Abstract
Colon cancer is a life-threatening disease all over the world and is linked to constant oxidative stress and inflammation. Epigallocatechin gallate (EGCG), is a naturally occurring flavone possessing health benefiting pharmacological properties including antioxidant, anti-inflammatory and free radical scavenging properties. Our study investigates the role of EGCG on N,N′-dimethylhydrazine (DMH), a toxic environmental pollutant, induced colon toxicity. To investigate the effect of EGCG, Wistar rats were given EGCG for 7 days at the two doses of 10 and 20 mg/kg body weight and DMH was injected on the seventh day in all the group rats except the control. Our results indicate that DMH administration increased the oxidative stress (MDA) and depleted the glutathione and antioxidant enzyme activities (SOD, CAT, GR, GST and GPx) which was significantly ameliorated by EGCG treatment. Additionally DMH treatment upregulated inflammatory markers expression (NF-κB, COX-2 and IL-6) and enhanced mucosal damage in the colon. EGCG treatment significantly reduced inflammation and restored the normal histoarchitecture of the colon. We can conclude from the present study findings that EGCG protects the colon from DMH toxicity through its antioxidant and anti-inflammatory potential.
Introduction
Colon cancer is the second cause of cancer-related deaths and the third most common in frequently detected cancers. 1 In epidemiological studies, dietary habits, high fat and protein diet, low intake of fiber, and carbohydrates were considered the major reasons behind an increase in incidences of colon cancer worldwide. 2 However, the cause of colon cancer is not fully known, in several studies, it has been reported that constant condition of oxidative stress and persistent inflammatory response in the colon may lead to colon toxicity, colitis and ultimately to colon cancer.3,4 Intestine serves as the biggest immune organ of the human body and its health condition is necessary for whole-body health. 5 Many studies have found that plant-derived products have the potential to prevent colon toxicity induced by DMH and chemotherapeutic drugs.6,7 Conventional treatments of organ toxicity and chemotherapeutics for cancer prevention has many drawbacks that demand alternatives treatments or methods.8–10 Therefore a need for natural compounds for preventing colon toxicity and search for chemopreventive agents appear.
Green tea is consumed as a beverage in most countries for many decades. Green tea is produced from
DMH and azoxymethane (AOM) are used in chemically induced colon carcinogenesis in rodents. DMH is reported to be more effective in the carcinogenesis model in terms of advanced stages of adenomas and carcinomas. 20 Moreover, In experimental studies, DMH-induced model is developed for exploring the chemopreventive effect and antioxidant properties of plant-derived compounds because it offers identical site and pathological changes similar to human sporadic colon cancer.21,22
Therefore, to combat the DMH-induced colon toxicity or better chemopreventive agents search to use in colon cancer chemopreventions is appeared. As far as we know there is no study, about the ameliorating effect of EGCG on DMH-induced colon toxicity. We designed this study to assess the effect of EGCG on colonic oxidative damage (MDA), the status of various antioxidants such as SOD, CAT, GSH, GST, GPX and GR, expression of inflammatory markers such as NF-κB, COX-2 and IL-6, goblet cell disintegration (Alcian blue staining) and morphological changes in colon tissue (histology).
Material and methods
Chemicals and reagents
Bovine serum albumin (BSA), ethylene diamine tetraacetic acid (EDTA), thiobarbituric acid (TBA), glutathione reductase, reduced glutathione (GSH), nicotinamide adenine dinucleotide phosphate oxidized (NADP), oxidized glutathione (GSSG), 1-chloro-2,4-dinitrobenzene (CDNB), nicotinamide adenine dinucleotide phosphate reduced (NADPH), 5,5′-dithio-bis-[2-nitrobenzoic acid] (DTNB), pyrogallol, poly-Llysine, and N,N′-Dimethylhydrazine dihydrochloride (DMH) were bought from Sigma (Sigma Chemical Co., St Louis, MO). Trichloroacetic acid (TCA), Hydrogen peroxide, Folin–Ciocalteau reagent (FCR), magnesium chloride, sulphosalicylic acid, perchloric acid, tween-20, sodium hydroxide, sodium di-hydrogen phosphate, sodium potassium tartrate, di-sodium hydrogen phosphate, isopropanol and chloroform were purchased from Merck (E. Merck Limited, India). EGCG was obtained from Cayman Chemicals (Ann Arbor, MI, USA). All other reagents and chemicals were of the highest grade and purity commercially available.
Animal maintenance
The animals used in this study were approved by the Institutional Animal Ethics Committee of Jamia Hamdard, New Delhi, India which is certified by the Committee for Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Govt. of India. Animals were obtained from the Central Animal House Facility (CAHF) of Jamia Hamdard, New Delhi. Male Wistar rats, 1-month-old (120–150 g body weight) were housed in propylene encloses under an artificial 12 hour light/dark period at constant room temperature. Animals were given standard chow and tap water ad libitum and left for 7 days to acclimatize before the experiment starts.
Experimental design and treatment protocol
Doses of EGCG and DMH were selected based on previous reports.23–25 Thirty male Wistar rats were allocated randomly into five groups maintaining six animals in each group (Figure 1).

Schematic representation of the experimental design.
Animals of group I (control group) were provided a standard lab diet and received a single injection of PBS intraperitoneally (i.p.) on daily basis for 7 days. Animals of group III, IV and V were given i.p. injection of EGCG at the dose of D1 (10 mg/kg), D2 (20 mg/kg) and D2 (20 mg/kg) respectively daily for 7 days. Animals of groups II, III and IV also received a single subcutaneous injection of DMH with pH 6.5 dissolved in EDTA on the seventh day after the last dose of EGCG. After 24 hours of DMH injection, animals were sacrificed to collect tissue and blood samples to process for experiments.
Post mitochondrial supernatant (PMS) preparation
After dissection rat colon was excised quickly and washed with chilled saline (0.85% NaCl) to make it free of irrelevant materials. Then the colon tissue was homogenized (10% Weight/Volume) using Potter-Elvehjem Homogenizer in chilled phosphate buffer (0.1 M, pH 7.4). The obtained homogenate was processed to disperse the nucleus debris in a Remi cooling centrifuge at 3000 rpm for 10 min at 4°C. The obtained aliquot was further processed in the centrifuge (12000 rpm for 20 min at 4°C) to get Post Mitochondrial Supernatant, which was used as a source of various enzyme assays.
Estimation of biochemical parameters
Estimation of lipids peroxidation (LPO)
The LPO was done by the method of Wright et al.
26
The reaction mixture consisted of 1.0 ml tissue homogenate, 1.0 ml TCA (10%), and 1.0 ml TBA (0.67%) in a total volume of 3.0 ml. Test tubes were kept in a boiling water bath for 45 min. The tubes were placed in an ice bath and then centrifuged at 2,500 ×
Superoxide dismutase (SOD) activity
SOD activity was measured by the method of Marklund and Marklund. 27 In short, the reaction mixture in a total volume of 3 ml contained 2.875 ml Tris–HCl buffer (50 mM, pH 8.5), pyrogallol (24 mM in 10 mM HCl) and 100 ml PMS. The enzyme activity was measured at 420 nm and was measured as units/mg protein.
Catalase (CAT) activity
CAT activity was measured by the method of Claiborne. 28 In short, the reaction mixture in a total volume of 3 ml contained 0.05 ml PMS, 1.0 ml of H2O2 (0.019 M), 1.95 ml phosphate buffer (0.1 M, pH 7.4). The enzyme activity was measured at 240 nm, and CAT activity was expressed as nmol H2O2 consumed/min per mg protein.
Glutathione peroxidase (GPx) activity
The GPx activity was estimated by the method of Mohandas et al. 29 In short, the reaction mixture consisted of 0.1 ml EDTA (1 mM), 0.1 ml sodium azide (1 mM), 1.44 ml phosphate buffer (0.1 M, pH 7.4), 0.05 ml GR (1 IU/ml), 0.05 ml GSH (1 mM), 0.1 ml NADPH (0.2 mM) and 0.01 ml H2O2 (0.25 mM) and 0.1 ml 10% PMS in a total volume of 2 ml. The depletion of NADPH at 340 nm was recorded at 25°C. The enzyme activity was measured as nmol NADPH oxidized/min per mg protein with the molar extinction coefficient of 6.22 × 103/M per cm.
Estimation of glutathione (GSH)
GSH estimation was determined by the method of Jollow et al. 30 A volume of 1.0 ml of 10% PMS mixed with 1.0 ml of (4%) sulphosalicylic acid, incubated at 4°C for a minimum time of 1 hour and then centrifuged at 4°C at 1200 g for 15 min. The reaction mixture of 3.0 ml consisted of 0.4 ml of supernatant, 2.2 ml phosphate buffer (0.1 M, pH 7.4) and 0.4 ml dithio-bis-2-nitrobenzoic acid (4 mg/ml). The yellow color developed was read immediately at 412 nm. GSH concentration was measured as nmol GSH conjugates/g tissue.
Glutathione-S-transferase (GST) activity
GST activity was measured by the method of Habig et al. 31 A total volume of 3.0 ml reaction mixture contained 2.5 ml phosphate buffer (0.1 M, pH 6.5), 0.2 ml GSH (1 mM), 0.2 ml 1-chloro-2,4-dinitrobenzene (1 mM) and 0.1 ml of the cytosolic fraction (10%). Changes in absorbance were read at 340 nm, and enzymatic activity was expressed as µmol 1-chloro-2,4-dinitrobenzene conjugated/min per mg protein using a molar extinction coefficient of 9.6 × 103/M per cm.
Glutathione reductase (GR) activity
GR activity was measured by the method of Carlberg and Mannervik. 32 In a total volume of 2 ml the reaction mixture contained 1.65 ml phosphate buffer (0.1 M, pH 7.6), 0.1 ml NADPH (0.1 mM), 0.05 ml GSSG (1 mM), 0.1 ml ethylenediaminetetraacetic acid (EDTA; 0.5 mM), and 0.1 ml 10% PMS. Enzyme activity was recorded at 25°C by measuring the disappearance of NADPH at 340 nm and was expressed as a nanomole of NADPH oxidized per minute per milligram protein using a molar extinction coefficient of 6.22 × 103 M−1 cm−1.
Estimation of protein
The protein concentration was estimated by the method of Lowry et al. 33 Protein content in all samples was estimated using BSA as standard.
Immunohistochemical (IHC) evaluation
Immunohistochemistry was done as described earlier. 34 Paraffin-embedded formalin-fixed tissues were sliced to make 5 µm thin sections which were smeared on poly-L-lysine coated microscopic slides. Sections were deparaffinized in xylene three times and rehydrated through ethanol to water. For antigen retrieval, sections were placed in 10 mM sodium citrate buffer (pH 6.0) and boiled at 95°C for 5 minutes. To limit non-specific staining, sections were incubated in 3% H2O2 for 20 minutes then washed three times with TBS (0.05% Tween 20). Sections were smeared with blocking solutions and washed with TBS then sections were incubated with recommended dilution primary antibodies, purified rabbit polyclonal antibody (Thermo Fischer) overnight at 4°C in a humidified chamber. Primary antibodies used for overnight incubation are NF-κB p65 antibody (dilution 1:100, Biolegend, catalog # 622601), anti-rat COX-2 polyclonal antibody (dilution 1:200, Abcam, catalog # ab 15191), anti IL-6 antibody (dilution 1: 100, Abcam, catalog # ab 7737). Sections after washing with TBS, incubated with a secondary antibody (Ultra vision plus detection system) for 25 minutes. Sections were again washed with TBS and incubated with streptavidin peroxidase plus (Ultra vision plus detection system, catalog # TP-015-HDX). Sections were processed to develop brown color with 3,3′-Diaminobenzidine then counterstained with Mayer’s Hematoxylin solution. Sections were allowed to dry then mounted with DPX, covered with a coverslip, and examined under the light microscope (Olympus BX51).
Alcian blue staining for detection of goblet cells
Paraffin-embedded formalin-fixed colon tissues were sliced into sections of 5 µm thickness. Sections were placed on microscopic slides that were coated with poly-L-lysine. Tissue sections were processed in xylene for de-paraffinization and then transferred to graded series of ethyl alcohol to water for rehydration. Tissue sections were processed in 3% acetic acid then stained in 1% alcian blue for half an hour. After washing sections with distilled water, sections were counterstained with neutral red for 5 minutes and dehydrated in alcohol. After mounting, slides were covered with a coverslip and examined using the light microscope (Olympus BX51) at 40× magnification. 34
Histology
The colon tissue was removed, cut longitudinally, and washed with saline. Colon tissues were fixed in 10% formalin and after 24 hours tissues were processed to make tissue sections. Tissue sections were stained with Hematoxylin and counterstained with Eosin then visualized in the microscope for histological examinations. 24
Quantitative evaluation of NF-κB, COX-2 and IL-6
Quantitative evaluation of NF-κB, COX-2, and IL-6 was done by the immune reactive scoring (IRS) method. 35 The IRS method is based on the scoring of intensity and quantity of immune staining in tissue. Following the staining intensity sections were graded as no reaction (0), mild (1), moderate (2), and intense (3) and the percentage of positive cells were evaluated as 0 (no positive cells), 1 (<10% positive cells), 2 (10–50% positive cells), 3 (51–80% positive cells), and 4 (>80% positive cells). The IRS was obtained by multiplying the staining intensity score and percentage of positive cells score. Finally, every tissue sample was classified into negative (IRS points 0–1), mild (2–3), moderate (4–8) or strong (9–12). All slides were examined by two independent observers who were unaware of the experimental protocol. Observation were done using a light microscope (Olympus BX15) at 40× magnification.
Statistical analysis
For statistical analysis, the data is presented as the mean ± SE of the individual groups. One way analysis of variance (ANOVA) was used to analyze differences between groups followed by Tukey’s multiple comparisons test. The Kruskal–Wallis test, followed by Dunn’s multiple comparison test, was used for immunohistochemical analysis. Values of p < 0.05 was set to be the minimum criterion for statistical significance. Statistical analysis was carried out using GraphPad Prism software (GraphPad Software Inc. V6.0c, San Diego, USA).
Results
EGCG reduces membrane damage (LPO)
MDA is the product of lipid peroxidation and a marker of oxidative damage. In colonic tissue of group II, DMH administration has significantly increased (***p < 0.001) the MDA level as compared to the control group while, pretreatment of EGCG at both the doses (10 mg/kg and 20 mg/kg) in group III and group IV significantly decreased (###p < 0.001 and ###p < 0.001) the MDA level as compared to group II. However, there were no significant changes in the MDA level in group V found (Figure 2).

EGCG reduces membrane damage (LPO) in the colon. MDA level was significantly elevated (***p < 0.001) in the DMH-treated group (II) as compared to the control group (I). EGCG treatment at both the doses (10 mg/kg and 20 mg/kg) reduced MDA level in group III and IV significantly (###p < 0.001 and ###p < 0.001) as compared to group II. There were no significant changes found between only EGCG treated group (V) and the control group. Results represent the mean ± SE of six animals per group. LPO was measured as nmol MDA formed/g tissue.
EGCG increases the activities of antioxidant enzymes
The effect of EGCG on SOD and catalase enzyme activities were observed. DMH exposure has significantly diminished (***p < 0.001) the activities of SOD and CAT in group II as compared to group I. EGCG treatment at both the doses significantly enhanced the activities SOD (###p < 0.001 and ###p < 0.001) and CAT (#p < 0.05 and ###p < 0.001) in group III and IV as compared to group II. In group V there were no significant differences found as compared to group I (Figure 3).

EGCG increases the activities of antioxidant enzymes SOD (a) and CAT (b) in the colon. Activities of SOD and CAT were significantly diminished (***p < 0.001) in the DMH-treated group (II) as compared to the control group (I). EGCG treatment at both the doses showed a significant increase in the activities of SOD (###p < 0.001 and ###p < 0.001) and CAT (#p < 0.05 and ###p < 0.001) when compared to group II. There were no significant changes found between only EGCG treated group (V) and the control group. Results represent the mean ± SE of six animals per group. SOD and CAT enzyme activities were measured as units/mg protein and nmol H2O2 consumed/min/mg protein respectively.
EGCG restores the level of GSH and GST, GPX and GR enzyme activities
The effect of EGCG on GSH level and GST, GPX and GR enzyme activities were determined. There were significant differences (***p < 0.001) in the level of GSH and GST, GPX and GR enzymes activities between DMH-treated group (II) and the control group (I). EGCG treatment at both the doses has significantly elevated the level of GSH (###p < 0.001 and ###p < 0.001), GST (#p < 0.05 and ##p < 0.01), GPx (#p < 0.05 and ###p < 0 .001) and GR (##p < 0.01 and ###p < 0.001) in group III and group IV colon tissues when compare to group II. There were no significant changes found in group V as compared to group I (Figure 4).

EGCG restores the level of GSH (a) and activities of GST (b), GPx (c) and GR (d) in the colon. DMH-treated group (II) exhibited a significant decline (***p < 0.001) in GSH, GST, GPx and GR as compared to control group (I). EGCG treatment at both the doses showed a significant increase in the level of GSH (###p < 0.001; ###p < 0.001), GST (#p < 0.05 and ##p < 0.01), GPx (#p < 0.05 and ###p < 0.001) and GR (##p < 0.01 and ###p < 0.001) when compared to group II. There were no significant changes found between only EGCG treated group (V) and the control group. Results represent the mean ± SE of six animals per group. GSH, GST, GPx and GR were measured as nmol GSH conjugates/g tissue, μmol CDNB conjugated/min/mg protein, mmol NADPH oxidized/min/mg protein respectively.
EGCG attenuates the goblet cell disintegration
Goblet cells produce mucin which protects the colon. Effect of EGCG pretreatment on DMH-induced disintegration of goblet cells was estimated by Alcian blue staining and results were found. DMH exposure leads to heavy destruction of goblet cells in group II as compared to group I. EGCG treatment in group III and group IV showed a reduction in goblet cell disintegration in comparison to group II. However, no differences in goblet cell disintegration were found in group V in comparison to group I (Figure 5).

Photomicrograph showing the effect of EGCG on the disintegration of goblet cells estimated by Alcian blue staining. DMH-treated group (II) exhibited heavy destruction of goblet cells as compared to control group (I) while EGCG treatment has prevented in groups III and IV as compared to group II. However, no differences in goblet cell disintegration were found in group V in comparison to the group I.
EGCG suppresses inflammation
The effect of EGCG on inflammatory markers (NF-κB, COX-2 and IL-6) were assayed by IHC evaluation. Results revealed that DMH exposure has increased the expression of these inflammatory markers in comparison to the control group. While EGCG treatment reduced the expression of these inflammatory markers in group III and group IV (Figures 6 to 8).

Photomicrograph showing immunohistochemical staining of NF-κB p65 (a). The colonic section of the DMH-treated group (II) has a higher expression of NF-κB than the control group (I). EGCG treatment in groups III and IV has reduced the expression of NF-κB when compared with group II. Scatter-dot plot showing the immuno-reactive score of NF-κB p65 and median is indicated by a line in the same groups (b). Significance was set **p < 0.01 when compared with group I, ns: nonsignificant and #p < 0.05, when compared with group II using Kruskal–Wallis followed by Dunn’s multiple comparison test (magnification: 40× and n = 6).
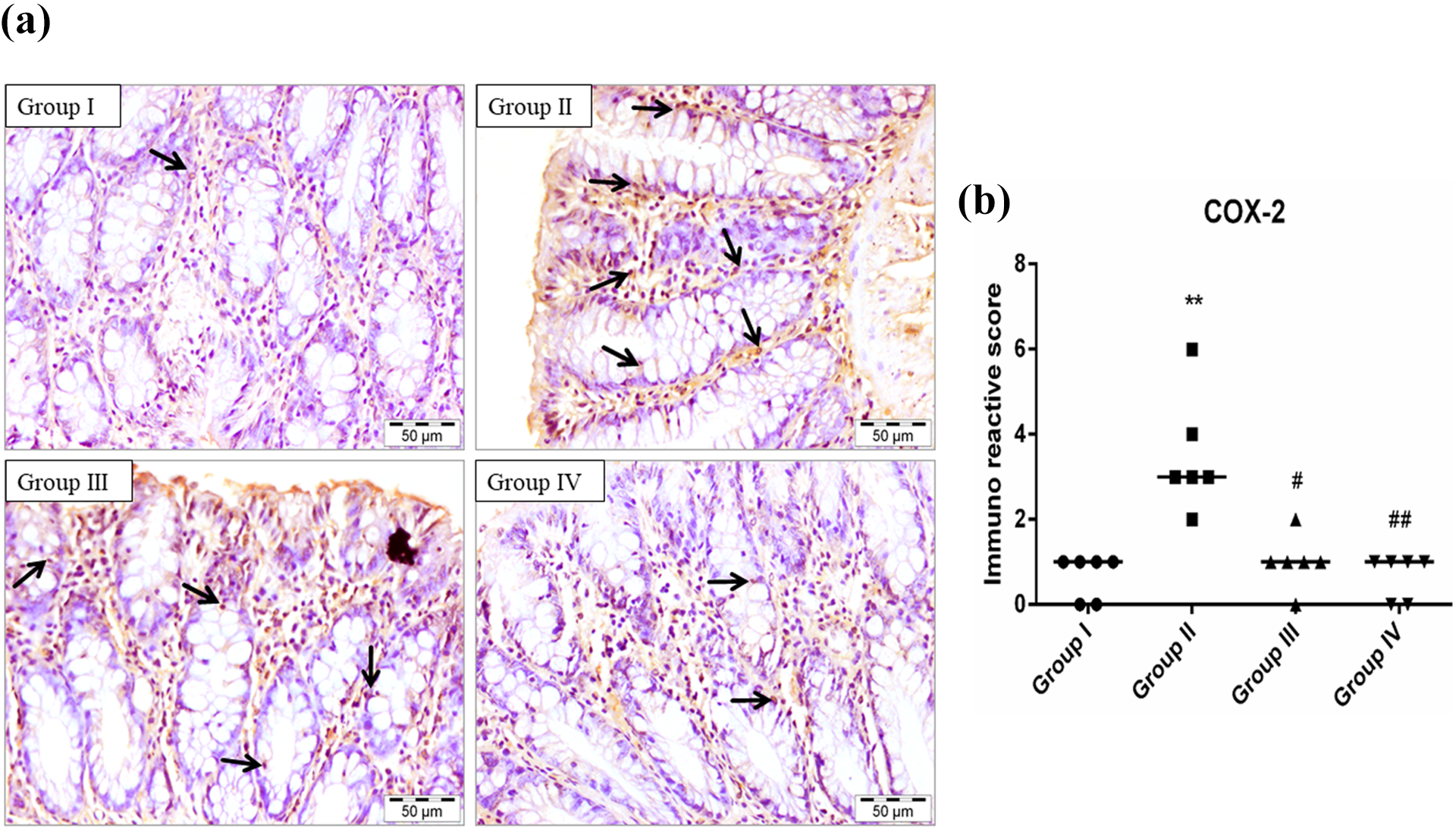
Photomicrograph showing immunohistochemical staining of COX-2 (a). The colonic section of the DMH-treated group (II) has a higher expression of COX-2 than the control group (I). EGCG treatment in groups III and IV has reduced the expression of COX-2 when compared with group II. Scatter-dot plot showing the immuno-reactive score of COX-2 and median is indicated by a line in the same groups (b). Significance was set **p < 0.01 when compared with group I, #p < 0.05 and ##p < 0.01, when compared with group II using Kruskal–Wallis followed by Dunn’s multiple comparison test (magnification: 40× and n = 6).
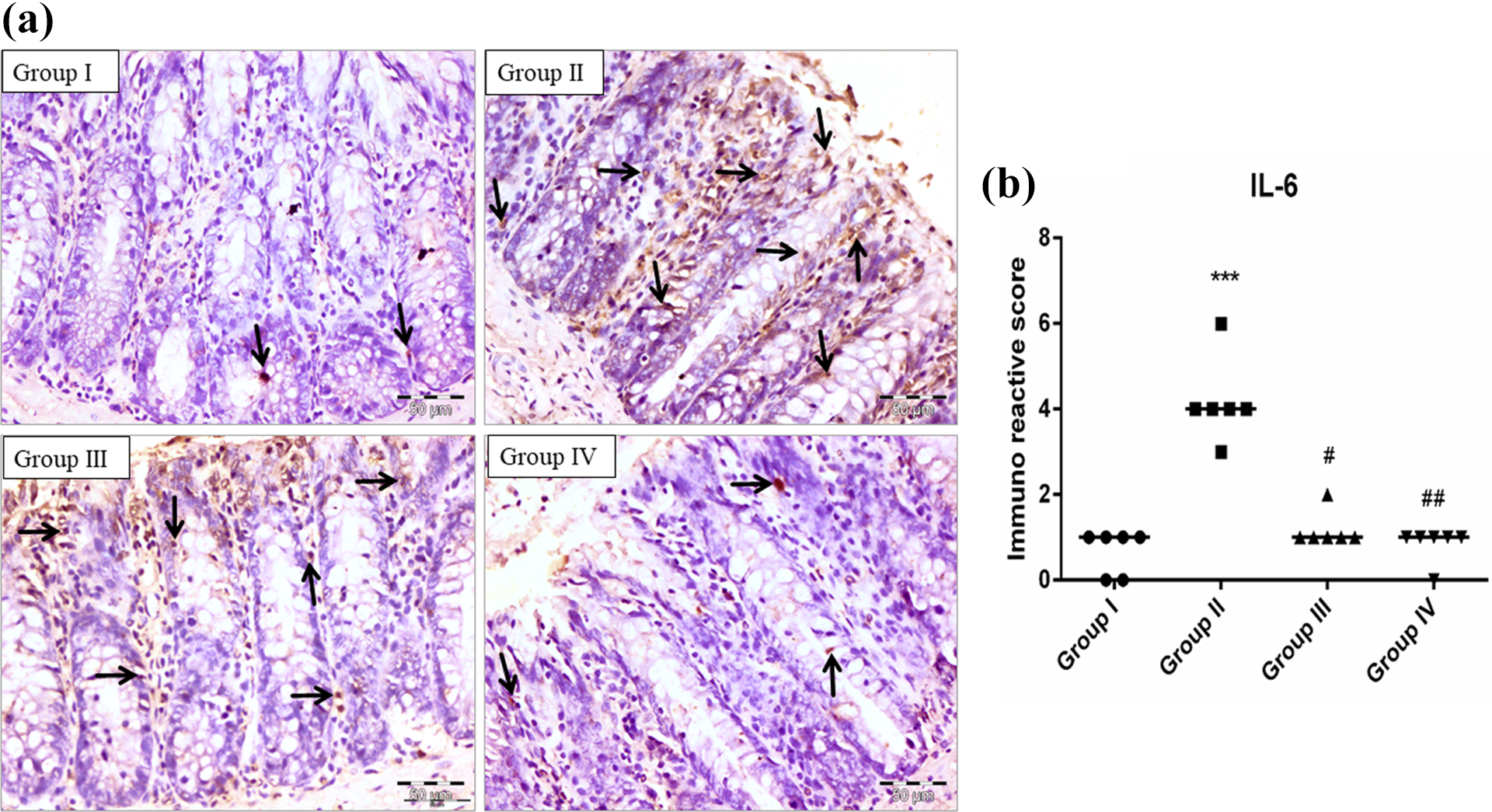
Photomicrograph showing immunohistochemical staining of IL-6 (a). The colonic section of the DMH-treated group (II) has a marked high expression of IL-6 than the control group (I). EGCG treatment in groups III and IV has reduced the expression of IL-6 when compared with group II. Scatter-dot plot showing the immuno-reactive score of IL-6 and median is indicated by a line in the same groups (b). Significance was set ***p < 0.001 when compared with group I, #p < 0.05 and ##p < 0.01 when compared with group II using Kruskal–Wallis followed by Dunn’s multiple comparison test (magnification: 40× and n = 6).
EGCG attenuates mucosal damage
Histological analysis of Hematoxylin & Eosin stained sections revealed the colonic damages. Sections of group I showed normal histoarchitecture of the colon and in sections of DMH-treated group II there is regional damage of mucosal layer, crypt ablation, and extreme infiltration of inflammatory cells were detected. While EGCG treatment has attenuated mucosal damage and crypt ablation in group III and group IV. There was a similar pattern of colon histoarchitecture found in group V when compared to group I (Figure 9).

Photomicrograph showing H&E staining of colonic sections. The colonic section of the control group (I) exhibited normal histology of the colon with mild infiltration of inflammatory cells (arrow). Colonic section of DMH treated group (II) have mucosal damage (triangle) crypt ablation (star) with irregular glandular structure and intense inflammatory cells, while colonic sections of EGCG treated groups (III and IV) showed moderate mucosal damage and less irregular glandular structure. However, only EGCG treated group (V) exhibited normal histology similar to group I.
Discussion
In recent times, various natural compounds possessing antioxidant and chemopreventive properties have been studied for the treatment and prevention of colon toxicity and cancers. 36 Gallic acid naturally found in figs, tea and berries with antioxidant activities have displayed the potential in the prevention of colon toxicity in Wistar rats. 6 Similarly, Naringenin flavonoid found in citrus fruits has demonstrated chemopreventive efficacy due to antioxidant and anti-inflammatory properties in experimentally induced colon cancer in rats. 7 Diet with natural compounds has been suggested as a better remedy for the amelioration of organ toxicity and in the prevention or delaying of cancer. Daily food ingestion has many beneficial and pharmacologically active compounds within and benefits the human body. In this study, we have reported that EGCG has protective efficacy in DMH-induced colon damage via its antioxidant and anti-inflammatory properties.
DMH and its isomers are mainly exposed to humans via surrounding nature and food chain. 6 Exposure to DMH creates teratogenic, mutagenic and embryo-toxic effects which lead to serious troubles to an individual’s health. 37 DMH is a pro-carcinogen and requires its metabolic activation in the liver before converting into an active metabolite. In the liver after various steps of N-oxidation and hydroxylation, DMH transformed into methyl azoxy methanol (MAM) metabolite. In the colon, MAM is cleaved by colonic enzymes and ultimately forms the methyl diazonium ion, an alkylating compound that is the root of mutagenesis and carcinogenesis in the colon. 38 This compound further leads to the formation of methyl free radicals and hydroxyl radicals and generates oxidative stress. 24
Oxidative stress occurs when the production of reactive oxygen species (ROS) goes above the protective potential of armory antioxidants in the body. 39 Earlier studies have established that oxidative stress and inflammation are linked to organ toxicity and eventually to cancer.40,41 Exposure of DMH also leads to the generation of peroxide radicals, superoxide radicals, and ROS which causes peroxidation of cell membrane lipids and forms malondialdehyde (MDA), a key marker of oxidative damage. 24 MDA has been found to react with DNA to form DNA adducts which are mutagenic and showed its role in carcinogenesis including colon cancer. 41 In this study, we also found enhanced MDA level in DMH-treated animals that are in support of previous findings. 24 However, EGCG treatment has diminished the MDA level in rat colon which may be due to its protective ability against oxidative damage and is consistent with previous reports. 42
Enzymatic and non-enzymatic antioxidants play a crucial role in protecting against free radicals, ROS, and oxidative stress in the living system. We evaluated the activity of SOD, CAT, GSH, GST and GR in DMH-treated group to advocating the antioxidant role of EGCG. Our results showed that DMH treatment depleted the activity of SOD, CAT, GSH, GST and GR as reported earlier. 43 However, EGCG treatment has ameliorated the activity of GSH, GST, GR, SOD, and CAT in the colon that support the defending capacity of EGCG against oxidative stress which is in agreement with previous results of EGCG. 44 Antioxidant activity of EGCG in this study may be related to its metal chelating property and formation of stable conjugates binding with thiol-bearing compounds. 45
Inflammation has been extensively studied for implications in the pathophysiology of DMH-induced colonic mucosa damage and cancer. 46 An excessive amount or overproduction of ROS causes the constant damage of cells in tissue and continued state subsequently results in inflammation. 47 Inflammatory cells generate chemokines and cytokines soluble mediators which can activate nuclear factor-kappa B (NF-κB), a protein that stimulates downstream proteins involved in inflammation. 48 In non-activated form, NF-κB dimers remain connected to an inhibitor of kappa B (IκB) and upon stimulation IκB kinase (IκK) phosphorylates the NF-κB and IκB complex. The further step involves ubiquitination then degradation of IκB which activates NF-κB and translocates NF-κB to the nucleus. 49 It has been considered that the modulation of the NF-κB pathway could be beneficial in the prevention of intestinal inflammation as NF-κB over activation is reported in inflamed colon tissue. 50 In this study, we found upregulated expression of NF-κB in DMH-treated animals that are in support of earlier findings. 6 EGCG treatment has downregulated the NF-κB in DMH-treated animals which are in agreement with previous findings on the anti-inflammatory effect of EGCG on cigarette smoke-induced inflammation in cardiomyocytes via inhibition of NF-κB. 51
NF-κB activation involves upregulation of downstream proteins such as COX-2 and pro-inflammatory cytokine IL-6 which are crucial in inflammation. 52 COX-2 is commonly over-activated in the tumors in all main types of cancers and COX-2 over activation stimulates carcinogenesis, cancer incidences, and lower survival chances. 53 It was evaluated that COX-2 expression was upregulated in experimental colon carcinogenesis and targeting with COX-2 inhibitor reduces the incidence and severity of colon cancer. 54 So, in this study, we evaluated the COX-2 expression and found that DMH treatment enhanced the expression of COX-2 that is in support of previous findings. 54 EGCG treatment reduced the expression of COX-2 in the colon which is in agreement with the previous findings of the anti-inflammatory effect of EGCG on doxorubicin-induced inflammation in human ovarian tissue. 55
A pro-inflammatory mediator, IL-6 has a significant function in inflammation. It has been found to induce inflammatory lesions and leukocyte presence during chronic inflammation. A genetic link incriminated between IL-6 and intestinal lesions and accepted that inhibition of IL-6 activity results in the reduction of colon cancer. 56 It is considered that stimuli that activate IL-6 are commonly related to tissue injury or stress. 57 In this study, DMH treatment elevated the expression of IL-6 in colon tissue that is in support of earlier findings. 34 While EGCG treatment downregulated the expression of IL-6 in colon tissue which is in agreement with the previous findings of EGCG effect on inhibition of IL-6 synthesis. 58 This anti-inflammatory effect of EGCG supports that a decrease in oxidative stress results in reduced production of pro-inflammatory cytokines (IL-6) which is evident with anti-inflammatory response and restoration of antioxidant enzyme level by EGCG. 11 Our results demonstrated that EGCG offers an effective anti-inflammatory impact on the colon via downregulating the expression of NF-κB, COX-2, and IL-6 in DMH-induced toxicity.
It has been evaluated that initial events associated with intestinal inflammation and histological changes include loss of goblet cells or mucin secretion. 59 It is well known that DMH toxicity causes the destruction of goblet cells which leads to colon damage. 60 Goblet cells named due to their cup-like looks, packed with mucin in the cytoplasm have specialized in the production and secretion of mucus. 61 Mucus is gel-like secretion mostly formed of water and mucin, a high molecular weight glycosylated protein that forms a protective layer in the intestine. 62 It has been suggested that upon stress stimuli goblet cells secrete mucin and persistent stress limits mucin accessibility. 63 In this study, DMH treatment result in immense damage to goblet cells in colon tissues that support the previous findings. 21 EGCG supplementation restored the destruction of goblet cells and mucin depletion which is in agreement with previous findings of EGCG on goblet cells protection in dry eye syndrome and hypersecretion of mucin in rats.64,65
Further histological investigation proves that the DMH treatment produced the distorted glandular structure, mucosal damage, and crypt ablation with intense infiltration of inflammatory cells in colon tissue which confirms colon injury and is in support of the earlier findings. 6 EGCG supplementation has prevented the DMH-induced colonic damages and restored normal histology of the colon which is in agreement with the protective effect of EGCG on renal injury in rats. 66
Conclusion
From the above findings, it can be concluded that EGCG protected the colon from DMH toxicity via ameliorating oxidative stress, antioxidants and inflammatory markers. This pavement the EGCG role as an antioxidant and anti-inflammatory agent and can be used in oxidative damage and inflammation generated gastric or colonic diseases. However further investigations are required to explore the molecular mechanisms of EGCG implicated in the amelioration of DMH-induced colon toxicity.
Footnotes
Acknowledgment
The authors gratefully acknowledge Dr Haider A Khan (Associate Professor), JH and Professor RP Singh, JNU for their suggestions and valuable comments. The authors are also thankful to the late Dr AK Tiwari, for his kind support in performing the histopathological studies. We also acknowledge the University Grant Commission (UGC), New Delhi, India for providing UGC BSR fellowship and Indian Council of Medical Research (ICMR), New Delhi, India for providing SRF fellowship (File No. 3/2/2/22/2019-NCD-III) to the first author Shekh Mohammad Afzal.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
