiangsu, China).
Hoechst 33342 staining
Briefly, cultured MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with 20 μM lobetyolin for 24 h, then cells were washed with fresh PBS for 3 times and stained with Hoechst 33342 dye with indicated dilution ratio for 30 min at room temperature. Cells were washed with fresh PBS for three times and morphology of nucleus was captured by Inverted Fluorescent Microscope (Nikon, Japan).
Preparation of cytoplasmic and mitochondrial protein
For immunoblot analysis, the extraction and isolation of nuclear protein was performed using the Cell Mitochondria Isolation Kit (Beyotime, China). After treatment with lobetyolin with indicated concentration, cells were washed with PBS and digested with trypsin-EDTA Solution. Then the cells were centrifuged at room temperature for 5–10 min. Pre-cooled PBS was used to gently resuscitate the cell precipitate. A small number of cells were taken for counting. The remaining cells were centrifuged at 600 g at 4°C for 5 min to precipitate the cells and then removed the supernatant. Added 1–2.5 ml mitochondrial separation reagent or mitochondrial separation reagent with PMSF added before use to 20–50 million cells, gently suspended the cells, and placed in the ice bath for 10–15 min. Transfered the cell suspension to a suitably sized glass homogenizer and homogenized about 10–30 times. The homogenate was centrifuged at 600 g at 4°C for 10 min. Carefully transfered the supernatant to another tube and centrifuged at 11,000 g at 4°C for 10 min. Removed the supernatant carefully to another tube, the supernatant was cytoplasm and the precipitation was the isolated cell mitochondria.
siRNAs, plasmid and transfection
siRNAs for SLC1A5 and C-MYC were purchased from Sangon Biotech (Shanghai, China). The sequences for effective siRNAs were as follows: siSLC1A5#1, GGATGAAATTGCACCTATT; siSLC1A5#2, GTCCAGGAGACTGTGTCAT; siC-MYC#2, GAGGAAATATGTACTACGA. Myr-AKT plasmid was purchased from GenePharma (Shanghai, China). Transfection of siRNAs and plasmid was performed using Lipofectamine 3000 (Thermo Fisher Scientific, USA). In brief, MDA-MB-231 or MDA-MB-468 breast cancer cells were seeded into six well plates till adherence in advance. For siRNAs transfection for a single well, mixed 100 µl Opti-MEM + 5 µl siRNA which labeled as A, then mixed 100 µl Opti-MEM + 5 µl Lipofectamine 3000 which labeled as B. Incubated A and B in room temperature for 10 min, then mixed A and B and incubated the mixture in room temperature for another 10 min. The mixture was added into six well plates for 48 h. For plasmid transfection for a single well, mixed 100 µl Opti-MEM + 2 µg plasmid + 5 µl P3000 which labeled as A, then mixed 100 µl Opti-MEM + 5 µl Lipofectamine 3000 which labeled as B. Incubated A and B in room temperature for 10 min, then mixed A and B and incubated the mixture in room temperature for another 10 min. The mixture was added into six well plates for 48 h.
Statistical analysis
Data were analyzed using the GraphPad Prism 8 software. Statistical evaluation was done by one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison test. P-value less than 0.05 was considered to be statistically significant.
Results
Lobetyolin exerts anti-cancer effect on breast cancer cells
To investigate whether lobetyolin (molecular structure is shown in Figure 1(A)) possessed anti-cancer effect on breast cancer cells, two breast cancer cell lines MDA-MB-231 and MDA-MB-468 were employed and the IC50 value of lobetyolin on each breast cancer cell line was counted, which was 24.55 μM for MDA-MB-231 cells and 19.25 μM for MDA-MB-468 cells (Figure 1(B)). When treated both cell lines with 10 μM, 20 μM, 40 μM lobetyolin for 48 h, the proliferation of both cell lines was inhibited in a dosage-dependent manner (Figure 1(C)). The proliferation curve test showed similar results that lobetyolin inhibited the proliferation of MDA-MB-231 and MDA-MB-468 breast cancer cells in a dosage-dependent manner (Figure 1(D)). In addition, the phosphorylation of 4EBP1 and p70S6k, the two markers of cell proliferation, was suppressed by lobetyolin in a dosage-dependent manner, indicating that lobetyolin inhibited the proliferation of both MDA-MB-231 and MDA-MB-468 breast cancer cells. Taken together, these results suggest that lobetyolin possesses the anti-cancer effect on MDA-MB-231 and MDA-MB-468 breast cancer cells.

Lobetyolin inhibited the proliferation of MDA-MB-231 and MDA-MB-468 breast cancer cells. (A) Molecular structural of lobetyolin. (B) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin with indicated concentration for 24 h, cell viability was measured using MTT assay and IC50 of lobetyolin in both cell lines was calculated by GraphPad Prism 8. (C) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, cell survival rate was measured by MTT. (D) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with 20 μM lobetyolin for 3 days. Cell number was counted every 24 h. (E) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, the expression of indicated protein was analyzed by immunoblot, with GAPDH as loading control. The data presented are representative of three independent experiments. Data are presented as mean ± SD. *P < 0.05 vs control group; **P < 0.01 vs control group; ***P < 0.001 vs control group; ****P < 0.0001 vs control group.
Lobetyolin disrupts ASCT2-mediated glutamine metabolism
Glucose and glutamine are two dominant substance of energy source for rapidly proliferating cancer cells. 15 To investigate whether the anti-cancer effect of lobetyolin on breast cancer cells was correlated with the dysfunction of glucose metabolism or glutamine metabolism, the effect of lobetyolin on glucose uptake and glutamine uptake was detected. As shown in Figure 2(A), lobetyolin did not influence glucose uptake in both MDA-MB-231 and MDA-MB-468 breast cancer cells. Consistently, the gene expression of SLC2A4, HK2 and PKM2, which were critical for glucose uptake and glycolysis, was also did not influenced by lobetyolin treatment (Figure 2(B)). However, lobetyolin inhibited glutamine uptake in a dosage-dependent manner in both MDA-MB-231 and MDA-MB-468 breast cancer cells (Figure 2(C)). Next, the expression of key enzymes for glutamine metabolism including SLC1A5, GLS1, GDH and GLUL was analyzed. The result showed that the mRNA expression of SLC1A5 which encodes ASCT2, the key glutamine transporter, was down-regulated by lobetyolin in a dosage-dependent manner in both MDA-MB-231 and MDA-MB-468 breast cancer cells (Figure 2(D)). Consistently, the protein expression of ASCT2 was also suppressed by lobetyolin in these two breast cancer cell lines (Figure 2(F)). In addition, as the direct source for biological energy source, the production of ATP was analyzed upon lobetyolin treatment. Lobetyolin inhibited the production of intracellular ATP in a dosage-dependent manner in both MDA-MB-231 and MDA-MB-468 breast cancer cells (Figure 2(E)). Collectively, these results suggest that lobetyolin could inhibit glutamine uptake in breast cancer cells.

Lobetyolin inhibited glutamine uptake and the expression of ASCT2 in MDA-MB-231 and MDA-MB-468 breast cancer cells. (A) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, glucose uptake was measured using Glucose Uptake Colorimetric Assay Kit. (B) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, RNA samples were harvested and the mRNA expression of SLC2A4, HK2, PKM2 was detected by RT-qPCR. (C) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, glutamine uptake was measured using Glutamine Assay Kit. (D) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, RNA samples were harvested and the mRNA expression of SLC1A5, GLS1, GDH and GLUL was detected by RT-qPCR. (E) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, intracellular ATP content was measured using ATP Assay Kit. (F) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, protein samples were harvested and the expression of ASCT2 was analyzed by immunoblot, with GAPDH as loading control. The data presented are representative of three independent experiments. Data are presented as mean ± SD. *P < 0.05 vs control group; **P < 0.01 vs control group; ***P < 0.001 vs control group.
Lobetyolin-caused oxidative stress and induced apoptosis in MDA-MB-231 and MDA-MB-468 breast cancer cells
Given glutamate is derived from glutamine and serves as a substrate for the synthesis of GSH, a key antioxidant inside of cells, 16 intracellular GSH level and ROS level then were detected after lobetyolin treatment. As shown in Figure 3(A) and (B), lobetyolin significantly inhibited intracellular GSH level and increased intracellular ROS level in a dosage-dependent manner in both MDA-MB-231 and MDA-MB-468 breast cancer cells. ROS is a common inducer of mitochondrial pathway-mediated apoptosis. 17 Thus, whether lobetyolin-induced apoptosis in MDA-MB-231 and MDA-MB-468 breast cancer cells was checked. As shown in Figure 3(C), the result of Annexin V/PI double staining showed that lobetyolin-induced apoptosis in a dosage-dependent manner. Hoechst 33342 staining showed that the nucleus of MDA-MB-231 and MDA-MB-468 breast cancer cells post lobetyolin treatment turned brighter, which consolidated that lobetyolin-induced apoptosis in MDA-MB-231 and MDA-MB-468 breast cancer cells. Besides, lobetyolin increased the cleavage of Caspase 3, Caspase 9 and PARP (Figure 3(E)) and promoted the release of Cytochrome C from mitochondria to cytoplasm (Figure 3(F)). In summary, these results demonstrate that lobetyolin could cause oxidative stress and induce apoptosis in breast cancer cells.

Lobetyolin-induced oxidative stress and apoptosis in MDA-MB-231 and MDA-MB-468 breast cancer cells. (A) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, intracellular GSH level was measured using a GSH Detection Kit. (B) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, intracellular ROS level was measured using a ROS Assay Kit. (C) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, then stained with Annexin/PI. Then cells were collected and analyzed by flow cytometry. (D) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with 20 μM lobetyolin for 24 h, then cells were stained with Hoechst 33342. The morphology of nucleus was captured using microscope. (E) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, protein samples were harvested and the expression of indicated protein was analyzed by immunoblot, with GAPDH as loading control. (F) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, cytoplasmic and mitochondrial protein were extracted using Cell Mitochondria Isolation Kit. The expression of Cytochrome C in cytosol and mitochondria was detected by immunoblot, with GAPDH as loading control. The data presented are representative of three independent experiments. Data are presented as mean ± SD. *P < 0.05 vs control group; **P < 0.01 vs control group; ***P < 0.001 vs control group; ****P < 0.0001 vs control group.
Lobetyolin exerted anti-cancer effect in MDA-MB-231 and MDA-MB-468 breast cancer cells via down-regulating ASCT2 expression
In order to investigate the role of ASCT2 played in the anti-cancer effect of lobetyolin, knockdown of SLC1A5 was performed using siRNAs. Specific siRNAs targeting SLC1A5 were screened and the result was shown in Figure 4(A) that siSLC1A5#1 and siSLC1A5#2 were efficiently suppressed ASCT2 expression in both MDA-MB-231 and MDA-MB-468 breast cancer cells. Thus, siSLC1A5#1 and siSLC1A5#2 were picked to perform following experiments. As shown in Figure 4(B), SLC1A5 knockdown significantly attenuated the inhibitory effect of lobetyolin on cell proliferation in both MDA-MB-231 and MDA-MB-468 breast cancer cells. The inhibition of lobetyolin on the phosphorylation of 4EBP1 and p70S6 k was also weakened by SLC1A5 knockdown (Figure 4(C)). Moreover, lobetyolin-induced apoptosis was also attenuated by ASCT2 knockdown (Figure 4(D)). In total, these results suggest that lobetyolin exerts anti-cancer effect in MDA-MB-231 and MDA-MB-468 breast cancer cells via down-regulating ASCT2 expression.
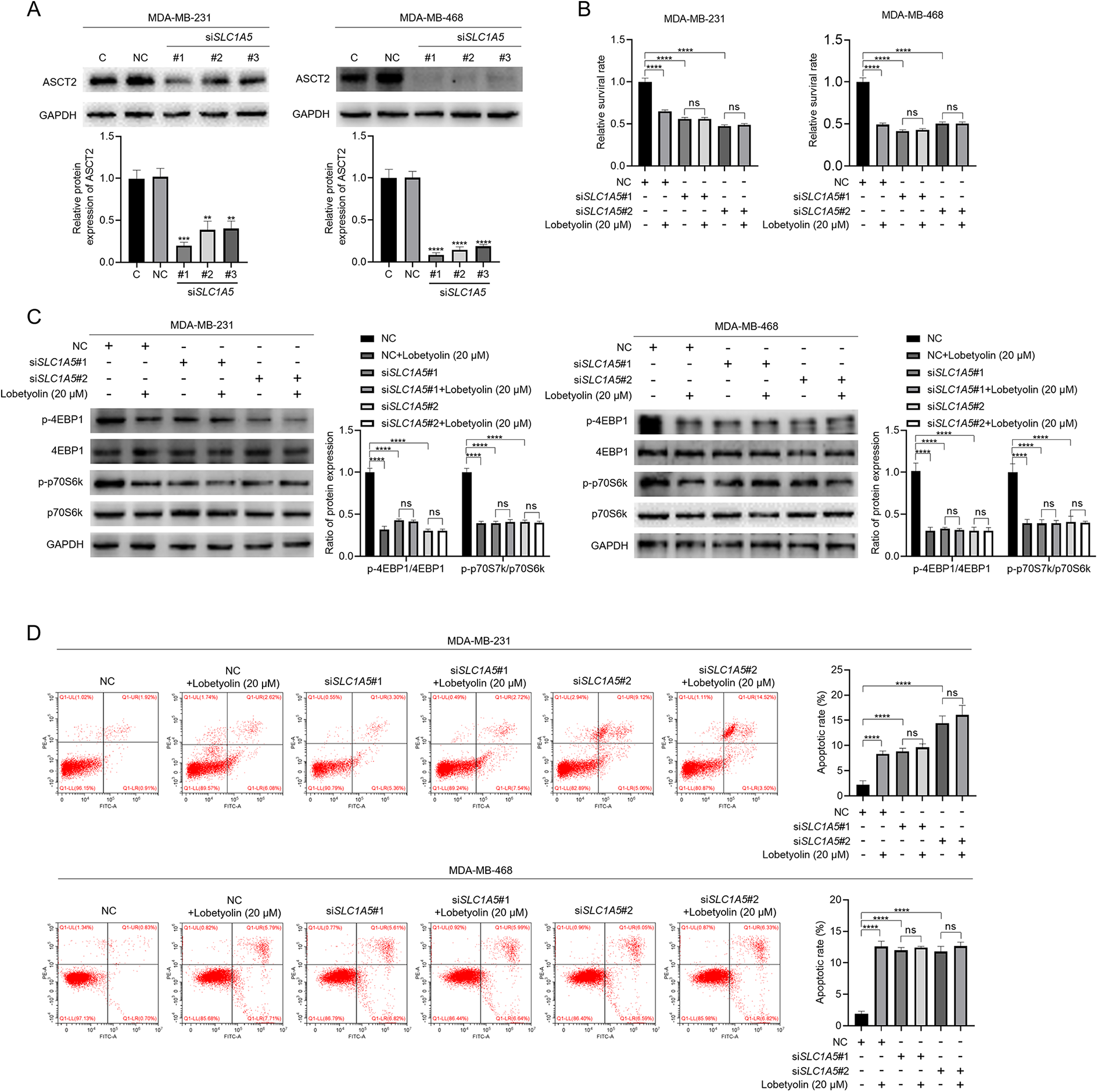
Knockdown of ASCT2 attenuated the anti-cancer effect of lobetyolin in MDA-MB-231 and MDA-MB-468 breast cancer cells. (A) MDA-MB-231 and MDA-MB-468 breast cancer cells were transfected with 100 nmol siRNA for 48 h, then protein samples were harvested and knockdown efficiency of ASCT2 was checked by immunoblot, with GAPDH as loading control. (B) MDA-MB-231 and MDA-MB-468 breast cancer cells were transfected with 100 nmol siSLC1A5#1 or siSLC1A5#2 for 48 h, then cells were treated with 20 μM lobetyolin for another 24 h, cell survival rates were measured by MTT. (C) MDA-MB-231 and MDA-MB-468 breast cancer cells were transfected with 100 nmol siSLC1A5#1 or siSLC1A5#2 for 48 h, then cells were treated with 20 μM lobetyolin for another 24 h, protein samples were collected and the expression of indicated proteins were analyzed by immunoblot, with GAPDH as loading control. (D) MDA-MB-231 and MDA-MB-468 breast cancer cells were transfected with 100 nmol siSLC1A5#1 or siSLC1A5#2 for 48 h, then cells were treated with 20 μM lobetyolin for another 24 h, cells were harvested and stained with Annexin V/PI. Then cell apoptosis was analyzed by flow cytometry. The data presented are representative of three independent experiments. Data are presented as mean ± SD. **P < 0.01 vs control group; ***P < 0.001 vs control group; ****P < 0.0001 vs control group.
Lobetyolin inhibits ASCT2 expression via suppressing c-Myc
As lobetyolin inhibited the expression of SLC1A5 at both mRNA and protein levels, we hypothesized that lobetyolin disrupted the transcription of SLC1A5. c-Myc, P53 and RNF5 are well established transcription factors of ASCT2. 18 Thus, the effect of lobetyolin on the expression of c-Myc, P53 and RNF5 was investigated. As shown in Figure 5(A), lobetyolin inhibited c-Myc expression in a dosage-dependent manner in both MDA-MB-231 and MDA-MB-468 breast cancer cells while had no significant effect on the expression of P53 and RNF5. To study whether it was c-Myc mediated lobetyolin-induced down-regulation of ASCT2, C-MYC knockdown was performed using siRNA in MDA-MB-231 and MDA-MB-468 breast cancer cells. The siRNA specifically targeting C-MYC was picked from a screening including three siRNAs, from which siC-MYC#2 was chosen in consideration of its acceptable knockdown efficiency in both MDA-MB-231 and MDA-MB-468 breast cancer cells (Figure 5(B)). As shown in Figure 5(C), the inhibitory effect of lobetyolin on ASCT2 was significantly attenuated by C-MYC knockdown. Collectively, these findings indicate that c-Myc mediates lobetyolin-induced ASCT2 down-regulation in MDA-MB-231 and MDA-MB-468 breast cancer cells.
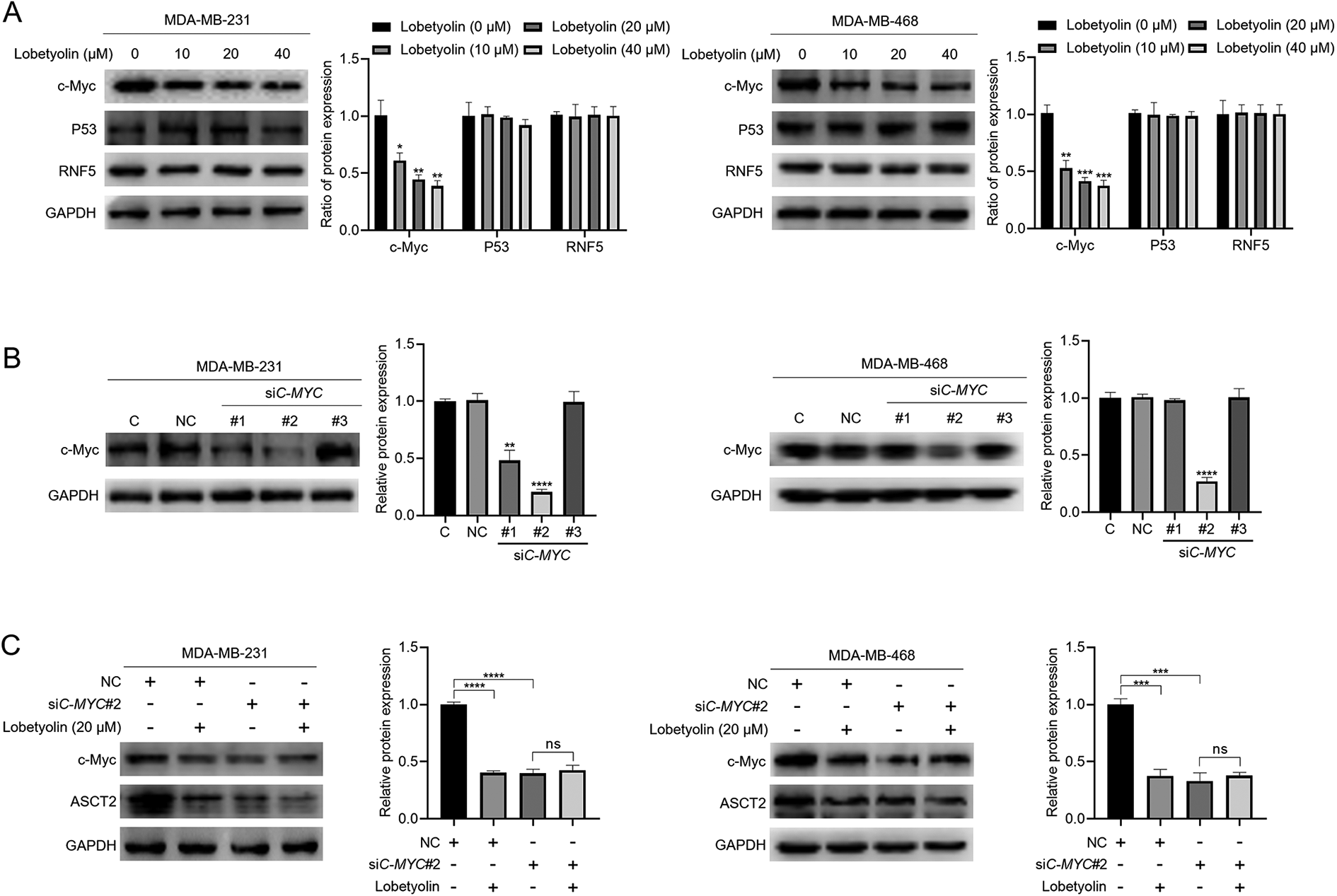
c-Myc mediated lobetyolin-induced ASCT2 down-regulation in MDA-MB-231 and MDA-MB-468 breast cancer cells. (A) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, protein samples were harvested and the expression of c-Myc, P53 and RNF5 was detected by immunoblot, with GAPDH as loading control. (B) MDA-MB-231 and MDA-MB-468 breast cancer cells were transfected with 100 nmol siRNA for 48 h, then protein samples were harvested and knockdown efficiency of c-Myc was checked by immunoblot, with GAPDH as loading control. (C) MDA-MB-231 and MDA-MB-468 breast cancer cells were transfected with 100 nmol siC-MYC#2 for 48 h, then cells were treated with 20 μM lobetyolin for another 24 h, protein samples were collected and the expression of ASCT2 and c-Myc were analyzed by immunoblot, with GAPDH as loading control. The data presented are representative of three independent experiments. Data are presented as mean ± SD. **P < 0.01 vs control group; ***P < 0.001 vs control group; ****P < 0.0001 vs control group.
Lobetyolin suppressed ASCT2 expression via AKT/GSK3β/c-Myc axis in MDA-MB-231 and MDA-MB-468 breast cancer cells.
Next, we set to investigate how lobetyolin regulated c-Myc expression. First, the effect of lobetyolin on the transcription of C-MYC was analyzed and the result showed that lobetyolin had no effect on the mRNA expression of C-MYC (Figure 6(A)), suggesting that lobetyolin might influence the stability of c-Myc protein. As expected, MG-132, the proteasome inhibitor, protected against lobetyolin-induced down-regulation of c-Myc and ASCT2 in both MDA-MB-231 and MDA-MB-468 breast cancer cells (Figure 6(B)). Given that the stability of c-Myc is regulated by the phosphorylation at Thr58 site, and the phosphorylation of c-Myc at Thr58 site is controlled by AKT/GSK3β signaling,19,20 the effect of lobetyolin on AKT/GSK3β signaling and phosphorylation of c-Myc at Thr58 was checked. As shown in Figure 6(C), lobetyolin increased the phosphorylation of c-Myc at Thr58 and decreased the phosphorylation of AKT and GSK3β in a dosage-dependent manner. Furthermore, Myr-AKT, a continuously active form of AKT, reversed decreased phosphorylation of GSK3β and down-regulation of c-Myc and ASCT2 in both MDA-MB-231 and MDA-MB-468 breast cancer cells (Figure 6(D)). Taken together, these results demonstrate that lobetyolin suppresses ASCT2 expression via AKT/GSK3β/c-Myc axis in MDA-MB-231 and MDA-MB-468 breast cancer cells.

Lobetyolin suppressed ASCT2 expression via AKT/GSK3β/c-Myc pathway in MDA-MB-231 and MDA-MB-468 breast cancer cells. (A) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, mRNA samples were harvested and the mRNA expression of C-MYC was detected by RT-qPCR. (B) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with 20 μM lobetyolin, 20 nM MG-132 or 20 μM lobetyolin combined with 20 nM MG-132, protein samples were harvested and the expression of ASCT2, c-Myc was analyzed by immunoblot, with GAPDH as loading control. (C) MDA-MB-231 and MDA-MB-468 breast cancer cells were treated with lobetyolin (10 μM, 20 μM, 40 μM) for 24 h, protein samples were harvested and the expression of indicated proteins was analyzed by immunoblot, with GAPDH as loading control. (D) MDA-MB-231 and MDA-MB-468 breast cancer cells were transfected with 2 µg Myr-AKT plasmid for 48 h, then cells were treated with 20 μM lobetyolin for another 24 h, protein samples were harvested and the expression of indicated proteins was analyzed by immunoblot, with GAPDH as loading control. The data presented are representative of three independent experiments. Data are presented as mean ± SD. **P < 0.01 vs control group; ***P < 0.001 vs control group; ****P < 0.0001 vs control group.
Discussion
Our study revealed that lobetyolin exerted anti-cancer effect via ASCT2 down-regulation-mediated apoptosis in breast cancer. Our data showed that lobetyolin inhibited ASCT2 expression by disturbing AKT/GSK3β/c-Myc axis. Therefore, our study highlights the potential value of lobetyolin for the treatment of breast cancer.
Our study first revealed the inhibitory effect of lobetyolin on the growth of MDA-MB-231 and MDA-MB-468 breast cancer cells. As rapid proliferation of cancer cells requires sufficient nutrients uptake including glucose and glutamine to meet the increased demand of biomacromolecule synthesis, 21 we investigated whether lobetyolin had an effect on glucose metabolism or glutamine metabolism. As expected, lobetyolin inhibited glutamine uptake and the expression of the glutamine transporter ASCT2. Knockdown of SLC1A5 weakened the anti-cancer effect of lobetyolin on MDA-MB-231 and MDA-MB-468 breast cancer cells. This finding is consistent with a previous study that lobetyolin-induced apoptosis via inhibiting ASCT2 expression in HCT-116 colon cancer cells. 8 However, the mechanisms of lobetyolin-induced ASCT2 down-regulation were different. In HCT-116 colon cancer cells, lobetyolin-induced ASCT2 down-regulation was mediated by P53, 8 but in MDA-MB-231 and MDA-MB-468 breast cancer cells it was c-Myc involved in lobetyolin-induced ASCT2 down-regulation. We speculate that the possible reason for this difference might be the genetic variation.
There are two apoptotic pathways, mitochondrial pathway and death receptor pathway and endoplasmic reticulum pathway. 22 ROS is a common inducer of mitochondrial pathway. In addition, Cleaved Caspase 3 and Cleaved Caspase 9 correlate closely with the activation of mitochondrial pathway and Bax/Bcl-2 regutale apoptosis by facilitating mitochondrial membrane permeabilization. 23 Our data showed that lobetyolin decreased intracellular GSH level and increased intracellular ROS level. Moreover, lobetyolin promoted the cleavage of Caspase 3 and Caspase 9 and regulated the expression of pro-apoptotic protein Bax and anti-apoptotic protein Bcl-2. Importantly, Cytochrome C, a mitochondrial content, was released from mitochondria to cytosol. These findings indicate that lobetyolin induces apoptosis via mitochondrial pathway in breast cancer cells.
Honestly, there are some limitations in our study. The anti-cancer effect of lobetyolin on breast cancer should be evaluated in vivo. This evaluation will be conducted in our future studies. Moreover, the anti-cancer effect of lobetyolin should be assessed in other cancer types.
In summary, our study demonstrates that lobetyolin exerts anti-cancer effect on breast cancer via ASCT2 down-regulation-induced apoptosis. Mechanistically, lobetyolin inhibits ASCT2 expression via AKT/GSK3β/c-Myc axis. Our present provide possibility for the application of lobetyolin in the treatment of breast cancer.
Footnotes
Authors’ contributions
Y.T.: design of the work and drafted the article. Y.C.: experiment operation, statistically analyzed data and reviewed the initial manuscript. Z.C., W.G.: helped to conduct a part of experiments. G.J., X.Z., X.L.: helped to revise the initial manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was financially supported by Jiangsu Medical Innovation Team (CXDT-37), Suzhou Clinical Medical Center Construction Project (Szzxj201503).
