Abstract
Background:
Visfatin acts as an oncogenic factor in numerous tumors through a variety of cellular processes. Visfatin has been revealed to promote cell migration and invasion in gastric cancer (GC). Snai1 is a well-known regulator of EMT process in cancers. However, the relationship between visfatin and snai1 in GC remains unclear. The current study aimed to explore the role of visfatin in GC.
Methods:
The RT-qPCR and western blot analysis were used to measure RNA and protein levels, respectively. The cell migration and invasion were tested by Trans-well assays and western blot analysis.
Results:
Visfatin showed upregulation in GC cells. Additionally, Visfatin with increasing concentration facilitated epithelial-mesenchymal transition (EMT) process by increasing E-cadherin and reducing N-cadherin and Vimentin protein levels in GC cells. Moreover, endogenous overexpression and knockdown of visfatin promoted and inhibited migratory and invasive abilities of GC cells, respectively. Then, we found that snai1 protein level was positively regulated by visfatin in GC cells. In addition, visfatin activated the NF-κB signaling to modulate snai1 protein expression. Furthermore, the silencing of snai1 counteracted the promotive impact of visfatin on cell migration, invasion and EMT process in GC.
Conclusion:
Visfatin facilitates cell migration, invasion and EMT process by targeting snai1 via the NF-κB signaling, which provides a potential insight for the treatment of GC.
Introduction
Gastric cancer (GC), one of the most common gastrointestinal tumors, ranks the second among all cancer-related death globally. 1 In 2018, more than 1,000,000 new GC cases have been diagnosed worldwide and approximately 800,000 deaths were caused by GC.1,2 In China, the high incidence and distant metastasis of GC have resulted in difficulties for the treatment of GC.1,3 Known factors such as smoking, obesity, helicobacter pylori infection and genetic factors are the leading causes of the initiation and progression of GC.4–6 Many methods have been developed for cancer therapy. Sentinel lymph-node scintigraphy is a helpful tool in personalized therapy for GC patients. 7 Moreover, next generation sequencing technologies contribute to the medical research providing new possibilities in personalized medicine and prognostic views.8,9 However, the etiology and pathology of GC remain obscure and it is urgent to explore more mechanisms in GC progression to improve therapeutic strategies of GC.
The adipokine visfatin, known as nicotinamide phosphoribosyltransferase (NAMPT), was reported to participate in the regulation of carcinogenesis.10–12 In detail, visfatin limits the biosynthesis of nicotinamide adenine dinucleotide (NAD). 13 NAD+ is essential for multiple cellular processes and tumor cells have a high rate of NAD+ turnover compared to normal cells. 14 In addition, recent studies has revealed the potential roles of visfatin in regulating cell migration and invasion by promoting epithelial-mesenchymal transition (EMT) process.15,16 During EMT process, epithelial cells are transformed into mesenchymal phenotypic cells by specific procedures, which is the first step for cell migration and invasion in tumors. 17 Tumor cells loss cell adhesion and obtain migratory and invasive abilities in EMT process. 18 The reduction of E-cadherin (primary marker of EMT) levels is the beginning of EMT, accompanied by the increase of N-cadherin and vimentin levels. 19 The EMT process is closely correlated with the tumor progression and metastasis. 20 Numerous evidences have demonstrated that visfatin can facilitate cell migration and invasion in several tumors such as breast cancer, colorectal cancer, melanoma and ovarian cancer.21–25 These previous findings suggested that visfatin facilitates cancer progression by promoting cell migration, invasion and EMT process. Visfatin inhibitor FK866 increases the chemosensitivity of gastric cancer cells to fluorouracil as well as kills GC cell lines with EMT-associated gene expression signatures.16,26 However, the role of visfatin in GC cellular processes needs a further exploration.
In the current study, we first measured expression of visfatin in GC cell lines. We then specially investigated the biological function of visfatin treatment or visfatin transfection on GC cell migration, invasion and EMT process. We innovatively explored the downstream signaling pathway of visfatin in GC cellular processes. This research may provide a potential novel insight for the treatment of GC.
Materials and methods
Cell lines and cell treatment
The human normal gastric mucosal epithelial cells (GES-1) and GC cell lines including SNU-601, HGC27, BGC823 and MGC803 (Chinese Academy of Science Cell Bank, shanghai, China) and incubated in Roswell Park Memorial Institute 1640 (RPMI1640; Sigma-Aldrich, St Louis, USA) medium containing 10% fetal bovine serum (FBS; Gibco, Beijing, China), 50 U/mL penicillin and 50 μg/mL streptomycin. The cells were incubated in a damp atmosphere containing 5% CO2 at 37°C. After 48 h of incubation, BGC823 and MGC803 cells were treated with different concentration gradients (0, 25, 50, 100, 200, 400 ng/ml) of recombinant visfatin (Sigma-Aldrich) for 48 h before Trans-well assays and western blot analysis. In western blot analysis, BGC823 and MGC803 cells were treated with 10 μM of BAY11-7082 (an inhibitor of NF-κB, Absin, shanghai, China) for 24 h to inactivate the NF-κB signaling. 27 The flowchart to show the research methodology was specified in Supplementary Figure 1(a).
Cell transfection
The small interference RNAs (siRNAs) targeting visfatin or snai1 (si-visfatin or si-snai1) with negative control (si-NC) were used to knock down the level of visfatin or snai1. The vector pcDNA3.1/visfatin was used to overexpress visfatin with empty pcDNA3.1 as a control. The transfection was performed with Lipofectamine 2000 (Invitrogen, USA) for 48 h following the manufacturer’s protocols. All vectors were obtained from GenePharma (Shanghai, China).
Reverse transcription quantitative polymerase chain reaction (RT-qPCR)
The extraction of total RNA from cells was performed by TRIzol reagent (Invitrogen) as described previously. 28 The extracted RNA was reverse-transcribed into complementary DNA (cDNA) using a cDNA Reverse Transcription Kit (Invitrogen). Subsequently, RT-qPCR was carried out with a standard SYBR-Green PCR kit (Invitrogen) using an ABI 7900 Real-Time PCR System under manufacturer’s protocol. The relative expression of visfatin and snail was normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH). The 2−ΔΔCt method was used for RNA expression calculation as previously described. 29
Trans-well assays
The Trans-well chamber with or without Matrigel were applied for the examination of cell invasion or migration. For cell invasion, diluted Matrigel (70 μL) was added into six-well plates. Then, serum-free RPMI1640 medium was used to prepare single cell suspensions with a density of 1 × 106 cells/mL. Afterward, 100 µL of cell suspensions were placed into the upper chamber and 600 µL of RPMI1640 medium with 10% FBS was added into the bottom chamber. Then, both upper and bottom chambers were incubated for 24 hours at 37°C with 5% CO2. The invaded cells were immobilized with 10% formaldehyde and stained with 0.2% purple crystal, and the cells still in the upper chamber were gently cleaned with a cotton swab. Finally, the invaded cells were counted under an inverted microscope in each field. For cell migration, similar procedures were performed without Matrigel.
Protein extract from cytoplasm and nucleus
The GC cells suspensions were prepared by treatment with Trypsin-EDTA (Gibco, Beijing, China). The proteins were extracted from cytoplasm and nucleus of GC cells using a kit from Pierce (Rockford, IL) according to the manufacturer’s protocols.
Western blot
Total proteins were extracted from BGC823 and MGC803 cells using Protein Lysis Buffer (Beyotime, Shanghai, China). Then, cell lysates were segregated by 10% SDS-PAGE and moved onto a polyvinylidene fluoride (PVDF) membrane. Afterward, the membrane was coated with defatted milk and cultured with primary antibodies overnight at 4°C. Next, the membrane was incubated with secondary antibody at room temperature for 2 hours after washed with phosphate buffered saline (PBS). Finally, the blots were visualized by the enhanced chemiluminescence Plus and analyzed by ImageJ software. The primary antibodies including visfatin (ab236874), snai1 (ab216347), E-cadherin (ab1416), N-cadherin (ab18203), p65 (ab16502), Vimentin (ab92547), Lamin B (ab194109) and GAPDH (ab128915) as well as secondary antibodies (ab150077) were purchased from Abcam (Cambridge, USA).
Statistical analysis
Data were expressed as mean ± standard derivation (SD) and analyzed by the SPSS 16.0 software system (SPSS, Chicago, IL). The differences between two groups were analyzed by Student’s t-test. The differences among more than two groups were evaluate by one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test. Each experiment was performed at least three times. p < 0.05 had statistical significance.
Results
High concentration of visfatin promotes migration, invasion and EMT process of GC cells
Visfatin has been widely reported to regulate the progression of diverse tumors.12,24,25,30 To explore the role of visfatin in GC, a range of experiments were designed and conducted. As shown in Figure 1(a), RT-qPCR analysis suggested that visfatin showed a higher expression in GC cells than in GES-1 cells. Furthermore, Trans-well assays indicated that the number of migrated and invaded cells was gradually increased by the treatment of increasing concentration of recombinant visfatin (Figure 1(b)–(e)). Finally, western blot analysis delineated that the treatment of increasing visfatin concentration triggered a gradual decline of E-cadherin protein level and increase of N-cadherin and Vimentin protein levels (Figure 1(f)).

High concentration of visfatin promotes GC cell migration and invasion. (a) The visfatin level in GC cells and GES-1 cells was examined by RT-qPCR analysis. ((b)–(e)) Trans-well assays were used to test migration and invasion of GC cells treated with different concentrations (0, 25, 50, 100, 200, 400ng/ml) of recombinant visfatin for 48 h. (f) Western blot analysis was used to measure E-cadherin, N-cadherin and Vimentin protein levels in GC cells treated with different concentrations (0, 25, 50, 100, 200, 400ng/ml) of recombinant visfatin for 48 h. *p < 0.05.**p < 0.01.
Visfatin promotes GC cell migration, invasion and EMT process
Then, loss- or gain-of-function assays were conducted to further explore the function of visfatin in GC cells. First, the mRNA level of visfatin was knocked down by si-visfatin and overexpressed by pcDNA3.1/visfatin, respectively (Figure 2(a) and (b)). Subsequently, Trans-well assays showed that the migration and invasion of GC cells were suppressed by visfatin silence but facilitated by visfatin overexpression (Figure 2(c) and (d)). Then, western blot analysis demonstrated that the transfection of si-visfatin triggered increase of E-cadherin protein level and decrease of N-cadherin and Vimentin protein levels. On the contrary, visfatin overexpression led to decrease of E-cadherin protein level and increase of N-cadherin and Vimentin protein levels (Figure 2(e) and (f)).
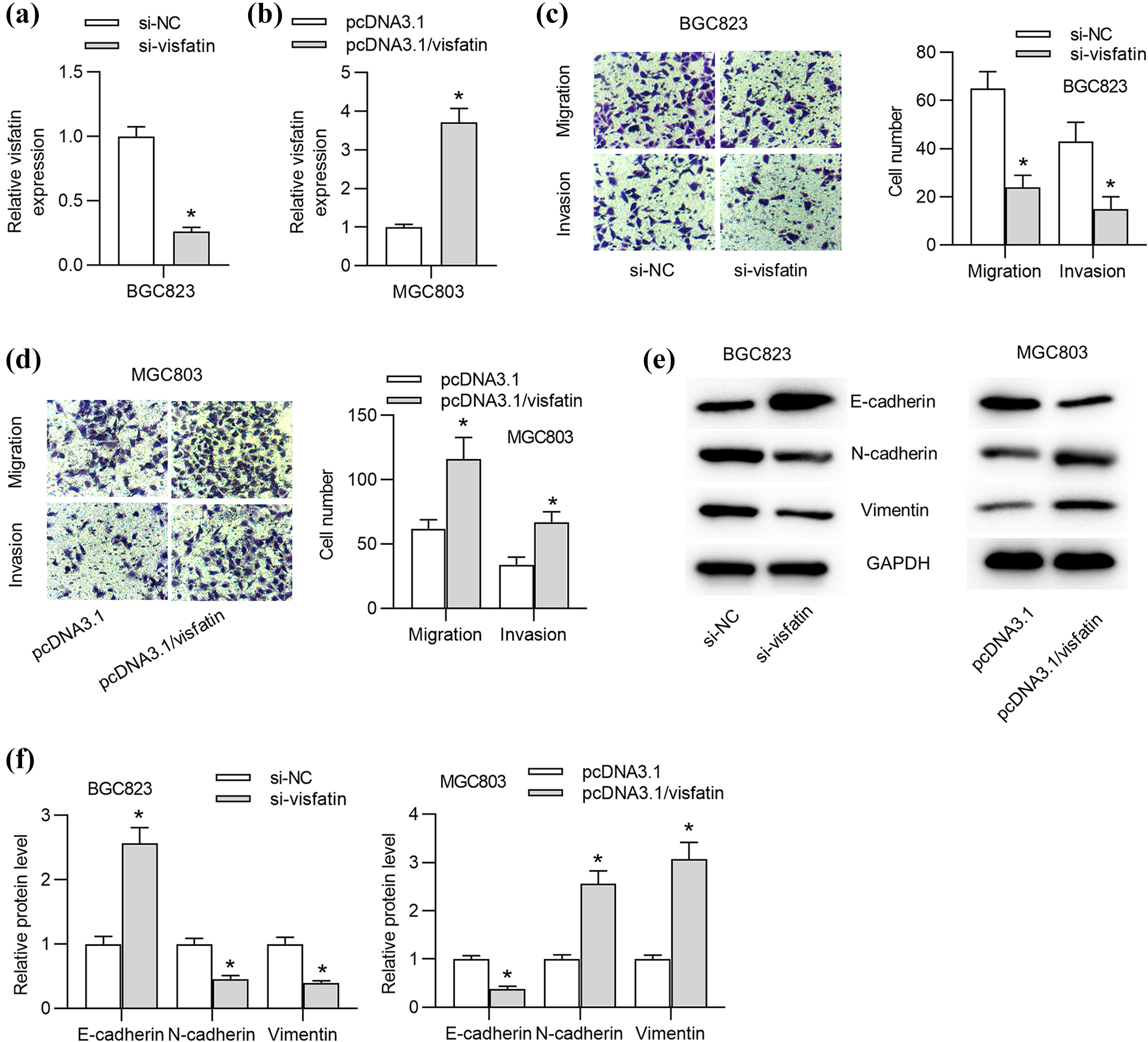
Visfatin promotes GC cell migration, invasion and EMT process. ((a) and (b)) The knockdown efficacy of si-visfatin and the overexpression efficacy of pcDNA3.1/visfatin were evaluated by RT-qPCR analysis. ((c) and (d)) Trans-well assays were carried out to assess GC cell migration and invasion under endogenous visfatin silencing or overexpression. ((e) and (f)) Western blot analysis was used to detect E-cadherin, N-cadherin and Vimentin protein levels in GC cells transfected with si-visfatin or si-NC as well as pcDNA3.1/visfatin or pcDNA3.1. *p < 0.05.
Visfatin positively regulates snai1 by activating the NF-κB signaling in GC cells
Visfatin was revealed to modulate snai1 level by activating the NF-κB signaling in osteosarcoma cells. 28 Thus, we anticipated that visfatin exerted regulation in the same way in GC cells. As shown in Figure 3(a), the knockdown of visfatin increased cytoplasmic p65 protein level and decreased nuclear p65 protein level in GC cells. On the contrary, overexpression of visfatin activated the nucleus translocation of p65 in GC cells. Additionally, the decrease of E-cadherin protein level and increase of N-cadherin and Vimentin protein levels under visfatin overexpression were reversed by BAY11-7802 treatment in MGC803 cells (Figure 3(b)). Moreover, the increase of snai1 protein level mediated by pcDNA3.1/visfatin was rescued by BAY11-7802 treatment (Figure 3(c) and (d)). Furthermore, in response to the treatment of increasing concentration of visfatin, both mRNA and protein levels of snai1 increased gradually (Figure 3(e)–(h)).

Visfatin positively regulates snai1 level in GC cells. (a) Western blot analysis was conducted to examine p65 protein level in cytoplasm or nucleus of GC cells under indicated transfection. C-p65: cytoplasm p65; N-p65: nucleus p65. *p < 0.05 vs. si-NC group. *p < 0.05 vs. pcDNA3.1 group. ((b)–(d)) The E-cadherin, N-cadherin, Vimentin and snai1 protein levels were examined by western blot analysis in GC cells transfected with pcDNA3.1 or pcDNA3.1/visfatin and treated with BAY11-7802 (10 μM) for 48 h. *p < 0.05 vs. pcDNA3.1 group. #p ≥ 0.05 vs. pcDNA3.1/visfatin group. ((e)–(h)) RT-qPCR and western blot analysis was used to detect snail mRNA and protein levels in GC cells treated with different concentrations of recombinant visfatin (0, 25, 50, 100, 200, 400ng/ml). *p < 0.05 vs. visfatin (0 ng/ml) group.
Visfatin promotes GC cell migration and invasion by modulating snai1
To validate whether visfatin promotes cell migration and invasion through modulating snai1, rescue assays were performed in BGC823 cells. First, cells were transfected with for 48 h. Then, RT-qPCR showed that snai1 level was knocked down by transfection with si-snai1 in BGC823 cells (Figure 4(a)). Next, Trans-well assays indicated that knockdown of snai1 countervailed the promotive effect of visfatin on GC cell migration and invasion (Figure 4(b) and (c)). Moreover, the decrease of E-cadherin protein level and the decrease of N-cadherin and Vimentin protein levels under visfatin treatment were reversed by knockdown of snai1 (Figure 4(d)). As shown in Figure 4(e), the treatment of recombinant visfatin or overexpression of visfatin facilitated the NF-κB (p65) nucleus translocation, and then mediated the upregulation of snai1, ultimately promoting GC cell migration, invasion and EMT process.
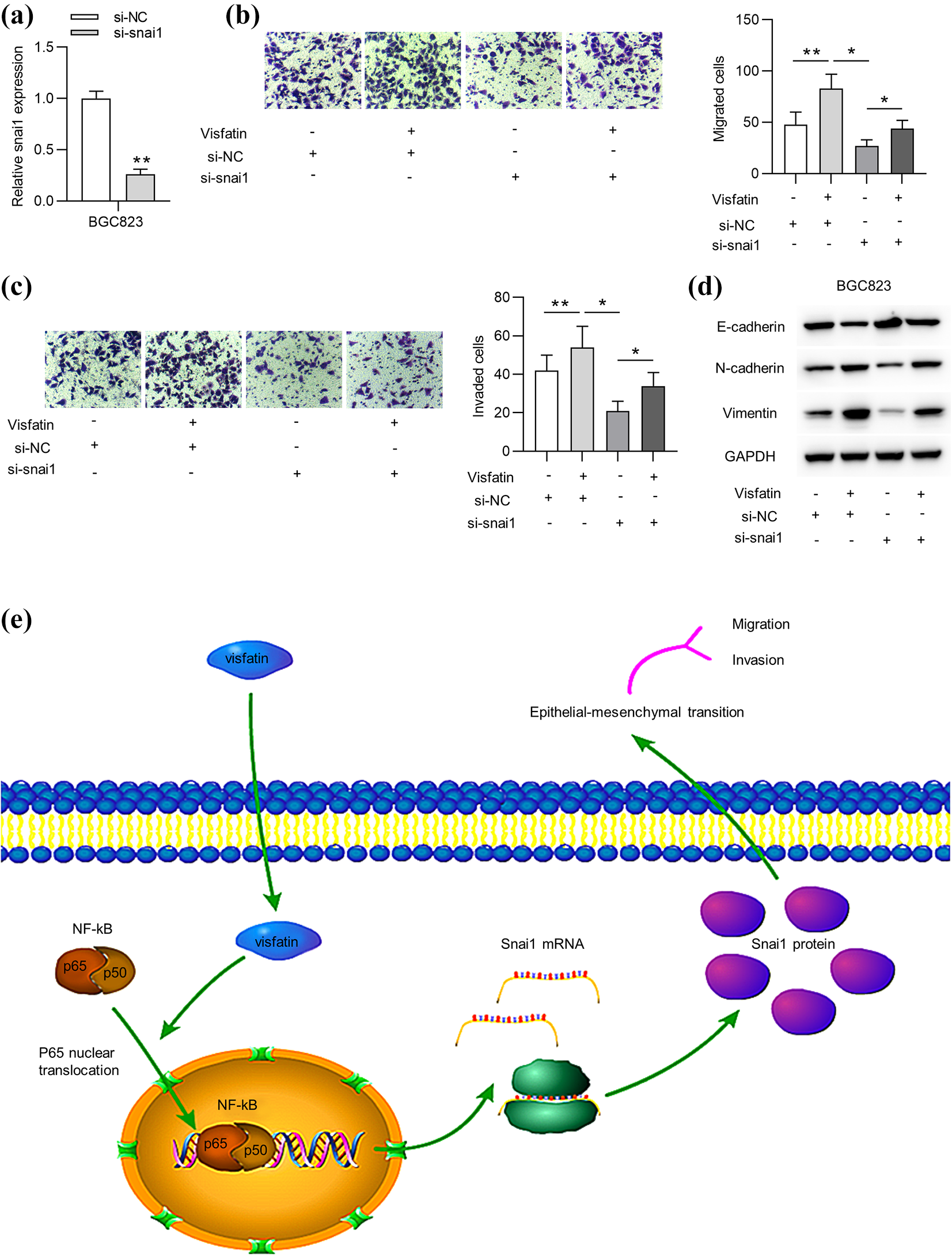
Visfatin promotes GC cell migration and invasion by modulating snai1. (a) The knockdown efficacy of si-snai1 in BGC823 cells was assessed by RT-qPCR. ((b) and (c)) Trans-well assays were carried out to assess cell migration and invasion of BGC823 cells under indicated treatment. (d) The protein levels of E-cadherin, N-cadherin, and Vimentin in BGC823 cells were examined by western blot analysis in each group. (e) Schematic model of current study: the treatment or endogenous overexpression of visfatin facilitated the NF-κB (p65) nucleus translocation, and then mediated the upregulation of snai1, ultimately promoting EMT process and migration and invasion in GC cells. *p < 0.05. **p < 0.01.
Discussion
Recently, great progress has been made in therapeutic methods for GC. Unfortunately, the overall survival rate of GC patients in 5-year remains low due to high recurrence rate.31,32 Thus, further exploration for the biomarkers of GC is extremely important for treatment of GC.
Numerous reports have revealed that visfatin is closely related to GC progression. For instance, the increased plasma visfatin level predicts poor prognosis of GC patients. 33 Particularly, a previous study has proposed that visfatin inhibitor FK866 regulates EMT process in GC cells.16,26 Similarly, our report revealed that visfatin was upregulated in GC cells compared to in normal control cells. Additionally, the increasing concentration of visfatin promoted cell migration, invasion and EMT process in GC. The results suggested that visfatin exerted a carcinogenic role in GC cells.
Snai1 has been widely reported to activate EMT process in carcinomas.34,35 For examples, FOXF1 induces the initiation of EMT by acting as a transcriptional factor of snai1 in colorectal cancer metastasis. 36 Snai1 reduces E-cadherin level to promote the aggressiveness of prostate cancer. 34 In addition, several molecules including circRBMS3, PCGEM1 and TRERNA1 can regulate snai1 expression to affect EMT process.37–39 Moreover, numerous studies have demonstrated that the NF-κB signaling promotes EMT process in GC progression.40–42 Previously, visfatin was proven to regulate snai1 by promoting the nucleus translocation of p65 in osteosarcoma cells. 28 Therefore, we hypothesized that visfatin exerted regulation on EMT process and snai1 expression by activating the NF-κB signaling in GC cells. In our report, the knockdown and overexpression of visfatin inactivated and activated the nucleus translocation of p65 in GC cells, respectively. Moreover, the EMT process and snai1 expression increased by pcDNA3.1/visfatin was rescued by BAY11-7802, the inhibitor of the NF-κB signaling. Furthermore, we found that snai1 mRNA and protein levels were positively regulated by visfatin. Eventually, rescue assays suggested that the knockdown of snai1 countervailed the effects of recombinant visfatin on migration, invasion and EMT process of GC cells.
In conclusion, we first proposed and verified that visfatin was upregulated in GC cells. We innovatively revealed that visfatin facilitated GC cell migration, invasion and EMT process by targeting snai1 via the NF-κB signaling. Our research may provide a potential novel insight into the treatment of GC. However, our investigation on the visfatin-mediated NF-κB signaling involved in GC progression is still in preliminary stages with limitations. Further in-depth functional investigations with advanced genomic technologies and animal experiments are still needed to evaluate the accuracy of the biomarker with large clinical samples and to explore the complex ceRNA molecular mechanisms involved in GC progression.
Supplemental material
Supplemental Material, sj-tif-1-het-10.1177_09603271211006168 - Visfatin facilitates gastric cancer malignancy by targeting snai1 via the NF-κB signaling
Supplemental Material, sj-tif-1-het-10.1177_09603271211006168 for Visfatin facilitates gastric cancer malignancy by targeting snai1 via the NF-κB signaling by D Cao, L Chu, Z Xu, J Gong, R Deng, B Wang and S Zhou in Human & Experimental Toxicology
Footnotes
Acknowledgment
We appreciate all participants in this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by 2019 Bengbu Medical College Key Natural Science Project (No. BYKY2019132ZD) and 2018 Anhui University Natural Science Research Project (KJ2018A0240). The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
