Abstract
Introduction:
Malignant pleural mesothelioma (MPM) is a malignant tumor that is associated mostly with asbestos exposure. The present study was to evaluates the diagnostic value of neopterin, periostin, YKL-40, Tenascin-C (TNC), and Indolamine 2,3-dioxygenase (IDO) as noninvasive markers of malign pleural mesothelioma.
Methods:
Included in the study were 30 patients diagnosed with malign pleural mesothelioma, and 25 people as a control group. Biomarker levels were determined using an enzyme immunoassay . A Mann-Whitney U test and Spearman correlation methods were used for the statistical analysis.
Results:
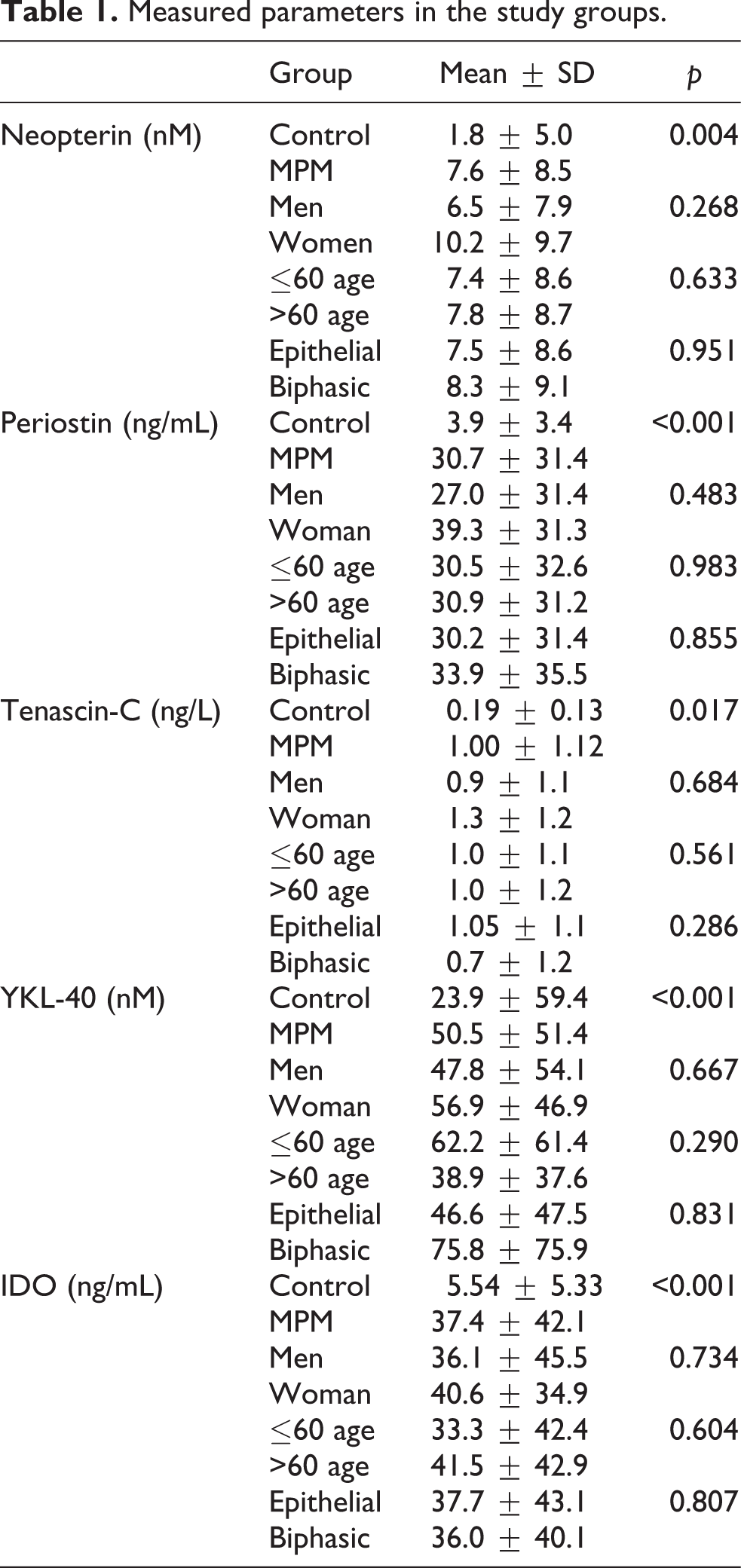
All evaluated biomarkers were found to be significantly higher in the MPM group than in the control group (p < 0.05). There was no effect of such variables as gender, age or MPMsubtype on the parameters (p > 0.05) in the patient group. All biomarkers were positively correlated with each other (p < 0.001).
Conclusions:
The current non-invasive biomarkers that can be used in the diagnosis of MPM yielded significant results and can make important contributions to the early diagnosis of MPM.
Introduction
Background
The global incidence of malignant pleural mesothelioma (MPM) has witnessed a steady increase over the past decade and its prevalence is expected to have reached a peak in 2020. MPM is an aggressive malignancy of the pleural surface that is caused predominantly by asbestos exposure. 1 The irregular use of asbestos has made MPM a global health problem.2–4 Most cases of MPM are attributed to asbestos exposure, which is usually over 40 years. 4 Since asbestos exposure mainly occurs in the workplace, MPM is classified as an occupational disease. 1 Erionite (a mineral found in rocks in Turkey), chest wall radiation and simian virus 40 (SV40) are among the other causes of MPM among which SV40, an oncogenic virus that blocks tumor suppressor genes, may act as a cofactor in MPM development, although studies of its direct effects have been inconclusive.1–3
The carcinogenesis mechanism in MPM is multifactorial. Asbestos fibers are inhaled and migrate to the pleural space where they cause irritation and a repetitive cycle of tissue damage and tissue repair begins. When free oxygen radicals released by the asbestos fibers are phagocytosed by macrophages, intracellular DNA damage and abnormal repair occurs. Asbestos fibers also penetrate mesothelial cells, interfering with mitosis, producing DNA mutations and altering chromosome structures. Mesothelial cells exposed to asbestos secrete inflammatory cytokines, including tumor growth factor-β, platelet-derived growth factor, and vascular endothelial growth factor (VEGF) 2 and these inflammatory cytokines create a favorable microenvironment for tumor growth. Asbestos induces the phosphorylation of various protein kinases (mitogen-activated protein and extracellular signal-regulated kinases 1 and 2), resulting in an increased expression of proto-oncogenes and a greater promotion of abnormal cellular proliferation.5,6
The majority of patients with MPM experience shortness of breath, chest pain and pleural effusion while other symptoms include fatigue, anorexia, weight loss, sweating and weakness. 7 MPM is a complex disease that causes significant morbidity and mortality.8,9 There are four main histological subtypes of MPM; epithelioid, sarcomatous, biphasic or mixed and desmoplastic.4,5 Since the diagnosis of MPM is difficult, many biomarkers have been evaluated for its diagnosis and prognosis8,9 as identiying a reliable diagnostic biomarker with high diagnostic sensitivity and specificity would contribute significantly to the diagnosis and prognosis of MPM. The markers evaluated to date include glycoproteins such as mesothelin, megakaryocyte potentiating factor, osteopontin, and fibulin-3, although their diagnostic ability is limited and has no clinical utility. 10
Periostin is a matricellular protein that is secreted by cancer-associated fibroblasts (CAFs), and that promotes cancer deviation, initiation and progression.11,12 Periostin upregulation has been reported for many types of cancer in clinical settings, including MPM, and has been identified as a potential tumor-promoting factor. 13
Neopterin is a derivative of pteridine that is produced by monocyte, macrophage, dendritic and endothelial cells, and that is activated by interferon-gamma (IFN-γ). It has been detected in various bodily fluids such as serum, cerebrospinal fluid, synovial fluid, urine and saliva, and its concentrations in these bodily fluids have used been as an indicator in evaluations of systemic immune and inflammatory responses in several different studies.14–16
Indoleamine 2, 3-dioxygenase (IDO) is an inducible enzyme that catalyzes the degradation of kynurenine, as the initial stage in tryptophan catabolism. In its normal functions, IDO plays an immunosuppressive role following inflammatory stimuli, preventing permanent immune activation and accompanying immunopathologies. IDO can be plundered by malignancies as an immune escape mechanism. 17
YKL-40 (a chitinase-like protein) is an inflammatory biomarker produced at the disease site by various cells, including cancer cells and cancer-associated macrophages, and is associated with the pathogenesis of lung lesions. 18 Although the true function of YKL-40 is unclear, its expression is associated with inflammation, extracellular tissue remodeling and hepatic fibrosis. 19
Tenascin-C is an extracellular matrix (ECM) glycoprotein with anti-adhesive properties, and is expressed in human malignant neoplasms. 20 TNC is highly expressed during organogenesis and in the accompanying interactions between cell proliferation, migration, epithelial-mesenchymal transition (EMT) and parenchyma mesenchyme. 21
While increased levels of TNC, neopterin, periostin, IDO and YKL-40 have been detected in many infectious lung diseases; tuberculosis, pneumonia, obstructive lung diseases; asthma, lung cancer, fibrotic lung disorders; cystic fibrosis, idiopathic pulmonary fibrosis) in various types of benign and malignant lung tumors, to our knowledge no studies have assessed neopterin, IDO, YKL-40 and TNC levels in MPM patients. Additionally, the prognostic utility of these markers has not been compared previously. The aim of the present study was to compare the the potential usefulness of serum IDO, neopterin, YKL-40, periostin and TNC levels with currently used tumor markers in asbestos induced-MPM patients.
Patients and methods
Study design and setting
This prospective cross-sectional study was carried out between November 2018 and April 2019 in the Chest Diseases, Thoracic Surgery Outpatient Clinic of Dicle University. The MPM group comprised 19 male and 11 female patients, while the control group comprised 25 cancer-free individuals. The study was carried out in accordance with the principles of the Declaration of Helsinki, and with the approval of the Inonu University Malatya Clinical Research Ethics Committee (approval number 2018/145). All participants gave informed consent for the use of their medical data for research purposes. The participants’ age (≤60 or >60 years), sex, asbestos exposure and duration of exposure (≤20 or >20 years), histopathological type, stage and survival status were recorded on standard forms. Radiologically, the affected side (right/left), identified through thorax ultrasonography and/or computed tomography findings, the presence of pleural fluid and cytology result, the diagnostic method (non-surgical procedures such as pleural/peritoneal biopsy or video-assisted thoracoscopic surgery [VATS], surgery such as thoracotomy, laparotomy methods), and the mesothelioma subtypes were evaluated as epithelioid, sarcomatoid or biphasic (mixed), according to the 1999 WHO/IASLC classification. The International Mesothelioma Interest Group (IMIG) classification was used for the staging of mesothelioma patients with a definitive diagnosis. Peripheral venous blood (1–2 ml) of all the participants in the study was taken into routine biochemistry tubes. After the blood samples were centrifuged at 3,500 rpm at room temperature for 15 min, the separated sera were taken into Eppendorf tubes and stored at -80° C until the time of analysis.
Neopterin, YKL-40, periostin, TNC and IDO were quantified in the serum samples using enzyme-linked immunosorbent assay kits (E3155Hu, E2063Hu, E3226Hu, E1414Hu, and E0796Hu; Bioassay Technology Laboratory, Ltd.). The measurement ranges of the assays for neopterin, YKL-40, periostin, TNC, and IDO were 0.1–38.0 nmol/L, 1–400 ng/mL, 0.5–150 ng/mL, 20–6000 ng/L and 0.3–90.0 ng/mL, respectively. The coefficients of variation for inter- and intra-assay precision were less than 10% and less than 8%, respectively, for all ELISA kits. Statistical analysis
A statistical analysis of the findings obtained in the study was made using SPSS for Windows (Version 11.5. Chicago, SPSS Inc.). The relationship between the measured parameters in the study groups was evaluated with a Spearman correlation test. A regression analysis was made in the preparation of calibration accuracy and calculations, since the data are not normally distributed, Mann Whitney U test was used for the comparison of the numerical data. p < 0.05 was accepted as statistically significant.
Results
The mean age of the control group was 38.6 ± 5.2 (Min-Max: 30–49). . The entire patient group grew up in a geography with known asbestos content, and so were exposed to asbestos from birth. The mean age of the patient group was 59.5 ± 10.6 (Min-Max: 35–76). Thoracoscopy was performed on 18 patients (M/F;14/4). Pleural fluid was examined in 10 patients with thoracoscopy (M/F;5/5). Axillary lymph node biopsy was performed in one female patient and thoracentesis was performed in another. A histopathological examination of the patient group classified 26 as epithelial and four with biphasic malignant mesothelioma. Six patients had a stage II of the disease, 15 had stage III, and 9 had stage IV. While 22 patients were inoperable, 7 patient refused surgery. Twenty patients who participated in the study are now dead, 10 patients are receiving chemoradiotherapy.
No statistically significant difference was noted in a comparison of periostin, TNC, neopterin, YKL-40 and IDO levels according to gender, age, and histopathological subtype in the patient group (p > 0.05) (Table 1).
Measured parameters in the study groups.
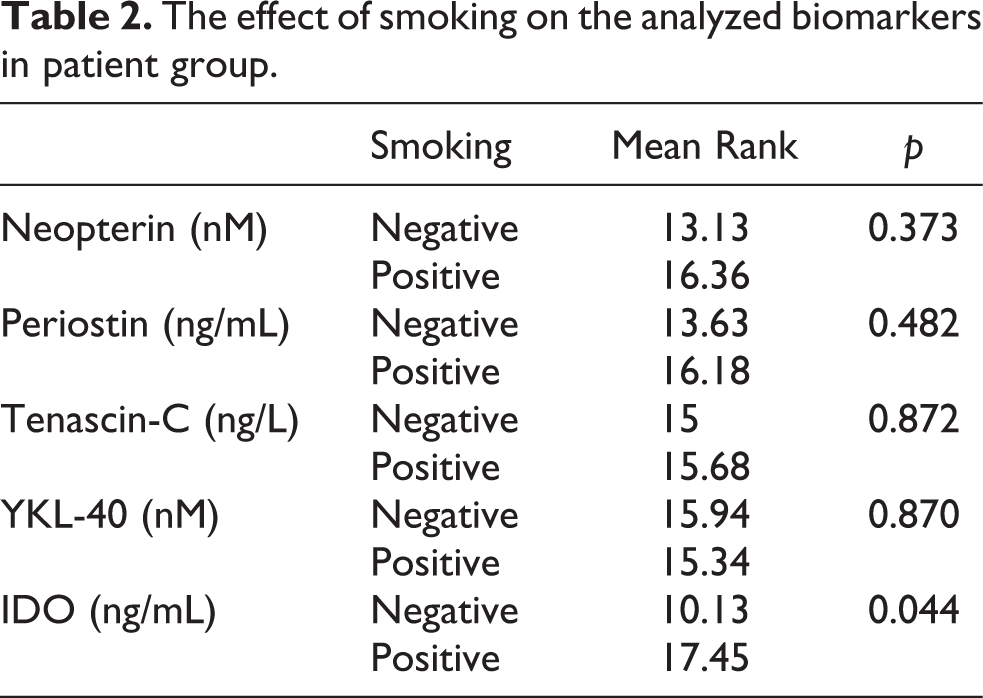
Among the parameters, only IDO is statistically significant in smoking (Table 2).
The effect of smoking on the analyzed biomarkers in patient group.
Correlations between parameters evaluated in the study groups are summarized in Table 3.
Correlations between biomarkers in the study groups.

rs: Spearman’s rho value.
** Correlation is significant at the 0.01 level (2-tailed).
Discussion
Malignant pleural mesothelioma is difficult to diagnose and is usually resistant to most treatments. Biomarkers would be useful in managing three aspects of the clinical management of MPM: diagnosis, prognosis, and treatment success prediction. 22 The markers analyzed in our study may be useful in diagnosis and insight into the molecular factors related to the diagnosis is required for effective therapeutic strategies. Literature contains many studies investigating the interactions between asbestos fibers and mesothelial cells. Asbestos interacts with mesothelial cells through direct and indirect mechanisms, with experimental studies, having revealed the translocation of fibers from the lung to the pleura. Asbestos is known to mediate the release of inflammatory factors and growth factors in the pleural space. 23
Higher YKL-40 levels have been identified in the serum and sputum of cystic fibrosis patients than in those of healthy individuals, and its levels are higher in severe exacerbation periods of the disease. 24 In a similar study, it has been suggested that YKL-40 is increased in the circulation and lungs of idiopathic pulmonary fibrosis patients, and that this glycoprotein is associated with the pathophysiology of idiopathic pulmonary fibrosis. 25 It has been further reported that an increase in serum YKL-40 occurs in systemic sclerosis with sarcoidosis and that pulmonary involvement, and serum YKL-40 is a potential biomarker of disease activity and ongoing fibrosis.26,27 Pleural YKL-40 levels have been found to be higher in patients with exudative pleural effusion than those with cardiogenic transudative pleural effusion, 28 and serum YKL-40 levels have been reported to predict poor prognosis in non-small cell lung carcinoma (NSCLC). 29 In another study, the correlations between YKL-40 and inflammatory, fibrosis markers and lung function indices were evaluated to further investigate the relationship of YKL-40 with asbestosis. While YKL-40 was negatively correlated with lung function capacity parameters, it was found to be positively correlated with inflammation markers and fibrosis markers. 30 Guan et al. found that YKL-40 inhibits cell apoptosis while promoting proliferation, migration and inflammatory cytokines of the human bronchial epithelial cell line BEAS-2B. They also suggested that the suitable concentration of YKL-40 treatment could stimulate the proliferation of BEAS’-2B2 and provide a new perspective for the treatment of allergic asthma. 31 When all these studies are evaluated together, it can be concluded that besides its etiology, YKL-40 increases under pathological conditions that lead to chronic inflammation and tissue remodeling in the lung. In our study, we found that while YKL-40 was found to be high in the MPM group; gender, age, and disease subtype did not affect YKL-40 levels. Likewise, a statistically significant difference was found between healthy individuals and patients in TNC levels. However, other variables had no effect on TNC levels.Tenascin-C expression is low or undetectable in healthy adult tissues, and has been shown to be increased in the inflamed respiratory tract of asthma patients and the epithelial lining fluid of patients with lung fibrotic disorders. 32 The expression of TNC in lung diseases is likely associated with the stimulating effect of inflammatory cytokines on the epithelium of asthma patients, and TNC expression has also been observed in various types of benign and malignant lung tumors. 20 Some researchers have suggested that Tenascin-C may function as a diagnostic tumor marker due to its overexpression in the stroma of most solid malignancies, and in their study they evaluated the potential importance of Tenascin-C as a predictor of tumor progression in the sera of NSCLC patients. Although they found serum Tenascin-C levels significantly higher in patients with NSCLC, they found the sensitivity and specificity of the quantification test to be low. However, based on their results, they emphasized that Tenascin-C could be used as a predictive prognostic marker for NSCLC patients. 33 Aside from these, it has been suggested that TNC may be used as an indicator of the severity of the disease in many chronic lung diseases, such as chronic obstructive pulmonary disease (COPD), bronchopulmonary dysplasia (BPD), idiopathic pulmonary fibrosis (IPF), 32 respiratory distress syndrome (RDS), asthma.34,35 and NSCLC. 33
Several studies suggest that IDO expression or activity in cancer patients is associated with reduced intratumoral T cell infiltration, disease progression, and shorter overall survival.36–38 In the tumor microenvironment, not only tumor cells, but also some tumor infiltrating cells such as dendritic cells, lymphocytes and monocytes can be the source of IDO. In NSCLC, a negative correlation was found between IDO-producing tumor infiltrating eosinophils and disease progression. IDO-induced tryptophan depletion from the tumor microenvironment may be due to elevated levels of the enzyme or increased consumption by both the host’s disordered antigen-presenting cells and tumor cells. 39 IDO expression caused by an increase in cancer cells is considered a critical immune escape mechanism and a new prognostic factor in cancer immunity, while serum IDO activity has been identified as a new prognostic factor in patients with pulmonary tuberculosis and community-acquired pneumonia. These findings suggest that IDO plays an important role in the pathogenesis of lung diseases and can be considered as a new therapeutic target. 40 While IDO levels were higher in the MPM group; age, gender, and disease subtype did not change IDO levels. When the effect of smoking was examined, it was found that only IDO levels increased in smokers. Similar to IDO, Significantly higher serum neopterin concentrations were identified in the MPM group than in the control group in the present study. Consistent with our findings, Prakova et al. reported serum neopterin levels to be high in miners exposed to asbestos and coal dust. 41 Increased neopterin levels have also been reported in miners exposed to bauxite dust, 42 while another study has suggested that levels are increased in tuberculosis which may be associated with the stimulation of cellular immune response. 43 Workers exposed to dust containing free crystalline silicon dioxide were found to have higher neopterin levels compared to the control group. This supports the theory of cellular immune response and activation of alveolar macrophages in workers exposed to inorganic dust aerosols. 44 In a study evaluating changes in serum neopterin levels in lung toxicity caused by paraquat, it was shown that the increase in serum neopterin levels reflects the severity of tissue damage. 45
Periostin modulates transcripts involved in oxidative phosphorylation, oxidative stress, fatty acid metabolism, and TNF-a NF-kB signaling pathways. 46 Periostin combines with other extracellular matrix proteins to support the formation of collagen fibers, and has been associated with fibrotic diseases such as bronchial asthma, scleroderma and atopic dermatitis. 47 An autopsy of a patient with familial idiopathic pulmonary fibrosis, revealed periostin to be highly expressed in the lungs, especially in the fibrotic interstitium and bronchiolar epithelium, but not in the healthy lung tissue. At the time of presentation to the clinic, serum periostin levels were normal, and periostin accumulation was detected in the lungs during the autopsy. 48 In the clinic, periostin upregulation has been reported for many types of cancer, including MPM, and has been identified as a potential tumor-promoting factor. It has also been suggested that periostin expression in MPM cells may be an independent prognostic factor for overall survival. 13 Periostin-activated signaling pathways may increase cellular survival, angiogenesis, and resistance to cell death caused by hypoxia, indicating its potential as, aprognostic biomarker of cancer. In addition, because the epitope can be accessed via the bloodstream, it is considered a potential target for tumor therapy. 49 Periostin levels were found to be increased in patients with MPM compared to control. It has also been observed that there is a positive correlation between all biomarkers examined, and their levels increase as the age increases.
Conclusions
Tumor markers have important uses in the early and rapid determination of the disease and the diagnosis and may be useful in screening cases of asbestos-induced MPM. The biomarkers evaluated in the present study were found to be significantly higher in the patient group, and also showed a correlation among themselves. Serum periostin, TNC, neopterin, YKL-40 and IDO levels were significantly higher in patients with MPM than in those without cancer. These results may help identify new markers for diagnosing cancers.
Footnotes
Author contributions
The conception and design of the study; the acquisition, analysis, and interpretation of data for the work; drafting the work and revising it critically for important intellectual content; and final approval of the manuscript were done by SU and MI. The drafting of the study and its critical revision for important intellectual content was performed by AS, FM and MI. Analysis, and interpretation of data for the study were made by ZT and HY.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Approval was obtained from the Inonu University Malatya Clinical Research Ethics Committee (date 2018/number 145). The procedures used in this study are in line with the principles of the Declaration of Helsinki.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Inonu University Department of Scientific Research Projects (Project Number: TDK-2019/1512).
Informed consent
This study was authorized by the medical authorities of our hospital and patients were informed and signed their consent to their data collection.
