Abstract
Colistin is an antimicrobial agent that is used in resistant gram-negative infections. Its most common dose-limiting adverse effect is nephrotoxicity. The objective of our study was to explore the possible effects of each of taxifolin and dapagliflozin alone and in combination on colistin-induced nephrotoxicity in rats. Sixty male rats were randomized into six groups: Control; colistin; colistin + taxifolin; colistin + dapagliflozin; colistin + carboxymethyl cellulose (CMC) and colistin + taxifolin + dapagliflozin. Dapagliflozin, taxifolin, and CMC were given daily for 7 days, 4 hours before colistin injection. Kidney weight/body weight ratio and renal function tests were determined. Renal tissue nerve growth factor-β (NGF-β), transforming growth factor beta 1 (TGF-β1), proinflammatory cytokines, nuclear factor (erythroid-derived 2)-like 2 (Nrf2), toll-like receptor 4 (TLR4), nuclear factor kappa B (NF-κB) p65, signal transducer and activator of transcription 3 (STAT3), oxidative stress parameters, beclin-1 and c-Jun NH2-terminal kinase (JNK) activities were measured. Kidneys were examined histopathologically and immunohistochemically. Taxifolin and/or dapagliflozin induced significant improvement in the renal functions and oxidative stress parameters with significant increase in tissue Nrf2, STAT3 and NGF-β accompanied with significant decrease in kidney weight/body weight ratio, tissue proinflammatory cytokines, TGF-β1, NF-κB (p65), TLR4, beclin-1 and JNK activities and improved the histopathological picture when compared to rats treated with colistin alone. This improvement was significant with taxifolin/dapagliflozin combination compared to rats treated with each of these agents alone. So, we concluded that the combined use of taxifolin and dapagliflozin may confer a therapeutic tool for attenuation of colistin-induced nephrotoxicity.
Introduction
Colistin, also known as “Polymyxin B,” is an antimicrobial agent that plays an important role in treatment of resistant gram-negative bacterial infections. 1 It represents a cornerstone in the most effective antibiotic regimens against infections caused by antibiotic-resistant micro-organisms. 2 However, the high incidence of nephrotoxicity even with the therapeutic doses represents an important obstacle against the use of colistin in management of bacterial infection. 3 Several studies were carried out to clarify the exact mechanisms that underlie this nephrotoxic effect. Most reports indicated that oxidative damage caused by reactive oxygen species (ROS) and affection of apoptosis are incriminated in the pathogenesis of colistin-induced renal damage. 4 In addition, colistin was proven to increase the expression of the different mediators involved in the inflammatory processes and inhibits the activity of the anti-inflammatory mediators with subsequent aggravation of the damage to the renal tissues. 5
Dapagliflozin is a drug that decreases glucose reabsorption in the kidney at a lower threshold via inhibition of sodium–glucose cotransporter 2 (SGLT2) in the distal convoluted tubules. 6 This effect increases glucose excretion in urine leading to renal glucosuria with subsequent reduction of blood glucose levels. 7 Phase II and III clinical trials had reported that dapagliflozin can be considered as a safe and an effective agent for treatment of non-insulin dependent diabetes mellitus. 8 Recent reports indicated that dapagliflozin has worthy antioxidant and anti-inflammatory properties that may have a positive impact on the pathogenic pathways of various types of human diseases. 9 Also, dapagliflozin was reported to affect a wide range of the mediators involved in autophagy and apoptosis. 10 In addition, dapagliflozin was found to counteract the toxic effects by which antibiotics may damage the renal tissues. 11 Taken together, these effects may confer a potential role for dapagliflozin in amelioration of colistin-induced nephrotoxicity.
Flavonoids are polyphenolic compounds that are present in considerable amounts in most fruits and vegetables. 12 They attract attention of researchers for years, owing to their biological and pharmacological characteristics that may aid in management of a wide variety of human diseases. 13 Taxifolin, also known as dihydroquercetin, is one of the flavonoids that is especially found in fresh fruits, green leafy vegetables, cocoa, and tea. 14 Recent reports had shown that the pharmacological activities of taxifolin may contribute to the management of diseases in which inflammation, apoptosis and oxidative stress play a mechanistic role. 15 Also, taxifolin was proven to interact with the regulators of cell cycle resulting in cell cycle arrest which may lead to regression of growth of the malignant tumors. 16 Taxifolin was reported to reach sufficient concentrations in the renal tissues that may protect the renal cells against oxidative damage. 17 In addition, taxifolin was proven to inhibit the expression of p-glycoprotein that is responsible for the renal uptake of many nephrotoxic agents.18,19 The purpose of this work was to explore the effects of each of taxifolin and dapagliflozin alone and in combination on colistin-induced nephrotoxicity in rats and to assess the possible mechanisms that may underlie these effects.
Methods and materials
Drugs and reagents used
Colistin sulfate (≥19,000 IU/mg) was obtained from Sigma-Aldrich Co., St. Louis, Missouri, USA (CAS # 1264-72-8). Taxifolin was purchased from Med Chem Express, NJ 08852, USA (CAS # 480-18-2). Dapagliflozin was obtained from Santa Cruz Biotechnology, Inc. (CAS # 461432-26-8). Carboxymethyl cellulose (CMC) was purchased from Dalian Richfortune Chemicals Co., Ltd (CAS # 9004-32-4). All other reagents and chemicals were obtained from Sigma-Aldrich Co., St. Louis, Missouri, USA. Colistin was dissolved in 0.9% sodium chloride solution. Taxifolin and dapagliflozin were suspended in 1% CMC solution.
Animals and groups
In the present study, 60 male adult mature Wistar rats aged 8 weeks weighing about 180–230 grams were used. Rats were housed in special cages at a constant temperature of 24 ± 3°C with relative humidity of 53 ± 7% and with exposure to 12 h light/dark cycle. Animals were given the standard diet and distilled water provided ad libitum. Diet constituents include crude protein (18.4%), crude fat (4.35%), crude fibers (3.15%), calcium (1.1%), phosphorus (0.5%), moisture (6%), total ash, and carbohydrates (62%). Animals were allowed to acclimatize for 2 weeks. All experiments were in compliance with the ARRIVE guidelines and were performed in accordance with the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines, EU Directive 2010/63/EU for animal experiments. Rats were randomly grouped into six groups of 10 rats each as follows: (a) Control rats, were injected intraperitoneally with 0.9% sodium chloride solution daily for 7 days; (b) Colistin group, was injected intraperitoneally with colistin sulfate in a dose of 480,000 IU/kg/day (0.5–1 ml) for 7 days 1 ; (c) colistin + taxifolin group, received taxifolin by oral gavage in a dose of 50 mg/kg (1–1.5 ml) daily 20 ; (d) colistin + dapagliflozin group, received dapagliflozin by oral gavage in a dose of 1 mg/kg (1–1.5 ml) daily 21 ; (e) colistin + CMC group, received 1% CMC solution by oral gavage (1–1.5 ml) daily; f) colistin + taxifolin + dapagliflozin group, received dapagliflozin (1 mg/kg) in combination with taxifolin (50 mg/kg) by oral gavage. Taxifolin, dapagliflozin, and CMC were administered daily for 7 days, 4 hours before colistin injection.
Determination of the renal function tests
After 7 days from starting colistin injection, animals were kept in metabolic cages for collection of urine for 24 hours. The obtained urine was centrifuged at 1500 rpm for 5 minutes and the supernatant was used for assay of the biochemical parameters. Twenty-four hours urinary protein levels were estimated according to Fujita et al. 22 Urinary albumin excretion rate (UAER) was assessed by multiplying total urine volume in 24 hours (L) by urinary protein levels (mg/l).
Urinary N-acetyl-β-d-glucosaminidase (NAG) activity was measured according to Jepson et al. 23 Urinary gamma glutamyl transpeptidase (GGT) activity was determined according to Mancinelli et al. 24 Urinary creatinine was determined according to Owen et al. 25 After that, animals were fasted overnight and weighed. Then, they were anaesthetized with intraperitoneal injection of thiopental sodium (Sigma-Aldrich Co., St. Louis, Missouri, USA) in a dose of 40 mg/kg body weight. 26 Blood was withdrawn from the retro-orbital plexus using capillary tubes and centrifuged at 3000 rpm for 15 minutes. The resulting serum was used for determination of serum creatinine according to Henry 27 and blood urea according to Coulombe and Favreau. 28 Also, creatinine clearance was determined using the following formula 29 :
Both kidneys were excised and weighed for determination of the kidney weight/body weight ratio. Then, slices from the left kidney were homogenized in 0.1 M sodium phosphate buffer of pH 7.4 in a ratio of 1 g tissue to 4 ml buffer in a potter homogenizer. Then, the resulting homogenate was centrifuged at 4000 rpm for 20 minutes at 4°C. 30 The supernatant was analyzed for determination of the biochemical parameters. Specimens from the right kidney were subjected to histopathological and immunohistochemical examination.
Determination of renal tissue oxidative stress parameters and nuclear factor (erythroid-derived 2)-like 2 (Nrf2) content
Tissue glutathione reductase (GR) and glutathione peroxidase (GPx) were assessed using kits supplied by BioVision, Inc., Milpitas, California, USA (CAT # E4625 and K762 respectively) according to the manufacturer’s protocol. Assessment of tissue malondialdehyde (MDA) was carried out according to Ohkawa et al. 31 Determination of tissue Nrf2 content was performed using kits purchased from CUSABIO Technology LLC, Houston, TX, USA (CAT # CSB-EQ027869RA) according to the instructions of the manufacturer.
Assessment of transforming growth factor beta 1 (TGF-β1), interleukin 15 (IL-15), tumor necrosis factor alpha (TNF-α), toll-like receptor 4 (TLR4), nuclear factor kappa B (NF-κB) p65 and signal transducer and activator of transcription 3 (STAT3) in the renal tissues
Assessment of tissue TGF-β1 was performed using kits obtained from Abcam, Cambridge, MA, USA (CAT # ab119558) according to the manufacturer’s protocol. Determination of tissue IL-15 was carried out using ELISA kits purchased from Novus Biologicals, LLC, Centennial CO, USA (CAT # NBP3-06798) according to the instructions of the manufacturer. Rat ELISA kits obtained from RayBiotech, Inc., USA (CAT # ELR-TNFa-1) were used for assessment of TNF-α according to the manufacturer’s protocol. Rat TLR4 was assessed using ELISA kits supplied by MyBioSource, Inc., San Diego, CA, USA (CAT # MBS705488) according to the manufacturer’s instructions. ELISA kits purchased from Abcam, Cambridge, MA, USA were used for assay of NF-κB (p65) and STAT3 (CAT # ab176648 and ab126459 respectively) according to the manufacturer’s guide.
Determination of caspase 3 activity and nerve growth factor beta (NGF-β) in the renal tissues
Specimens of the renal tissues were prepared by homogenization and caspase-3 activity was determined colorimetrically according to Gurtu et al. 32 NGF-β was assessed using kits supplied by MyBioSource, Inc., San Diego, CA, USA (CAT # MBS824760) according to the manufacturer’s instructions.
Assessment of the c-Jun NH2-terminal kinase (JNK) activity and the levels of beclin-1 in the renal tissues
Determination of JNK activity and beclin-1 levels in the renal tissues was executed by using ELISA kits obtained from RayBiotech, Peachtree Corners, GA, USA (CAT # PEL-JNK-T183-2 and PEL-BECLIN1-S234-1 respectively) according to the manufacturer’s guide.
Histopathological examination
Specimens from the right kidney were fixed in 10% neutral buffered formalin and embedded in paraffin blocks. Then, 5–7 µm thickness sections were cut, stained with hematoxylin and eosin (H & E) and examined under light microscope. A histological score for assessment of the extent of acute tubular necrosis was performed by examination of 10 fields of the outer strip of the renal cortex and medulla under 200× magnification. The percentages of the tubules showing cell necrosis, cast formation, loss of the brush border or tubular dilation was counted and graded as follows: 0 means no changes, 1 means more than 10% of the tubules showed one or more of these changes, 2 means 11–25% of the tubules showed one or more of these changes, 3 means one or more of these changes appeared in 26–45% of the tubules, 4 means the changes involved 46–75% of the tubules, and 5 means more than 76% of the tubules showed one or more of these changes. 30
Immunohistochemistry for cytochrome c (Cytc) expression
Sections of 4 µm thickness were cut from the paraffin-embedded blocks. Then, these sections were deparaffinized, rehydrated and incubated with 1% hydrogen peroxide in methanol for 30 min to block the endogenous peroxidase activity. After that, these sections were rinsed with phosphate-buffered saline. Rabbit polyclonal antibody for c-cas-3 obtained from Zytomed Systems GmbH, Berlin, Germany (CAT # RBK009-05) was used as the primary antibody. A rabbit polyclonal antibody, cytochrome c (7H8.2C12) purchased from Abcam, Cambridge, MA, USA (CAT # ab13575) was used in a 1:100 dilution in phosphate-buffered saline for 17 h at 7°C. The secondary antibody used was biotinylated goat-anti rabbit IgG obtained from Boster Bio, Pleasanton, CA, USA (CAT # BA1003) and diluted as 1:100 in phosphate-buffered saline for 30 min at room temperature. 0.3% hydrogen peroxide incubated in phosphate-buffered saline for 30 min at room temperature was used to quench the endogenous peroxidase activity. 11 The intensity of positive immunostaining of Cytc was determined using IHC profiler tool in image J software (1.49v) national institute of health, USA and was graded as (+) refers to mild, (++) refers to moderate, and (+++) refers to marked positive immunostaining of Cytc.
Statistical analysis
Results were statistically analyzed using the statistical package for the social sciences (SPSS) version 22.0. Multiple measures analysis of variance (ANOVA) followed by Tukey’s multiple comparison test were used for multiple comparisons. Data obtained were presented as mean ± standard error of mean (SEM). P-values less than 0.05 were considered statistically significant.
Results
Effect of taxifolin and/or dapagliflozin on the kidney weight/body weight ratio
The kidney weight/body weight ratio was significantly increased with colistin injection compared to the control group (p < 0.001). Each of taxifolin or dapagliflozin induced significant decrease in the kidney weight/body weight ratio when compared to rats treated with colistin alone (p < 0.001). CMC induced non-significant change in the kidney weight/body weight ratio compared to colistin group (p = 0.65). The use of taxifolin/dapagliflozin combination had a better effect on the kidney weight/body weight ratio than when each of these agents is administered alone (p < 0.01) (Figure 1).
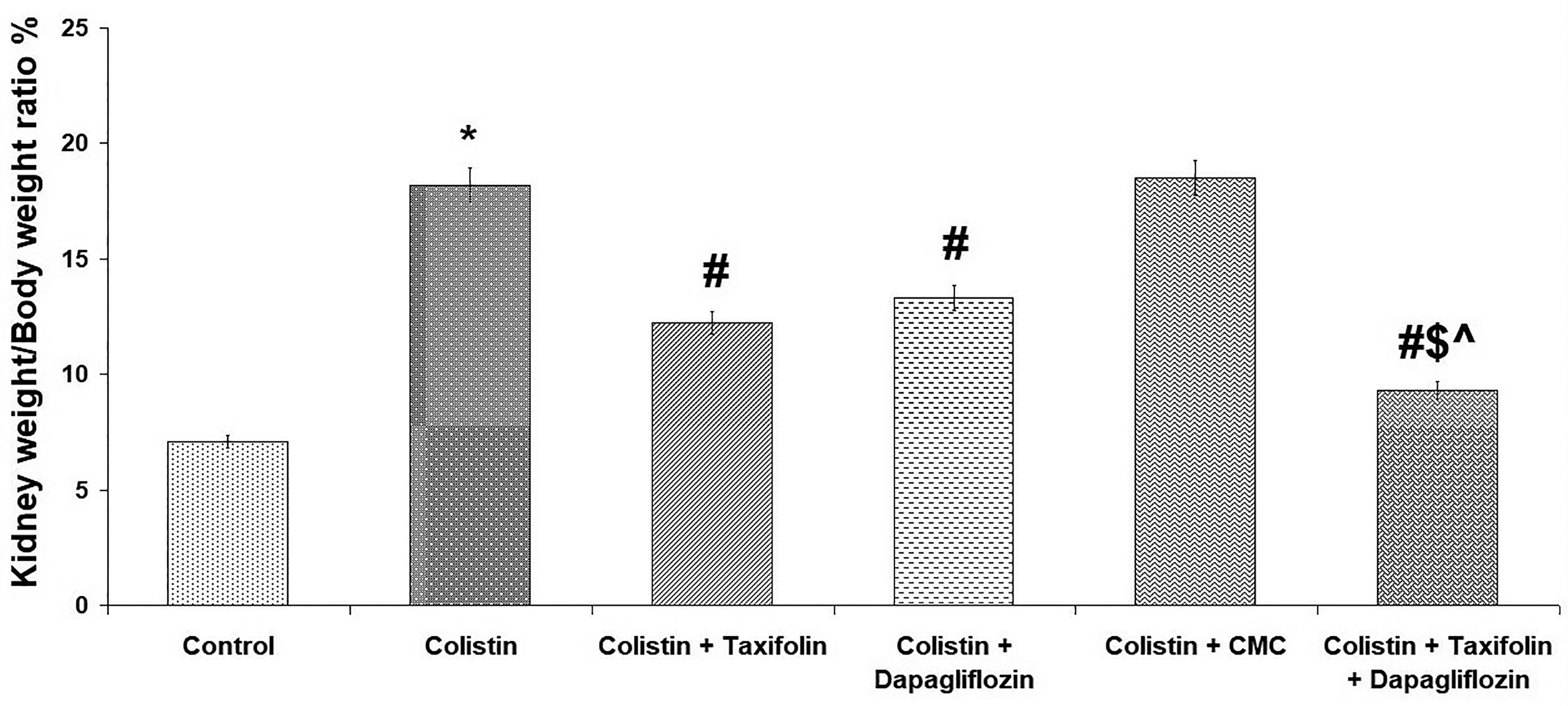
Effect of taxifolin and/or dapagliflozin on the kidney weight/body weight ratio %.
Effect of taxifolin and/or dapagliflozin on the renal function tests
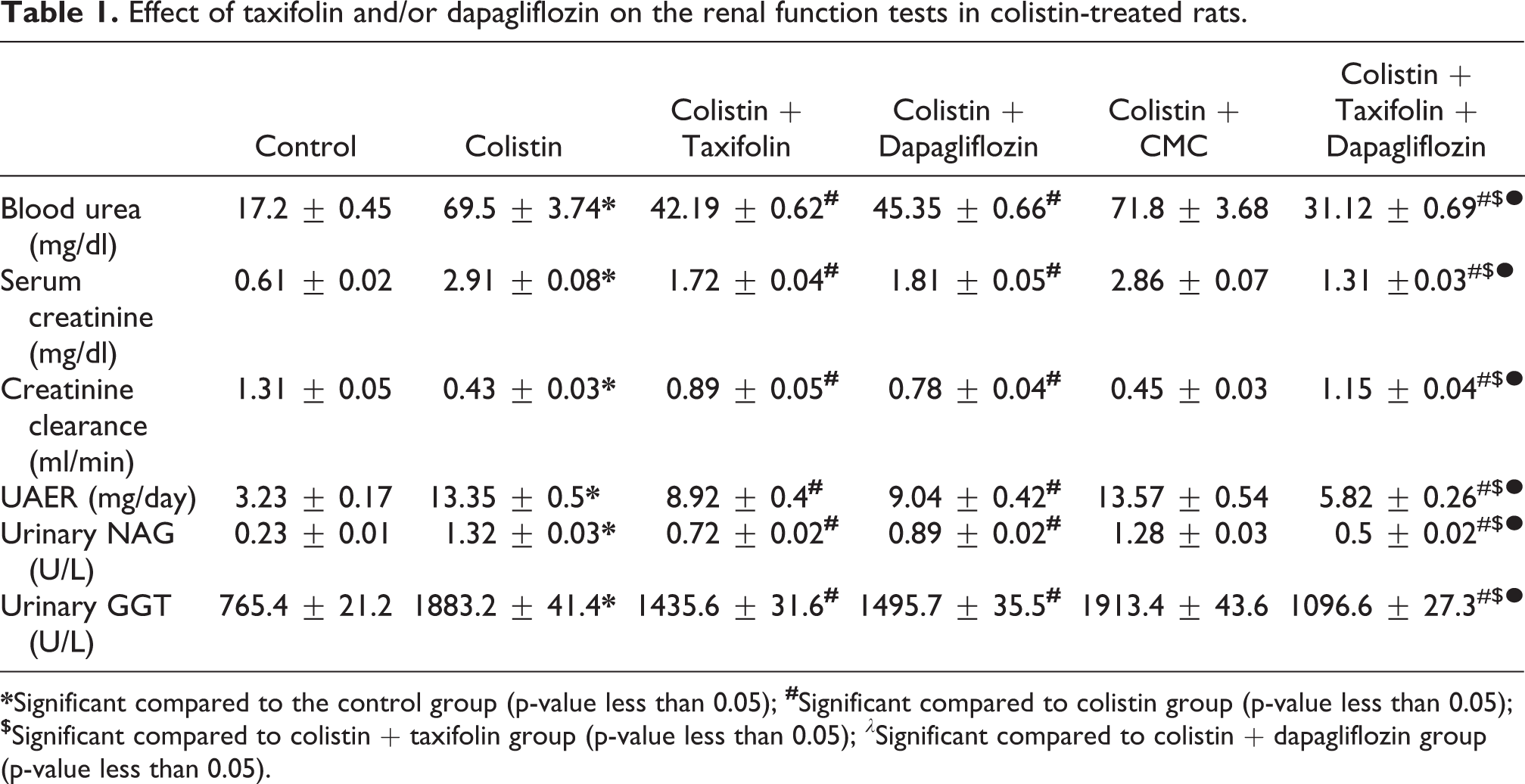
Colistin group showed significant increase in blood urea, serum creatinine, urinary GGT, urinary NAG and UAER associated with significant decrease in creatinine clearance compared to the control group (p < 0.001). CMC induced non-significant effect on the renal function tests in comparison to colistin group (p = 0.72, 0.84, 0.51, 0.66, 0.19, and 0.35 respectively). Treatment with either taxifolin or dapagliflozin induced significant improvement in the above-mentioned renal function tests compared to animals injected with colistin alone (p < 0.001). This improvement was significant with taxifolin/dapagliflozin combination group compared to the use of each of these agents alone (p < 0.01) (Table 1).
Effect of taxifolin and/or dapagliflozin on the renal function tests in colistin-treated rats.
Effect of taxifolin and/or dapagliflozin on the renal tissue GR, GPx, MDA and Nrf2 content
Significant decrease in tissue Nrf2, GR and GPx associated with significant increase in tissue MDA were observed in rats treated with colistin alone in comparison to the control group (p < 0.001). CMC induced non-significant effect on these measurements compared to colistin group (p = 0.32, 0.53, 0.64, and 0.39 respectively). Each of taxifolin or dapagliflozin induced significant improvement in tissue oxidative stress parameters and Nrf2 content compared to rats treated with colistin alone (p < 0.001) but taxifolin/dapagliflozin combination showed a superior effect (p < 0.01) (Table 2).
Effect of taxifolin and/or dapagliflozin on renal tissue nuclear factor (erythroid-derived 2)-like 2 (Nrf2), malondialdehyde (MDA), glutathione reductase (GR) and glutathione peroxidase (GPx) in colistin-treated rats.

Effect of taxifolin and/or dapagliflozin on the renal tissue TGF-β1, IL-15, TNF-α, TLR4, NF-κB (p65) and STAT3 expression
Significant increase in tissue levels of TGF-β1, IL-15, TNF-α, TLR4 and NF-κB (p65) expression associated with significant decrease in STAT3 levels was observed in rats injected with colistin alone in comparison to the control group (p < 0.001). No significant differences were noted in these parameters in rats treated with CMC + Colistin compared to rats treated with colistin alone (p = 0.61, 0.47, 0.77, 0.18, 0.27, and 0.63 respectively). Significant decrease in tissue TGF-β1, IL-15, TNF-α, TLR4 and NF-κB (p65) associated with significant increase in tissue STAT3 was reported with the use of each of taxifolin or dapagliflozin alone compared to colistin group (p < 0.001) but their combination had the upper hand (p < 0.01) (Table 3).
Effect of taxifolin and/or dapagliflozin on renal tissue transforming growth factor beta 1 (TGF-β1), interleukin 15 (IL-15), tumor necrosis factor alpha (TNF-α), toll-like receptor 4 (TLR4), nerve growth factor beta (NGF-β), nuclear factor kappa B (NF-κB) p65, signal transducer and activator of transcription 3 (STAT3), and caspase-3 activity in colistin-treated rats.

Effect of taxifolin and/or dapagliflozin on the renal tissue NGF-β and caspase-3 activity
Significant increase in tissue caspase-3 activity accompanied with significant decrease in tissue NGF-β were noted in colistin-treated rats in comparison to the control group (p < 0.001). No significant differences were noted in these parameters in rats treated with CMC + Colistin compared to rats treated with colistin alone (p = 0.54 and 0.43 respectively). Significant decrease in tissue caspase 3 activity and significant increase in tissue NGF-β were observed in rats treated with taxifolin or dapagliflozin compared to colistin group (p < 0.001) but their combination had the superior hand over the use of each of these agents alone (p < 0.01) (Table 3).
Effect of taxifolin and/or dapagliflozin on the renal tissue beclin-1 and JNK activities
Beclin-1 and JNK activities showed significant elevation with colistin use compared to the control group (p < 0.001). CMC administration to colistin-treated animals induced non-significant effect on the forementioned parameters in comparison to animals injected with colistin alone (p = 0.67 and 0.38 respectively). Significant decrease in beclin-1 and JNK activities was observed in rats that received either taxifolin or dapagliflozin compared to rats treated with colistin alone (p < 0.001) but taxifolin/dapagliflozin combination was better (p < 0.01) (Figures 2 and 3).

Effect of taxifolin and/or dapagliflozin on tissue beclin-1 (ng/mg tissue).

Effect of taxifolin and/or dapagliflozin on tissue JNK activity (% of control).
Histopathological findings
Administration of colistin induced significant degeneration of the renal glomeruli with formation of hyaline casts, marked vascular congestion, severe dilatation of the tubules and heavy infiltration of the renal tissues with inflammatory cells (Figure 4(b)) with significant increase in acute tubular necrosis score compared to the control group (p < 0.001) (Figure 5). This picture was improved with administration of either taxifolin or dapagliflozin with restoration of the normal structure of the renal glomeruli with minimal congestion, minimal casts, and significant reduction in the inflammatory cellular infiltrate (Figure 4(c) and (d) respectively) associated with significant decrease in acute tubular necrosis score (Figure 5) compared to rats treated with colistin alone (p < 0.001). CMC administration to colistin-treated rats induced non-significant effect in comparison to rats injected with colistin alone (p = 0.71) (Figures 4(e) and 5). Rats treated with taxifolin/dapagliflozin combination showed restoration of the normal architecture of the renal tissues with minimal inflammatory cellular infiltration (Figure 4(f)) with significant decrease in acute tubular necrosis score (Figure 5) compared to rats treated with each of these agents alone (p < 0.01).
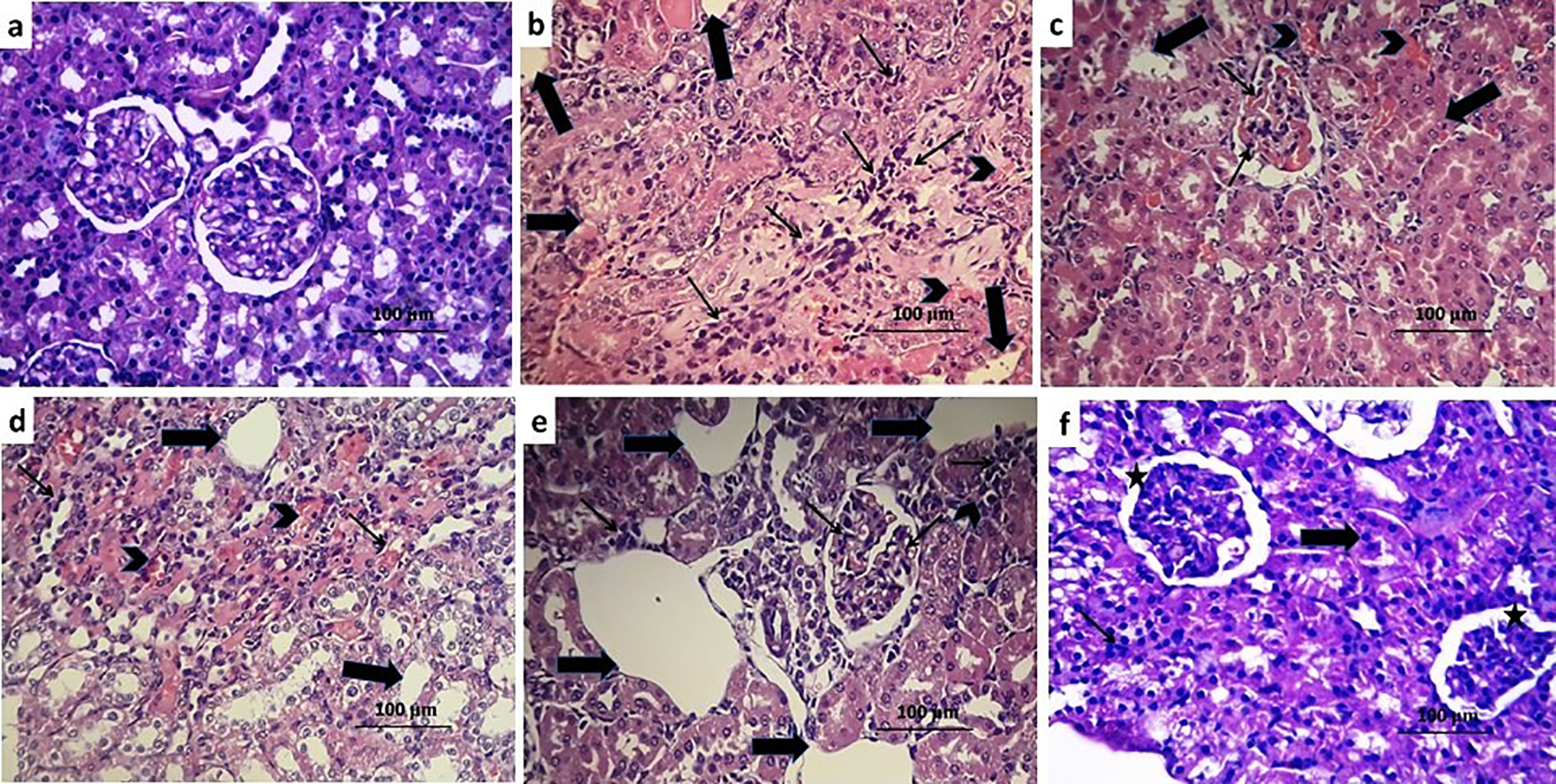
Hematoxylin & eosin stained sections from the kidney of (a) control group showing with normal appearance of the glomeruli, tubules and interstitial tissues; (b) colistin group with marked dilatation of the tubules (thick arrows), diffuse infiltration with inflammatory cells (thin arrows) with marked vascular congestion (arrow head); (c) colistin + taxifolin group with significant decrease in the inflammatory cellular infiltration of the glomeruli (Thin arrows) and vascular congestion (Arrow head) with scanty tubular dilatation (thick arrows); (d) colistin + dapagliflozin group with mild inflammatory cellular infiltration (thin arrows) and decrease in vascular congestion (arrow head) with scattered tubular degeneration (thick arrows); (e) colistin + CMC group showing huge dilatation of the tubules (thick arrows) with massive inflammatory cellular infiltration (thin arrows) and areas of interstitial hemorrhage (arrow head); (f) colistin + taxifolin + dapagliflozin group showing apparently normal glomeruli (asterix) with mild tubular dilatation (thick arrow).

Effect of taxifolin and/or dapagliflozin on acute tubular necrosis score.
Evaluation of the immunohistochemical expression of cytochrome c (Cytc)
The glomeruli and the renal tubular cells of the control group showed mild expression of Cytc (Figure 6(a)). There was marked positive expression of Cytc in the renal tissues of rats treated with colistin alone (Figure 6(b)) or colistin + CMC (Figure 6(e)). Administration of either taxifolin or dapagliflozin to rats treated with colistin significantly reduced Cytc expression to be of moderate degree (Figure 6(c) and (d) respectively) but taxifolin/dapagliflozin combination produced mild Cytc expression relative to the use of each of these agents alone (Figure 6(f)).

A photomicrograph of Cytc immunostaining of renal sections from (a) the control group showing mild Cytc expression (+); (b) colistin group showing marked positive Cytc expression (+++); (c) colistin + taxifolin group showing moderate positive Cytc expression (++); (d) colistin + dapagliflozin group showing moderate positive immunostaining for Cytc (++); (e) colistin + CMC showing marked positive Cytc expression (+++); (f) colistin + taxifolin + dapagliflozin group showing mild positive immunostaining for Cytc (+).
Discussion
Nephrotoxicity is one of the common side effects of colistin that represents an obstacle against its use in treatment of infectious diseases. 3 Up till now, the exact mechanism underlying the pathogenesis of colistin-induced renal damage is unclear. 4 However, increased production of reactive oxygen species (ROS), enhancement of the inflammatory pathways in the renal microenvironment and affection of the apoptotic pathways were suggested as precipitating factors. 33 Coinciding with these data, administration of colistin in the present study induced significant deterioration of the renal functions associated with significant increase in the kidney weight/body weight ratio and significant histopathological changes compared to the control group. Eadon et al. 34 and Lee et al. 35 attributed the increase in the kidney weight/body weight ratio encountered with colistin administration to the decrease in the total body weight with relative increase in the kidney weight. This decrease in the body weight may be due to arrest of the cell cycle induced with colistin with affection of apoptosis and inhibition of cellular proliferation.
TGF-β1 is a multifunctional cytokine that belongs to the transforming growth factor superfamily. 36 Recent reports had proven that TGF-β1 represents the key stone in most of the pathogenic events that occur in the renal tissues with administration of colistin. 35 Dai et al. 37 stated that TGF-β1 interacts with NF-κB resulting in increased expression of the proinflammatory cytokines with significant damage to the renal tissues. Also, colistin-induced expression of TGF-β1 was positively correlated with enhancement of the production of ROS and inhibition of the free radical scavenging mechanisms in the renal cells. 38 Moreover, TGF-β1 was reported to affect STAT3/MAP kinase signaling pathway with subsequent activation of the fibrogenic mechanisms leading to significant damage of the renal tissues. 39 In addition, increased expression of TGF-β1 was reported to increase the activity of caspase 3 and 9 and decrease the production of several antiapoptotic proteins resulting in enhancement of the programmed cell death. 40 This was in accordance with our results where colistin-mediated increase in TGF-β1 expression was associated with increased levels of NF-κB, enhanced production of the proinflammatory cytokines, inhibition of the activity of the antioxidant enzymes and STAT3 expression and enhancement of apoptosis compared to the control group.
Administration of taxifolin in the present study to colistin-treated rats was able to decrease the expression of TGF-β1, TNF-α and IL-15 and increase STAT3 levels in comparison to animals injected with colistin alone. This may be due to the effect of taxifolin on NF-κB expression which is the key regulator of the production and activity of TGF-β1. 41 Also, coinciding with our results, dapagliflozin was reported to affect STAT1/TGF-β1 signaling pathways in the renal tubulointerstitial tissues with subsequent inhibition of the inflammatory and the fibrogenic cascades. 42
Nrf2 is a transcriptional factor that was proven to be the master regulator of the protective responses against oxidative stress in various body tissues. 43 In the present study, colistin induced significant decrease in Nrf2 levels with subsequent decrease in the activity of the antioxidant enzymes and elevation of MDA levels compared to the control group. This was in the same line with the results of Dai et al. 44 who stated that colistin significantly suppresses the gene expression of Nrf2 resulting in deprivation of the renal tissues from the main protective mechanisms against oxidative and nitrosative damage. Recent trials had proven that taxifolin may have a positive impact on mRNA and the protein expression of Nrf2 with subsequent inhibition of oxidative stress-induced apoptosis. 45 Also, dapagliflozin in our study had the ability to increase Nrf2 content of the renal cells which may increase the gene expression of the antioxidant enzymes with subsequent amelioration of the renal damage mediated by free radicals. 46
TLR4 is a type I transmembrane protein that was reported to represent the key stone in recognition of the pathogens, activation of the innate immunity, generation of the proinflammatory cytokines and regulation of apoptosis. 47 Colistin in the present study significantly increased the expression of TLR4 in comparison to the control group. This was in agreement with Smith 48 who suggested that there is an interplay between NF-κB and TLR4 expression which may be considered as the main mechanism that underlies the pathogenic events that occur with administration of colistin. In addition, a positive correlation was found between TLR4 production and the expression of caspase 3 and 9 which supports the role of TLR4 in regulation of apoptosis. 49 The ability of taxifolin to affect nuclear factor kappa B expression may be the reason by which taxifolin may affect TLR4 production with subsequent inhibition of chemotaxis and tissue infiltration with inflammatory cells. 50 Coinciding with our results, Arow et al. 51 found that dapagliflozin by its ability to suppress the genes responsible for ROS overproduction may decrease TLR4 expression resulting in amelioration of the inflammatory cascade.
Affection of NGF-β signaling was demonstrated to play a fundamental role in the glomerular and tubular responses to damage induced by colistin. 4 In harmony with the results of the present study, colistin-mediated reduction of NGF-β expression was reported to be associated with increased caspase 3 immunostaining and affection of the expression of the mediators of autophagy. 37 These effects were attenuated in our study with taxifolin administration which was proven to enhance the expression of the genes responsible for NGF-β production with subsequent amelioration of apoptosis. 52 Also, the effect of sodium-glucose cotransporter 2 inhibitors on insulin production by the beta cells of the pancreas may be mediated through increased production of NGF-β with subsequent inhibition of apoptosis and autophagy. 53
c-Jun N-terminal Kinase (JNK) is a member of three families of mitogen-activated protein (MAP) kinases that may represent an important mediator of the functions of the immune system. 54 Recent studies indicated that JNK may increase apoptosis in the renal cells, possibly by enhancing the expression of the pro-apoptotic genes via enhancement of c-Jun/AP1-dependent or p53/73 protein-dependent mechanisms. 55 Moreover, suppression of JNK activity was proven to be correlated with significant decrease in the expression of beclin-1 which in turn will affect the gene expression of a group of proteins that may play an important role in the signaling pathways related to apoptosis and autophagy. 56 This agreed with the results of our study where colistin group showed significant increase in tissue JNK, beclin-1 and caspase-3 compared to the control group. Dapagliflozin in our study was able to ameliorate these changes which was in accordance with the reports of El-Sahar et al. 10 who stated that STGL2 inhibition may modulate the expression of the antiapoptotic proteins with subsequent affection of the cellular events that underlie the pathogenesis of autophagy and apoptosis. Also, the effect of taxifolin encountered in the present study on JNK, beclin-1 and caspase-3 may be explained by its effect on the signaling pathways in which MAP kinases play a vital role with subsequent inhibition of apoptosis and autophagy. 57
Cytochrome c (Cytc) is one of the intermediates of the electron transport chain that plays a key role in energy production and ATP formation. 58 Also, it has an impact on the production of ROS which in turn will affect the generation of the inflammatory mediators. 59 Moreover, it oxidizes cardiolipin during apoptosis leading to affection of life and death decisions of the body cells. 60 This agreed with our results where colistin enhanced the expression of Cytc compared to the control group. This was accompanied with significant increase in tissue caspase 3 activity, supporting the role of Cytc in apoptosis. Taxifolin in our study was able to decrease the expression of Cytc and caspase 3 compared to rats treated with colistin alone which was in the same line with Vladimirov et al. 61 who stated that taxifolin has a direct effect on Cytc production and caspase 3 activity on the gene expression level. Also, the levels of immunostaining of Cytc and caspase 3 activity were significantly reduced in the present study with administration of dapagliflozin. This supports the reports that indicated that dapagliflozin suppresses apoptosis of the renal cells via affection of the electron transport chain mediators and modulation of the expression of caspases.62,63
The reports obtained by the present study indicated that taxifolin/dapagliflozin combination had the upper hand in amelioration of colistin-induced nephrotoxicity over the use of each of these agents alone. This may be attributed to their synergistic effects against the oxidative damage induced by ROS generated by colistin which in turn affected the inflammatory processes with subsequent inhibition of apoptosis and modulation of autophagy. In addition, recent reports suggested that dietary flavonoids including taxifolin may inhibit the intestinal sodium-glucose co-transporters which may potentiate the effect of dapagliflozin. 64 In addition, taxifolin was proven to be inhibit the functions of P-glycoprotein, which may be responsible for the decreased bioavailability and the limited tissue distribution of dapagliflozin. 19
Conclusion
Taxifolin/dapagliflozin combination may confer a novel therapeutic tool for amelioration of colistin-induced nephrotoxicity. This may be due to their synergistic effects on oxidative stress and the inflammatory microenvironment created by colistin in the glomerular and the tubulointerstitial tissues. In addition, they have synergistic inhibitory effects on a wide range of mediators involved in autophagy and apoptotic cascade with subsequent amelioration of the nephrotoxicity induced by colistin. Further studies are needed to evaluate the possible implication of these findings in the clinical practice.
Footnotes
Acknowledgment
The authors greatly thank Taif University for providing funds for this study through Taif University Researchers Supporting Project number (TURSP‐2020/52), Taif University, Taif, Saudi Arabia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Taif University through Taif University Researcher Supporting Project (No. TURSP-2020/52).

