Abstract
Cryptorchidism (CPT), the most common male congenital abnormality, is variably associated with other male reproductive tract problems. We evaluated if cryptorchid rats develop enhanced testicular susceptibility to dibutyl phthalate (DBP) or acrylamide (AA) after extended exposure. Three studies with rats were performed: (1) in utero and postnatal exposure to DBP or AA; (2) establishment of CPT and orchiopexy; and (3) in utero and postnatal exposures to DBP or AA associated with CPT/orchiopexy. Seminiferous tubules were histologically scored according to the severity of lesions: (1) Rats exposed to DBP (score 1.5) or AA (score 1.1) presented mostly preserved spermatogenesis. Some seminiferous tubules showed vacuolated germinative epithelium, germ cell apoptosis, and a Sertoli cell-only (SCO) pattern. (2) CPT (score 3.3) resulted in decreased absolute testes weights, degenerated and SCO tubules, and spermatogenesis arrest that were reversed by orchiopexy (score 1.1). (3) Exposure to DBP or AA with CPT/orchiopexy led to atrophic testes, spermatogenesis arrest, germ cell exfoliation/multinucleation, and SCO tubules (both chemicals score 2.5). Exposure to chemicals such as DBP or AA prevented the recovery of cryptorchid testes by orchiopexy. The possible role of environmental contaminants should be considered when looking for factors that modulate human testicular disorders associated with CPT.
Introduction
Human male reproductive disorders have increased globally over the past five decades. 1,2 Such disorders may be observed in newborns as cryptorchidism (CPT) and hypospadias as well as in young adults as impaired spermatogenesis (infertility) and testicular germ cell tumors. While the role played by genetic factors is unclear, the geographical and temporal variations in the prevalence of the aforementioned disorders are probably due to large variation of environmental exposures, nutrition, and lifestyles among different populations. 1 –6
Phthalate diesters are a group of chemicals used to make plastics flexible and are found in household and personal care products, toys, soaps, drug capsules, and medical equipment. The widespread use of phthalates results in continuous human exposure through air, water, and food. 7 The potential mechanisms underlying phthalate toxicity remain unclear but endocrine disruption is a possibility. 8 In particular, exposure to dibutyl phthalate (DBP) has been reported to be associated with reduced levels of testosterone and reduced anogenital distance (AGD) in newborns, a sexually dimorphic landmark indicative of altered fetal androgen levels. 9,10 Perinatal exposure of rats to some phthalates, including DBP, results in a range of reproductive tract defects in male offspring, including reduced AGD, CPT, multinucleated gonocytes, and reduced semen quality. 7,11 –13 In addition to its effect on germ cells, DBP also affects the Sertoli cells 13 and may also have a deleterious influence on spermatogenesis.
Another known environmental toxicant is acrylamide (AA), a polymer with varied applications such as treatment of wastewater, gel electrophoresis, processing of paper, and permanent press fabrics. AA is also formed during smoking and cooking common starchy foods under high temperatures. 14 –16 AA has a wide toxicologic profile that encompasses geno-, neuro-, reproductive-, and immunotoxicities and carcinogenicity. 14,15,17 Male rats administered AA exhibited a variety of testicular toxicities including vacuolation of the germinative epithelium, germ cell degeneration, multinucleation, and apoptosis, with aberrant sperm morphology and decreased sperm count and motility. 18 –21 The toxicologic responses with DBP and AA have been observed in animals administered relatively high doses.
CPT is the most common congenital abnormality in male newborns. 22,23 Although most cryptorchid testes later spontaneously descend to the scrotum, a considerable number continue into puberty and adulthood, variably associated with testicular morphological anomalies, impairment of germ cell maturation, infertility, and testicular germ cell neoplasia. 1,3 Various animal models of CPT have been established through surgical intervention, hormonal manipulation, or transgenic techniques. 24 –26 Testes of rats that underwent these different models of experimental CPT generally show progressive delay of growth, atrophy of seminiferous tubules, and germinative epithelium disruption with most tubules lined by Sertoli cells with only a few spermatogonia and spermatocytes but no elongated spermatids or spermatozoa, that is, maturation arrest. 25 –27 To maintain fertility and prevent malignancy in humans, it has been recommended that surgical relocation of the testes to the scrotum—orchiopexy—should be performed between 6 and 18 months of age. 28 Similarly, it has been suggested that experimentally cryptorchid rats should receive orchiopexy as early as possible to reestablish normal spermatogenesis. 24,25,29
Taking these observations into account, three studies were performed to evaluate morphologically the testicular effects of the association between phthalate (DBP) or AA exposures in Sprague-Dawley rats surgically made cryptorchid. To our knowledge, there is no report in the literature about the effects of the association of experimental surgical CPT and exposure to testicular toxicants. The results indicate that exposure to chemicals such as DBP or AA prevent the recovery of cryptorchid associated testicular lesions by orchiopexy. The potential role of environmental contaminants should be taken into consideration when looking for factors that modulate human testicular disorders associated with CPT.
Material and methods
Chemicals and doses selection: gavage and dietary exposure
DBP 99% pure (CAS 84-74-2; Sigma-Aldrich, Saint Louis, MO, catalog 524980) and AA 99% pure (CAS 79-06-1; Sigma-Aldrich, catalog A9099) were used. Exposures to DBP or AA through gavage, breast feeding, or diet were the same in experiments 1 and 2(b). Dams were exposed through gavage to 500 mg/kg DBP in corn oil (100 g/L) (CAS 8001-30-7; Sigma-Aldrich, catalog C8267) or to 10 mg/kg AA in deionized water (2 g/L) from gestational days (GD) 12 to 21. The DBP dose level was based on reports indicating that 500 mg/kg by gavage (embryonic days 11.5 to 20.5) was sufficient to induce rat germ cell abnormalities. 11,13 Also, exposure of male rats to AA at doses above 10 mg/kg through gavage or drinking water caused impairment of spermatogenesis and dominant lethal mutation as well as reduction of body weight and neurotoxicity. 18 –20,30 Control (CTL) dams received corn oil by gavage according to their body weights.
To treat the offspring through the diet with the same amount of chemicals provided by gavage to the dams, an estimated dose was calculated based on daily ingestion reported for the adult rat, 31,32 which resulted in concentrations of 6000 ppm DBP and 120 ppm AA.
Experiment 1: Treatment with DBP or AA with no surgical intervention
This experiment was performed to evaluate adult testes on postnatal day (PND) 112 after in utero and continuous postnatal exposure to the testicular toxicants DBP or AA. The study protocol was approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Nebraska Medical Center (UNMC, Omaha, Nebraska, USA), protocol no. 17-047-05-EP.
Animals
Female (
Female rats were mated overnight at the proportion of two females to each male rat. Vaginal smears were collected daily and the day of sperm detection was considered as day 0 of gestation (GD 0).
Experimental design
Pregnant dams were randomly placed into three groups and treated daily by gavage from GD 12 to GD 21 with plain corn oil (
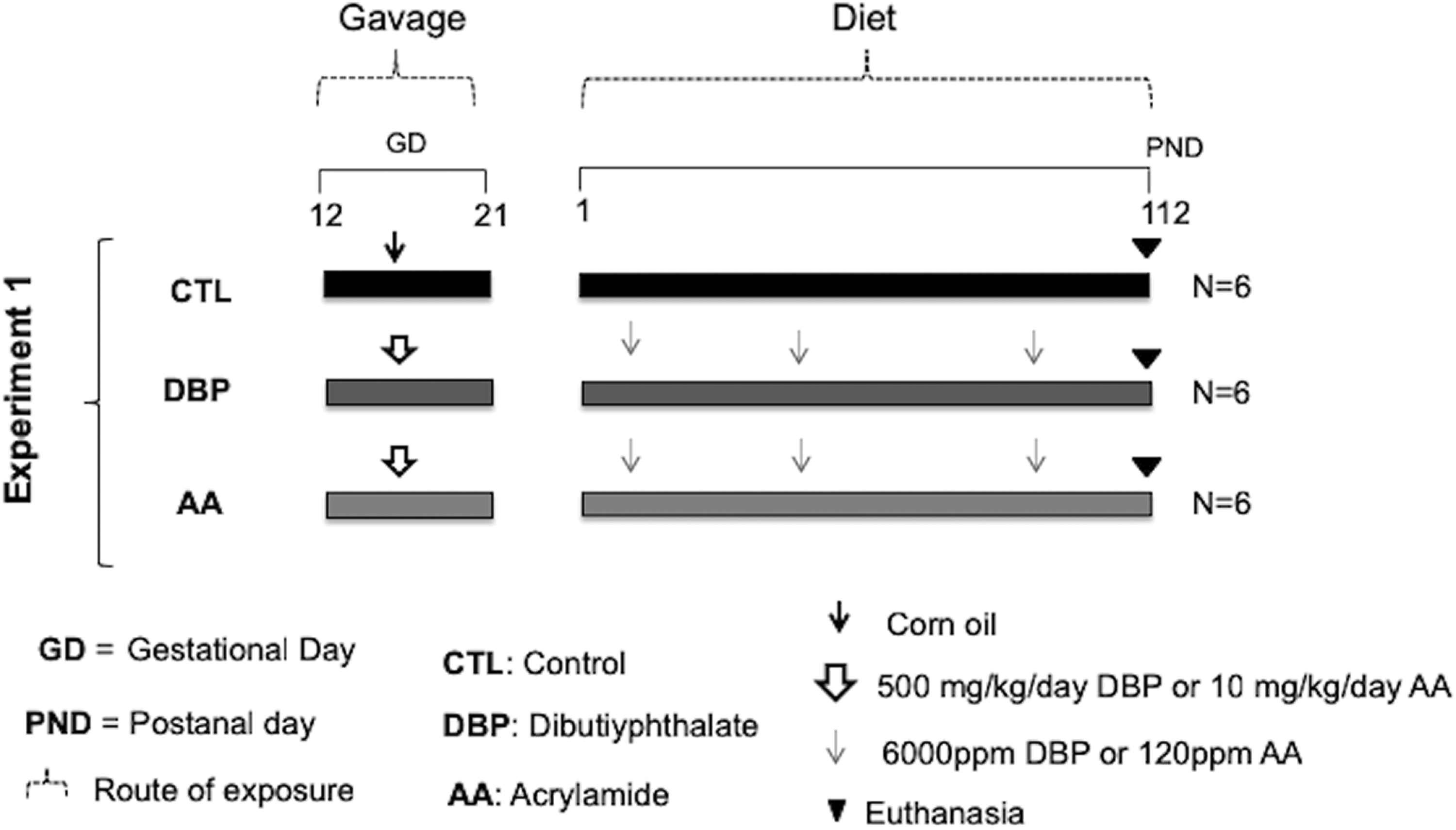
Experiment 1 design. Animals exposed in utero through the dams (gavage), and postnatally to DBP (500 mg/kg/day) or AA (10 mg/kg/day) through the diet. CTL animals received only corn oil. Euthanasia performed on PND 112.
On PND 4, 33,34 the distance between the anus and the genital tubercle (AGD) was measured using calipers. To avoid possible errors caused by body size differences, the AGD of each animal was divided by the cube root of body weight, 35 generating a normalized AGD (nAGD). Litters were adjusted to eight pups per dam at PND 4, with a preference for males. The total number of DBP-treated, AA-treated, and CTL rats who had their AGD measured eventually became greater than the total number of animals indicated in the experimental protocol because all male pups from each litter had their AGD measured, despite whether or not they were included in the experiment.
Euthanasia, necropsy, and sample collection
All F1 animals were euthanized on PND 112 by an overdose of Fatal Plus (Vortech Pharmaceuticals, Dearborn, Michigan, USA), 150 mg/kg body weight. At necropsy, the testes were removed, weighed, and fixed in modified Davidson’s fixative for 24 h. 36 Following fixation, the tissues were processed for paraffin embedding in the UNMC Tissue Sciences Core Facility. Approximately 4–5-μm sections were cut transversely from the middle portion and stained with hematoxylin and eosin (H&E) for histological evaluation.
Experiment 2: Surgically induced CPT and orchiopexy with or without exposure to DBP or AA
This experiment was approved by the Committee for Ethics in Animal Experimentation of the Botucatu Medical School, UNESP, SP, Brazil, protocol no. 926/2012.
Animals
Thirty female and 15 male Sprague-Dawley rats (4 weeks old) were obtained from the Multidisciplinary Center of Biological Investigations (CEMIB UNICAMP, Campinas, SP, Brazil) and kept under a 12-h light/dark cycle and with a targeted temperature of 22 ± 2°C. Standard pelleted food (Presence; Evialis, Paulínia, SP, Brazil) and water were provided ad libitum. The animals underwent a 4-week acclimation period upon arrival before starting the experiment. Rats were mated overnight at the proportion of two females to each male. Vaginal smears were collected daily and the day of sperm detection was considered as GD 0.
Experiment 2(a)
CPT and orchiopexy. The objective of this study was to document morphological changes in adult testes following surgically induced CPT performed as early as possible during postnatal life and its reversal by orchiopexy.
At weaning (PND 21), F1 male rats were randomly allocated to four groups: (1) bilateral CPT surgery on PND 21 and euthanasia on PND 98 (CPT group; 10 males from 9 dams); (2) sham-operation on PND 21 and euthanasia on PND 98 (CTLCPT; 5 males from 3 dams); (3) bilateral CPT surgery on PND 21, followed by orchiopexy on PND 42 and euthanasia on PND 98 (CPT/R; 7 males from 3 dams); and (4) sham operation for CPT (PND 21) and for orchiopexy (PND 42) and euthanasia on PND 98 (CTLCPT/R; 5 animals from 2 dams). No more than three pups from the same litter were used to constitute each group (Figure 2).

Experiments 2(a) and 2(b) designs. Experiment 2(a) (euthanasia performed on PND 98): group CPT—CPT surgically established on PND 21; group CTLCPT—CTL animals underwent only sham surgery; group CPT/R—CPT established on PND 21 and orchiopexy on PND 42; group CTLCPT/R—CTL animals underwent only to sham surgeries. Experiment 2(b) (euthanasia performed on PND 112): animals exposed in utero through the dams (gavage) and postnatally to DBP (500 mg/kg/day) or AA (10 mg/kg/day) through the diet, underwent cryptorchid surgery on PND 21 (groups DBP-CPT and AA-CPT), and orchiopexy surgery on PND 42 (groups DBP-CPT/R and AA-CPT). CTL animals (group CTL) received corn oil and underwent sham surgeries.
Surgical procedures
On PND 21, between 8 a.m. and 9 a.m., rats were anesthetized with ketamine (30 mg/kg intraperitoneal) and xylazine (4 mg/kg intraperitoneal). Ketoprofen (10 mg/kg subcutaneous) and abdominal lidocaine injections (7 mg/kg subcutaneous) were used as an analgesic and local anesthetic, respectively. The abdominal cavity was aseptically opened by a small midline incision, and both testes were translocated from the scrotum into the abdominal cavity through the inguinal rings. Care was taken to avoid twisting of the spermatic cord, which could lead to testicular ischemia. The testes were fixed to the dorsolateral abdominal wall with two stitches passing through the tunica albuginea in the cranial and caudal regions of the testes using a 5-0 blunt needle and non-absorbable suture material (NL50CR13 Nylon, Bioline, Brazil). The muscle and skin layers were then closed, and the animals were kept at 30°C for 30 min to minimize the deleterious effects of hypothermia induced by anesthesia. 24,26
For orchiopexy, the anesthetic, analgesic, and asepsis procedures were the same as for the CPT surgery. On PND 42, a midline abdominal incision was made, and the sutures which held both testes in the dorsolateral abdominal wall were carefully removed. The inner surface of the scrotum wall was clamped, guided up through the inguinal canal, and reversed to facilitate the manipulation; the testes were sutured through the tunica albuginea to the inner wall of the scrotum by a 5-0 blunt needle (NL50CR13 Nylon, Bioline) and then gently guided down through the inguinal canal and the inner layer of the scrotum back to its natural position. The abdominal muscle and skin layers were closed, cleaned, and the animals left to recover under the same conditions as described above. All animals received antibiotic (enrofloxacin, 5 mg/kg/day subcutaneous) for 3 days following orchiopexy.
CTL animals underwent sham surgeries under the same anesthesia, analgesia, and aseptic conditions as described above. They had only the abdominal skin and muscle layers opened and then sutured.
Experiment 2(b)
Treatment with DBP or AA combined with CPT and orchiopexy. This study was designed to evaluate whether experimental surgical CPT, performed as early as possible during postnatal life, could enhance testicular susceptibility to DBP or AA.
Pregnant dams were randomly placed into three experimental groups and treated daily by gavage from GD 12 to GD 21 with plain corn oil (
All male pups exposed to the chemicals in utero, during lactation and by diet during post-weaning life underwent the same surgical procedures as described in experiment 2(a): CPT on PND 21 and orchiopexy on PND 42. Due to the complexity of surgical procedures and the large number of animals, they were euthanized on PND 112 instead of the PND 98 in experiment 2(a). CTL animals were also sham-operated at the same time points as described for the experimental groups.
Anogenital distance
On PND 1, 37,38 all male offspring from each litter had their AGD measured using calipers (35), as described for experiment 1.
Euthanasia, necropsy, and sample collection
Animals from experiments 2(a) and 2(b) were anesthetized with ketamine (30 mg/kg i.p.) and xylazine (4 mg/kg ip) and euthanized by exsanguination via heart puncture. The testes were immediately removed, weighed, and placed in modified Davidson’s fixative for 24 h. 36 The testes followed the same histological processing as in experiment 1. Testicular calcification observed on H&E sections was confirmed by von Kossa staining.
Histologic examination
The histological analyses of the seminiferous tubules observed in the transverse sections of each testis were performed blinded without identification of the experimental group. Hence, a two-step analysis of the H&E testicular sections was performed. First, the seminiferous tubules were analyzed qualitatively according to a standardized nomenclature of rat testicular microscopic lesions. 39 Next, a four-class ranking system developed at this laboratory based on the most frequent histological tubular alterations 40 was applied. Accordingly, an average of 250 rounded sections of seminiferous tubules per animal were classified according to increased severity of lesions as follows: class 1, tubules showing normal spermatogenesis; class 2, tubules with spermatid-like and sperm mature cells but also showing alterations such as intra-epithelial vacuoles, apoptotic bodies, and/or multinucleated germ cells; class 3, tubules with the same elements of epithelial damage as class 2 but presenting with only spermatocytes and spermatogonia (germ cell maturation arrest/epithelial layer atrophy); and class 4, increased amount of Sertoli cells-only (SCO) tubules. The results obtained by this class-ranking system were adjusted by an adapted H-score. This is the same methodology used for semi-quantitative evaluations in immunohistochemistry analyses. 41,42 The incidence of tubules (%) in each class per animal was calculated by multiplying the number of tubules in that class by 100 and dividing by the total number of tubules counted in the testis section. The final H-score for each group was generated by adding the number of tubules in class 1 times 1, the number of tubules in class 2 times 2, the number of tubules in class 3 times 3, and the number of tubules in class 4 times 4, dividing each result by the total number of tubules counted in the respective section.
Statistical analyses
Since the experimental groups used more than one pup from the same litter, both the pups and dams were included in the statistical analyses. Variables were analyzed by a generalized linear mixed effect model with gamma distribution followed by a post hoc Sidak correction
43
; the study experimental units were the F1 rats, adjusted by their dams. Body weights at birth and AGD were analyzed using the Mann–Whitney test followed by a post hoc Dunns’ test. All analyses were performed with the IBM SPSS 22.0 Statistics software (Statistical Package for Social Science; SPSS Incorporation). A
Results
Body weights and AGD at birth
Body weights and nAGD at PND 1 and 4 of Sprague-Dawley F1 males exposed in utero to DBP or AA.

CTL: control; AGD: anogenital distance; nAGD: normalized anogenital distance; DBP: dibutyl phthalate; CPT: cryptorchidism; AA: acrylamide.
aMale offspring exposed in utero either to DBP (500 mg/kg/day) or to AA (10 mg/kg/day). CTL dams received corn oil.
bSince all offspring were exposed in utero, body weights and AGD were measured in all male pups, resulting in a number of animals (
cSignificantly different from the respective CTL,
Location of the testes at PND 21, when CPT was established
Terminal body and testes weights
Final body weights and absolute and relative testicular weights of F1 male Sprague-Dawley rats from experiments 1, 2(a), and 2(b).a
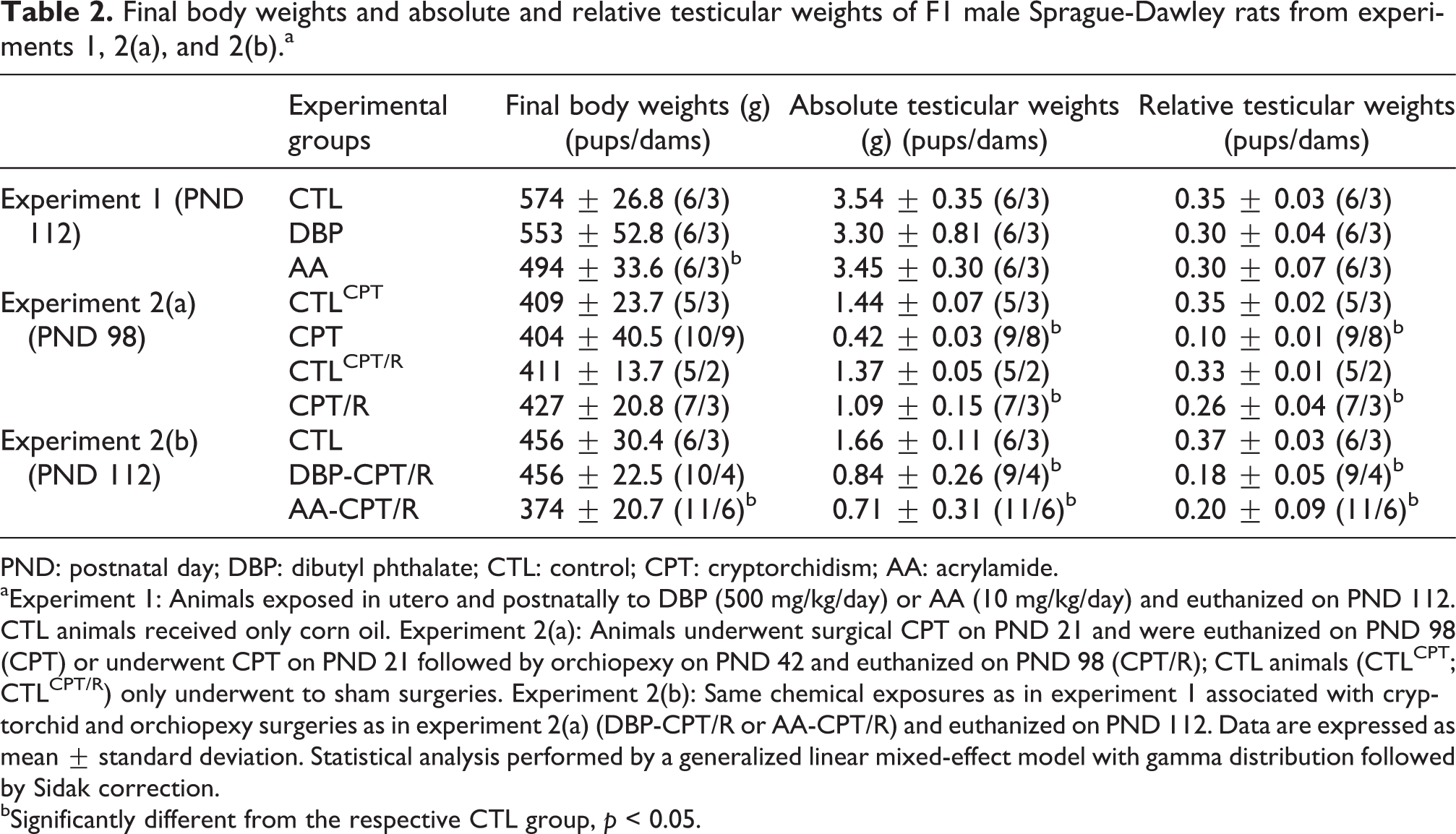
PND: postnatal day; DBP: dibutyl phthalate; CTL: control; CPT: cryptorchidism; AA: acrylamide.
aExperiment 1: Animals exposed in utero and postnatally to DBP (500 mg/kg/day) or AA (10 mg/kg/day) and euthanized on PND 112. CTL animals received only corn oil. Experiment 2(a): Animals underwent surgical CPT on PND 21 and were euthanized on PND 98 (CPT) or underwent CPT on PND 21 followed by orchiopexy on PND 42 and euthanized on PND 98 (CPT/R); CTL animals (CTLCPT; CTLCPT/R) only underwent to sham surgeries. Experiment 2(b): Same chemical exposures as in experiment 1 associated with cryptorchid and orchiopexy surgeries as in experiment 2(a) (DBP-CPT/R or AA-CPT/R) and euthanized on PND 112. Data are expressed as mean ± standard deviation. Statistical analysis performed by a generalized linear mixed-effect model with gamma distribution followed by Sidak correction.
bSignificantly different from the respective CTL group,
Testicular histology
Histological analyses performed at the end of the experiment (PND 98 and 112) indicated that every animal with surgical CPT and/or exposed to DBP or AA developed seminiferous tubule alterations. These alterations were not homogenous among the animals or within the same animal with damaged tubules intermixed with apparently normal ones. The epithelial lesions observed in the seminiferous tubules included intraepithelial vacuoles, germ cell exfoliation, multinucleation, apoptosis, spermatogenesis arrest, and SCO type of tubules. The SCO tubules were considered to be the end-stage of the tubular degeneration/atrophy process. In addition, a few animals presented rare intratubular calcification. The seminiferous tubule damage was classified into four classes according to the scoring system described in “Material and methods.”
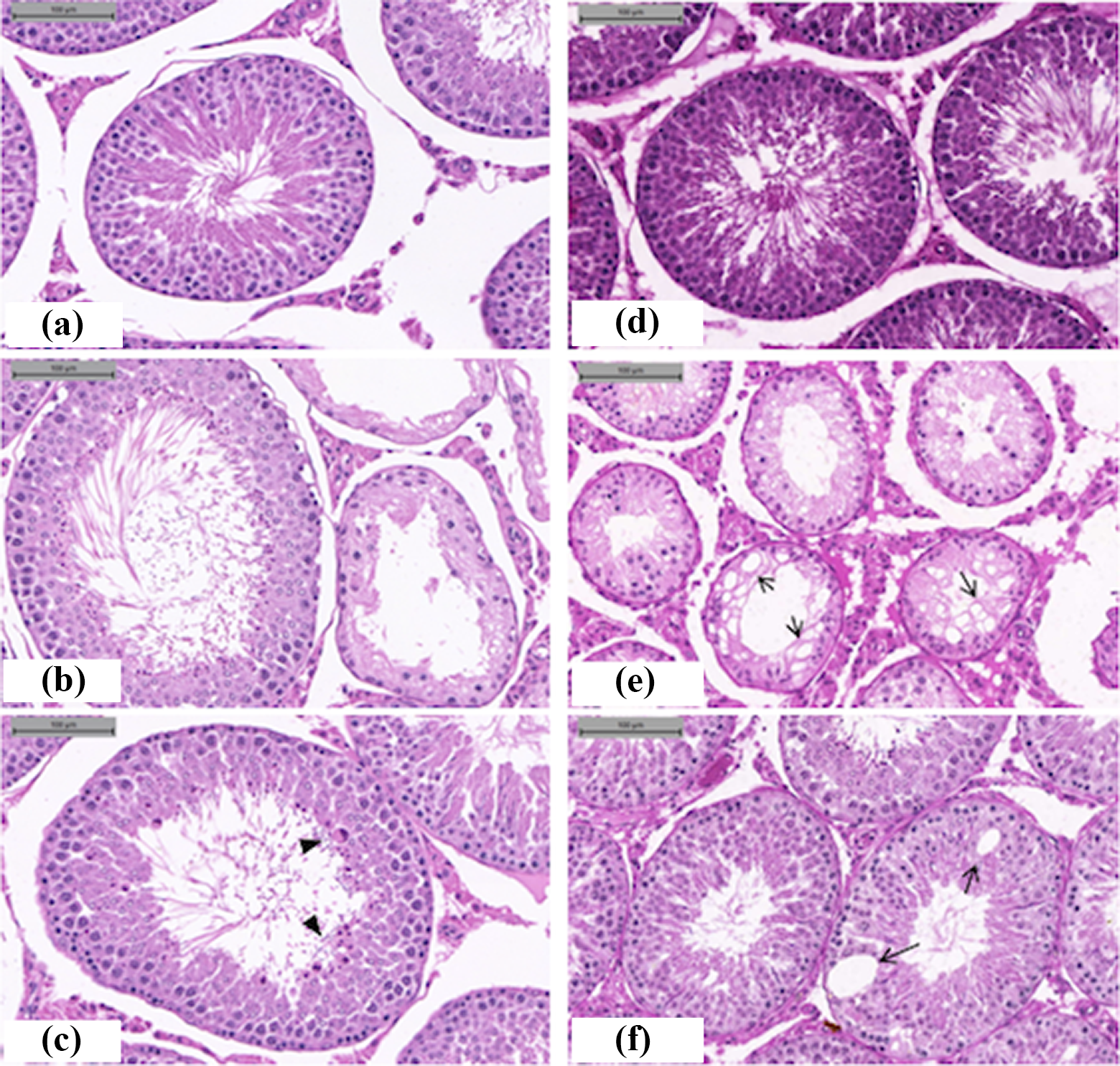
Histology of the testes of Sprague-Dawley rats (H&E). Experiments 1 and 2(a). (a) CTL animals received corn oil and were euthanized on PND 112; normal seminiferous tubules. (b) Animals exposed in utero and postnatally to DBP and euthanized on PND 112; mostly normal-appearing tubules (left) among with some SCO-type seminiferous tubules (right). (c) Animals exposed in utero and postnatally to AA and euthanized on PND 112; normal-appearing tubules with germ cell differentiation through the stage of sperm, although vacuoles, apoptotic bodies (arrow head), and a few SCO-type tubules were also present. (d) CTL animal underwent sham surgeries for CPT and orchiopexy; seminiferous tubules show normal spermatogenesis. (e) Cryptorchidism performed on PND 21 and euthanasia on PND 98; depletion of germ cells, intraepithelial vacuolation (arrow), and absence of sperm in the tubular lumen, where apoptotic bodies and multinucleated germ cells were observed; some SCO tubules. (f) Cryptorchidism performed on PND 21 followed by orchiopexy on PND 42 and euthanasia on PND 98; structural recovery of the testes with predominance of normal tubules showing an organized seminiferous epithelium and sperm in the lumen; occasional apoptotic bodies and scattered tubules with vacuolated epithelium (arrow) present. CTL: control; CPT: cryptorchidism; PND: postnatal day; DBP: dibutyl phthalate; AA: acrylamide; SCO: sertoli cell only; H&E: hematoxylin and eosin.
Experiments 1, 2(a), and 2(b)—Testicular histological scores (see “Material and methods”) determined for each group of male Sprague-Dawley rats.a

PND: postnatal day; DBP: dibutyl phthalate; CTL: control; CPT: cryptorchidism; AA: acrylamide.
aExperiment 1: Animals exposed in utero and postnatally to DBP (500 mg/kg/day) or AA (10 mg/kg/day) and euthanized on PND 112. CTL animals received only corn oil. Experiment 2(a): Animals that underwent surgical CPT on PND 21 and were euthanized on PND 98 (CPT) or underwent surgical CPT on PND 21 and orchiopexy on PND 42 and euthanized on PND 98 (CPT/R); CTL animals (CTLCPT; CTLCPT/R) underwent sham surgeries. Experiment 2(b): Same chemical exposures as in experiment 1 associated with the cryptorchid and orchiopexy surgeries as in experiment 2(a) (DBP-CPT/R or AA-CPT/R) and euthanized on PND 112.
bScore values are expressed as the median (Q1–Q3). Statistical analysis performed by a generalized linear mixed-effects model with gamma distribution followed by Sidak correction.
cSignificantly different from the respective CTL,

Histology of the testes of Sprague-Dawley rats (H&E). Experiment 2(b); in utero and postnatal exposure to DBP or AA associated with CPT performed on PND 21, orchiopexy on PND 42 and euthanasia on PND 112. DBP-CPT/R treatment: (a) Spermatogenesis arrest, intraepithelial vacuoles (arrow), and germ cell exfoliation (arrowhead); (b) atrophic seminiferous tubules (*); (c) atrophic tubules with intratubular calcification (dotted arrow). AA-CPT/R treatment: (d) Multinucleated germ cells (arrow); (e) maturation arrest—enlarged germ cells with hyperchromatic nuclei and pleomorphic cytoplasm (arrowhead); (f) atrophic tubules with luminal debris and calcification (dotted arrow). CTL: control; CPT: cryptorchidism; PND: postnatal day; DBP: dibutyl phthalate; AA: acrylamide; SCO: sertoli cell only; H&E: hematoxylin and eosin.
Discussion
The three complementary studies reported here show that exposure to the chemicals DBP or AA prevents the complete reversal of testicular alterations that occurred during an experimental 3-week long surgically induced CPT in Sprague-Dawley rats performed as early as possible during the postnatal period. The alterations of the seminiferous tubules were not reversed 10 weeks after the orchiopexy surgery on PND 42, that is, at the 6th postnatal week.
The time frame of the experiments was established to cover more than one cycle of spermatogenesis, which in rats lasts approximately 50 days. 44 However, no attempt was made to assess the spermatogenesis stages along the spermatogenesis cycle because the focus of the analyses was on the histological lesions of the germinative epithelium (i.e. vacuoles, germ cell sloughing, multinucleation, etc) as described elsewhere. 39 Because of the type and extent of the lesions, we were unable to use the conventional Johnsen score which has been used to evaluate spermatogenesis in humans 45,46 and in rats. 27 The semiquantitative procedure used for scoring the histological lesions in these studies allowed us to discriminate between the experimental groups at the end of the studies and revealed significant variations of the frequency and severity of the tubular lesions among the groups.
When CPT was not included in the treatment (experiment 1), the effects of in utero and postnatal exposure to DBP or AA on the seminiferous tubules were not statistically significant on PND 112. The respective histological scores were 1.5 (DBP) and 1.1 (AA), similar to the CTL score of 1.0 (Table 3). The testicular alterations observed in the animals exposed to each chemical included SCO-type seminiferous tubules, intraepithelial vacuolation, and apoptotic bodies (Figure 3(a) to (c)), which are variably described in rats exposed either to DBP 7,11 –13 or to AA. 19,21,30,47,48 These observations indicate that the experimental conditions of the present studies did not result in characteristic histological alterations of the seminiferous tubules that would be indicative of which chemical (DBP or AA) was used, despite their different modes of action on the germinative epithelium.
However, the higher score in the DBP-treated group relative to the respective CTL group resulted from these animals having a higher number of SCO-type tubules, that is, seminiferous tubules showing an almost complete depletion of germ cells. The influence of DBP on the testes may be mostly a result of the in utero anti-androgenic activity of this phthalate. As in humans, the rat AGD is influenced by fetal testosterone levels and is a characteristic phenotype of each sex. If the levels of testosterone are decreased during the critical developmental window, a decreased AGD will occur. 12,49 In the current studies, the anti-androgenic activity of DBP was demonstrated by the decreased nAGD measured on PND 4 in experiment 1, which was significantly different compared to CTLs. The nAGD was not decreased in experiment 2(b) possibly due to the measurements being done in younger animals (PND 1) and the large variability of the measurements (Table 1).
The nAGD was not altered in AA-exposed animals. This finding supports the absence of clear anti-androgenic effects in literature reports, with levels of rat serum testosterone after AA exposures reported as either decreased 19,21 or increased 20 using different experimental designs. Depending on the dose levels, the putative toxicologic influences of AA on the seminiferous tubules are quite varied and include oxidative stress, altered expression of genes related to nucleic acid-binding, cell proliferation and differentiation, cytoskeletal actin filament organization, calcium ion signaling, and impairment of chromosome segregation during cell division, and apoptosis. These various and non-mutually exclusive effects may lead to the reported alterations of spermatogenesis and sperm health, altered fertility and mating, dominant lethal mutations, and neurotoxicity. 19 –21,30,47 Considering all of these reported effects, it is noteworthy that the alterations induced by AA alone on the seminiferous epithelium did not differ compared to the respective CTL group on PND 112 (Table 3).
In experiment 2(a), animals that were cryptorchid (CPT) from PND 21 to PND 98 (14 weeks) developed atrophic testes with a statistically significant decrease in testicular weights (71%; Table 2). The severe histological testicular dysplasia resulted in a final score of 3.3 (Table 3, Figure 3(e)). The histologic alterations were similar to those described in other rat models of CPT. 24 –26 There are two hypotheses about the dysplastic picture of the human undescended testes. One proposes that both maldescent and testicular morphologic alterations are caused by a common primary testicular defect during embryo fetal development. 1,3 The other proposes that a potentially normal testis for whatever reason fails to descend and the high intra-abdominal temperature leads to secondary changes in the testes’ morphology. 27,50 Since the rat bilateral CPT established in experiment 2(a) differs from the human congenital undescended testes in that it was surgically established early in the postnatal life in otherwise normal animals, it should be assumed that the intra-abdominal temperature was the major stress responsible for the impairments of testicular development and of spermatogenesis, as indicated elsewhere, to explain these cryptorchid alterations. 50
In addition, in experiment 2(a), orchiopexy performed on PND 42 (CPT/R), after a 3-week long CPT, resulted in an almost complete restoration of the testes observed at the end of the experiment, on PND 98 (14th week; Table 2; Figure 3(f)) The difference between the mean testes weights in the reversal group (CPT/R) compared to its CTL group (CTLCPT/R) was smaller than for the non-reversal group (CPT) compared to its CTL group (CTLCPT). Although the testes weights remained about 20% smaller than the CTL, the recovery of the germinative epithelium was evident with the histologic score in the reversal group, the same as in its respective CTL group (score 1.1; Table 3). These data indicate that orchiopexy is potentially able to restore at least morphologically, the cryptorchid testes in rats when performed at an appropriate time point, which in the present case was in the 3rd week after CPT on PND 42, when the male animal was going into puberty or sexual maturity. 51
In experiment 2(b), animals that underwent CPT and orchiopexy with continuous exposure to DBP or AA presented with severely damaged testes at the end of the study on PND 112 (Table 2). The absolute and relative testes weights were reduced by approximately 50% in both DBP- or AA-exposed animals compared to the CTL group, and vacuolation, germ cell exfoliation, spermatogenesis arrest, multinucleation, and calcification were observed (Figure 4). In the DBP- and AA-exposed groups, the histological lesion scores were 2.5 and 2.9, respectively, while the CTL group score was 1.0. Therefore, both chemicals did inhibited the efficacy of orchiopexy in reversing testicular CPT-dependent lesions. In other words, when the rats were exposed to DBP or AA, orchiopexy was not able to completely restore the cryptorchid testes morphology, even if performed at a proper animal age.
The calcified intratubular amorphous material observed in the testes of a few animals exposed either to DBP-CPT/R or to AA-CPT/R was confirmed with the von Kossa stain. It has been suggested that these structures correspond to accumulation of cellular debris, followed by glycoproteins and calcium deposition, 52 a process known as dystrophic calcification. In humans, intratubular calcification has been described in prepubertal testes in conjunction with a wide variety of clinical and pathological conditions, including cryptorchid testis, infertility, and atrophic seminiferous tubules at the periphery of primary germ cell tumors. 52 The occurrence of dystrophic calcification in animals that underwent CPT and also exposed to chemicals documents the severity of the damage that occurred when chemical exposures and CPT treatments were both included.
These three studies show that exposure alone to the chemicals DBP or AA did not induce severe testicular lesions, since only class 1 alterations were observed at the end of experiment 1. However, CPT itself induced more severe class 3 lesions, but these were almost completely reversed by orchiopexy. When rats were exposed to both treatments, chemical exposure and CPT, orchiopexy was not effective in restoring testicular integrity, with histological scores in these groups classified as class 3 alterations (scores 2.5 for DBP and 2.9 AA). This may be the result of additive or synergistic interactions of the mechanisms of the chemical exposure and the CPT.
In conclusion, the present results indicate that exposure to chemicals such as DBP or AA prevents the complete recovery of cryptorchid testes by orchiopexy. As already indicated, there are various animal models of CPT, each with its own merits and limitations. 24 –26 Although these studies did not aim to reproduce completely human CPT, the present observations should be taken into consideration when looking for modulating factors of human testicular disorders associated with CPT.
Footnotes
Acknowledgements
The authors are grateful to Carlos Márcio Nóbrega de Jesus, Botucatu Medical School, UNESP—Univ Estadual Paulista, Botucatu Campus, SP, Brazil, for assistance with the surgical procedures; Paulo Roberto Cardoso and Paulo Cesar Georgete for helpful technical assistance; Fred and Pamela Buffett Cancer Center Tissue Sciences Facility Shared Resource, supported by the National Cancer Institute under award number P30 CA036727, Center for Evaluation of Environmental Impact of Human Health (TOXICAM) and São Paulo State University (UNESP).
Authors’ note
AP Ferragut Cardoso is now affiliated to Department of Pharmacology and Toxicology, University of Louisville, Louisville, KY, 40292, USA.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All the authors were associated with academic institutions when the paper was prepared.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) [Grant No. 2012/09873-4]; Conselho Nacional de Pesquisa (CNPq) [Grant 132667/2013-4].
