Abstract
The most widespread neoplasm of the pleura is malignant pleural mesothelioma (MPM) with low prevalence rate. The mechanistic target of rapamycin signaling pathway, inhibited by RAD001, was shown to be deregulated in MPM development and considered a novel target for the MPM therapy. The EF24, a curcumin analog, also affects several signaling pathways and kills cancer cells as a single agent or in combination with classical drugs. We aimed to evaluate possible effects of RAD001, EF24, cisplatin, and oxaliplatin treatments on both malignant pleural mesothelioma (MSTO-211H) and nonmalignant mesothelial (Met-5A) cell lines. The effects of the agents on MSTO-211H and Met-5A cells were evaluated in terms of cell viability, cytotoxicity, DNA synthesis rate, quantitation of apoptotic DNA fragmentation, and cleaved caspase 3 levels. Moreover, quantitative messenger RNA (mRNA) analysis of apoptotic (CASP9) and antiapoptotic (BCL2L1 and BCL2) genes were also performed. We found that both EF24 and RAD001 alone treatments decreased only MSTO-211H cell viability, but cisplatin and oxaliplatin affected both cell lines. Pretreatment with EF24 or RAD001 followed by cisplatin increased the effects of cisplatin alone application. EF24 and RAD001 pretreatment decreased DNA fragmentation rate when compared with cisplatin alone treatment in Met-5A cells. Sequential treatments resulted in a significant increase of CASP9 mRNA expression in MSTO-211H cells but not in Met-5A cells. Our preliminary results suggest that pretreatment with EF24 or RAD001 may reduce cytotoxic effect of cisplatin on nonmalignant mesothelial cells and increase cell death response of MPM cells. Further analyses using animal models are needed to confirm these findings in vivo.
Introduction
Malignant pleural mesothelioma (MPM) is the most common neoplasm of the pleura, but its incidence rate is very low. 1 This extremely lethal cancer has a poor prognosis and the median survival of MPM patients is around 1 year. 2 Due to the fact that single modality therapies (surgery, radiotherapy, and chemotherapy) have no impact on survival of MPM patients, multimodality therapy is suggested to treat patients with MPM. 3 The combined use of cisplatin (cis-diamminedichloroplatinum(II); CDDP) and pemetrexed is the current standard first-line chemotherapy in practice worldwide for MPM patients. Moreover, carboplatin can be used instead of cisplatin due to its diminished risk of toxicity. 4
As reviewed by Oh and Jacinto, the mechanistic target of rapamycin (mTOR) is a protein kinase that coordinates the regulation of cell growth and proliferation, cell cycle progression, and angiogenesis. 5 Due to the fact that elevated levels of pmTOR expression is displayed in malignant pleural mesothelioma cell lines 6 and MPM tumors, 7 mTOR inhibition is suggested to be one of the promising approaches for the development of novel strategy against MPM. 8 Moreover, mTOR is one of the regulators of autophagy induction and its inhibition with RAD001 may also cause induction of autophagic cell death. 9
Rapamycin, also known as sirolimus, was isolated from an Easter Island (Rapa Nui) soil bacterium in the early 1970s. 10 Till date, three well-studied derivatives of sirolimus are synthesized: temsirolimus (CCI-779), everolimus (RAD001), and deforolimus (AP23473). Rapamycin and its analogs are also known as rapamycins or rapalogs. 11 Temsirolimus and everolimus are approved by the Food and Drug Administration for use in renal cell carcinoma. Moreover, everolimus in combination with exemestane is also approved for use in breast cancer. 12 Pretreatment of RAD001 has also been shown to sensitize tumor cells to platinum-based chemical agents in vitro. 13 –16
It was previously shown that curcumin can inhibit cell growth, proliferation, angiogenesis, and metastasis. 17 Because of the suppression of mTOR-mediated molecular pathways by curcumin, Beevers et al. 18 suggested that curcumin may be grouped as a new class of mTOR inhibitor. Owing to curcumin’s poor absorption, rapid metabolism, and consequently its low bioavailability, researchers are trying to develop novel analogs with antineoplastic property. 19
Diphenyl difluoroketone (EF24) is one of the novel synthetic curcumin analogs. 20 The effects of EF24 alone or combination with classic chemotherapeutic has been displayed in a numerous human cancers, such as ovarian 21 –24 colon, 25 prostate, 26 breast, 22,27 and lung. 28 The effects of EF24 on mesothelial and mesothelioma cells has not been studied so far and thus we also want to explore this situation.
In the present study, we aimed to find out whether, RAD001 and EF24 pretreatment alone and combination with cisplatin or/and oxaliplatin can induce apoptotic pathways in both malignant pleural mesothelioma (MSTO-211H) and nonmalignant mesothelial (Met-5A) cell lines.
Materials and methods
Cell culture
Cell lines (MSTO-211H (biphasic human MPM) and Met-5A (nonmalignant human pleural mesothelioma)) were purchased from the American Type Culture Collection (Manassas, Virginia, USA). MSTO-211H cells were grown in Roswell Park Memorial Institute-1640 medium, and Met-5A cells were cultured in Dulbecco’s modified eagle’s medium/F12 medium. Both media were supplemented with 10% fetal bovine serum (10% FBS), 2 mM
Cell viability test
Cell viability rate was determined after sequential administration of EF24 (Sigma Aldrich, St Louis, Missouri, USA), everolimus (RAD001; Fluka, Hamburg, Germany), oxaliplatin (1-OHP; Tocris Bioscience, Bristol, United Kingdom), and cisplatin (CDDP; Sigma Aldrich, Missouri, USA) by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Stock solutions of EF24 and RAD001 were prepared in dimethyl sulfoxide (DMSO) at various concentrations. CDDP and 1-OHP were dissolved in distilled water. The final concentration of DMSO was <0.1%. The cells were seeded on a 96-well culture plate containing 2 × 10 4 cells/well for MSTO-211H and Met-5A cells. Both cell lines were administered EF24 (0.5–32 µM), RAD001 (1–300 nM), CDDP (1–100 µM), and 1-OHP (1–100 µM), and dose-dependent effects of the chemical agents on the cells were defined after 24 h. Since it was previously shown that 20 μM cisplatin induced apoptosis in MSTO-211H cells, we decided to use this concentration for the experiments in this study. 29 It was shown that cisplatin is inactivated by DMSO. 30 Because EF24 and everolimus were dissolved in DMSO, we sequentially administrated the drugs. The cells were treated with selected doses of EF24 (1–16 µM) or RAD001 (1–200 nM) for 24 h and fixed concentrations of CDDP (20 µM) and 1-OHP (80 µM) were administered for an additional 24 h. As a result of MTT assay data, 2 µM EF24 and 200 nM RAD001 concentrations were selected for further analysis. After the incubation period, 10 µl MTT solutions (5 mg/ml) was added to the medium and kept at 37°C for 4 h. DMSO was used for dissolving formazan crystals, and the absorbance values were measured at 570 nm using Spectramax M3 microplate reader (Molecular Devices, Silicon Valley, California, USA). All experiments were performed in quadruplicate for each dose, and the mean absorbance values were calculated.
Cytotoxicity assay
As a marker of cytotoxicity, lactate dehydrogenase (LDH) release into culture media was measured with Cytotoxicity Detection KitPlus (Roche Applied Science, Mannheim, Germany). Cells were seeded at a density of 2 × 10 4 cells/well in 96-well plates. Single-agent treatments were performed for 24 h. For the sequential administration, cells were treated with selected doses of EF24 and RAD001 for 24 h, following fixed concentration of CDDP and 1-OHP for additional 24 h.
After treatment of the cells with agents at various concentrations, kit protocol was performed as described by the supplier. Optical density (OD) was measured at 490 nm using Spectramax M3 microplate reader (Molecular Devices). The results were expressed as percentage cytotoxicity relative to the untreated control cells.
Cell proliferation assay
To determine the effects of the agents on DNA synthesis rate, a commercially available kit (Cell Proliferation ELISA, BrdU colorimetric, Roche Applied Science, Mannheim, Germany) was used following the instructions of the manufacturer. MSTO-211H and MET-5A cells were seeded in 96-well culture plates at a density of 2 × 10 4 cells/well and treated with selected doses of EF24 and RAD001 for 24 h, following fixed concentration of CDDP and 1-OHP for additional 24 h. Single agent treatments were performed for 24 h. At the end of the incubation period, BrdU was added to each well and incubated for 2 h. Absorbance of each sample was measured at 450 nm using Spectramax M3 microplate reader (Molecular Devices).
Quantitation of DNA fragmentation
To quantitate DNA fragmentation as a marker for apoptosis, The Cell Death Detection ELISAPlus kit (Roche Applied Science) was used according to the guidelines of the manufacturer. MSTO-211H and Met-5A cells were plated into 96-well plates (2 × 10 4 cells/well). Single-agent treatments were performed for 24 h. After pretreatment with EF24 and RAD001 for 24 h, fixed concentration of CDDP and 1-OHP was added to the wells followed by 24 h incubation. OD was measured at 405 nm with Spectramax M3 microplate reader (Molecular Devices).
Detection of cleaved caspase 3 level
To verify the presence of apoptosis in agent-treated cells, cleaved caspase 3 levels were detected using the PathScan Cleaved Caspase-3 (Asp175) Sandwich ELISA kit (Cell Signaling, Beverly, Massachusetts, USA). Single-agent treatments were performed for 24 h. For the sequential administration, cells treated with selected doses of EF24 and RAD001 for 24 h, following fixed concentration of CDDP and 1-OHP for additional 24 h. After various treatments of agents, cell lysates were obtained. Then, protein content of the lysates was determined using the bicinchoninic acid (BCA) protein assay kit from Pierce Chemical (Pierce, Rockford, Illinois, USA). First, 100 µl lysates, containing 30 µg total protein, were transferred to microplates coated with total caspase 3 antibody. Second, biotinylated-cleaved caspase 3 antibody was added to the wells. Third, horseradish peroxidase (HRP)-linked streptavidin and ultimately HRP substrate were transferred. OD was measured at 450 nm with Spectramax M3 microplate reader (Molecular Devices).
Quantitative mRNA analysis
Total RNA was isolated using High Pure™ RNA Isolation Kit (Roche Applied Science) according to the manufacturer’s recommendations. RNA quantitation was determined using the Nanodrop spectrophotometer (NanoDrop ND-1000, Montchanin, Delaware, USA). Complementary DNA (cDNA) synthesis was performed using Transcriptor First Strand cDNA synthesis kit (Roche Applied Science) according to the supplier’s guidelines. cDNA reaction was carried out in an Eppendorf Mastercycler ep gradient S thermal cycler (Eppendorf, Hamburg, Germany).
Gene-specific intron spanning primers and probes were designed for the transcripts of the BCL2L1, BCL2, and CASP9 genes with the Universal Probe Library (UPL) Assay Design Center (https://www.universalprobelibrary.com). The sequences of the primers and UPL probe numbers are shown in Table 1. Crossing point values were determined using Light Cycler®480 (Roche Diagnostics GmbH). The quantitative polymerase chain reaction (qPCR) conditions were as follows: 50 cycles, 95°C for 15 s and 60°C for 20 s and ultimately, the samples were cooled to 40°C. The mRNA level of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene was used to normalize expression levels of genes of interest. All qPCR reactions were repeated three times.
Forward and reverse primer sequences and UPL probe numbers used in qPCR.
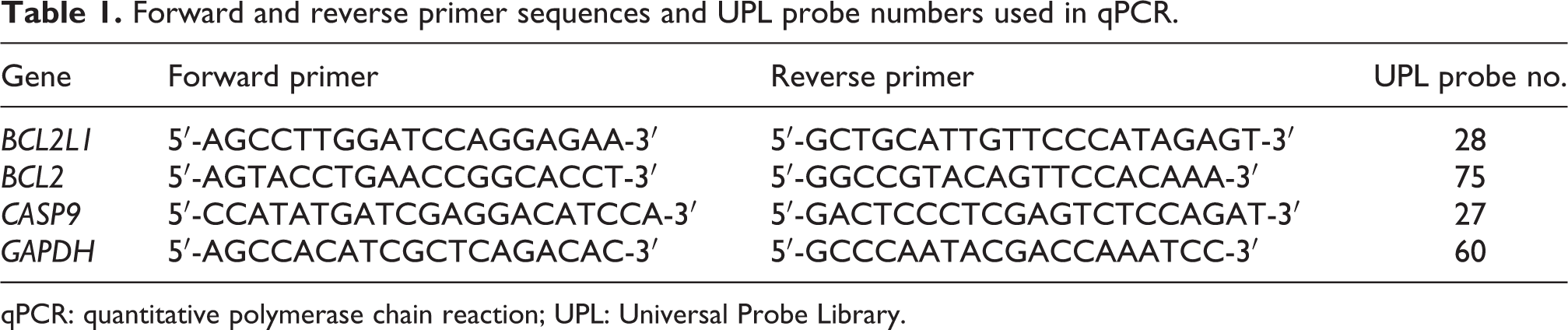
qPCR: quantitative polymerase chain reaction; UPL: Universal Probe Library.
Statistical analysis
mRNA expression levels of BCL2L1, BCL2 and CASP9 were compared through “REST (2009 V2.0.13)” software using “Pair-wise Fixed Reallocation Randomization” statistical analysis test. 31 Other parameters were compared using one-way analysis of variance test by SigmaStat v3.5. The values of p < 0.05 were considered as statistically significant. Single-agent treatments were compared with untreated control cells. The control group of sequential treatments was C20 for E2/C20 and R200/C20 combinations. The E2/O80 and R200/O80 sequential treatments were compared with O80 group.
Results
Growth of the MSTO-211H cells were only inhibited at concentrations of EF24 higher than 2 µM, whereas these decreases were not observed in Met-5A cells after 24 h incubation (Figure 1(a)). Similarly, RAD001 alone treatment decreased the cell viability of MSTO-211H but Met-5A cells continued proliferation (Figure 1(b)). Both cisplatin alone and oxaliplatin alone slightly diminished the cell viability of MSTO-211H and Met-5A cells (Figure 1(c) and (d)). EF24 and RAD001 pretreatment followed by 20 µM CDDP reduced cell growth of MSTO-211H and Met-5A more than single-agent alone treatment. However, EF24 and RAD001 did not sensitize both cells to 1-OHP (Figure 1(e) and (f)).

Cell viability rates after (a) EF24, (b) RAD001, (c) CDDP, (d) (1-OHP) incubation of the MSTO-211H and Met-5A cells. (e) Effect of EF24, CDDP, 1-OHP alone treatment, and sequential administration of EF24 and CDDP and/or 1-OHP in both studied cells. (f) Effect of RAD001, CDDP, 1-OHP alone treatment, and sequential administration of RAD001 and CDDP and/or 1-OHP in both studied cells. † p < 0.05 compared with control; *p < 0.05 compared with CDDP. CDDP: cisplatin; 1-OHP: oxaliplatin. C20: 20 µM cisplatin; 080:80 µM oxaliplatin; E2:2 µM EF24; R200:200 nm RAD001.
As shown in Figure 2, EF24 administration was more effective in MSTO-211H cells than MET-5A. EF24 pretreatment apparently diminished the cytotoxic effect of CDDP and 1-OHP in Met-5A cells ( p < 0.05; Figure 2(a)). RAD001 pretreatment resulted in downregulation of cytotoxic effects of 1-OHP in both cell lines ( p < 0.05). Moreover, after RAD001 administration for 24 h, we detected the downregulation of the cytotoxicity of CDDP in Met-5A ( p < 0.05), whereas this effect elevated in MSTO-211H cells (Figure 2(b)).

(a) Cytotoxic effects of selected doses of EF24, CDDP, 1-OHP, and sequential administration of EF24 and CDDP and/or 1-OHP in both studied cells. (b) Cytotoxic effects of selected doses of RAD001, CDDP, 1-OHP, and sequential administration of RAD001 and CDDP and/or 1-OHP in both studied cells. Single-agent treatments were compared with untreated control cells. The E2/C20 and R200/C20 sequential treatments were compared to C20 group and the E2/O80 and R200/O80 sequential treatments were compared to O80 group. *p < 0.05 compared with CDDP; ‡ p < 0.05 compared with 1-OHP. CDDP: cisplatin; 1-OHP: oxaliplatin. C20: 20 µM cisplatin; 080:80 µM oxaliplatin; E2:2 µM EF24; R200:200 nm RAD001.
As depicted in Figure 3, all selected treatments significantly decreased the proliferation of both cells when compared with the control cells ( p < 0.05). EF24 pretreatment elevated DNA synthesis in MSTO-211H cells as compared to cisplatin alone administration. When correspond to 1-OHP treatment with EF24 + 1-OHP, replication was upregulated in MET-5A cells ( p < 0.05; Figure 3(a)). When corresponded to control cells, all selected treatments strikingly diminished the DNA synthesis rate in MSTO-211H cells ( p < 0.05). Similarly, replication rate was also decreased in all treatments ( p < 0.05) except RAD001 alone treatments in Met-5A. Sequential administration of RAD001 and CDDP in MST0-211H, DNA replication rate was increased as compared to CDDP alone administration ( p < 0.05; Figure 3(b)).

(a) DNA synthesis rate after treatment of selected doses of EF24, CDDP, 1-OHP, and sequential administration of EF24 and CDDP and/or 1-OHP in both studied cells. (b) DNA synthesis rate after treatment of selected doses of RAD001, CDDP, 1-OHP, and sequential administration of RAD001 and CDDP and/or 1-OHP in both studied cells. Single-agent treatments were compared to untreated control cells. The E2/C20 and R200/C20 sequential treatments were compared with C20 group and the E2/O80 and R200/O80 sequential treatments were compared with O80 group. † p < 0.05 compared with control; *p < 0.05 compared with CDDP; ‡ p < 0.05 compared with 1-OHP. CDDP: cisplatin; 1-OHP: oxaliplatin. C20: 20 µM cisplatin; 080:80 µM oxaliplatin; E2:2 µM EF24; R200:200 nm RAD001.
1-OHP alone, EF24 + CDDP, and EF-24 + 1-OHP administration resulted in an increased DNA fragmentation rate in MSTO-211H ( p < 0.05). When compared with the control cells, EF24, CDDP, 1-OHP alone treatment elevated DNA fragmentation rate in Met-5A ( p < 0.05). Compared with CDDP alone treatment, EF24 pretreatment overwhelmingly reduced DNA fragmentation rate in Met-5A cells ( p < 0.05). Similarly, EF24 + 1-OHP administration decreased the effect of 1-OHP alone in Met-5A ( p < 0.05) However, EF24 + 1-OHP treatment increased the effect of 1-OHP alone in MSTO-211H cell line ( p < 0.05) (Figure 4(a)). Compared with the control cells, RAD001 pretreatment and oxaliplatin alone administration resulted in an increased DNA fragmentation rate in MSTO-211H ( p < 0.05). When compared with control cells, CDDP and 1-OHP treatment alone elevated DNA fragmentation rate in Met-5A cells ( p < 0.05). RAD001 significantly sensitized in MSTO-211H cells to CDDP treatment ( p < 0.05). On the contrary, RAD001 pretreatment resulted in a diminished DNA fragmentation rate compared to CDDP and/or 1-OHP alone treatment in Met-5A cells ( p < 0.05; Figure 4(b)).

(a) DNA fragmentation rate after treatment of selected doses of EF24, CDDP, 1-OHP, and sequential administration of EF24 and CDDP and/or 1-OHP in both studied cells. (b) DNA fragmentation rate after treatment of selected doses of RAD001, CDDP, 1-OHP, and sequential administration of RAD001 and CDDP and/or 1-OHP in both studied cells. Single-agent treatments were compared to untreated control cells. The E2/C20 and R200/C20 sequential treatments were compared to C20 group and the E2/O80 and R200/O80 sequential treatments were compared to O80 group. † p < 0.05 compared with control; *p < 0.05 compared with CDDP; ‡ p < 0.05 compared with 1-OHP. CDDP: cisplatin; 1-OHP: oxaliplatin. C20: 20 µM cisplatin; 080: 80 µM oxaliplatin; E2: 2 µM EF24; R200: 200 nm RAD001.
When corresponded to control cells, 1-OHP, EF24 + 1-OHP, and RAD001 + 1-OHP administration increased the level of active caspase 3 levels in MSTO-211H ( p < 0.05). However, CDDP, 1-OHP, RAD001, and EF24 alone treatments did not significantly change cleaved caspase-3 level in MSTO-211H cells ( p > 0.05). Sequential administration of EF24 and 1-OHP in MST0-211H, strikingly elevated active caspase 3 levels as compared to 1-OHP alone ( p < 0.05; Figure 5).
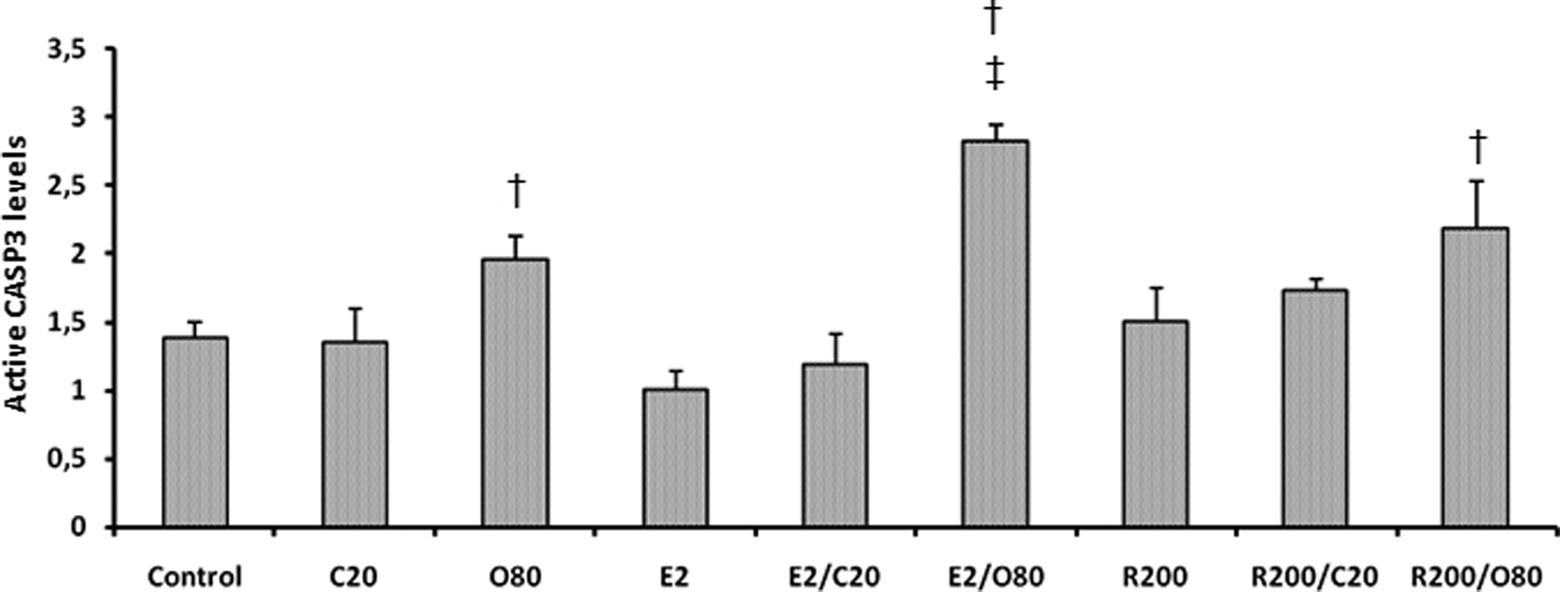
Cleaved caspase-3 protein levels of each agent alone treatment and pretreatment of EF24 and/or RAD001, followed by CDDP and/or 1-OHP in MSTO-211H cells. Single agent treatments were compared to untreated control cells. The E2/C20 and R200/C20 sequential treatments were compared to C20 group and the E2/O80 and R200/O80 sequential treatments were compared to O80 group. † p < 0.05 compared with control; ‡ p < 0.05 compared with 1-OHP. CDDP: cisplatin; 1-OHP: oxaliplatin. C20: 20 µM cisplatin; 080: 80 µM oxaliplatin; E2: 2 µM EF24; R200: 200 nm RAD001.
In MSTO-211H cells, CDDP, 1-OHP, and RAD001 alone treatments strikingly decreased the expression levels of both antiapoptotic BCL2L1 and BCL2 genes when compared with the control cells ( p < 0.05). Yet CASP9 mRNA expression level is not affected. EF24 alone treatment resulted in downregulation of BCL2L1 expression as compared to the control cells ( p < 0.001). In MSTO-211H cells, EF24 pretreatments followed by CDDP and 1-OHP overwhelmingly diminished the mRNA expression of both antiapoptotic BCL2L1 and BCL2 genes when compared with the control cells ( p < 0.05). Moreover, RAD001 pretreatments followed by CDDP and 1-OHP elevated the expression level of CASP9 gene when corresponded to control cells ( p < 0.05). RAD001 + 1OHP treatment resulted in downregulation of BCL2L1 and BCL2 expression as compared to the control cells ( p < 0.05). Compared with the CDDP alone treatment, EF24 pretreatment increased the CASP9 expression by approximately twofold in MSTO-211H cells ( p = 0.002). Moreover, EF24 + 1-OHP administration resulted in decreased BCL2L1 and increased CASP9 expression levels as compared to 1-OHP alone treatment ( p < 0.05). RAD001 + CDDP administration was resulted in upregulation of the CASP9 expression (2.22-fold) level compared to CDDP alone treatment ( p < 0.001). Sequential administration of RAD001 and 1-OHP when compared with 1-OHP alone treatment, mRNA expression level of CASP9 gene was found to increase by 4.7-fold ( p < 0.001; Figure 6(a)).

(a) Comparison of the mRNA expression levels of BCL2L1, BCL2, and CASP9 genes in MSTO-211H cells. (b) Comparison of the mRNA expression levels of BCL2L1, BCL2, and CASP9 genes in MET-5A cells. Expression levels of the target genes were normalized to the mRNA expression level of GAPDH. Single-agent treatments were compared with untreated control cells. The E2/C20 and R200/C20 sequential treatments were compared with C20 group and the E2/O80 and R200/O80 sequential treatments were compared with O80 group. † p < 0.05: compared with control; *p < 0.05: compared with CDDP; ‡ p < 0.05: compared with 1-OHP. CDDP: cisplatin; 1-OHP: oxaliplatin. C20: 20 µM cisplatin; 080: 80 µM oxaliplatin; E2: 2 µM EF24; R200: 200 nm RAD001. mRNA: messenger RNA; MSTO-211H: malignant pleural mesothelioma cell line; MET-5A: nonmalignant mesothelial cell line; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.
Compared with the control cells, CDDP and 1-OHP treatment alone diminished the expression level of both BCL2L1 and BCL2 approximately by twofold in MET-5A cells ( p < 0.05). Whereas the administration of EF24 and RAD001 alone in MET-5A cells, the expression levels of studied genes did not change. When corresponded to CDDP treatment with EF24 + CDDP, it was observed that mRNA expression level of BCL2L1 gene was elevated by about twofold ( p < 0.05). Yet RAD001 pretreatment did not significantly change the expression level of studied genes compared to CDDP and/or 1-OHP alone treatment in Met-5A cells (Figure 6(b)).
Discussion
In this study, we showed that inhibition of mTOR signaling pathway by RAD001 produced significant antiproliferative response in MSTO-211H cells rather than nonmalignant mesothelial (Met-5A) cells. The antiproliferative effects of mTOR inhibition have also been shown in other solid tumors. From this point of view, pretreatment of A549 lung carcinoma cells with RAD001 was found to result in enhanced cisplatin-induced apoptosis by suppression of p53-activated p21 expression. 13 Moreover, Mabuchi et al. 14 displayed that pretreatment with RAD001 sensitized ovarian cancer cells to cisplatin in AKT/mTOR-dependent manner. In addition, RAD001 has an additive-to-synergetic effect on antitumor activity of cisplatin in both cisplatin-resistant and sensitive nasopharyngeal carcinoma cell lines. 15 Hirashima et al. 16 displayed that RAD001 increased in an additive manner the effect of cisplatin in human esophageal squamous cell carcinoma cell lines. Furthermore, it was demonstrated that RAD001 pretreatment of the cells before oxaliplatin was effective only at high concentrations of oxaliplatin in cholangiocarcinoma cells. 32
Several studies showed the fundamental importance of PI3K/Akt/mTOR pathway in MPM cells, and inhibition of mTOR signaling showed synergistic effects with chemotherapeutics. For example, hyperactivation of the PI3K/Akt/mTOR pathway, especially overexpression of mTOR, found to contribute to acquisition of apoptosis resistance in MPM spheroids. 33 With respect to dual drug treatment, Hoda et al. 8 indicated synergistic antiproliferative effect of temsirolimus and cisplatin in six human MPM cell lines. Temsirolimus, also, affected cisplatin-resistant MPM cells more than cisplatin sensitive cells. Moreover, temsirolimus in combination with cisplatin exerted more survival rate and reduction in tumor mass in mice xenografted MPM cell line. Elevated mTOR expression in primary cells from 15 human MPM patients grown in culture as spheroids was also detected. In these cells, rapamycin diminished gemcitabine resistance and stimulated apoptotic cell death. 34 In another study, the combined treatment with sirolimus and cisplatin increased cell death in four MPM cell lines, as compared to either drug alone administration. 35 In concert with these previous findings, our results also showed that sequential administration of RAD001 with CDDP and 1-OHP increased the antiproliferative effect of CDDP and 1-OHP as supported by MTT and LDH assays. While RAD001 pretreatment before CDDP increased the cytotoxic response on MSTO-211H cell, nonmalignant mesothelial cells (Met-5A) were protected from damaging effects of CDDP and 1-OHP by this sequential treatment. Basically, the protective effect may be resulted from the differences between MSTO-211H and Met-5A cells with regard to metabolic activity, growth rate, DNA synthesis rate, and any other unknown mechanisms. Further analyses are required to uncover this situation.
The several anticancer effects of EF24 were also evaluated in some tumors originating from different tissues including ovarian 21,23,24 , colon, gastric, 25 skin, and prostate cancers. 26 According to the current literature search, we were unable to find any study investigating the effects of EF24 on MPM tumors both in vivo and in vitro. However, there were two studies assessing the effect of curcumin rather than EF24 in MPM. In one of them, it was shown that although curcumin alone treatment diminished MPM cell growth in both dose- and time-dependent manners, curcumin pretreatment followed by cisplatin overwhelmingly elevated the activity of cisplatin. Additionally, oral administration of curcumin also inhibited the growth of the MPM cell-derived xenograft growth in vivo. Moreover, apoptosis-promoting effect of curcumin was also shown both in vitro and in vivo analyses. 36 In another study, Yamauchi et al. 37 showed that curcumin induced autophagic cell death instead of apoptosis in human malignant pleural mesothelioma cell line (ACC-MESO-1). Our results also showed that sequential administration of EF24 with CDDP increased the antiproliferative effect of CDDP. While EF24 treatment followed by CDDP and 1-OHP increased the LDH-releasing effect on MSTO-211H cells, nonmalignant mesothelial cells (Met-5A) were partly kept safe from damaging effects of CDDP and 1-OHP by this successive treatment. Basically, the protective effect may result from differences between MSTO-211H and Met-5A cells with regard to metabolic activity, growth rate, DNA synthesis rate, and any other unknown mechanisms.
As a conventional marker for apoptosis, we also analyzed cleaved caspase 3 levels and DNA fragmentation rate. Our findings showed a significant increase in cleaved caspase 3 levels and DNA fragmentation ratio after sequential treatment of EF24 and 1-OHP in MSTO-211H cells. Similar results were also found in ovarian carcinoma cells, and EF24-promoted caspase-mediated cell death was also shown. 23
Moreover, a significant increase in CASP9 mRNA was determined after sequential treatments in MSTO-211H cells. Furthermore, downregulation of antiapoptotic BCL2L1 and BCL2 mRNA expression may also support activation of programmed cell death. In relation to the presence of apoptotic markers in EF24 + 1-OHP-treated cells, our results were correlated with those of Wang et al., 36 EF24 may also show a similar apoptotic effect to curcumin.
Although we found an increase in DNA synthesis in MSTO-211H cells after EF24 + CDDP treatment as compared to CDDP alone, this effect may be attributed to the effect of EF24 on cell cycle progression. For example, EF24 was shown to accumulate ovarian cancer cells in G 2/M phase. 21 Moreover, in colon and gastric cancer lines, Subramaniam et al. 25 showed that EF24 suppresses cell proliferation and colony formation and induces G 2/M arrest. Also, another study displayed that curcumin alone or combination with cisplatin treatment resulted in S and G 2/M phase arrest in mesothelioma cancer cells. 36 In relation to EF24 and CDDP administration, by allowing cancer cells to enter S phase, EF24 may enhance the interaction of CDDP to DNA strands and result in more cell killing response than CDDP alone. According to our findings, RAD001 also affected MSTO-211H cells like EF24 with regard to the DNA replication with unknown mechanisms.
In conclusion, our preliminary results showed that pretreatment with EF24 or RAD001 enhanced the effects of platinum-based drugs, diminished cytotoxic effect of these agents on nonmalignant mesothelial cells, and also increased cell death response of MPM cells. As in vitro studies can’t replicate in vivo conditions and may not exactly reflect the tumor environment, further studies using animal models are needed to reveal the mechanisms of effect of EF24 and RAD001 responses in MPM.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
This study was financially supported by Gazi University Research Foundation (Project code no. 01/2010-23).
