Abstract
A positive association of lead exposure with clinical cardiovascular outcomes has been identified. Epigallocatechin-3-gallate (EGCG) is one of the active polyphenols in green tea, it has not been reported as an antioxidant against lead toxicity. This study was carried out to investigate whether EGCG could protect the ventricular myocytes of rats against lead-induced oxidative damage. Isolated ventricular myocytes were exposed to lead and/or EGCG, then activities of superoxide dismutase (SOD), catalase (CAT), and malondialdehyde (MDA) and the levels of hydroxyl radical (OH·), hydrogen peroxide (H2O2), and superoxide anion (O2 ·−) were measured. The results showed that lead induced a significant decrease of SOD and CAT activities, while the levels of MDA increased significantly. Increases in intracellular OH·, O2 ·−, and H2O2 were found as well. These processes were concentration-dependent and statistically significant different when compared to 2.0 µM lead exposure. The activity of SOD and CAT increased while the levels of MDA and reactive oxygen species (ROS) decreased after treatment with EGCG. While there were progressive benefits with increasing EGCG concentrations, there was no statistical significance at a 30 µg/mL dose when compared with the control. These results will provide more evidence for lead toxicity to tissue, cell, and biological macromolecule.
Introduction
Lead is one of the most abundant heavy metals polluting the environment. It originates from numerous sources and has become an environmental concern due to its human health hazards. 1 Lead is toxic to many organs and was associated with many diseases, it has been observed that the levels of lead were high in the scalp hair samples of patients with chronic kidney disease, 2 diabetes mellitus, 3 and myocardial infarction. 4 A positive association of lead exposure with clinical cardiovascular outcomes has been identified since the links between lead exposure and cardiovascular outcomes were first reported 100 years ago. 5,6 Lead was shown to induce highly reactive oxygen species (ROS) such as O2 ·−, OH·, H2O2, and lipid peroxides (LPO°). 7 Until now, lead toxicity was attributed to oxidative stress by ROS 8,9 ; however, Navas-Acien proposed that the evidence was suggestive but insufficient to infer a relationship between lead exposure and clinical cardiovascular outcomes. 10
Epigallocatechin-3-gallate (EGCG) as a natural antioxidant can protect cardiovascular system. Green tea contains many polypenolic compounds that are generally known as catechins. These compound include (−) epicatechin (EC), (−) epigallocatechin (EGC), (−) epicatechin gallate (ECG), and (−) epigallocatechin-3-gallate (EGCG). EGCG is the most active of the major polyphenols and is primarily responsible for the effects of green tea. 11,12 A wealth of literature has suggested EGCG has the most potent free radical scavenging abilities among catechins. 13–15 Many studies have described the effectiveness of EGCG inhibition of the LPO, and as a scavenger of H2O2 and O2 .- radicals in vitro. The protective mechanism of EGCG on the cardiovascular system has been studied 100 years ago. The most popular hypothesis is that there is a direct antioxidant effect by means of polyphenol interaction with O2 ·−, OH·, H2O2, and other ROS. 16
The goal of this study was to determine whether the lead toxicity on ventricular myocytes is a result of oxidative stress. To achieve this goal, selected oxidative stress parameters were determined in ventricular myocytes of rats. Malondialdehyde (MDA) was used as an endpoint of lipid peroxidation. Superoxide dismutase (SOD) and catalase (CAT) activity were evaluated as important components of the intracellular defense system of ventricular myocytes against oxidative attack. The levels of OH·, O2 ·−, and H2O2 were analyzed and used as indices of oxidative stress related to lead toxicity.
Methods
Cell isolation
Single ventricular myocytes were obtained from the hearts of adult rats (250−300 g) by a modified enzymatic dissociation technique. Rats were purchased from Experimental Animal Center of Shanxi Medical University (Grade II, Certificate No. 070101). The animals were stunned with a heavy blow to the head, and the hearts quickly excised and mounted on a Langendorff perfusion apparatus for perfusion through the aorta. Hearts were perfused for 5 minutes at 37°C with a nominally Ca2+-free Tyrode’s solution containing (mM): NaCl 137, KCl 5, MgCl2 1, NaH2PO4 0.33, CaCl2 1.8, HEPES 10, glucose 10, and with a pH 7.4. The perfusate was then replaced with enzymatic digestion which was initiated by 25 minutes of perfusion with 50 mL Ca2+-free Tyrode’s solution containing 15 mg collagenase (Type P, Boehringer Mannheim, Roche). At the end of enzyme perfusion, the heart was sequentially washed with 0.2 mM Ca2+ Tyrode’s solution containing 1 mg/mL bovine serum albumin (50 mL), then the heart was removed from the cannula. The atria were discarded and the ventricles were chopped and stirred in a small vessel in ‘Krafteburhe’ (KB) solution containing (mM): L-Glu 50, KCl 30, Tau 20, KH2PO4 30, MgCl2 1, HEPES 10, glucose 10, EGTA 0.5, and with a pH 7.4. The striated myocytes dissociated from the tissue pieces. Myocytes were harvested after filtering the cell-containing suspension through a nylon mesh (200 μm). They were washed three times in storage solution and then maintained at room temperature in KB solution.
The study involving experimental animals was conducted in accordance with national and institutional guidelines for the protection of animal welfare.
Experimental design
Myocytes dissociated from the tissue pieces were exposed to Tyrode’s solution containing lead (Pb2+ from lead nitrate) and/or EGCG for 1 hour, centrifuged at 4000 rpm at 4°C for 10 minutes, washed three times using Tyrode’s solution; finally, a 1 mL aliquot of ventricular myocytes suspension was vortexed strongly to break the membranes. The mixture was then centrifuged at 15,000 rpm at 4°C for 15 minutes and supernatant obtained was used for assays of SOD, CAT, MDA, and total protein. Lead nitrate (≥99.8%) and EGCG (≥99.8%) were purchased from Sigma and dissolved in the extracellular solution. Stock solution of lead nitrate (Pb2+) 2.0 mol/L and 2.0 mg/mL EGCG were prepared. Lead concentrations of 1.0, 2.0, 5.0 µM and EGCG of 10, 30, 50 µg/mL were used for the test experiment, which was replicated three times.
Assay of H2O2, OH·, and O2 ·−
The levels of H2O2, OH·, and O2 ·− were determined spectrophotometrically according to the methods described by Wei et al. 17
Assay of SOD
Total SOD activity in ventricular myocytes was determined using modification of a previously described method. Briefly, the reaction mixture contained 20 μM cytochrome c and 50 μM xanthine in 50 mM phosphate buffer (pH 7.8); the reaction was initiated with xanthine oxidase. SOD activity was determined by adding lysate containing 40 μg of protein to the reaction mixture. In order to standardize SOD activity, a curve was generated by plotting known concentrations of bovine SOD. The results of this enzymatic assay were shown in units of SOD (U/mL) where 1 unit of SOD was defined as the amount able to inhibit 50% cytochrome c reduction at 25°C. Absorbance measurements were recorded at 550 nm with a Hitachi model U-3010 dual beam spectrophotometer (Hitachi Instruments, San Jose, California, USA).
Assay of CAT
Catalase activity in ventricular myocytes was assayed according to the following procedure at room temperature. A tube containing tissue extract (100 µL) was placed in an ice bath for 30 minutes and then for another 30 minutes at room temperature. To each tube was added Triton-X 100 (10 µL). In a cuvette containing a mixture of phosphate buffer (200 µL) and tissue extract (50 µL) was added 0.066 M H2O2 in phosphate buffer (250 µL), and a decrease in optical density was measured at 240 nm for 60 seconds. The molar extinction coefficient of 43.6 M/Cm was used to determine CAT activity. One unit of activity was equal to the µmol of H2O2 degraded/min per mg protein.
Lipid peroxidation determination
The lipid peroxidation product in ventricular myocytes was determined by thiobarbituric acid reactive substances (TBARS) expressed as the extent of MDA production. 18 LPO was expressed as mg of MDA formed/mL cell suspension.
All results were expressed as mean ± standard deviation or mean ± standard error of the mean in figures. The analysis of variance (ANOVA) was used to determine whether significant difference existed between the control and treatment groups. A value of p <0.05 was chosen as the minimal level of statistical significance.
Results
Change of activities of antioxidant enzymes during exposure of lead and EGCG
The results in Figure 1 showed the effects of lead on the activity of SOD and the protective effect of EGCG. Lead exposure alone at different concentrations led to a decrease in SOD activities in ventricular myocytes of rats. SOD activities decreased with an increase in lead concentration (control at 49.44 ± 0.45 U/mL; 48.37 ± 1.29 at 1.0 µM; 39.66 ± 1.25 U/mL at 2.0 µM; and 25.01 ± 1.21 U/mL at 5.0 µM) in a notable concentration-dependent manner (r > 0.99). Compared with the control, a significant increase in SOD activity was observed in ventricular myocytes exposed to 2.0 µM lead (p < 0.05; Figure 1a). In the EGCG with lead exposure group, SOD activities increased in correlation with EGCG concentration (from 10, 30, to 50 µg/mL). There was no significant difference at EGCG 30 µg/mL compared with the control (p > 0.05), but the difference was significant when compared with the 2.0 µM lead exposure group (Figure 1b).
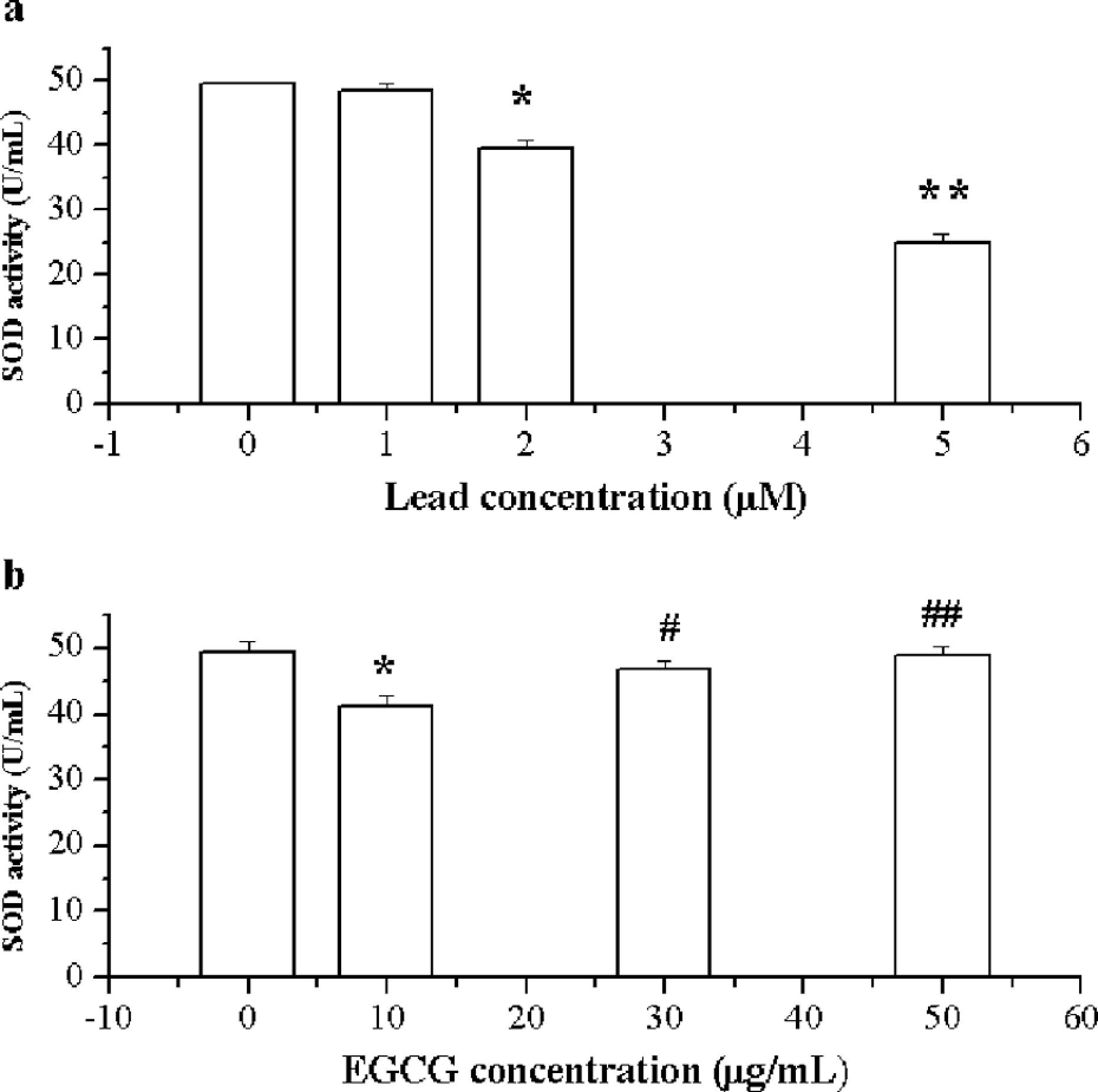
Effects of lead and/or epigallocatechin-3-gallate (EGCG) on superoxide dismutase (SOD) activity. A, Effects of lead on SOD activity. B, Effects of lead and EGCG on SOD activity. *p < 0.05, **p < 0.01 vs control, #p < 0.05, ##p < 0.01 vs lead 2.0 μM exposure.
Figure 2 showed the effects of lead on CAT activity and the protective effects of EGCG in ventricular myocytes. CAT activity increased slightly in the low lead exposure group (1.0 µM); however, there was a statistically significant decrease in lead 2.0 µM exposure group. Lead 5.0 µM exposure significantly inhibited the CAT activity by nearly 67% (Figure 2a). When ventricular myocytes exposed to lead were treated simultaneously with EGCG, the enzyme activity was inhibited by only 25% in the group treated with 30 µg/mL EGCG. CAT activity was increased 0.7-fold when compared to the lead 2.0 µM value. Lead exposure was unable to inhibit CAT activity and there was no statistically significant difference in the group treated with 50 µg/mL EGCG compared with the control, but the difference was significant compared with the lead 2.0 µM exposure group (p < 0.05 or 0.01; Figure 2b).

Effects of lead and/or epigallocatechin-3-gallate (EGCG) on catalase (CAT) activity. A, Effects of lead on CAT activity. B, Effects of lead and EGCG on CAT activity. *p < 0.05, **p < 0.01 vs control, #p < 0.05, ##p < 0.01 vs lead 2.0 μM exposure.
Effects of EGCG on LPO Level
MDA was measured as an important parameter in evaluating oxidative stress induced by lead. In ventricular myocytes exposed to 1.0, 2.0, 5.0 µM lead, the LPO level increased and the effect reached statistical significance in 1.0 exposure group (p < 0.05; Figure 3a). After subsequent EGCG treatment, the MDA level became reversed and it was at nearly the normal level with 50 µg/mL EGCG. The differences between the 2.0 µM lead exposure group and both EGCG treatments at 30 and 50 µg/mL were statistically significant (p < 0.05 or 0.01; Figure 3b).

Effects of lead and/or epigallocatechin-3-gallate (EGCG) on level of malondialdehyde (MDA). a, Effects of lead on level of MDA. b, Effects of lead and EGCG on level of MDA. *p < 0.05, **p < 0.01 vs control, #p < 0.05, ## p < 0.01 vs lead 2.0 μM exposure.
Effects of EGCG on OH· formation
The results in Figure 4a showed the effects of lead on OH· formation. Lead resulted in an increase of intracellular OH· in a concentration-dependent manner. The increase was significant at low concentration (lead 2.0 µM) compared with the control (P<0.05). After the treatment with 10, 30, or 50 µg/mL EGCG, OH· decreased with the increase of EGCG concentration, with enhancement values of 50%, 8.2% and 3.6%, respectively. The concentration of OH· in ventricular myocytes of rats at 50 µg/mL of EGCG was similar to the control (p > 0.05), but it was significantly different from the exposure group of 2.0 µM lead (p < 0.05; Figure 4b).

Effects of lead and/or epigallocatechin-3-gallate (EGCG) on the formation of OH·. a, Effects of lead on the formation of OH·. b, Effects of lead and EGCG on the formation of OH·. *p < 0.05, **p < 0.01 vs control, #p < 0.05, ##p < 0.01 vs lead 2.0 μM exposure.
Effects of EGCG on O2 ·− Formation
Figure 5 showed the effects of EGCG on the O2 ·− formation in ventricular myocytes. O2 ·− level increased with the increase of lead exposure concentration. The concentration of O2 ·− was nearly 125% of the control at a 2.0 µM level of lead (p < 0.01). When ventricular myocytes were treated with EGCG after lead exposure, the concentration of O2 ·− decreased and the amplitude was 6.5% of the control at 30 µg/mL of EGCG, lead was not able to inhibit the increase of O2 ·− in 50 µg/mL treatment of EGCG (p > 0.05), in which the O2 ·− concentration was 329.9 ± 11.02 U/g prot. This result was significant compared with 2.0 µM lead exposure alone (p < 0.01).

Effects of lead and/or epigallocatechin-3-gallate (EGCG) on the formation of O2.- a, Effects of lead on the formation of O2 .-. b, Effects of lead and EGCG on the formation of O2 .-. *p < 0.05, **p < 0.01 vs control, #p < 0.05, ##p < 0.01 vs lead 2.0 μM exposure.
Effects of EGCG on H2O2 formation
As compared with the control, H2O2 were increased up to 163%, 287%, and 373% of the control, respectively, when exposed to 1.0, 2.0, and 5.0 µM lead. Significant increase was found at low lead concentration 1.0 µM compared with the control (p < 0.05; Figure 6a ). When ventricular myocytes were treated with EGCG at different concentrations, H2O2 formation was attenuated. There were no statistical significance between the control ventricular myocytes and those treated with 30 µg/mL EGCG (p > 0.05); however, there was a significant difference when the treatment group was compared with the 2.0 µM lead exposure group (Figure 6b; p < 0.05 or 0.01).

Effects of lead and/or epigallocatechin-3-gallate (EGCG) on the formation of H2O2. a, Effects of lead on the formation of H2O2. b, Effects of lead and EGCG on the formation of H2O2. * p < 0.05, **p < 0.01 vs. control, #p < 0.05, ## p < 0.01 vs lead 2.0 μM exposure.
Discussion
Lead is one of the major hazards for human health and is widely distributed in the environment. It has been suggested that lead levels of 0.8 µg/L and 0.1 µg/L in drinking water can induce atherosclerosis in animal models, and lead levels of 0.5−10 μM may induce the proliferation of vascular smooth cells and fibroblasts in vitro. 19,20 Recent lead-toxicity studies have emphasized the minimum dose of lead that induces toxicity. In order to help compare these results with other studies, exposure concentrations of lead 1.0, 2.0, and 5.0 μM were chosen for this study.
There was a significant decrease in SOD and CAT activity in the ventricular myocytes upon lead exposure, in parallel with an increase in MDA level. SOD is believed to play a major role in the first line of antioxidant defense by catalyzing the dismutation of O2 .- radicals to form H2O2 and molecular oxygen O2. 21 The decrease of SOD could be explained by the massive production of O2 .-, which overrides enzymatic activity and leads to the fall of SOD concentration in ventricular myocytes. On the other hand, copper and zinc usually serve as cofactors of many enzymes, they could be replaced by lead, thereby making the copper and zinc deficiency and the enzymes inactive (Donaldson, 1991). 22 Furthermore, the decrease of SOD activity could also be due to the inactivation of the enzyme by crosslinking or damage to DNA by ROS. Decreased CAT activity in ventricular myocytes was also noted following lead exposure. CAT activity was observed to increase in the low lead exposure group (1.0 µM), but fell quickly with an increase in lead concentration. This is because initially and when the stress was moderated, the cells react by increasing the CAT, but if the stress lasts a long time, the enzyme is exhausted and its concentration falls. In this study, lead induced a significant increase in LPO, a process that may relate to deterioration of polyunsaturated fatty acids, resulting in altered membrane integrity, permeability, and function. This result was in agreement with Patra et al., who reported an increase in MDA level in the liver and the brain of rats exposed to lead. 23
Lead toxicity has been attributed to oxidative stress by ROS, in which there is increased lipid peroxidation and inhibition of enzymes responsible for prevention of such oxidative injury. 24–26 In order to further support this conclusion, we measured the O2 .-, H2O2, and OH·. The data from our studies of in vitro isolated ventricular myocytes indicated that lead exposure significantly increased intracellular ROS formation. The production of O2 .-, H2O2, and OH· increased after lead exposure in a concentration-dependent manner. These results showed oxidative stress appears to be a possible mode of the molecular mechanism of lead toxicity. This cellular deleterious effect occurs when generation of free radicals exceeds the capacity of antioxidants defense mechanisms.
EGCG has been proposed to protect the cardiovascular system against oxidative damage in vitro by means of polyphenols' interactions with O2 .- and other ROS. This study was carried to determine whether the lead toxicity on ventricular myocytes is a result of oxidative stress. EGCG is usually applied at 10−100 μM (approximately 5−50 µg/mL) concentrations in cellar models, and specific effects on cell signaling and apoptotic control have been observed under these circumstances consistently. 27 In this study, the effect of low concentration EGCG (10, 30, 50 µg/mL) were studied.
This study showed the protective effect of EGCG against lead-induced damage in ventricular myocytes. In the ventricular myocytes of rats exposed to lead then treated with EGCG, we found that EGCG could reverse the oxidative stress of lead-exposed group due to its antioxidant effects. Administration of EGCG (30 µg/mL) significantly increased the activities of SOD and CAT, and lowered MDA levels in ventricular myocytes that were exposed to lead. EGCG also suppressed the formation of O2 .-, H2O2, and OH·. EGCG dissolved in solution, as a scavenger, cleared away the ROS such as O2 .-, H2O2, and OH· radicals, inhibited the LPO, and protected the ventricular myocytes against the oxidative damage of lead. EGCG may also act as more than a scavenger, as several studies have found that tea polyphenols are able to induce enzymes that play important roles in cellular antioxidant defense mechanisms. 28 For example, CAT activity in aortas of spontaneously hypertensive rats was found to be stimulated by 2 weeks of oral consumption of green tea polyphenols. 29 EGCG induced the antioxidative enzymes and scavenged the ROS; both may be part of the mechanisms by which EGCG protected the ventricular myocytes of rats. However, it has been proposed that EGCG is auto-oxidized under cell culture conditions, and the half-life of EGCG is rather short, EGCG is oxidized by molecular oxygen of form O2 - and EGCG quinine. 30 But, there is no evidence for the auto-oxidation of EGCG in the mouse. It remains not clear whether EGCG auto-oxidation occurs in special tissues. In the future studies with EGCG, auto-oxidation should be investigated and avoided by adding SOD.
In conclusion, the results of this study suggest the protective effects of EGCG on ventricular myocytes of rats are by means of interacting with O2 ·−, OH·, H2O2, and other ROS. As a natural and excellent free radical scavenger, EGCG reversed the effects of lead on oxidative stress parameters, further supporting the hypothesis that oxidative stress is involved in lead toxicity. These results will provide more evidence for lead damage to tissue, cell, and biological macromolecule and provide evidence of an antioxidant at a time when many studies are underway to identify protective food in lead toxicity and cardiovascular disease risk.
Footnotes
This study was supported by grant No. 20677035 and No. 20477023 from the National Natural Science Foundation of China and by grant 20031092 from the Natural Science Foundation of Shanxi Province.
