Abstract
Objective:
To discuss the effects of Epstein-Barr virus (EBV)-encoded BamHI A rightward transcript (BART) microRNA (miR-BART6-5p) by targeting Dicer1 on biological properties and radiosensitivity of nasopharyngeal carcinoma (NPC).
Methods:
NPC patients (n = 96) treated with radiotherapy were collected from Jan 2010 to Jan 2011. Real-time quantitative PCR (qRT-PCR) and western blot were carried out to measure the expression of miR-BART6-5p and Dicer1. Dual luciferase reporter gene assay verified that miR-BART6-5p targeted Dicer1. CCK8, wound-healing, Transwell and Annexin-FITC/PI were employed to evaluate the effects of Dicer1 mediated by miR-BART6-5p on biological characteristics of NPC cells. The radiosensitivity of miR-BART6-5p targeting Dicer1 was assessed in vitro and in vivo.
Results:
Increased miR-BART6-5p and decreased Dicer1 were discovered in NPC patients, displaying a close association with T-stage, clinical stage, as well as Pre-DNA of NPC. While elevated Dicer1 and miR-BART6-5p down-regulation in NPC patients were found after effective radiotherapy. Both miR-BART6-5p and Dicer1 were prognostic factors of NPC. Down-regulation of miR-BART6-5p could enhance Dicer1 expression and inhibit NPC cell proliferation, invasion and migration with promoted apoptosis. Clone formation assay also showed miR-BART6-5p down-regulation reduced planting efficiency (PE), which further decreased with the increased dose of irradiation. Injection with miR-BART6-5p inhibitors in nude mice after 6-Gy irradiation contributed to the overexpression of Dicer1 and the inhibition of tumor growth.
Conclusions:
EBV-miR-BART6-5p may target Dicer1 to facilitate proliferation and metastasis of NPC cells and suppress apoptosis, thus being a new target for NPC therapy.
Introduction
Nasopharyngeal carcinoma (NPC), a head-neck malignancy derived from epithelium, has a high prevalence in Southern China, Southeast Asia, North Africa and Alaska, with the incidence of 30 per 100,000 NPC cases a year in China, but only 1 per 100,000 in western countries. 1 Multiple causes, including genetic susceptibility, pathognostic environment factors and Epstein-Barr virus (EBV) infection, have been evidenced to contribute to the pathogenesis of NPC. 2,3 Although radiotherapy, especially intensity-modulated radiotherapy (IMRT), is currently the priority and mainstay for the treatment for NPC patients, and the acute radiation reaction in radiotherapy may seriously affect patients’ quality of life and interfere with the treatment, as well as the course of the disease, which directly results in radiation resistance, ultimately leading to recurrence and metastasis. 4,5 Therefore, it exhibits clinical significances to explore new potential intervention targets to improve curative and survival rate of NPC.
MicroRNA (miRNA) is a small single-stranded noncoding regulatory RNA to be used by mammals, plants and viruses, while EBV, exerting potential carcinogenic effects, is recognized as the first human virus coding miRNA by immortalizing B lymphocytes and human primary epithelial cells. 6 –8 So far, more than 40 mature miRNAs encoded by EBV have been discovered to be grouped into two major clusters: BHRF1 (Bam HI fragment H rightward open reading frame 1) and BART (Bam HI-A region rightward transcript). 9 Notably, EBV-encoded miR-BARTs could target both viral and cellular genes to be involved in cancer development, 9 including NPC. For instance, miR-BART17, miR-BART3, miR-BART7 and miR-BART13 are upregulated in NPC, which promoted epithelial-mesenchymal transition and metastasis of NPC cells and facilitated tumor growth. 10 –13 Moreover, Deng et al. observed that miR-BART6 suppressed the colony unit formation and polyploidization of megakaryocyte and decreased CD41 expression, 14 and miR-BART6-3p suppressed the invasion and metastasis of gastric cancer cells by targeting lncRNA (long non-coding RNA) LOC553103. 9 The miR-BARTs exerted a role in the expression of virus gene and host cell genes by means of decrease transcriptional and post transcriptional level to influence tumor cell growth and apoptosis. 15
Dicer, a key enzyme in miRNA synthesis that cleaves pri-miRNAs to release hairpin-shaped pre-miRNAs, 16 has been shown to present low expression in multiple human malignant tumors, including ovarian carcinoma, 17 lung cancer, 18 colon cancer 19 and NPC. 20 In particular, evidence pointed out an important role of EBV miR-BART6-5p through targeting Dicer1 to influence the latent state of EBV viral infection. 21 However, researches about mechanisms and effects of miR-BART6-5p and Dicer1 on the prognosis and radiotherapy of NPC remain scarce. Considering the above, we conducted the experiment to examine the expressions of miR-BART6-5p and Dicer1, and to further elucidate the potential effects of miR-BART6-5p-mediated host Dicer1 on biological characteristics and radiosensitivity of NPC.
Materials and methods
Ethics statement
All subjects in the current study were all informed and signed the informed consent, and it was approved by the Ethics Committee of clinical trials of Xiangya Hospital of Central South University. In addition, all the procedures in this study were strictly conformed to the guidelines and principles of the Declaration of Helsinki. 22
Subjects
A total of 96 cases of patients, diagnosed with NPC in Xiangya Hospital of Central South University, were enrolled from January 2010 to January 2011. There were 73 males and 23 females included in this study. The patients’ age ranged from 25 to 80 years (average age: 49.40 ± 16.34 years) with 47 cases under 45 years and 49 cases above 45 years. Of these patients, 36 cases were with smoking (37.50%), 12 cases with drinking (12.50%), 27 cases with family histories (28.13%), and 43 cases with Pre-DNA ≥ 1355 copies/mL (44.79%), respectively. All patients were classified following the TNM staging system of 7th edition of NPC published by the International Union against Cancer/American Joint Committee on Cancer (UICC-AJCC). 23 There were 5 cases in I stage, 20 cases in II stage, 46 cases in III stage and 25 cases in IVA–B stage. Besides, there were 53 cases in T1–2 stage and 43 cases in T3–4 stage as well as 37 cases in N0–1 and 59 cases in N2–3. Inclusion criteria: (1) patients newly diagnosed with NPC based on histopathology; (2) with normal heart, liver, lung and kidney function, and hemoglobin > 90 g/L; (3) KPS score > 70; (4) without infected and metastatic lesions. Exclusion criteria: (1) patients suffered from other malignant tumors; (2) received radiotherapy or chemotherapy previously; (3) combined with severe dysfunction or other medical conditions cannot be treated; (4) women during pregnant or breast-feed stage; (5) cognitive impairment; (6) had contraindications to chemotherapy or radiotherapy. Serum samples from the NPC patients were collected before radiotherapy for real-time quantitative PCR (qRT-PCR) detection. Additionally, 107 healthy controls with physical examination at the same period were recruited, ranging from 20 to 82 years old (average age: 52.75 ± 17.86 years), including 82 males and 25 females. There were 31 cases of smoking (28.97%), 17 cases of drinking (15.89%) and 18 cases with family history (16.82%) included in control group. There was no significant difference between the two groups according to age, gender, smoking, drinking and family history (all P > 0.05).
Treatment
All patients involved in the research were treated with the conclusively IMRT regimen at our hospital. A planning computed tomography characterized by contrast and high-resolution was employed to scan the parts from the vertex down to 2 cm below sternoclavicular joint (slice-thickness, 3 mm). The radiation doses prescribed at 2.12∼2.43 Gy/fraction to the planning target volume (PTV) of the primary gross tumor volume (GTVnx) was 66∼72 Gy, the PTV of the GTV of infected lymph nodes (GTVnd) was 64∼70 Gy, the PTV of high-risk clinical target volume (CTV1) was 60∼63 Gy, and the PTV of low-risk clinical target volume (CTV2) was 54∼56 Gy. When launching the treatment, radiotherapy alone was recommended for stage I, coinstantaneous chemoradiotherapy for stage II, and concurrent chemoradiotherapy (CCRT) ± neoadjuvant (NCT)/adjuvant chemotherapy (ACT) for stage III to IVA–B according to institutional guidelines. Neoadjuvant or adjuvant chemotherapy containing cisplatin with 5-fluorouracil (PF) or cisplatin with docetaxel (TP) was implemented every 3 weeks for 2 or 3 cycles. Concurrent chemotherapy consists of cisplatin was carried out weekly or on weeks 1, 4 and 7 of radiotherapy. 24
Short-term efficacy and follow-up
Patients were followed-up periodically in our hospital and be treated with comprehensive assessments including physical examination, MRI for nasopharyngeal and neck, nasopharyngoscopy, blood test, chest X-ray and abdominal sonogram routinely every 3 months in the first 3 years, and every 6 months in the 4 to 5 years. The efficacy was evaluated when treatment was completed for 3 months by Response Evaluation Criteria In Solid Tumors (RECIST) showing as below: complete response (CR): visible lesions completely disappear over the course of 1 month; partial response (PR): tumor size is reduced by more than 50% in no less than 4 weeks; no change (NC): tumor size cut back by less than 50% or is increased by no more than 25%; progression of disease (PD): tumor size is increased by more than 25% or new lesions are formed. Total efficiency was calculated in line with the following formula: total efficiency = (CR + PR)/total number of cases × 100%. 25 The follow-up duration was an average of 48 months (3∼89 months). The duration of OS began the day of radiotherapy completion and ended the day of death or the last follow-up.
Cell culture
The NPC cell lines CNE-2 and HONE-1 purchased from American Type Culture Collection (ATCC, USA) was cultured in DMEM (Hyclon, USA), containing 10% inactivated fetal bovine serum (FBS, Gibco, USA) with addition of 100 units/mL penicillin-streptomycin (Gibco, USA) in an incubator (Thermo, USA) at 37°C with 5% CO2. Cells were routinely cultured and then subjected to subsequent experiments with 0.25% trypsin (Gibco, USA) for digestion and passage when the cells were covered with 80% of the microscopic field.
Dual luciferase reporter gene assay
The human Dicer1 3′-UTR fragment containing miR-BART6-5p binding site was amplified and inserted into the pmir GLo vector (Ambion) to construct a wild-type plasmid (Dicer1-WT) and a mutant plasmid (Dicer1-Mut). MiR-BART6-5p mimics negative control (NC) and miR-BART6-5p mimic was bought from Shanghai GenePharma (Shanghai, China). The CNE-2 and HONE-1 cells were grouped into NC + Dicer1-WT group, NC + Dicer1-Mut group, mimic + Dicer1-WT group and mimic + Dicer1-Mut group. CNE-2 and HONE-1 cells were plated in 96-well plates (4 × 104 cells/well) for a day before transfection. Fresh medium was replaced in 1 hour before transfection. The culture medium in the plate was discarded and then the transfection mixture which was prepared according to Lipofectamine 2000 (Invitrogen, USA) kits was added. Then the cultures were incubated for 24 h at 37°C, and the double luciferase reporter assay kit (Promega) was utilized to analyze the transcriptional activity, namely firefly luciferase activity / Renilla luciferase activity.
Cell grouping and transfection
MiR-BART6-5p inhibitor and si-Dicer1 which were demanded for transfection were also purchased from Shanghai GenePharma (Shanghai Zhangjiang Hi-Tech Park, Shanghai, China). The transfection was conducted using Lipofectamine™ 2000 (Invitrogen, Carlsbad, USA) as the cell grouping into Mock group, NC group, mimics group, inhibitors group, si-Dicer1 group and inhibitors + si-Dicer1 group, and cells were cultured in plates for routine incubation in 24 h prior transfection.
qRT-PCR
Total RNA samples were extracted using TRIzol (Invitrogen, USA) according to procedures of miRNeasy Mini kit (Qiagen, Germany). The concentration and purity of the extracted RNA was determined via NanoDrop2000 (NanoDrop 2000c, Thermo, USA), and then it was stored at −80°C for preparation. PCR premiers (Table 1) were designed in light of the gene sequences published in Genbank with the aid of Primer5.0 software by Shanghai GenePharma. qRT-PCR was carried out on an ABI Prism 7900HT (Applied Biosystems) using 2 × Quantifast SYBR Green PCR Master mix (QIAGEN) with 1 µmol/L primers. Primer Assay systems (Qiagen, Valencia, CA) of Hs_Dicer1_1_SG QuantiTect and miR-BART6-5p_1 miScript were introduced in the study. U6 and β-actin were used as the reference genes for miR-BART6-5p and Dicer1, respectively. Thermal cycling was run with an initial denaturation for 5 min at 95°C, followed by 40 cycles of 95°C for 10 s and 60°C for 30 s. Melting curve was adopted to evaluate the PCR data, and a CT value (inflection point of amplification curve) was obtained for each curve. And the formula 2−△△Ct was introduced to relative expression of target gene. 26 The experiment was repeated three times independently.
The primer sequences for qRT-PCR.

Western blot
BCA Protein Assay Kit (Pierce, Rockford, IL) was introduced to determine the concentration of the extracted cell protein. The extracted proteins with loading buffer were boiled at 95°C for 10 min, and the 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) electrophoresis (Boster, Wuhan, China) with 30 µg loading sample for each well was performed for protein isolation. Wet transmembrane (PVDF membrane) was conducted transferring 80 V electrophoresis to 120 V at a voltage of 100 mv for 45∼70 min. Afterward, the membranes were blocked at room temperature for 1 h with 5% BSA, and then incubated with addition of corresponding primary antibodies of Dicer1 (1:100, ab82539, Abcam) and β-actin (1:500, ab8226, Abcam) overnight at 4°C. When incubation finished, the membranes were rinsed triplicate with TBST for 5 min each time and then incubated with the second antibody (1:1000, ab131368, Abcam) at room temperature for 1 h, washing the membranes with TBST as stated above. After that, the membranes were filmed with chemiluminescence. The β-actin was selected as the loading control. Bio-Rad Gel Doc EZ Imager (Bio-Rad, California, USA) was applied for imaging, and target bands were optioned for gray level analysis using Image J. The experiment was repeated three times independently.
CCK-8 assay
Cells in the logarithmic phase from each group were collected and inoculated in 96-well plates with 3,000 cells suspended in 100 µL complete medium for each well. The culture plates were placed in a 37°C incubator and the cell were detected at 24, 48, 72 and 96 h, respectively. With addition of 10 µL CCK-8, the cell cultures were then incubated for another 3 h. Microplate reader (Bio-Rad, California, USA) was applied to detect optical density (OD) of at 450 nm for cell growth curve. The experiment was repeated three times.
Wound healing assay
Cells were plated in 6-well plates and cultured at 37°C after transfection. Sterile tips were utilized to draw lines with even scratch when the cells were covered with plate bottom, and the plate covers were marked for locating a same field when taking pictures at 0 h. Afterward, culture medium was removed when the plates were incubated at 37°C for 24 h, and the plates were rinsed triplicate with PBS to eliminate cell debris from the scratches. With addition of serum-free medium, plates were photographed at 48 h. An inverted microscope (CKX31, Japan) was employed to take photographs, and 6 visual fields were selected when it was a fixed position. The click-counting software was utilized to calculate cell numbers migrated to the scratch for statistical analysis. This experiment was conducted in triplicate.
Transwell invasion assay
The transwell chamber (Corning, Tewksbury, MA, USA) was added with matrigel (3.9 mg/mL, 60∼80 μL) and cultured at 37°C for 30 min until matrigel coagulated. The culture plates with addition of preheated medium were placed in a 37°C incubator for 2 h to allow complete hydration. Transwell chamber in each well was added with 0.5 mL cell suspension with a density of 5 × l04/mL, which was then incubated at 37°C for 24 h. The chamber was taken out to wipe out the cells on upper surface. With PBS washing, the chamber was immersed in pre-cooling methanol for 30 min to stabilize the cells transferred to the lower side of the chamber. After stained with 0.1% crystal violet solution for 10 min, an inverted microscope was used for observation as above to analyze the cell number, which moved to the lower chamber of Transwell. This experiment was repeated three times.
Annexin V/propidium iodide (PI) assay
After transfection for 48 h, cells were then harvested, washed, and resuspended in 500 µL of binding buffer containing 5 µL FITC-annexin V and 5 µL propidium iodide (PI) (Keygen, Nanjing, China). The cells were subjected to gentle vortexing and incubated for 15 min at 25°C in the dark followed by the assessment with flow cytometry (BD Biosciences, CA, USA). The positive cells labeled with Annexin V were viewed as the apoptotic cells, and cells showing PI positive and Annexin V negative were the necrotic, while apoptosis rate = (Annexin V+PI+ cells + Annexin V+PI− cells)/104 × 100%. This experiment was conducted in triplicate.
Clone formation assay
CNE-2 and HONE-1 cells were digested into monoplast suspension and adjusted to 200 cells in each well (6-well plate) with 3 repeated wells. Cells were X-irradiated with 0, 2, 4, 6, 8 Gy for each group and incubated at 37°C with 5% CO2 until visible clones appeared, and then fixed with 100% methanol and stained with 1% crystal violet. The number of colonies containing over 50 cells was counted under a microscope, and the planting efficiency (PE) was calculated as follows: PE = number of clones/number of inoculated cells × 100%. This experiment was repeated three times.
Radiosensitivity tests in vivo
The BALB/c female nude mice purchased from Shanghai Experimental Animal Center of Chinese Academy of Sciences, aging 4∼6 weeks and weighting 18∼22 g, were raised and tested in specific–pathogen free (SPF) experimental animal center. CNE2 cells was adjusted to single-cell suspension with a density of 5 × 106/mL using serum-free RPMI 1640 medium, and 0.1 mL cell suspension was implanted subcutaneously in the left groin of the mice. The tumor-bearing nude mice were grouped randomly into 6 groups when GTVnx reached 80 mm3 in 10∼14 d as below: Blank group (without any treatments), RT group, NC + RT group, mimics + RT group, inhibitors + RT group and si-Dicer1 + RT group with 6 mice for each group. NC, miR-BART6-5p mimics, miR-BART6-5p inhibitors or si-Dicer1 was intratumorally injected into the tumor weight at multiple sites of each mouse, respectively, and the mice in RT groups were all endured a 6 Gy dose of ionizing radiation next day. There was another 6 Gy dose of ionizing radiation encountered by all RT groups after 14 days. And NC, miR-BART6-5p mimics, miR-BART6-5p inhibitors or si-Dicer1 was injected twice a week, and the RT group was treated with equal amount of saline simultaneously. The treatment for all groups continued for about 4 weeks. Caliper was utilized to measure tumor volume (in mm3) which was calculated using the modified ellipse formula (volume = length × width2/2) every 3 to 4 days. Nude mice were euthanized by decapitation and the xenograft tumors were immediately removed after death.
Immunohistochemistry
The tumor tissues embedded with paraffin were cut into slices to dewax and incubated with 3% H2O2 at room temperature for 5∼10 min. Afterward, the sliced tumor was rinsed with distilled water and soaked twice in PBS for 5 min. The normal 5∼10% goat serum (diluted by PBS) was blocked and incubated at room temperature for 30 min, discarding the serum without rinsing. Primary antibody of Dicer 1 (1:20, ab82539, Abcam) was added dropwise and incubated at 4°C overnight, with PBS washing for 5 min in triplicate. An appropriate amount of biotin-labeled secondary antibody was later added with incubation at 37°C for 2 h. After rinsed with PBS for 5 min in triplicate, (HRP)-conjugated streptavidin working solution was added, incubated at 37°C for 30 min, and then rinsed with PBS, following by diaminobenzidine (DAB) staining for 5 min and tap water rinsing. Thereafter, the samples were counterstained, dehydrated, cleared and mounted. The immunohistochemistry kit was purchased from Vector (PK-6100). Micrographs were acquired with an Olympus CKX31 microscope (Olympus, NSW, AU). Dicer1 protein is overexpressed in cytoplasm.
Statistical analysis
The statistical analyses were conducted with SPSS 22.0. Representative experiments were independently repeated three times. Chi-square test or Fisher’s exact text was introduced for the enumeration data analysis, and Wilcoxon Rank Sum test was for nonparametric data. All measurement data accorded with the normal distribution was presented by mean ± standard deviation (SD). Correlation between two variables was conducted with Pearson’s correlation analysis, and means differences between two groups were compared by means of independent-sample t test. Comparisons of multiple independent groups were analyzed using One-way ANOVA followed by Turkey’s HSD test. ROC curve was involved to evaluate the diagnostic efficiency of miR-BART6-5p and Dicer1 on radiotherapy efficacy of NPC. Univariate and multivariate survival analysis was carried out using Kaplan-Meier method and COX regression risk model, respectively. P < 0.05 was considered as statistically significance.
Results
Expressions of miR-BART6-5p and Dicer1 in NPC patients and its association with clinicopathological features
qRT-PCR results revealed that expression of miR-BART6-5p was apparently higher while Dicer1 presented lower expression in patients with NPC than healthy controls (all P < 0.05, Figure 1A–B). The expression of miR-BART6-5p and Dicer1 in NPC tissues was negatively correlated (r = −0.287, P = 0.005) according to Pearson’s correlation analysis (Figure 1C). Using the median expression level as the cut-off value, qRT-PCR made clear that the expressions of miR-BART6-5p (median = 2.30) and Dicer1 (median = 0.51) were not associated with age, gender, smoking, drinking, family history and chemotherapy of NPC patients (all P > 0.05), but were intimately correlated with T stage, clinical stage and Pre-DNA (all P < 0.01, Table 2).
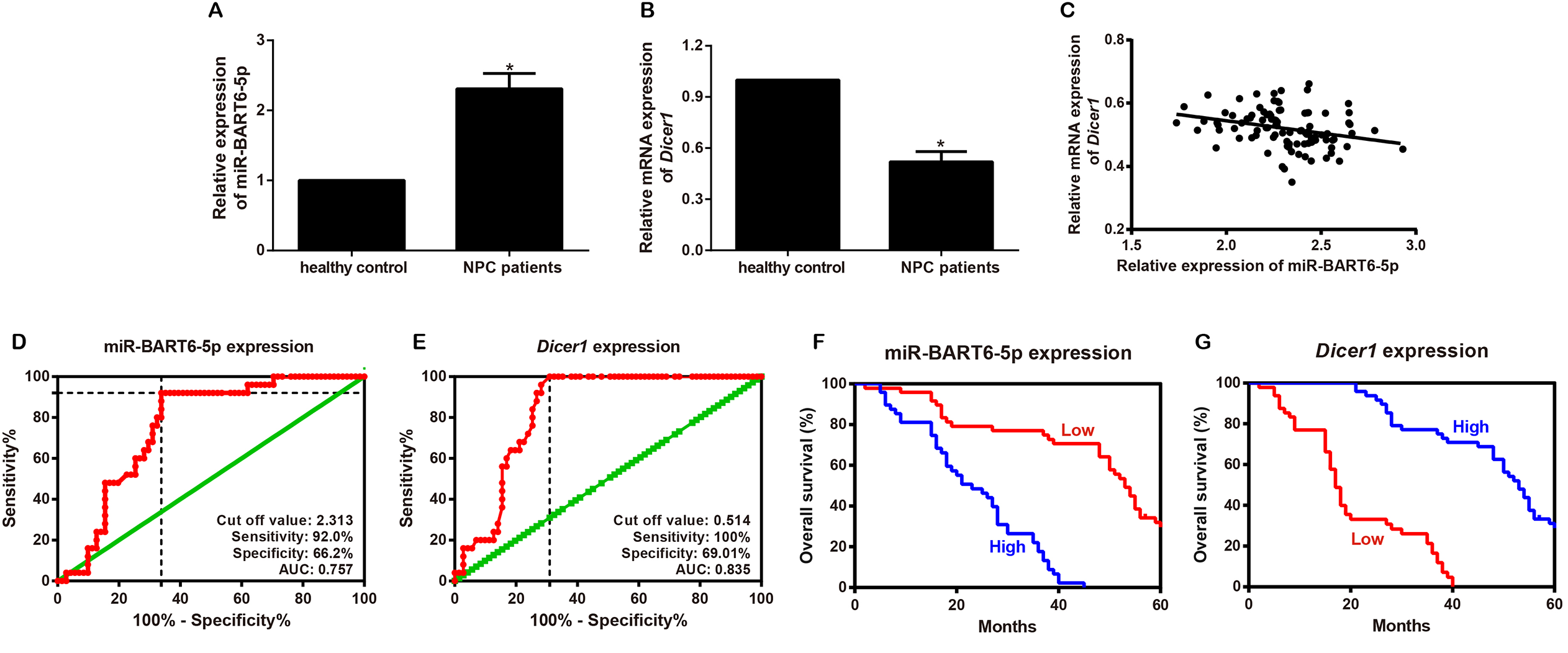
Associations of expression of miR-BART6-5p and Dicer1 in serum with clinicopathological feature and prognosis. Note: A–B, Comparisons of expression of miR-BART6-5p (A) and Dicer1 (B) between NPC patients and healthy controls, *P < 0.05; C, Correlation analysis of miR-BART6-5p with Dicer1 in serum of NPC patients; D–E: Diagnostic efficacy of expression of miR-BART6-5p (D) and Dicer1 (E) on NPC therapy evaluated by ROC curve; E–F: Influence of expression of miR-BART6-5p (F) and Dicer1 (G) analyzed by Kaplan-Meier univariate survival analysis on NPC prognosis.
Association of expression of miR-BART6-5p and Dicer1 in serum with clinicopathological feature of NPC (n (%)).
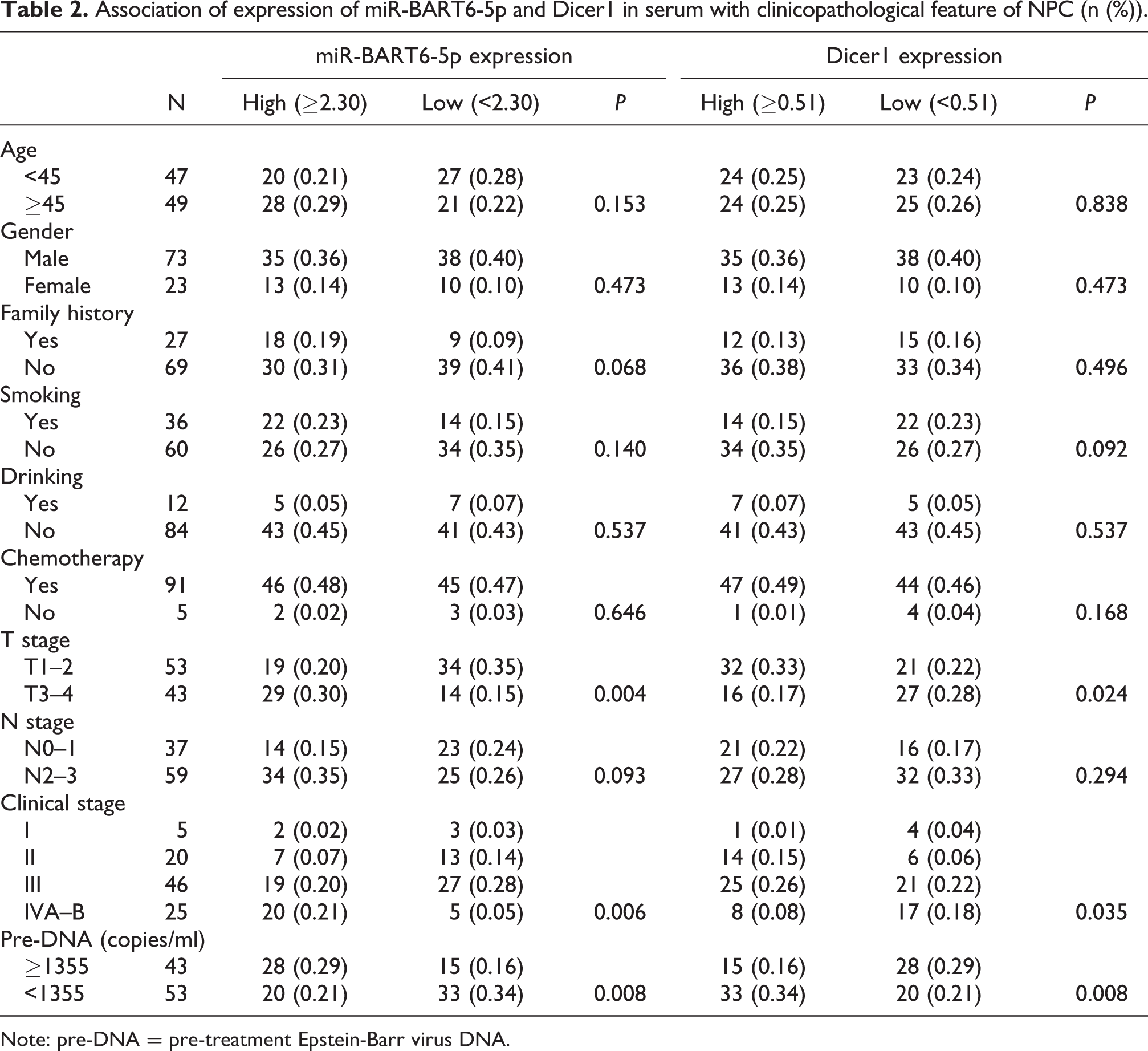
Note: pre-DNA = pre-treatment Epstein-Barr virus DNA.
Effects of miR-BART6-5p and Dicer1 on radiosensitivity and prognosis for NPC patients
The overall efficiency was 73.96% (71/96) after 3 months of treatment. As expected, the expression of miR-BART6-5p was reduced and Dicer1 increased in effective group (miR-BART6-5p: 2.26 ± 0.23; Dicer1: 0.54 ± 0.06) when compared to the non-effective group (miR-BART6-5p: 2.44 ± 0.99; Dicer1: 0.47 ± 0.04) (both P < 0.001). It turned out by ROC curve (Figure 1D–E) that the area under curve (AUC) for miR-BART6-5p was 0.757 (0.659∼0.854), and the sensitivity and specificity was 92.00% and 66.20%, respectively. In the meantime, the AUC for Dicer1 was 0.835 (0.757∼0.913), with sensitivity of 100.00% and specificity of 69.01%. Kaplan-Meier univariate survival analysis showed that the expressions of miR-BART6-5p and Dicer1 were prognostic factors of NPC (miR-BART6-5p: χ2 = 48.981, Dicer1: χ2 = 67.210, all P < 0.001) (Figure 1F–G). Without chemotherapy, high miR-BART6-5p expression, and low Dicer1 expression were the independent risk factors for NPC prognosis by multivariate Cox regression analysis (all P < 0.05, Table 3).
Prognostic factors for NPC with radiotherapy using multivariate Cox regression analysis.

Note: B: regression coefficient; S.E.: standard error; Wald: Chi-Square value; df: degree of freedom; Exp(B): hazard ratio (HR); 95%CI: 95% confidence interval; pre-DNA: pre-treatment Epstein-Barr virus DNA.
MiR-BART6-5p directly targeted Dicer1
Dicer1 was identified as one of the target genes of miR-BART6-5p using a computational search, namely as Diana Tools (http://diana.imis.athena-innovation.gr/DianaTools/index.php?r=microtv4/index), an algorithm for miRNA target prediction, which showed that miR-BART6-5p had four potential binding sites at the 3′UTR of Dicer1 (Figure 2A–B

MiR-BART6-5p targeted Dicer1 by using Diana Tools and dual-luciferase reporter assay. Note: A, Diana Tools predicted the binding sites of miR-BART6-5p with Dicer1, in which the red was the 4 bind sites, the blue was sequence primers and linker primers, and the blue italic was the enzyme loci of Spe1 and Pml, respectively; B, The luciferase reporter plasmid containing the four 5p strand target sites; C, MiR-BART6-5p regulating Dicer1 expression based on dual-luciferase reporter gene assay, * compared with the NC + Dicer1-WT group, P < 0.05.
Expression of miR-BART6-5p and Dicer1 in each transfected groups of NPC cells
The expression of miR-BART6-5p and Dicer1 in CNE-2 and HONE-1 cells presented no significant differences analyzed by qRT-PCR in the Mock and NC groups. When compared with the Mock group, miR-BART6-5p expression was 3-fold increase in the mimic group, and Dicer1 expression was decreased at least 70% in the mimic group and the si-Dicer1 group. Moreover, miR-BART6-5p expression at least 3-fold lower was found in the inhibitor group and the inhibitor + si-Dicer1 group, and Dicer1 expression at least 4-fold higher in the inhibitor group, as compared with NC group (all P < 0.05, Figure 3A, B). Western blot analysis revealed that the protein expression trends of Dicer1 in all groups were consistent with the gene expression (Figure 3C, D).

Expression of miR-BART6-5p and Dicer1 in NPC cells in each group. Note: A–B, Expression of miR-BART6-5p (A) and Dicer1 (B) were detected by qRT-PCR; C–D, Protein expression of Dicer1 was analyzed using Western blot; the same letters referring to no significant differences (P < 0.05), and different letters representing significant differences (P < 0.05).
MiR-BART6-5p promotes proliferation and inhibits apoptosis of NPC cells by targeting Dicer1
As compared to the Mock group, NPC cell proliferation (at 48 h, 72 h and 96 h) was increased and apoptosis rates was decreased both in the mimic group and the si-Dicer1 group, while the inhibitor group showed depressed ability of proliferation and promoted apoptosis of NPC cells (all P < 0.05). Additionally, higher cell proliferation and lower apoptosis rate in the inhibitor + si-Dicer1 group were found than the inhibitor group (all P < 0.05), which was shown in Figure 4.

MiR-BART6-5p promotes proliferation and suppresses apoptosis of NPC cells by targeting Dicer1. Note: A–B, CCK8 was used to detect proliferation of CNE-2 cells (A) and HONE-1 cells (B); C–D, Annexin-FITC/PI was performed to evaluate apoptosis of NPC cells; the same letters referring to no significant differences (P < 0.05), and different letters representing significant differences (P < 0.05).
MiR-BART6-5p facilitates invasion and metastasis of NPC cells by targeting Dicer1
The wound healing assay and Transwell invasion assay demonstrated that the cell number of migration and invasion of Mock, NC and inhibitors + si-Dicer1 groups had no significant differences (all P > 0.05), which were increased in mimics group while reduced in inhibitors group (all P < 0.05) when compared with the Mock group. The cell migration and invasion ability in inhibitors + si-Dicer1 group were much stronger than inhibitors group (all P < 0.05, Figure 5).
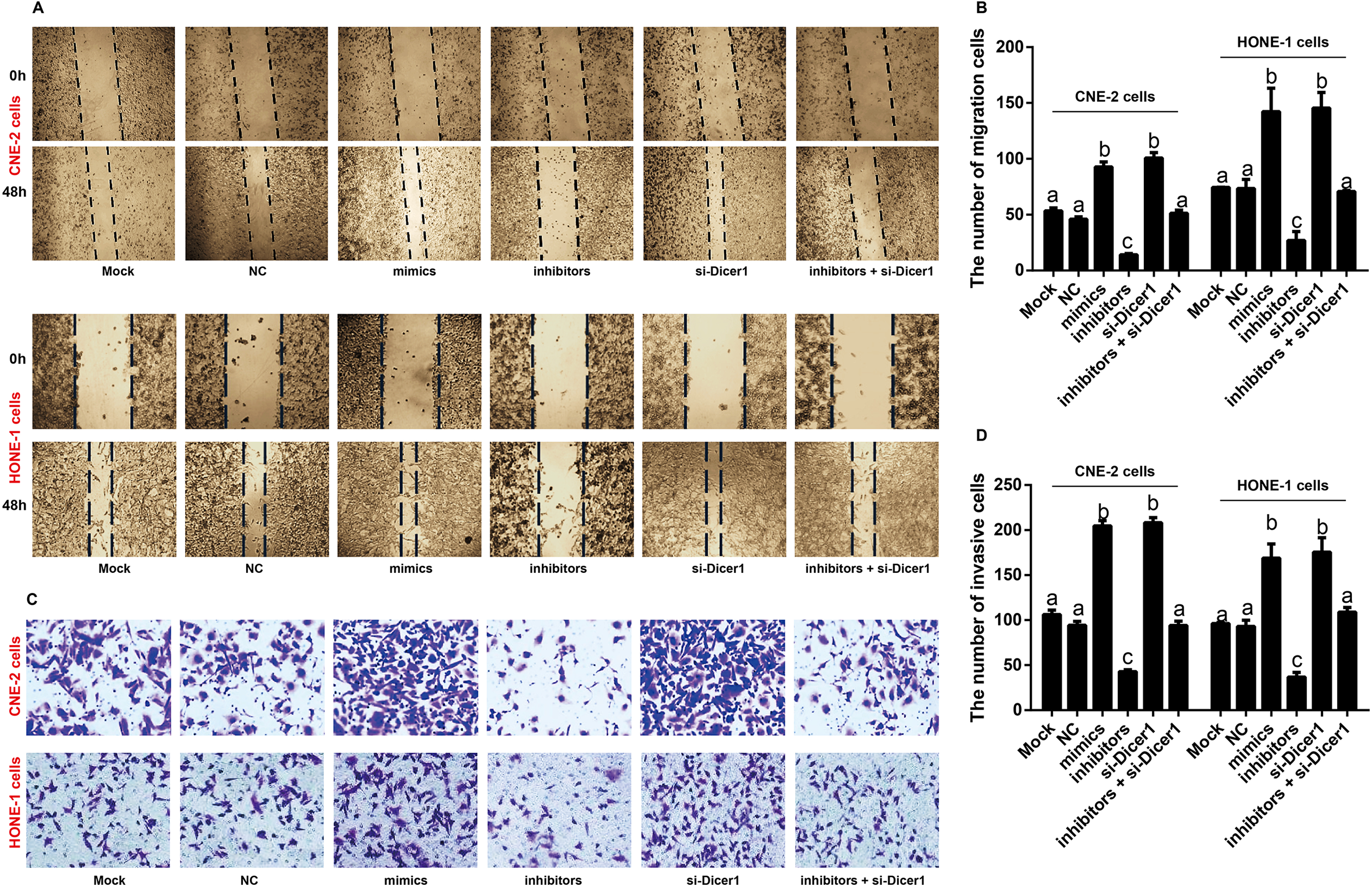
MiR-BART6-5p targeting Dicer1 to facilitate invasion and migration of NPC cells. Note: A–B: Cell migration was assessed using scratch wound healing assay; C–D: Cell invasion were investigated using Transwell invasion assay; the same letters referring to no significant differences (P < 0.05), and different letters representing significant differences (P < 0.05).
Effects of miR-BART6-5p on radiosensitivity of NPC cells by targeting Dicer1
The clone formation assay was conducted to assess the effects of miR-BART6-5p targeting Dicer1 on radiosensitivity of NPC cells (CNE-2 and HONE-1) treated by X-ray irradiation with different doses of 0, 2, 4, 6, and 8 Gy. Compared with the Mock group, the planting efficiency (PE) of NC group showed no significant differences (all P > 0.05), but which was significantly increased in mimic group, and decreased in inhibitor group, meanwhile, it changed more obvious with the increase of radiation dose (all P < 0.05). In addition, the PE was higher in inhibitor + si-Dicer1 group than that in inhibitor group (all P < 0.05, Figure 6). Results from in vivo experiments revealed that all nude mice were alive. Immunohistochemical staining revealed that Dicer1 expression presented no significant differences among Blank group, mimics + RT group and si-Dicer1 + RT group, as well as between RT group and NC + RT group, furthermore, inhibitor + RT group has the highest Dicer1 expression of all groups, and the mimics + RT and si-Dicer1 + RT group both showed lower Dicer1 expression than in RT and NC + RT group (Figure 7). In addition, the tumor volume and tumor weight of mice in inhibitor + RT group were significantly decreased than those of the other 5 groups, and the mimics + RT and si-Dicer1 + RT group both had larger tumor volume and bigger tumor weight than RT group (all P < 0.05, Figure 7B, C).

Effects of miR-BART6-5p targeting Dicer1 on radiosensitivity of NPC cells by clone formation assay in vitro.

Effects of miR-BART6-5p on radiosensitivity of NPC by targeting Dicer1 in vivo. Note: A, Dicer1 expression detected by immunohistochemistry in nude mice (× 200); B, Comparison of tumor volume at different time in each group; C, Comparison of tumor weight at different time in each group.
Discussion
NPC was found to be a consequence of multi-factor interactions concerning the expression and activation of oncogene and tumor suppressor gene in the host. While EBV miRNAs used as an oncogene or tumor suppressor, present great significance in regulating the incidence and progression of various cancers, including NPC.
Extensive studies discovered that plenty of BART miRNAs, such as miR-BART10-3p, miR-BART1-5p, miR-BART7-3p and miR-BART6-5p, were obviously increased in NPC, 27 –29 but the tumor suppressor gene Dicer1 was verified to have lower expression in multiple tumors, including ovarian cancer, lung cancer, as well as NPC, 17,18,20,30 which was in line with our current findings that miR-BART6-5p expression was upregulated but Dicer1 expression was downregulated in serum of NPC patients, indicating miR-BART6-5p could function as an oncogene to upregulate the expression of the cancer suppressor gene Dicer1 to be participated in the occurrence and development of NPC. Furthermore, our results found that expressions of miR-BART6-5p and Dicer1 had an intimate relationship with T stage, clinical stages and EBV-DNA, all of which were proved to be prognostic factors of NPC. In the past investigations, numerous miR-BARTs, for instance, miR-BART1, miR-BART7-3p, and miR-BART10-3p, have been pointed out to be correlated with clinical stages or prognosis of NPC, 12,27,29 and low expression of Dicer1 was associated with high clinical stages and poor prognosis to be observed in patients with ovarian or gastric cancer. 31,32 Besides, EBV-DNA, as a tumor marker, is highly expressed in NPC, positively correlated with advanced clinical stages, further reflecting the tumor progression and prognosis of NPC from the perspective of biological behaviors. 24,33
A Bioinformatic tool, DIANA-microT 3.0, identified Dicer as one of the target genes of miR-BART6-5p RNAs, 21 which was confirmed to the notion that miR-BART6-5 targeted Dicer1 in human NPC CNE2 cells in accordance with dual luciferase reporter assay. To our knowledge, miRNA mimics, a kind of small, chemically-modified double-stranded RNA molecules, are designed to mimic endogenous mature miRNAs, whereas miRNA inhibitors are sequence-specific and chemically modified oligonucleotides to specifically target and knockdown miRNA molecule. 34 In this paper, miR-BART6-5p mimics significantly decreased Dicer1 expression while miR-BART6-5p inhibitors elevated Dicer1 expression distinctly in CNE-2 and HONE-1 cells, which further declared that miR-BART6-5p could directly target Dicer1 to be important for NPC progression.
MiRNAs play crucial roles in regulating tumor cell adhesion, extracellular matrix degradation and angiogenesis by regulating transcription and translation of downstream target genes, namely metastasis-related genes, leading to invasion and metastasis of malignant tumors, which were the principal causes of patients’ death. 35,36 MiR-BART6-5p and si-Dicer1 was found to facilitate invasion and migration of NPC cells in our study, similar to miR-BART1, miR-BART9 and miR-BART10-3p in NPC by inhibition of their target mRNAs. 27,29,37 Meanwhile, evidence showed the mature miRNA level was declined as the function of Dicer was impaired, which may promote tumor growth and metastasis by inducing epithelial-mesenchymal transition. 20,38 Therefore, it was speculated that miR-BART6-5p may affect the tumor cell adhesion in NPC by targeting Dicer1, thus promoting cell metastasis. Further, increasing investigations stating that the network, composed of miRNA and its target gene, was participated in modulating the vital life activities, such as cell proliferation, apoptosis and metabolism, while the imbalance of cell proliferation and apoptosis conspire to promote tumorigenesis. 39,40 For example, a present study confirmed that there were at least 132 genes related to apoptosis regulated by multiple miR-BARTs in type III latent infection. 41 Chen X et al. demonstrated that miR-18a, another miRNA, could also target Dicer1 to promote the cell proliferation and apoptosis of NPC. 20
To date, radiotherapy is the major therapy of NPC, and the radiosensitivity of NPC had relationship with numerous gene expressions in tumor cells, which may lead to the radiation resistance. 42 We brought to light that the radiotherapy could reduce miR-BART6-5p expression in patients with NPC, which was identical to the findings of Zhang G et al. that miR-BART7 and miR-BART13 expressions were markedly decreased in patients suffered NPC after radiotherapy. 10 Furthermore, Selever et al. expounded that Dicer overexpression was not conductive to breast cancer chemotherapy. 43 In this study, we discovered that Dicer1 was highly expressed in NPC patients with effective radiotherapy, supporting the hypothesis that miR-BART6-5p could act as an oncogene gene influenced the NPC radiotherapy efficacy by regulating the activity of tumor suppressor gene Dicer1. In addition, the experiments in vivo in our study verified the effects of miR-BART6-5p and Dicer1 on radiosensitivity for NPC, and it turned out that upregulation of miR-BART6-5p and downregulation of Dicer1 evidently increased the clone formation rate, thus the tumors were suppressed in nude mice injected with miR-BART6-5p inhibitors and then enhanced radiotherapy, suggesting that inhibition of miR-BART6-5p could inhibit cell proliferation and tumor growth to thereby strengthen radiosensitivity of NPC by targeting Dicer1. Yet, the specific mechanism of miR-BART6-5p targeting Dicer1 to influence the downstream pathways remains unknown and more comprehensive and profound explorations are urgently required. Besides, although depletion or supplementation of miRNA, as well as targeted silencing of disease-associated genes by siRNA hold considerable promise as novel therapeutic strategies, 44 the possibility of off-target effects is highly associated with the miRNA and gene therapy. The microRNA-like off-target silencing is the extent to which it affects the specificity of therapeutic siRNAs in vivo because of the similarity in function of an siRNA guide strand and a miRNA. 45,46 Considering the above issue, the invention relates to a method of minimizing off-target effects should be further explored to validate the result of our experiment.
In conclusion, enhanced miR-BART6-5p and reduced Dicer1 were found in NPC patients, associated with clinical stages, EBV-DNA and prognosis. And miR-BART6-5pcould contribute to proliferation, migration and invasion of NPC cells, accompanied by suppression of apoptosis as well as reduction of radiosensitivity via targeting Dicer1, which provided novel clues for NPC therapy.
Footnotes
Authors’ note
Jing Tang and Zhao-Yang Liu are the first co-authors.
Acknowledgments
The authors want to thank for all the people for their help in the paper editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
