Abstract
The application of gold nanoparticle-peptide conjugates as theranostic agents for colorectal cancer shows much promise. This study aimed at determining the neurotoxic impact of 14 nm gold nanoparticles (AuNPs) functionalized with colorectal cancer-targeting peptides (namely p.C, p.L or p.14) in a rat model. Brain tissue samples, obtained from Wistar rats that received a single injection of citrate-capped AuNPs, polyethylene glycol-coated (PEG) AuNPs, p.C-PEG-AuNPs, p.L-PEG-AuNPs or p.14-PEG-AuNPs, and sacrificed after 2- and 12-weeks, respectively, were analysed. Inflammation marker (tumour necrosis factor-α, interleukin-6, interleukin-1β), oxidative stress (superoxide dismutase, catalase, glutathione peroxidase) and apoptotic biomarker (cytochrome c, caspase-3) levels were measured. Gold nanoparticle-treated groups sacrificed after 2-weeks did not exhibit any significant inflammatory, oxidative stress or apoptotic effects in brain tissue compared to the untreated control group. In brain tissue from rats that were exposed to citrate-capped AuNPs for 12-weeks, tumour necrosis factor-α and interleukin-6 levels were significantly increased compared to the untreated control. Exposure to PEG-AuNP, p.C-PEG-AuNP, p.L-PEG-AuNP and p.14-PEG-AuNP did not elicit significant toxic effects compared to the control after 12-weeks, as evidenced by the absence of inflammatory, oxidative stress and apoptotic effects in brain tissue. We thus report on the safety of PEG-coated AuNP-peptide conjugates for potential application in the diagnosis of colorectal cancer; however, exposure to citrate-capped AuNPs could induce delayed neuro-inflammation, and as such, warrants further investigation.
Introduction
Globally, colorectal cancer (CRC) is the third most diagnosed cancer and is one of the leading causes of cancer-related deaths; correlating to 10.2% of the total number of cases diagnosed in both men and women. 1 Current detection methods for CRC include stool-based tests (faecal occult blood tests) and structural examinations (colonoscopy). 2 Limitations associated with faecal occult blood tests include false-positive results due to diet and medication, and a low sensitivity for the detection of adenomas.3–5 The main drawbacks associated with colonoscopy is that it is an expensive procedure that involves extensive bowel preparation; in addition to the increased risk of intraperitoneal bleeding, puncture of the colon or infection.2,6–8 Accordingly, there is a need for an innovative CRC screening tool that is reliable, inexpensive, non-invasive, sensitive and highly specific for early detection of CRC to improve survival and quality of life for patients with CRC.
One prospective avenue of research in the early detection and treatment of CRC is the application of gold nanoparticles (AuNPs) as a theranostic agent; as gold is considered inert and biocompatible, and possesses unique electrochemical and optical properties.9–13 The ability of AuNPs to bioconjugate to a wide variety of ligands, polymers or biomolecules make them ideal candidates for molecular imaging, chemical and biological sensing, targeted drug and gene delivery vehicles, cancer therapy and HIV therapy.11,14–19
The toxicological behaviour of AuNPs is influenced by various physicochemical aspects; with size, shape, surface charge and surface functionalization being the most determinant factors.20–26 Other factors, such as the dose administered, route of exposure, as well as the immune response to nanoparticles may also influence the toxicity. 27
A fundamental prerequisite for high therapeutic efficacy is the safe entry of the nanoparticle into the cell. 28 Nanoparticles are typically incorporated into the cell via the endocytic pathway in a size-dependent manner; however, many adverse effects might arise once AuNPs interact with cellular components.29–31 The AuNP-cell interaction could elicit an inflammatory response, oxidative stress, cellular genotoxicity or apoptosis.26,32–40
Compared to all the organs in the body, the brain is the most susceptible to oxidative damage due to the high oxygen consumption rate, high content of easily peroxidizable unsaturated fatty acids, and relative paucity of antioxidant enzymes. 41 The ability of nanoparticles to cross the blood-brain barrier, where they may accumulate and cause detrimental effects, and the sensitivity of nerve cells to toxicity, highlight the importance of neurotoxic evaluation of nanoparticles.42,43
Various peptides that target human colon cancer have been identified by phage-display libraries. 44 For this study, three peptides, namely p.C, p.L and p.14, were studied.45–47 In vitro studies by Wang et al. and Mazyambe showed that p.C, p.L and p.14 bind selectively to CRC cell lines.47,48 The application of AuNPs functionalized with these peptides thus makes them attractive candidates as potential theranostic tools for CRC.
The synthesis and characterization of the AuNP-peptide conjugates (p.C-PEG-AuNP, p.L-PEG-AuNP, p.14-PEG-AuNP) used in this study have been reported previously.49,50 A pilot study analysing the localization of these AuNP-peptide conjugates in vitro and in vivo, reported no obvious side-effects up to 48 hours; while in a parallel study, the toxic effects of the AuNP-peptide conjugates on the liver, kidney and spleen of Wistar rats were investigated 2-weeks and 12-weeks respectively, after a single intravenous injection.49,51 The present study was aimed at investigating the neurotoxic potential of citrate-capped AuNPs, polyethylene glycol-coated-AuNPs (PEG-AuNPs), p.C-PEG-AuNP, p.L-PEG-AuNP and p.14-PEG-AuNP in Wistar rats, after a single intravenous injection. In this regard, we examined: i) the ability of the AuNPs to induce inflammation by measuring levels of tumour necrosis factor α (TNF-α), interleukin 1β (IL-1β) and interleukin 6 (IL-6)—key pro-inflammatory cytokines 52 ; ii) the effect on superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx), well-known biomarkers of oxidative stress53,54; and iii) the effect on caspase-3 activity and cytochrome c levels—key apoptotic signalling pathway-related proteins. 55
Materials and methods
Synthesis, functionalization and characterization of AuNP-peptide conjugates
The citrate-reduction method, as described by Turkevich et al. and Frens, was used to synthesize the spherical citrate-capped 14 nm AuNPs; while the functionalization of the relevant AuNPs was performed as previously described.56–58 The characterization of the AuNP-peptide conjugates has been reported previously and was determined by UV-Vis spectroscopy, high resolution transmission electron microscopy, Fourier transform infrared radiation spectroscopy, dynamic light scattering and zeta potential measurements.49,50 All chemicals used were of high purity or analytical grade.
Animals and treatment
Seventy-two healthy adult Wistar albino rats (12 weeks of age, male) were used. Thirty-six rats were obtained from the animal house of North West University, South Africa; while a second group of 36 rats was obtained from SA Vaccine Producers (Animal Unit), South Africa. The animals were kept under controlled conditions of 22°C (±3°C), with a 12-hour light dark cycle. All animals were fed standard commercial rat chow and given distilled water ad libitum. Ethical approval was sought from and granted by the Nelson Mandela University Research Ethics Committee: Animal (Ref: A15-SCI-BCM-002). After 2 weeks of acclimatization, the animals were divided into two main experimental groups. Each experimental group was further divided into six sub-groups, consisting of six animals each. The sub-groups included an untreated control group and groups of AuNP-treated animals. Control animals were injected with phosphate-buffered saline (PBS); while the AuNP-treated groups comprised animals that were given a single intravenous administration of citrate-capped AuNP, PEG-AuNP, p.C-PEG-AuNP, p.L-PEG-AuNP or p.14-PEG-AuNP, respectively. The AuNP treatment (prepared at a concentration of 100 μg/ml in sterile PBS) was administered intravenously through the tail vein, at a dose of 100 µg/kg body weight, which is within the acceptable range of intravenous injection in rats. 59 Body weight was recorded 24 h after injection and thereafter weekly; while behavioural changes were monitored for the first 4 h, then over a period of 24 h, and thereafter daily throughout the experiment. Observations were performed to assess general health status, symptoms of toxicity, and mortality throughout the 2- and 12-week period of the study.
Animals from the first and second experimental group were sacrificed 2-weeks and 12-weeks post-injection, respectively, by quick exposure to diethyl ether. Control animals were sacrificed concurrently with the treated groups. Subsequently, brains were harvested, rinsed in ice-cold 0.9% sodium chloride, blotted with filter paper, weighed, wrapped in aluminium foil, snap-frozen in liquid nitrogen and stored at −80°C for biochemical analyses.
Sample preparation
Tissue samples from the brain were homogenized as previously described, with minor modifications.60,61 Briefly, brain tissue (100 mg) was homogenized (20% w/v) in ice-cold cell lysis buffer (Biovision, Milpitas, USA) using a Dounce homogenizer (Carl Roth, Karlsruhe, Germany) on ice. The homogenate was centrifuged at 10,000 × g for 10 min at 4°C, and the resultant supernatant stored at −80°C for TNF-α, IL-1β, IL-6, SOD, CAT, GPx, caspase-3 and cytochrome c determinations.
Inflammation marker determinations
Concentrations of TNF-α, IL-1β and IL-6 in homogenate supernatants were measured using ELISA kits from Elabscience (catalogue numbers: E-EL-R0019, E-EL-R0012 and E-EL-R0015, Wuhan, China) respectively, according to manufacturer’s instructions.
Oxidative stress assessments
The activities of anti-oxidative stress enzymes (SOD, CAT and GPx) were measured using colorimetric activity assay kits from Invitrogen (catalogue numbers: EIASODC and EIACATC, Carlsbad, USA) and Abcam (catalogue number ab102530, Shanghai, China) respectively, in accordance with manufacturer’s instructions.
Quantitative estimation of apoptotic markers
Caspase-3 activity was determined using the Caspase-3 Assay Colorimetric Assay Kit (Abcam, Shanghai, China); while cytochrome c levels were determined using the Rat Cyt-C (Cytochrome C) ELISA kit (Elabscience, Wuhan, China), according to manufacturer’s instructions.
Protein concentration determination in tissue lysates
Cytokine, antioxidant enzyme activity and cytochrome c levels in each sample were normalized to the tissue lysate protein content; as measured by the bicinchoninic acid assay (Pierce BCA kit, catalogue number 23227, Rockford, IL, USA).
Statistical analysis
Data are presented as the mean ± SD of four experiments (n = 4). Analysis of data was performed using GraphPad Prism v8.0 software (GraphPad Software Inc., San Diego, C.A., U.S.A.) and students t-test. One-way analysis of variance (ANOVA) followed by Tukey’s posthoc test was used to analyse differences among groups. Comparisons between groups were deemed significant for values of p < 0.05.
Results
General morphology, behaviour and physiology of rats
As reported previously, 49 no abnormalities relating to behavioural changes, food and water intake, body weight, organ weights or skin colour changes in rats exposed to the AuNP treatments throughout the 2- and 12-week period of the study were noted. Furthermore, no mortalities occurred. 49
Effect on pro-inflammatory biomarker levels
To assess the effect of the AuNP treatments on levels of pro-inflammatory cytokines (TNF-α, IL-1β an IL-6), ELISA was used. TNF-α levels in brains of rats (Figure 1) sacrificed 2-weeks after treatment with the relative AuNP treatments were not significantly different compared to the PBS control (p > 0.05, respectively). Moreover, no significant difference in TNF-α levels was noted among the AuNP-treated groups (p > 0.05, respectively). In brain tissue from rats sacrificed after 12-weeks (Figure 1), citrate-AuNP-treated rat brains showed significantly increased levels of TNF-α compared to the PBS control (p = 0.0076); whereas TNF-α levels in rats treated with PEG-AuNP, p.C-PEG-AuNP, p.L-PEG-AuNP or p.14-PEG-AuNP did not differ significantly from the PBS control (p > 0.05, respectively). Furthermore, after 12-week exposure to citrate-AuNPs, TNF-α levels in brain tissue were significantly higher than those of p.C-PEG-AuNP-, p.L-PEG-AuNP- and p.14-PEG-AuNP-treated rats (p < 0.05, respectively). It is noteworthy that TNF-α levels in the brain tissue of citrate-AuNP-treated rats at week 12 were significantly higher than those in rats exposed to PBS (p = 0.0030), citrate-capped AuNPs (p = 0.0011), PEG-AuNP (p = 0.0019), p.C-PEG-AuNP (p = 0.0019), p.L-PEG-AuNP (p = 0.0006) or p.14-PEG-AuNP (p < 0.0001) at week 2.

Effect of AuNP treatments on TNF-α levels in brain tissue of Wistar rats 2- and 12-weeks after a single intravenous injection. Tumour necrosis factor α levels were normalized to the total protein content in tissue lysates and are reported as mean ± SD values (n = 4). All AuNP treatments (100 µg/kg body weight) did not differ significantly (p > 0.05) when compared to the PBS control, except citrate-AuNP (12 weeks). Significantly different at p < 0.05 relative to: *PBS control (12 weeks); #Citrate-AuNP (12 weeks).
The effect of the AuNP treatments on IL-6 levels in rat brain (2-week exposure) was not significantly different (p > 0.05) when compared to the PBS control (Figure 2). After 12-week exposure to citrate-AuNPs, IL-6 levels in brain tissue were significantly higher than those of the PBS control, p.C-PEG-AuNP and p.L-PEG-AuNP (p < 0.05, respectively); while PEG-stabilized AuNPs did not significantly affect IL-6 levels in relation to the PBS control at week 12 (p > 0.05, respectively). Moreover, IL-6 levels in the brain tissue of citrate-AuNP-treated rats at week 12 were significantly higher than those in rats exposed to PEG-AuNP (p = 0.0258), p.C-PEG-AuNP (p = 0.0046), p.L-PEG-AuNP (p = 0.0128) and p.14-PEG-AuNP (p = 0.0181) at week 2.
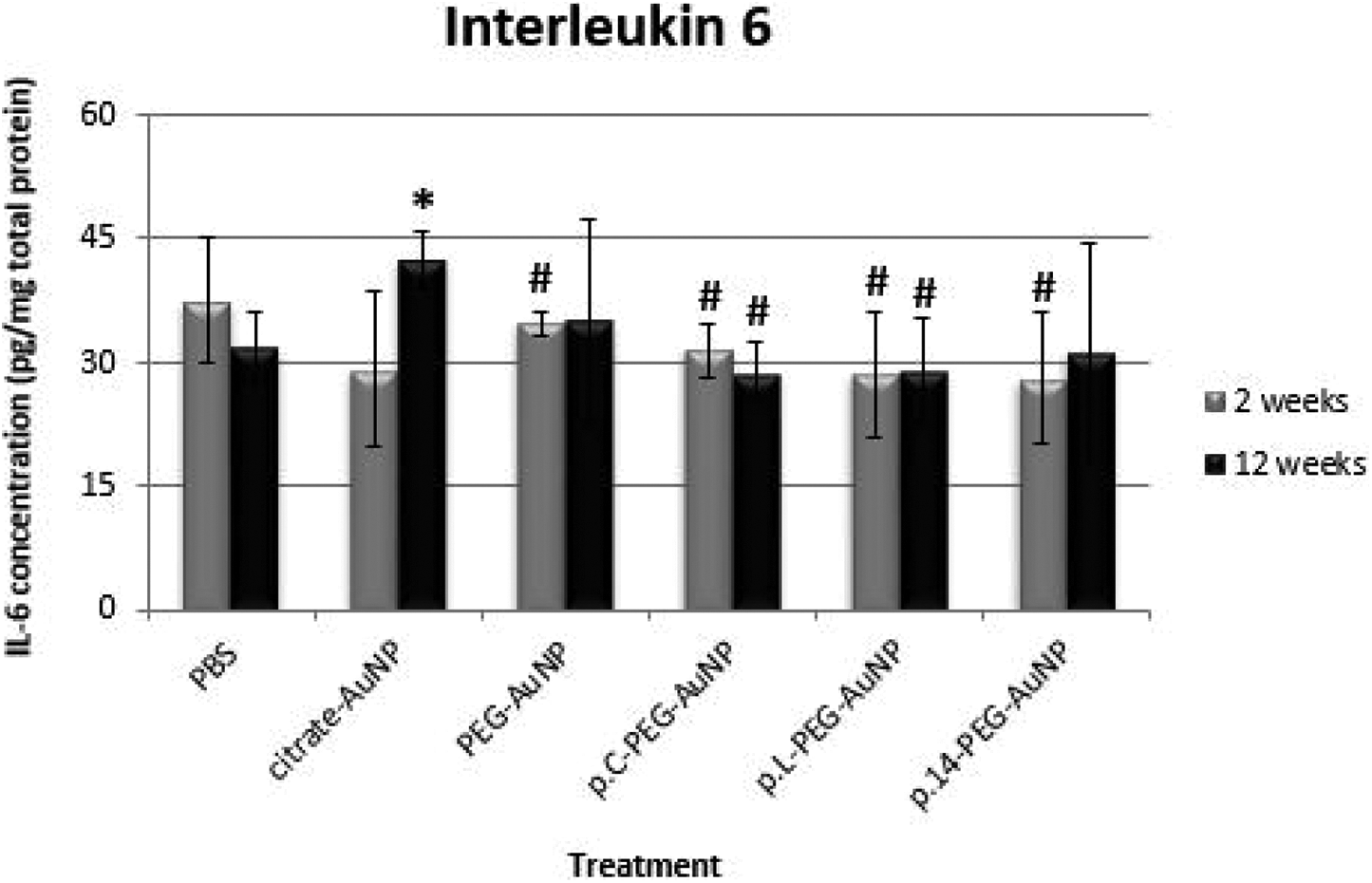
Effect of AuNP treatments on IL-6 levels in brain tissue of Wistar rats 2- and 12-weeks after a single intravenous injection. Interleukin-6 levels were normalized to the total protein content in tissue lysates and are reported as mean ± SD values (n = 4). All AuNP treatments (100 µg/kg body weight) did not differ significantly (p > 0.05) when compared to the PBS control, except citrate-AuNP (12 weeks). Significantly different at p < 0.05 relative to: *PBS control (12 weeks); #Citrate-AuNP (12 weeks).
None of the treatments (2- and 12-weeks exposure) impacted IL-1β levels significantly compared to the PBS control (Figure 3, p > 0.05, respectively), with no significant differences in IL-1β levels among AuNP-treated groups detected (p > 0.05, respectively).

Effect of AuNP treatments on IL-1β levels in brain tissue of Wistar rats 2- and 12-weeks after a single intravenous injection. Interleukin-1β levels were normalized to the total protein content in tissue lysates and are reported as mean ± SD values (n = 4). All AuNP treatments (100 µg/kg body weight) did not differ significantly (p > 0.05) when compared to the PBS control.
Effect on antioxidant enzyme activity
To assess the effect of the AuNP treatments on oxidative stress (Table 1), antioxidant enzyme activities (SOD, CAT and GPx) were determined. None of the AuNP treatments (2- or 12-weeks exposure) significantly affected SOD activity compared to the PBS control (p > 0.05, respectively), and there was no significant difference in the activity of SOD among AuNP-treated groups (p > 0.05, respectively).
Influence of the respective AuNP treatments on antioxidant enzyme activity in brain tissue of Wistar rats 2-weeks and 12-weeks after a single intravenous injection.
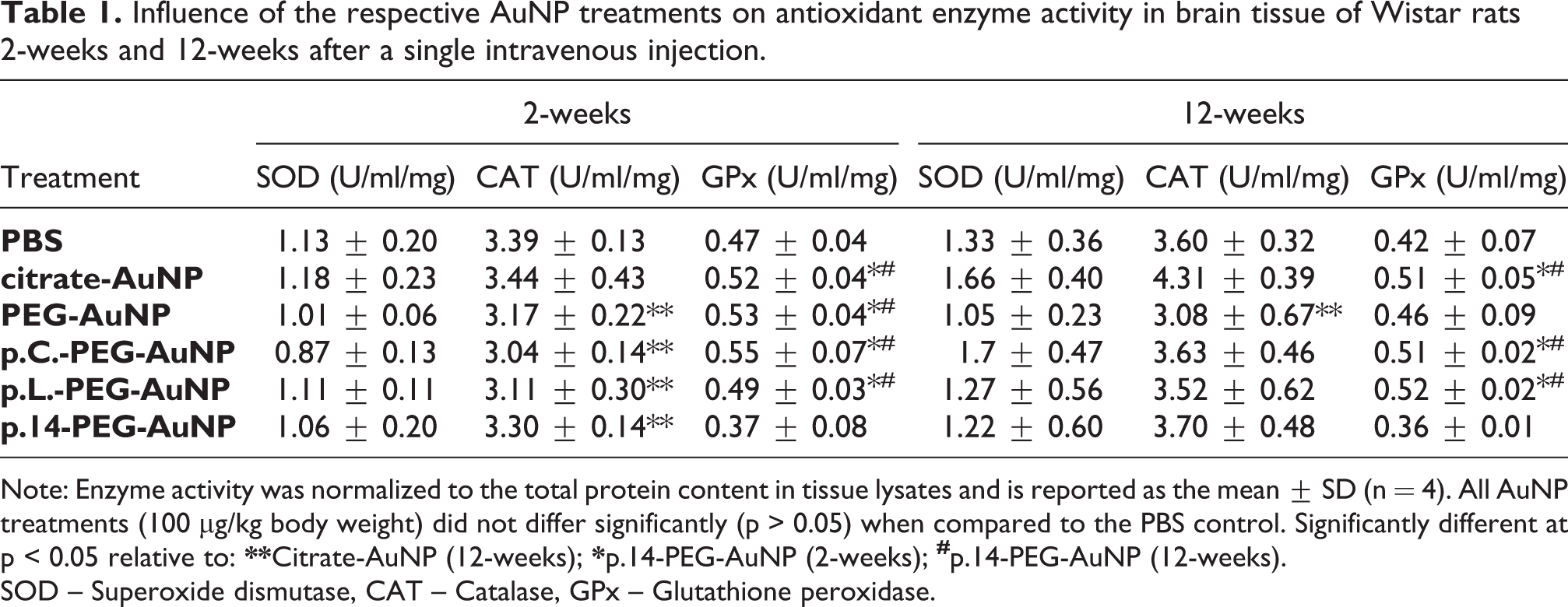
Note: Enzyme activity was normalized to the total protein content in tissue lysates and is reported as the mean ± SD (n = 4). All AuNP treatments (100 µg/kg body weight) did not differ significantly (p > 0.05) when compared to the PBS control. Significantly different at p < 0.05 relative to:
SOD – Superoxide dismutase, CAT – Catalase, GPx – Glutathione peroxidase.
Catalase activity (Table 1) was not significantly affected by the AuNP treatments in relation to the PBS control (p > 0.05) following 2- or 12-week exposure respectively; however, 12-week exposure to PEG-AuNPs significantly reduced CAT activity when compared to citrate-AuNPs (p = 0.0307). Moreover, a comparison between the different treatments over the 2- and 12-week periods indicated that exposure to PEG-stabilized AuNPs tended to decrease CAT activity in brain tissue at 2 weeks when compared to treatment with citrate-AuNPs at week 12. Consequently, there was a significant difference compared to that of rats exposed to PEG-AuNP (p = 0.0124), p.C-PEG-AuNP (p = 0.0036), p.L-PEG-AuNP (p = 0.0067) and p.14-PEG-AuNP (p = 0.0417) at 2-weeks.
The effect of the AuNP treatments on GPx activity (Table 1) in rat brain (2- and 12-week exposure) did not differ significantly compared to the PBS control (p > 0.05, respectively). Comparisons among groups indicated that p.14-PEG-AuNP-treated rats (2- and 12-week exposure) were associated with a significant reduction in GPx activity relative to the different AuNP-treated groups (p < 0.05, respectively).
Effect on the induction of apoptosis
No significant differences were observed in the cytochrome c levels (Figure 4) or caspase-3 activity (Figure 5) in all AuNP treatments (2-weeks and 12-weeks) when compared to the PBS control (p > 0.05, respectively). Similarly, no significant differences among AuNP-treated groups (2- and 12-week exposure) were noted (p > 0.05, respectively).
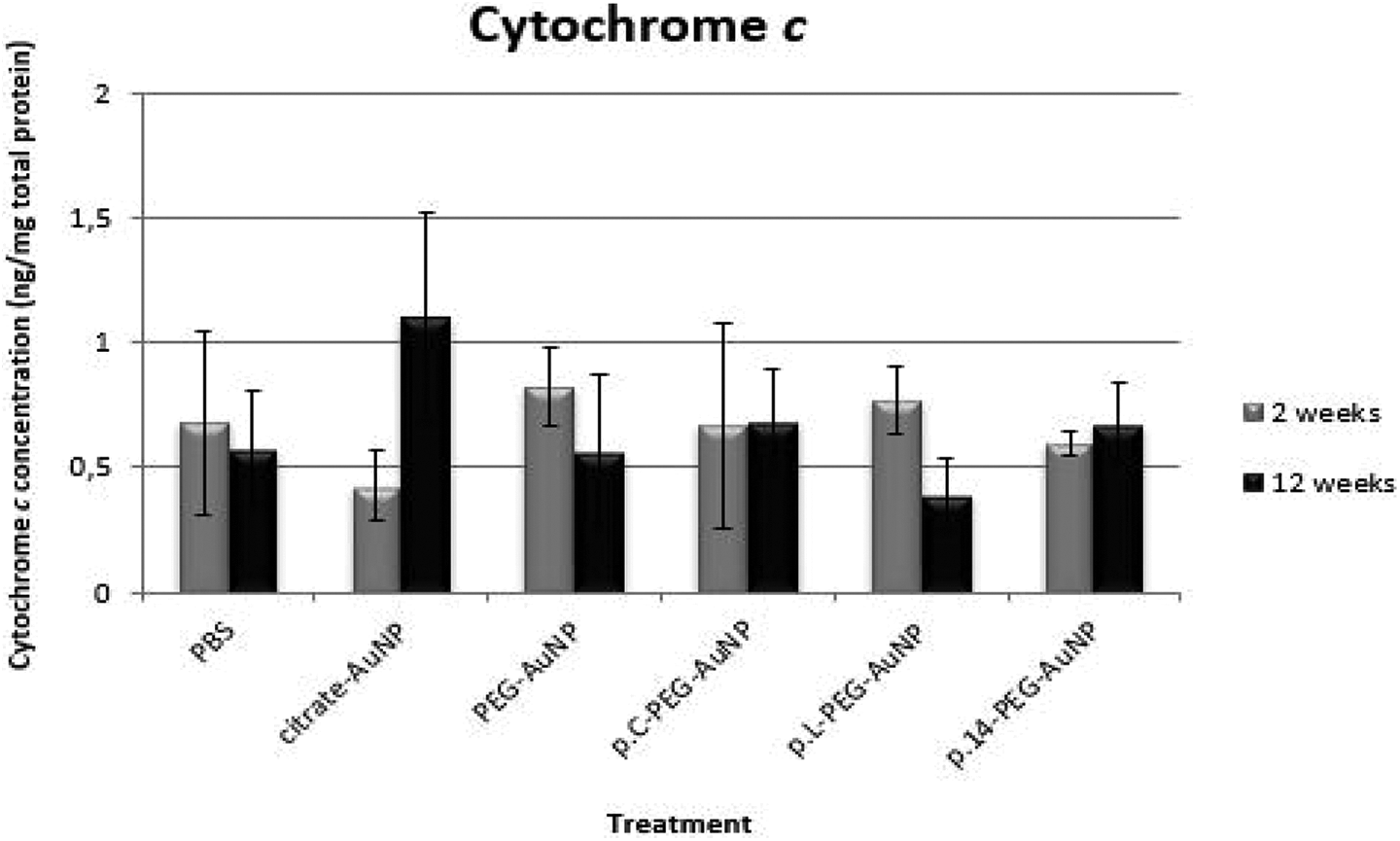
Effect of AuNP treatments on cytochrome c levels in brain tissue of Wistar rats 2- and 12-weeks after a single intravenous injection. Results were normalized to the total protein content in tissue lysates and are reported as mean ± SD values (n = 4). All AuNP treatments (100 µg/kg body weight) did not differ significantly (p > 0.05) when compared to the PBS control.

Effect of AuNP treatments on caspase-3 activity in brain tissue of Wistar rats 2- and 12-weeks after a single intravenous injection. Enzyme activity was calculated as the fold increase compared to the PBS control, and results are reported as the mean ± SD (n = 4). All AuNP treatments (100 µg/kg body weight) did not differ significantly (p > 0.05) when compared to the PBS control.
Discussion
The challenge in developing theranostics is to prevent side-effects on other vulnerable organs. The brain is relatively protected from most blood-borne molecules,62,63 but little is known about the effects of 14 nm AuNP with different coatings on the brain. Some studies have demonstrated the ability of AuNPs to cross the blood-brain barrier; investigations by Sonavane et al. and De Jong et al. indicated that AuNPs ranging in size from 10 nm to 50 nm were able to cross the blood–brain barrier and that the accumulation of AuNPs is size-dependent.64,65 Moreover, a study analysing the ability of insulin-coated gold nanoparticles (20, 50 and 70 nm) to penetrate the blood-brain barrier by targeting insulin receptors, found that 20 nm AuNPs displayed the most wide-spread biodistribution and accumulation within the brain. 66
Numerous nano-neurotoxicity studies implicate oxidative stress, apoptosis and the inflammatory response as the main mechanisms underlying the neurotoxicity of metallic nanoparticles.67–70 These mechanisms may function autonomously or may well be inter-related; where oxidative stress could trigger inflammation through the release of cytokines, DNA damage, autophagy or apoptosis. 71
The findings of this study indicated that acute exposure to 14 nm citrate-capped AuNPs for 12-weeks resulted in a marked increase in pro-inflammatory cytokine (TNF-α and IL-6) levels in rat brain compared to the untreated control and other AuNP-treated groups (Figures 1 and 2, respectively). This increase was significantly more pronounced at week 12, indicating that exposure to citrate-capped AuNPs may promote inflammation in the brain after an extended time.
It is noteworthy that although none of the antioxidant enzyme activity levels changed significantly in any group compared to the PBS control (Table 1), there was an overall tendency of lower levels of CAT activity in all the groups treated with AuNPs compared to the citrate-capped AuNP group. Further, it should be noted that AuNPs possess antioxidant properties 72 and could thus contribute towards reducing oxidative stress; consequently, less SOD, CAT or GPx would then be synthesized. Interestingly, the citrate-capped AuNPs used in our study had a zeta potential of −31 mV, the zeta potential of PEG-AuNP was −26 mV, while that of the peptide-PEG-AuNPs was lower than −11 mV. 49 Given that citrate-capped AuNPs (13 nm) have been reported to contribute to oxidative stress in cell lines 73 ; it seems therefore that PEG-stabilized AuNPs tend to rather reduce oxidative stress.
Considering that treatment with PEG-AuNPs or PEG-peptide-conjugated AuNPs did not cause inflammation (Figures 1 to 3), oxidative stress (Table 1) or apoptosis (Figures 4 and 5) in relation to the untreated control, these findings thus point to the neurotoxic potential of citrate-AuNP’s – which could be attributed to differences in the surface coating of the respective AuNPs analysed in this study.
Many studies have demonstrated that the physical and chemical properties of AuNPs with different surface coatings play a crucial role in determining the toxicological behaviour of AuNPs. A study analysing the physical properties of AuNPs suggested that one possible way of circumventing significant toxicity is to coat the AuNPs with biocompatible polymers. 74 Another study investigating the toxicity of citrate-capped AuNPs reported that naked AuNPs (or citrate-capped AuNPs) are significantly toxic both in vitro and in vivo, while surface coating (such as protein- or polymer-coating) may partially or significantly prevent these toxic effects. 75 According to Fratoddi, PEG and its derivatives are among the main polymeric ligands used to coat and stabilize AuNPs; while functionalization with peptides through surface modification enhances the properties and characteristics of nanoparticles. 76 These reports emphasize the significance of nanoparticle surface coating in toxicity studies and agrees with our findings.
In this study, citrate-capped AuNPs were coated with a combination of 1% PEG-biotin and 99% PEG-OH, followed by functionalization with streptavidin and subsequent conjugation to biotinylated p.C, p.L or p.14.49,50 The PEG-OH promotes AuNP stabilization by preventing its aggregation in aqueous solution; while PEG-biotin serves as a linker agent between streptavidin and the biotinylated peptides. 58 Notably, the findings from a parallel study analysing the short- and long-term post-exposure effects of citrate-AuNP, PEG-AuNP, p.C-PEG-AuNP, p.L-PEG-AuNP or p.14-PEG-AuNP in vivo, supported the use of 99% PEG-OH and 1% PEG-biotin, as no toxicity was noted in the liver, kidney and spleen of Wistar rats 12-weeks post-exposure. Further, no evidence of short- and long-term post-exposure toxicity was noted with citrate-capped AuNPs in blood parameters, serum marker enzymes or the investigated peripheral tissue. 49 These results do not correlate with the citrate-AuNP-induced toxicity observed in the brain in our present study; however, it could be possible that the citrate-AuNPs crossed the blood-brain barrier and interacted with the cerebral microvasculature, thereby producing a pro-inflammatory cascade 77 ; hence neurotoxicity.
Although exposure to PEG-AuNPs, p.C-PEG-AuNP, p.L-PEG-AuNP and p.14-PEG-AuNP (for 2- or 12-weeks) after a single intravenous injection did not induce significant inflammatory (Figures 1 to 3), oxidative stress (Table 1) or apoptotic effects (Figures 4 and 5), it was noted that GPx activity in brain tissue from p14-PEG-AuNP-treated rats (2- and 12-weeks) was significantly lower than that of the AuNP-treated groups (Table 1).The overall extent of the antioxidant enzyme system response was not significant relative to the untreated group – this is most likely related to differences in surface modification of the AuNP treatments. When considering the terminal (free) end of the amino acid sequence of p.C (CVFSSSYSSSG), p.L (LTVSPWY) and p.14 (PDHERPM), it is noted that the terminal amino acid of p.14 is methionine. 49 According to Aledo, methionine fulfils an important antioxidant role, stabilizes the structure of proteins, participates in the sequence-independent recognition of protein surfaces and can act as a regulatory switch through reversible redox reactions. 78 Nevertheless, methionine residues are susceptible to spontaneous oxidation to methionine-sulphoxide, which is reduced enzymatically by methionine-sulphoxide reductase; however, chronic or severe oxidative stress produces methionine sulphone that cannot revert to the reduced state. 79 The reduced GPx activity after treatment with p.14-PEG-AuNP (compared to that of p.C-PEG-AuNP and p.L-PEG-AuNP) could be a result of an adapted response of the enzyme to low-level oxidative stress, due to the cyclic oxidation and reduction of methionine. In this regard, further studies are warranted to elucidate the unknown mechanism of action of these peptides in vivo.
It is important to highlight that numerous discrepancies regarding AuNP toxicity have been reported. For instance, Siddiqi et al. revealed that oxidative stress was induced in brain tissue of rats that were exposed to 20 nm AuNPs, 20 μg/kg body weight, for 3 days (surface coating not defined). 68 Moreover, lipid peroxidation, 8-hydroxydeoxyguanosine, caspase-3 and heat shock protein 70 levels were significantly increased and correlated negatively with glutathione peroxidase activity; which further alluded to the inflammatory and apoptotic potential of AuNPs in brain tissue. 68 In agreement with these findings, Noor et al. reported that lipid peroxidation increased significantly in rat brains 24 hours after injection with 100 μg/kg body weight of 20 nm AuNPs (surface coating not defined); however, levels of glutathione and nitric oxide, including glutathione S-transferase activity (that was induced after early exposure to the AuNPs) normalized after 2 weeks. 80 Other in vivo-based studies reporting on AuNP-induced neurotoxicity found that AuNPs may cause astrogliosis, increased seizure activity, and cognition defects; as well as DNA damage in the rat cerebral cortex after exposure to 10 nm and 30 nm citrate-capped AuNPs at 70 μg/kg body weight.81,82
In contrast, a study analysing the effects of citrate-capped AuNPs (20 nm) on Alzheimer’s disease in a rat model, used the protein phosphatase inhibitor, okadaic acid, to induce neurotoxicity. Subsequent long-term AuNP treatment (2.5 mg/kg body weight every 48 hours for 21 days) inhibited neurotoxin-induced oxidative stress by restoring antioxidant enzyme activity and glutathione levels. 83 Another study found that daily intraperitoneal administration of citrate-capped AuNPs (12.5 nm) in mice for 8 days, at varying doses between 40 and 400 μg/kg/day, resulted in AuNP uptake in different tissues and that the AuNPs were able to cross the blood-brain barrier; however, no histological evidence of toxicity was observed in the relevant tissues examined. 84
Given the relative wealth of toxicity studies on gold nanoparticles, it is essential to clarify the toxic effects of nanoparticles in various tissues; especially when considering the wide variability of assay conditions, the physico-biochemical factors that influence the toxicity of AuNPs,85,86 as well as the unpredictable nature of AuNPs in biological systems.
Conclusion
The results from this study thus suggest that therapeutic application of citrate-capped AuNPs should be carefully considered and point to the safety of PEG-stabilized, CRC-targeting-peptide AuNP conjugates for potential application in the diagnosis of CRC. Nonetheless, the application of AuNP-peptide conjugates in preclinical cancer theranostics is an incipient field, while research concerning combinatorial applications of multi-functionalized AuNPs in cancer therapy is limited. 87 Accordingly, further investigation of AuNP-peptide conjugates in vivo and their potential application as theranostic agents should be explored – to investigate the fate and clearance efficiency of functionalized AuNPs within the brain – thereby increasing the therapeutic index of nano-based drugs approved for clinical use.
Footnotes
Acknowledgements
M Pereira gratefully acknowledge Nelson Mandela University, Port Elizabeth, South Africa, for support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Research Foundation, NRF-Innovation Grant 112019, Pretoria, South Africa.
