Abstract
Background:
Hexagonal boron nitride nanoparticles (hBN NPs) are encouraging nanomaterials with unique chemical properties in medicine and biomedical fields. Until now, the optimal hBN NP’s dosage and biochemical mechanism that can be used for in vivo systems has not been fully revealed. The main aim of this article is to reveal characteristics, serum and tissue interactions and any acute cytotoxic effect of different dose of hBN NPs for the first time.
Methods:
hBN NPs at concentrations varying between 50–3200 µg/kg was administered by intravenous injection to Wistar albino rats (n = 80) divided into seven dosage and control groups. Blood and tissue samples were taken after 24 hours.
Results:
Our findings suggested that higher doses hBN NPs caused oxidative stress on the serum of rats dose-dependently. However, hBN NPs did not affect thiol/disulfide homeostasis on kidney, liver, spleen, pancreas and heart tissue of rats. Furthermore, hBN NPs increased serum disulfide formation by disrupting the thiol/disulfide balance in rats. Also, LOOH and MPO levels increased at high doses, while CAT levels decreased statistically.
Conclusion:
The results revealed that hBN NPs induce oxidative stress in a dose-dependent manner by modulating thiol/disulfide homeostasis in rats at higher concentrations
Introduction
With the developing nanotechnological possibilities today, the use of nanomaterials in different spectrum on living organisms has increased considerably, and it is a matter of concern whether they have toxic effects on human health. 1 Many researchers have revealed that different nanoparticles (NPs) can have different toxic effects, both in terms of their functional groups and chemical components, as well as their physical properties. 2 Due to its large surface area and very small dimensions, NPs produce reactive oxygen species that attack DNA directly, thereby causing oxidative damage. 3 The different nanotoxic effects that different nanomaterials may have are still not fully known and toxic mechanism researches are still ongoing.
Boron nitride (BN) nanoparticles have greatly attracted significant interest due to its superior chemical, physical and thermal properties. With these features, it is used in many engineering applications. Recent research has revealed the potential for use in the biomedical field. Despite numerous studies in engineering applications for industries, studies to evaluate boron nitride for biomedical applications are still limited. There is a growing interest among researchers on the interaction of living systems and boron nitride in terms of biomedical engineering applications. This interest has raised curiosity about the cytotoxic effect of boric oxide and boron nitride materials and studies have been conducted on this.4–7
In this study, it was aimed to investigate the toxic properties of hBN NPs in vivo depending on its concentration, structure and shape dimensions. XRD and FTIR were used for the structural characterization. Scanning Electron Microscopy (SEM) and Transmission Electron microscopy (TEM) were conducted for revealing the morphological characterization. Also, possible toxic effects of hBN NPs were investigated by measuring thiol/disulfide homeostasis, lipid hydroperoxide (LOOH) levels, myeloperoxidase (MPO) and catalase (CAT) activities on rats.
Materials and methods
Synthesis and characterization of hBN
Synthesis: The novel synthesized hBN nanoparticles were provided from BORTEK, Boron Technologies, and Mechatronics Inc., Turkey. hBN powder is manufactured in agglomerate form through the direct reaction of boron oxide with ammonia gas. Highly crystalline hBN nanoparticles was obtained with sieved under 150 microns after milling by a planetary ball mill at 400 rmp in a 500 mL stainless steel jar together with 3 mm stainless steel balls as grinding media and characterized.
Characterization: The particle size analyses of samples were determined with Malvern Master Sizer 2000 Hydro G2000. X-Ray powder diffraction (XRD) patterns were obtained on a Rigaku Miniflex X-ray diffractometer with CuKα radiation (λ = 1.5418 Å), the 2θ range from 10–60° with the scan speed of about 0.5°/min. FTIR analysis was conducted with the Broker Tensor II spectrometer. SEM studies were conducted on a Zeiss Supra 50VP operated at 20 kV. For SEM analysis, samples were diluted with propan2-ol and sonicated for 5 minutes. Then the samples were deposited on lamella and heated at 80°C in the oven to remove propan2-ol. Samples were coated with Au/Pd sputter. Transmission electron microscopy (TEM) studies were conducted with a JEOL-JEM 2100F field emission TEM equipped with a STEM high angle annular dark-field (HAADF), an energy dispersive spectrometer (EDX, JEOL-JED2300T) and precession electron diffraction (PED, Nanomegas) with a 0.7 of precession angle.
Animals and experimental design
All animals included in our study were obtained from Eskişehir Osmangazi University Medical and Surgical Experimental Animals Application and Research Center. Our experimental protocol was approved by the ethical local committee of Eskişehir Osmangazi University, Eskişehir (Protocol No: 662). Turkey according to National Institutes of Health guidelines. All experimental procedures performed on rats were applied according to our previous study. 6 Eighty male Wistar albino Rats were grouped as follows: the first group used as control, and the other groups were received hBN NPs at concentrations varying between 50–3200 µg/kg was administered by intravenous injection. After the experiment procedure, rats were anesthetized by ketamine–xylazine, then sacrificed via exsanguination, and blood and tissue samples were gathered for biochemical analyses.
Thiol-disulfide homeostasis assay
Thiol-disulfide homeostasis parameters were made by modifying and automatizing Ellmann’s method. 8 Native thiol (NT) shows thiol (–SH) groups, and disulfit shows oxidized thiols (–S–S). Total thiol refers to the sum of both NT and disulfide. The ratio of disulfides to native and total thiole and the ratio of native thiol to total thiol gives information on thiol-disulfide balance. It was analyzed with native thiol (µmol/mg protein) and total thiol (µmol/mg protein) sodium borohydride (NaBH4) and 5,5′-dithiobis-(2-nitrobenzoic) acid (DTNB; Merch, Darmstadt, Germany) solutions. Disulfide (µmol/mg protein) level is obtained by subtracting native thiol from total thiol and dividing it into two.
LOOH assay
It is based on the principle of measuring LOOH by oxidation reaction of ferrous ions with xylenol orange. 9 LOOH levels was expressed as µmol/mg protein in tissue and µmol/L in serum.
MPO assay
It is based on the principle that MPO is oxidized with O-dianicide and when the hydrogen peroxide (H2O2) is added, measuring yellowish color 460 nm. 10 MPO activity was expressed as U/mg protein in tissue and U/L in serum.
CAT assay
Catalase (CAT) enzyme activity was measured with a method modified by Goth. 11 Serum (0.2 ml) was incubated in 1 mL substrate (65 µmol per H2O2 in 60 mmol/L sodium–potassium phosphate buffer, pH 7.4) at 37°C for 60 seconds. This enzyme catalyzed reaction was stopped with 1 mL of 32.4 mM ammonium molybdate and the yellow color of molybdate and hydrogen peroxide (H2O2) was detected at 405 nm against a blank. One CAT unit decomposes 1 µmol of H2O2 min−1 in this state. The activity of CAT was expressed as kU/mg protein in tissue and kU/L in serum.
Statistical analysis
Data from all groups were analyzed with SPSS 21 (SPSS Inc., Chicago, IL) and Graph-Pad Prism 7 (San Diego, CA, USA) statistical software then represented as the mean ± SD (standard deviation). One-way ANOVA test was used for normally distributed data, and Tukey test was performed for multiple comparisons. The value of p < 0.05 was regarded as the statistical significance.
Results
Hexagonal boron nitride nanoparticle characterization results
The XRD analysis of hBN powder and milled hBN was shown in Figure 1. The five characteristic peaks at 2θ values are attributed to (002), (100), (101), (004) and (103) crystallographic planes of the hexagonal form of boron nitride (JCPDS No: 034-0421). The d-spacing value of nanoparticles calculated for the peak at 2θ value of 26.64° was 0.34 nm. The sharp and intense peaks were shown the powder is well-crystallized hBN NPs. Any impurity caused by the milling wasn’t detected in the XRD pattern.

The XRD analysis of boron nitride nanoparticle.
Figure 2(a) shows the schematic representation of one layer of hBN NPs. To confirm the OH formation on hBN nanoparticles, a Fourier Transform Infrared Spectroscopy (FTIR) study was conducted. In Figure 2(b), the peak at 780 cm−1 corresponds to B–N vibrations and the band at 1370 cm−1 is due to the stretching vibrations of B–N. The hydroxyl group form a wide peak between 2525 and 3400 cm−1. But we did not observe the OH functional group on the particle surface.

(a) Schematic illustration of 1D hBN and (b) FTIR spectrum of hBN NPs.
The hBN nanoparticles were characterized by imaging and spectroscopic techniques. Before the SEM imaging was carried out Au-Pd sputtered for the 40 sec to obtaining a conductive layer of over the particles. Figure 3(a) shows the SEM images of the hBN particles and a more uniform structure with a lateral size dimension between 50 and 200 nm with a round shape was obtained as seen in the image. TEM images in Figure 3(b) and (c) were verified the hexagonal crystalline structure of boron nitride and the size of particles in the different intervals between the diameter of 50–200 nm and a thickness of 15–50 nm. Figure 3(c) shows the parallel straight-line like crystalline features of hBN particles.

(a) The SEM image hBN NPs, (b and c) TEM image of boron nitride nanoparticle, corresponding SAED pattern (d).
Hexagonal boron nitride nanoparticle change thiol-disulfide homeostasis
Thiol–disulfide homeostasis variables in serum treated with varying hBN NPS doses are shown in Figure 4. As shown in Figure 4(a) and (b), the treatments with 1600 and 3200 µg/kg of hBN NPs caused a significant decrease total thiol and native thiol levels on serum exposed to increasing concentrations of hBN NPs compared to control (all p < 0.05). However, statistically significant increases in disulfide levels were observed in serum after 1600 and 3200 µg/kg hBN NPs treatments (24.59 ± 3.09 and 24.78 ± 4.47 µmol/L, respectively) compared with control (16.03 ± 4.40 µmol/L) in Figure 4(c) (p < 0.05). As seen in Figure 4(d) to (f), at 1600 and 3200 µg/kg, thiol/disulfide balance in serum was impaired and shifted toward disulfide form when compared with the control (p < 0.001). Table 1 shows the effects of hBN NPs on thiol-disulfide parameters on tissues. There was no significant statistical difference in thiol/disulfide parameters on liver and kidney as in serum results. However, disulfide formation was observed surprisingly in 800 µg/kg hBN NPs application in heart and spleen tissues.
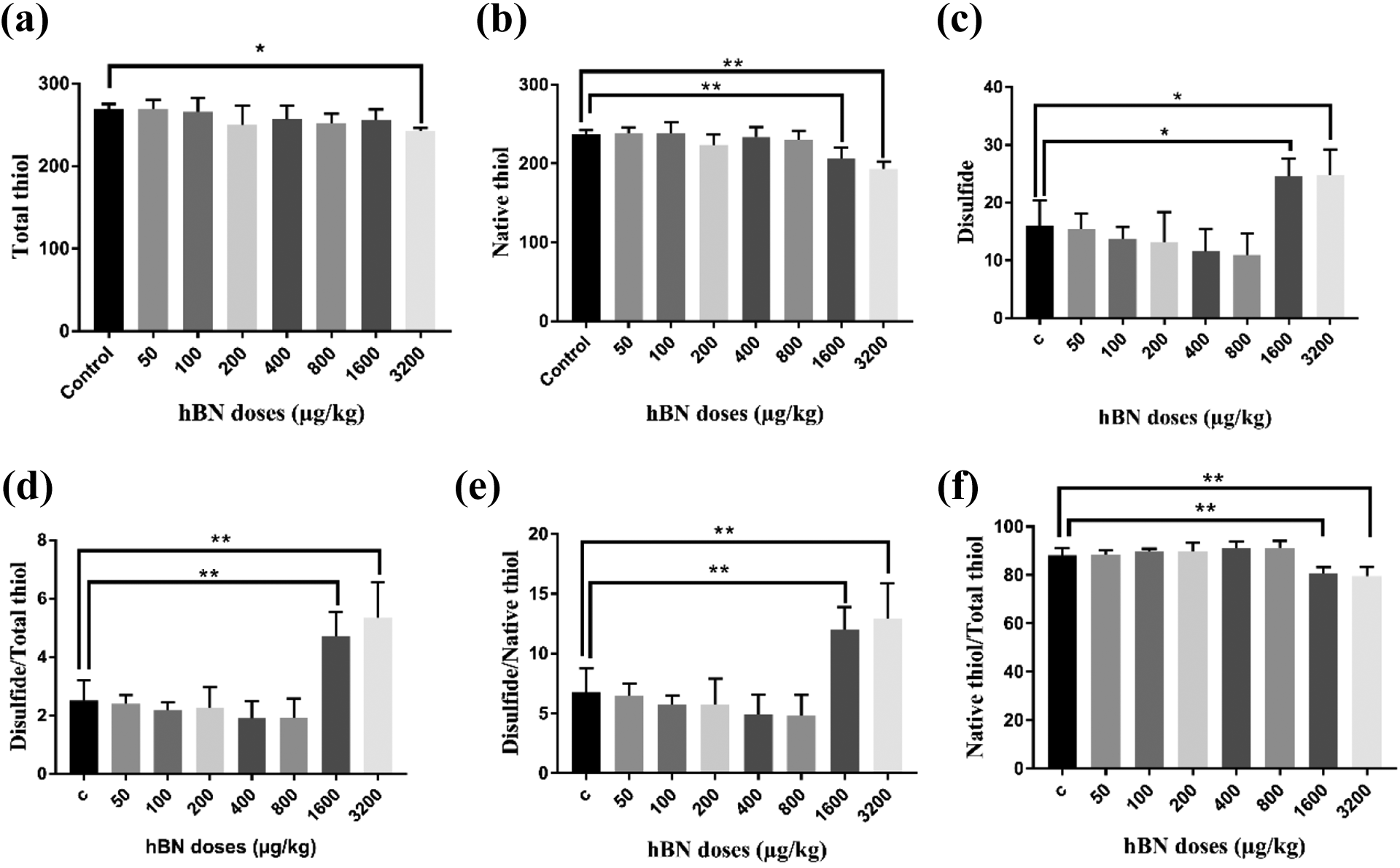
Thiol–disulfide homeostasis variables in serum treated with varying hBN NPs doses: (a) total thiol, (b) native thiol, (c) disulfide, (d) disulfide/total thiol, (e) disulfide/native thiol, and (f) native/total thiol. *P < 0.05; **P < 0.01 when compared to control and between hBN NPs doses. Native thiol and total thiol levels was expressed as µmol/mg protein in tissue and µmol/L in serum. The results are displayed as mean ± SD (n = 10).
Thiol-disulfide homeostasis in tissues.

Thiol–disulfide homeostasis variables in tissues treated with varying hBN doses. TT: total thiol; NT: native thiol; DS: disulfide; DS/NT: disulfide/native thiol; DS/TT: disulfide/total thiol; NT/TT: native/total thiol. The results are displayed as mean ± SD (n = 10). *P < 0.05; **P < 0.01 when compared to control and between hBN NPs doses.
The effects of hexagonal boron nitride nanoparticle on LOOH levels
LOOH levels in serum and tissues of rats treated with varying hBN NPS doses are shown in Figure 5. As shown in Figure 5(a) and (c), the treatments with 1600 and 3200 µg/kg of hBN NPs caused a significant increase LOOH levels on serum and kidney tissue compared to control (p < 0.05). In the liver tissues, LOOH levels were determined at higher rates in rats which in 200 µg/kg (p < 0.05) and above (p < 0.01) hBN NPs groups than the control. In the cardiac tissues, only 3200 µg/kg dose groups’ LOOH levels were statistically higher than the control (p < 0.05), and LOOH levels were higher in the spleen and pancreas tissues at both 1600 µg/kg (p < 0.05) and 3200 µg/kg (p < 0.05 in spleen, p < 0.01 in pancreas) doses compared to the control group.

LOOH levels treated with varying hBN doses: (a) serum, (b) liver, (c) kidney, (d) heart, (e) spleen, and (f) pancreas. LOOH levels was expressed as µmol/mg protein in tissue and µmol/L in serum. The results are displayed as mean ± SD (n = 10). *P < 0.05; **P < 0.01 when compared to control and between Con A doses.
The effects of hexagonal boron nitride nanoparticle on MPO levels
As the indicators of neutrophil infiltration and inflammation, MPO activities were examined in both serum and tissues of rats and no statistical difference was observed between the control and hBN NPs groups up to 1600 µg/kg hBN NPs dose (Figure 6). There was a statistically significant increase in MPO activities in liver (Figure 6(b), p < 0.05) and heart (Figure 6(d), p < 0.01) tissues of rats administered 1600 and 3200 µg/kg hBN NPs compared to the control group. MPO activities in kidney (Figure 6(c)), spleen (Figure 6(e)), pancreas (Figure 6(f)) tissues and serum (Figure 6(a)) samples of 3200 µg/kg hBN NPs groups were statistically higher from the control group (p < 0.05).

MPO levels treated with varying hBN doses: (a) serum, (b) liver, (c) kidney, (d) heart, (e) spleen, and (f) pancreas. MPO activity was expressed as U/mg protein in tissue and U/L in serum. The results are displayed as mean ± SD (n = 10). *P < 0.05; **P < 0.01 when compared to control and between hBN NPs doses.
The effects of hexagonal boron nitride nanoparticle on CAT levels
When CAT activities, one of the important enzymes of antioxidant defense system, were examined, it was seen that activity decreased as the dose of hBN NPs increase (Figure 7). Although it was observed that CAT activities increased in serum samples of rats treated with 100 µg/kg hBN NPs dose (Figure 7(a)) compared to the control group, there was no statistically significant difference. In rats treated with 1600 and 3200 µg/kg hBN NPs, CAT activities in all tissues and serum decreased compared to the control group, due to increased oxidative damage (Figure 7).

CAT levels treated with varying hBN doses: (a) serum, (b) liver, (c) kidney, (d) heart, (e) spleen, and (f) pancreas. The activity of CAT was expressed as kU/mg protein in tissue and kU/L in serum. The results are displayed as mean ± SD (n = 10). *P < 0.05; **P < 0.01 when compared to control and between hBN NPs doses.
Discussion
The hBN nanoparticles were characterized by imaging and spectroscopic techniques to understand the morphology and surface characteristics of BN particles. The XRD results show the well-crystallized hBN nanoparticle with high purity. (Figure 1). In the 2θ and d-spacing value of hBN peaks did not observe any changes caused by ball milling if compared to starting powder. There is no contamination or phase change has been detected. While B and N atoms form 2D hexagonal layers with strong covalent bonds with each other in the basal plane. The layers are stacked on each other with weak Van der Walls forces and creates 3D structure. This arrangement is affected by all stages of the process, starting with the precursor. There are several synthesis methods reported in the literature for hBN. 12 All these methods include a variety of boron and nitrogen precursors under different conditions. The synthesis methods influence particle shape, size, number of layers and crystallinity of BN.13,14 The surface characteristics are also depending on the production method. In the medical application, properties such as purity, biocompatibility and toxicity gain importance. In this study, considering that BN morphology is related to all these features, serum and tissue interactions and the acute cytotoxic effect of BN with round-shaped were investigated.
The growth kinetics of BN affect the particle shape. A layer of BN is composed of a one-atom-thick sheet and grew with boron and nitrogen atoms in the hexagonal arrangement (Figure 2(a)). The layers come together to form a platelet-like morphology. While the OH ions are on the surface of the basal plane, nitrogen ions are located periphery of the plate. FTIR results did not show us any OH functional group on the particle surface. The reactivity of used hBN powder is low due to the absence of OH ions on its surface (Figure 2(b)). The reason for the lack of OH ions on the particle surface is thought to be the role of surface energy on the formation of BN. BN layers are thought to be formed by the model in Figure 2(a) by surface energy that maximizing the edges of ending with Nitrogen. Thus, the particle tends to be a platelet structure. The platelet structure depending on the production methodology can be considered to mean minimum ROS formation. The change in physicochemical properties of nanoparticle makes the investigation of their toxic effects complex and challenging. 15
In addition to growth kinetics, post-treatment of the particle is also responsible for the changes in purity and surface properties. However, the characterization of these parameters is a key indicator of the resulting reactive oxygen species response. The morphology and purity of boron nitride play a critical role in chemical activity.BN particles having a multi-layered structure with a lateral size of 50–200 nm are similar in shape to platelets as seen from Figure 3. We did not observe any particle breaking on the basal plane caused by the ball milling. The milling can cause break the basal plane and activated the surface radicals.16,17 It is important because OH formation can be triggered by the milling responsible for particle size reduction or exfoliation. Exfoliation methods are used to increase the activity of the particle. These methods are used to separate the layers from each other. The separation of layers causes the formation of new OH radicals. Nanomaterials such as fullerenes, CNT or metal oxides have been shown to induce oxidative stress.18,19 When the silica particle breaks, it produces a large number of surface-bound ROS. 20 In this study, for BN produced as nanoparticles, grinding can be considered as dispersing of agglomerates. Therefore, OH radicals were not found and BN did not cause oxidative stress at low doses. Similarly, Taskin et al. showed that the round-shaped BNs they used in their studies did not generate ROS at low doses. 21 Also a recent study on hBN particle with round shape showed that hBNs led to increases of TAS levels and decreases of TOS levels in Parkinson Disease model. 5 In this case, the reliability of the medical grade hBN may be related to the morphological structure.
The thiol compound, which is an important part of the antioxidant defense system in living things, plays an important role in removing free radicals by taking place in both enzymatic and non-enzymatic pathways with the R–SH group it contains. Thiol compounds are oxidized under oxidative stress to form disulfide bonds and when oxygen radicals are removed from the environment, the continuity of thiol/disulfide homeostasis is preserved by reducing R–SH form again.8,22 In this approach, it thought that the morphological properties of the platelet-like nanoparticles used in this study affect the total thiol-disulfide concentration in serum. No significant change in total thiol/disulfide was observed in serum at low doses. In high concentrations of hBN NPs, thiol levels decrease while disulfide levels increase. This situation explained by the lower content of surface radicals on the particles. It thought that the presence of OH increases depending on the dosage. In another hypothesis, thiol is presented as protein thiols such as albumin, glutathione (GSH), cysteine, homocysteine and, non-protein thiols. 23 Conjugation of GSH with lipid peroxidation end products aldehydes, can decrease GSH levels (as an antioxidant). 24 Increased oxidative stress may also increase the levels of homocysteine and cysteine, another thiol incorporating molecules. 25 This is the first study to examine the effects of both nanoparticles and hexagonal boron nitride on thiol/disulfide balance. Hormetic response; It is a mechanism that helps the balanced management and preservation of cell and intra-organism reactions (homeostasis) in a way to maintain life. Various signaling pathways in the cell play a role in the beneficial effects of toxic agents on the organism at low doses. In the light of this information, the hormetic effect was emphasized in the dose studies conducted previously.26–30 We argue that high concentrations of hBN can alter the levels of these major thiol carrying molecules which subsequently triggers oxidative stress.
Lipid peroxidation involves oxidative catabolism of polyunsaturated fatty acid and it is an accepted parameter of cellular damage and death. 31 Reactive oxygen species generated in cells may attack directly to DNA and at the same time, they react with polyunsaturated fatty acids in cellular membrane which leads to generation of lipid peroxidation products and ultimately, to cell death. 32 This damage to cell membrane can be determined by measuring several reaction products. A common method is measuring the levels of lipid hydroperoxides which (LOOH) are stable lipid peroxidation products, in biological samples. 33 In our study the toxic effects of hBN NP’s on rats were demonstrated through LOOH measurements. Assays on serum and several tissues types showed that especially 1600 ve 3200 µg/kg hBN NPs doses considerably increased LOOH levels. In rat livers on the other hand, 200 µg/kg hBN NPs dose significantly increased LOOH levels compared to controls. Silver nanoparticles (Ag-NP) were found to cause dose-dependent increases of LOOH levels in rat liver tissue samples. 34 Liver is the main center for xenobiotic detoxification and its Kupffer cells are particularly involved in this task. Cytokines and other molecules released by activated Kupffer cells may induce liver damage. 35 Because of this, significant LOOH levels were observed moderate and higher doses caused significant LOOH increase in liver cells whereas in other tissue samples these significant increases were only observed in highest dose groups.
MPO activity which is a primary marker of neutrophil infiltration and inflammatory processes is widely measured parameter in studies of in vivo nanoparticle toxicity.36,37 In addition to inflammatory marker MPO, disruptions redox balance can also be determined by measuring the activities of CAT, one of the main antioxidant defense system enzymes. 38 We carried out MPO and CAT measurement assays in rat serum and tissue samples to determine toxic effects hBN NPs doses. MPO activities were found to be significantly elevated in 1600 and 3200 µg/kg hBN NPs dose groups but same dose hBN NP’ CAT activities were found considerably increased compared to controls. hese results indicate that high doses of hBN NPs administration disrupts redox balance in favor of oxidative damage which leads to depletion of antioxidant defense system and inflammation in tissues.
We did not encounter any other study about the effects of hexagonal bor nitrur on LOOH, MPO and CAT parameters however, boron nitrite (bor nitrur) was found to have a dose-dependent protective effect against 1-methyl-4-phenylpyridinium (MPP+) toxicity in an in vitro Parkinson model. 5 Against MPP+ toxicology, it was found that cell viability decreased due to increasing dose of boron nitrite and total oxidative stress increased. Boric acid and BN particles were reported to have dose-dependent variable effects on oxidative damage ratio through mediating antioxidant–prooxidant balance. In previous study, when human periferic blood cultures were exposed to 15, 50 and 500 mg/L doses of boric acid, increased SOD, CAT, GSH, GPx ve GST activities were determined in 15 mg/L dose group while 50 and 500 mg/L dose groups increased the levels of malondialdehyde, a lipid peroxidation by product, and decreased total antioxidant levels. 39 In a fetal alcohol syndrome model, authors argued that 100mg/kg moderate dose of boric acid may decrease lipid peroxidation by increase S-adenosylmethionine/S-adenosylhomocysteine ratio and it can also detoxify alcohol derived hydrogen peroxide into peroxy boric acid and, also may decrease by keeping CAT enzyme in its original form. 40 Büyükgüzel et al. observed dose-dependent three to four times increased MDA levels compare to controls in Galleria mellonella specimens treated with 156 or 620 ppm boric acid concentrations. 41 Similarly, 115 mg/kg, 250 mg/kg and 450 mg/kg oral doses of boric acid caused dose-dependent increase of MDA levels and again dose-dependent decrease of GSH levels in testicular tissue. 42
Conclusion
For the first time in scientific literature, we examined the toxic effects of hBN nanoparticles on in both serum and various tissues of rats through thiol/disulphide homeostasis, LOOH, MPO and CAT assays in this study. We found that high concentrations of hBN NPs given to rats crated oxidative stress and toxic effects through modulation of thiol/disulphide homeostasis and induction of inflammation respectively. Our study on acute effects of hBN NPs will be reference point for further studies especially about chronic toxic effects of high dose hBN NPs exposure.
Footnotes
Acknowledgment
The authors would like to thank BORTEK Boron Technologies and Mechatronic Inc. for their material support and Dr Umut SAVACI for his help to use Transmission Electron Microscopy (TEM) (Eskisehir Technical University, Turkey).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Eskişehir Technical University Scientific Research Projects Commission [Project No: 19ADP163].
