Abstract
Cytarabine (Ara-C) is a nucleoside analogue used in the treatment of cancers and viral infections. It has teratogenic potential and causes a variety of birth defects in fetuses. Alpha-lipoic acid (ALA) is a natural antioxidant offers protection against the developmental toxicity induced by drug- or toxicant-exposure or pathological conditions. This study was aimed at evaluating the protective effect of ALA against Ara-C induced developmental toxicity in rat fetus. Pregnant rats divided into five groups and received normal saline, ALA200 mg/kg, Ara-C12.5 mg/kg, Ara-C25 mg/kg and, Ara-C25 mg/kg plus ALA200 mg/kg respectively from gestational day (GD) 8 to GD14 and sacrificed on GD21. Ara-C treatment led to a significant and dose-dependent decrease in food intake, weight gain, placental weight, and an increase in oxidative stress in pregnant rats. Further, the in-utero exposure to Ara-C led to an increase in fetal mortality, resorptions, oxidative stress, external morphological anomalies and limb abnormalities, and impaired ossification. Co-administration of ALA resulted in amelioration of the footprints of Ara-C induced toxicity in pregnant rats as well as the fetus. These findings indicate that the ALA supplementation offers protection against developmental toxicity caused by Ara-C prenatal exposure in rats.

Keywords
Introduction
Cancer is standing as the second leading cause of death globally after cardiovascular disease for more than a decade. 1 However, as cardiovascular disease is likely to decrease substantially, cancer is expected to become the leading cause of death globally in the near future. 2,3 Further, cancer reported as the second leading cause of deaths in the female during the childbearing years. The incidence of cancer in pregnancy is about 1 in 1000 (0.07% to 0.1% of) pregnancies and emerged as the second leading cause of maternal mortality after pregnancy-related vascular complications. 4 –6 The management of cancer in pregnancy is very complex, as it requires balancing the short- and long-term maternal and fetal risks associated with it. 4 The use of chemotherapy should be avoided in the first trimester as the rate of fetal malformation, and the mortality is very high whereas it can be used during second and third trimesters, as the teratogenic risk is less. However, chemotherapy in the second and third trimester reported causing fetal toxicity like premature delivery, stillbirth, myelosuppression, impairment in growth, cognition, skeletal system, and fertility. 4,7 The use of chemotherapy during pregnancy has progressively increased over the last 20 years. 8 On the other hand, hematological malignancies are one of the commonly diagnosed cancers in pregnancy along with breast and cervical cancers 9 with an incidence of approximately 1 in 10,000 pregnancies. Acute leukemia mainly acute myeloid leukemia (AML) is the most common hematological malignancies diagnosed in pregnancy. 10
Cytarabine (cytosine arabinoside, 1-β-D-arabinofuranosylcytosine, Ara-C) is a well-known pyrimidine (deoxycytidine) analog and is the most widely used chemotherapeutic agent for the treatment of hematological malignancies. 7,11,12 For more than four decades, Ara-C is being considered as the backbone for AML treatment and it is also used for the treatment of solid tumors in combination with other antineoplastic drugs. 13,14 The triphosphate nucleotide metabolite of Ara-C (Ara-CTP) is responsible for its cytotoxic effects. Ara-CTP acts as a competitive inhibitor of deoxycytidine triphosphate (dCTP) and gets incorporated into DNA in place of dCTP which inhibits DNA polymerase thereby inhibits DNA synthesis during replication and repair which ultimately causes cell death. 15 Ara-C cause genotoxic and oxidative stress that induces apoptosis. 12,16,17 Albeit cytarabine is established as the cornerstone of AML therapy, its use is reported to cause a wide variety of side effects including genotoxicity, myelotoxicity, neurotoxicity, gastrointestinal toxicity, gonadal toxicity and teratogenicity in a dose-dependent manner. 18,19 Several clinical and experimental studies revealed the teratogenic potential of Ara-C. Prenatal exposure to Ara-C reported causing several anomalies in external morphology and skeletal system development of the fetus in rodents. The Ara-C induced anomalies include resorption, fetal death, growth retardation, cleft palate, cleft lip, micrognathia, kinky tail, hydrocephalus, encephalocele, club foot, phocomelia, hemimelia, limb anomalies including digital defects (oligo-, poly-, syn-, brachy-, ectro-, a-dactyly and diplopodia), narrowing or complete disappearance of joint spaces, impaired ossification of the bones, distortion, and fusion of the bones of the axial and appendicular skeletal system (Table 1). Owing to its inevitable use in some leukemia cases despite its adverse effects, it is essential to intervene a safe protective agent.
Experimental studies reported teratogenic potential of Ara-C.

GD, Gestation day.
Alpha-lipoic acid (thioctic acid, ALA, 1,2-dithiolane-3-pentanoic acid) is a naturally occurring organosulfur compound. 39 ALA, an antioxidant produced in mitochondria of plants, animals including humans plays an essential role in several metabolic processes. 40 ALA and its metabolite dihydrolipoic acid are regarded as “universal antioxidant” and acts as biological antioxidant, metal chelator, re-generator of other antioxidants, and modulator of several signaling pathways. 41,42 Copious research is emphasizing that ALA with its pleotropic effects offers therapeutic benefit in various pathological conditions including drug-induced toxicities. 43 The use of ALA in pregnancy found to be very promising in improving pregnancy outcomes. 40,44,45 Several experimental studies revealed the protective effect of ALA on the fetus in maternal diabetic or drug- or toxicant-exposure conditions. 40,46,47 Further, ALA reported to promote bone formation and prevent bone loss in various pathological conditions. 40,48 –58
As the use of natural antioxidants in alleviating chemotherapy-induced toxicity is gaining much attention and seems to be promising, 59 we explored the potential of ALA in offering protection against Ara-C induced developmental toxicity in rats.
Methodology
Drugs and chemicals
Cytarabine and ALA were obtained from Sigma Aldrich, USA. All other reagents (analytical grade) used for the experiment were purchased locally.
Animals and experimental design
The Institutional Animal Ethics Committee approved the experimental protocol and the experiments were performed in accordance with the guidelines of committee for the purpose of control and supervision of experimentation on animals. All the animals were housed in controlled environmental conditions (22 ± 2°C temperature, 50 ± 10% humidity and 12 hrs light/12 hrs dark cycle). Animals had unlimited access to standard laboratory animal feed (purchased from commercial supplier) and water and they were acclimatized to the experimental conditions for 1-week prior to the experiments.
The female rats were selected for mating after a careful visual examination of external genitalia. The female rats with swollen, pink and moist vaginal opening with striations or wrinkling were considered in estrous. The female rats in estrous, weighing 180–220 grams (3–4 months age) were allowed for mating with male rats in 2:1 ratio. The female rats observed on next day morning and rats having vaginal plug and/or traces of sperm in vaginal smears considered as pregnant (Gestation Day, GD0). The animals kept for acclimatization and non-pregnant rats separated from the pregnant rats on GD7. On GD8, the rats divided in to five groups each containing six pregnant rats: control, ALA, Ara-C 12.5, Ara-C 25, and protection groups and have received normal saline, ALA 200 mg/kg, Ara-C 12.5 mg/kg, Ara-C 25 mg/kg and, Ara-C 25 mg/kg plus ALA 200 mg/kg respectively from GD8 to GD14. The food intake and body weight of the pregnant rats were recorded daily. Each dam was examined at least once in a day for gross pathological and clinical signs like morbidity, secretions, lesions, abortions, mucous membrane status, behavior, nervous signs and mortality. On GD21, the dams were sacrificed and laparotomy was done. The uterine horns of the dams were examined for fetal distribution, and resorption sites. A caesarian section was done and fetuses were carefully separated from placenta. Placenta were collected, weighed and used for estimation of maternal oxidative stress biomarkers. Fetus were examined carefully to determine the following parameters: a) number of live or dead fetus, b) growth retardation (body weight, body length (crown–rump length), and tail length), c) morphological anomalies d) oxidative stress, e) skeletal system anomalies by using Alizarin S red and Alcian blue (ARAB) double (differential) staining and mineral (calcium, phosphorous) analysis.
Determination of skeletal abnormalities
The procedure for double (differential) staining with ARAB dyes was adopted from the method used by Wallin J et al. 1994. 60 In brief, fetal skin was removed and evisceration was done. Then, the fetuses were subjected to following sequence of events: fixation in 100% ethanol (EtOH) for 24 hours at room temperature; incubation in 100% acetone for 24 hours at room temperature; staining with ARAB solution (consists of 0.3% Alcian blue in 70% EtOH, 0.1% Alizarin red S in 95% EtOH, Glacial acetic acid, 70% EtOH in 1:1:1:17 ratio) for 3–4 days at 37°C; one time rinsing with water; incubation in 1% KOH (in water) for 3–5 hours; 2–3 washings with 1% KOH after every 8–10 hours; washings with 20%, 50% and 80% glycerol (in 1% KOH) 24 hours for each concentration. The fetuses stained with ARAB were evaluated for skeletal system development by examining the portions of Alizarin S Red (stains ossified portion) and Alcian blue (stains cartilage or non-ossified portion) staining in the bones. The fetus that exhibits less intensified or absence of Alizarin S Red of staining as compared to control fetus is considered to possess less ossification which indicates the impaired development of skeletal system. Further, the number of ossification centers 61 –63 and percent ossification 64 was calculated. The number of ossification centers was calculated in sternum, centrum and arches of thoracic, lumbar, sacral and coccygeal vertebrae, pelvic bones, carpals and metacarpals and percent of ossification was calculated in long bones including clavicle, humerus, radius, ulna, femur, fibula and tibia.
Bone mineral analysis
The femur and tibia of hind limbs were carefully isolated, cleared of soft tissue residues, air-dried and incubated with ethylene diamine tetra acetic acid (EDTA) for demineralization for a period of 24 hrs. 65 The EDTA supernatant was analyzed for calcium and phosphate content using colorimetric methods as described by 66 and 67 respectively.
Determination of oxidative stress (OS) markers
The placenta of dams and fetal liver were used for estimation of OS markers including malondialdehyde (MDA), glutathione (GSH), glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), catalase (CAT). 68 The isolated placenta and liver were homogenized using 0.1-M phosphate buffer (pH 7.4) containing 3-mM EDTA. Then, it was centrifuged (700 g for 10-min) and the supernatant was used for the determination of OS markers, including malondialdehyde (MDA), glutathione (GSH), glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), catalase (CAT) and protein content were measured according to references 69 –74 respectively. The MDA levels were expressed as µmol/mg protein. The GSH and GSH-Px levels were expressed as nmol/mg protein whereas CAT, and SOD levels were expressed as µmol of H2O2/min/mg protein.
Results
Maternal toxicity
No clinical abnormalities, morbidity and mortality were observed in dams of all the groups. The dams of control and ALA groups found to have normal food intake (Figure 1a), and weight gain (both weekly and overall) during treatment period (Figure 1b–d). However, there was a significant and dose-dependent reduction in food intake, and weight gains of dams treated with Ara-C as compared to control group. Interestingly, a significant increase was observed in food intake and weight gains of dams co-administered with ALA 200 mg/kg and Ara-C 25 mg/kg as compared to dams received Ara-C 25 mg/kg alone. The trends of percentage weight gain (Figure 1e) in the groups were found to be similar with the trends of weight gain.
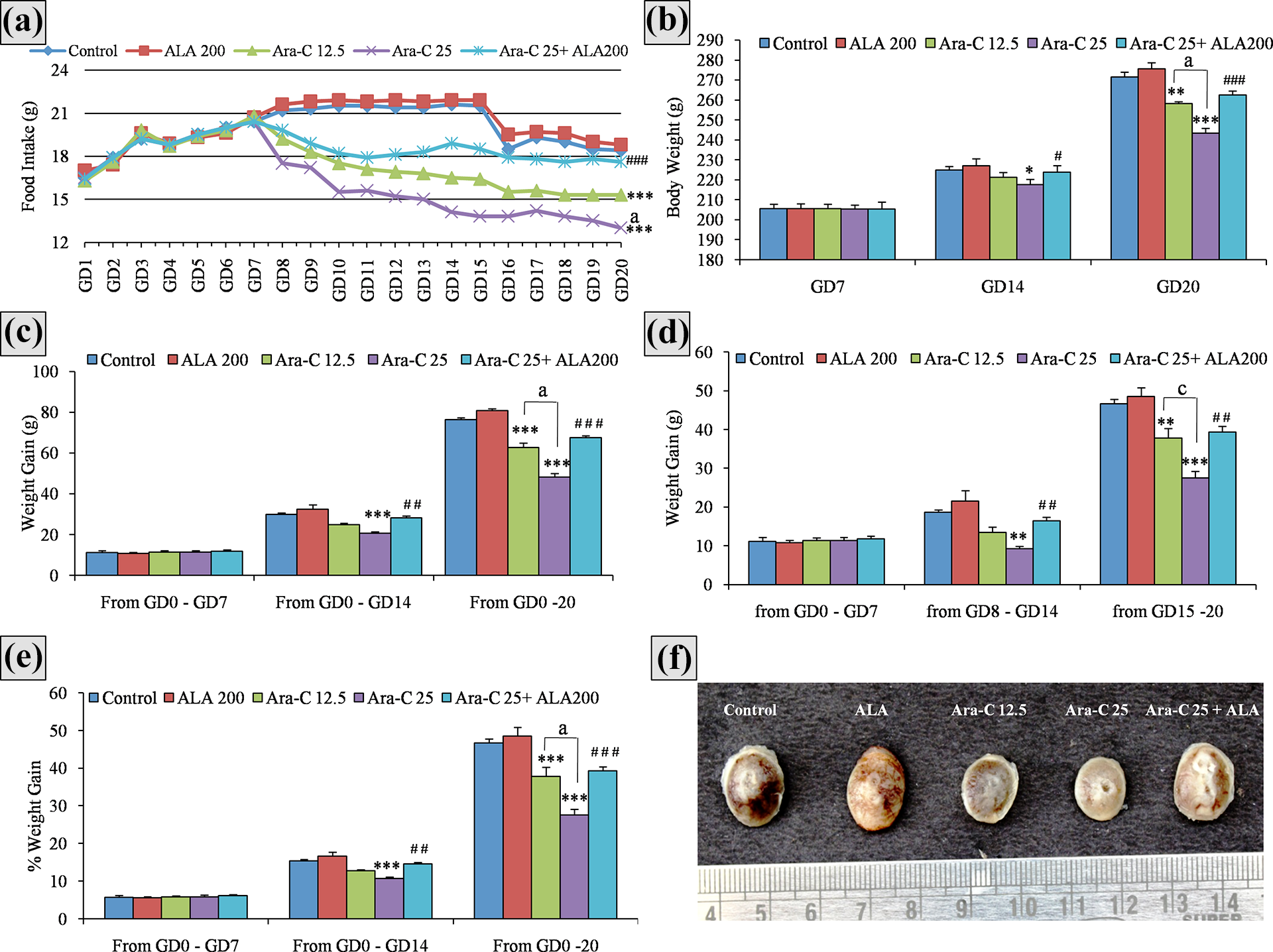
Effect of Ara-C and ALA treatments on (a) food intake, (b) body weight, (C) weight gain, (d) weekly body weight gain, (e) %weight gain, and (f) placenta size. The trends of food intake, body weight gain, and placenta size clearly indicating a significant and dose-dependent maternal toxicity in groups received Ara-C as compared to control group and amelioration of the same in Ara-C+ALA combination group as compared to Ara-C 25 mg/kg alone group. All the values are expressed as mean ± SEM, (n = 6), *** P < 0.001 vs. control, ** P < 0.01 vs. control) * P < 0.05 vs. control; # P < 0.05 vs. Ara-C25 mg/kg, ## P < 0.01 vs. Ara-C25 mg/kg, ### P < 0.001 vs. Ara-C25 mg/kg; a P < 0.001 vs. Ara-C12.5 mg/kg, b P < 0.01 vs. Ara-C12.5 mg/kg, c P < 0.05 vs. Ara-C12.5 mg/kg. In graph (a) due to space limit P values are not indicated against each day. The P value indicators placed at GD20 represents the P value indicators for all the days from GD8-GD20. Ara-C, cytarabine; ALA, alpha-lipoic acid 200 mg/kg, GD, gestation day; g, grams.
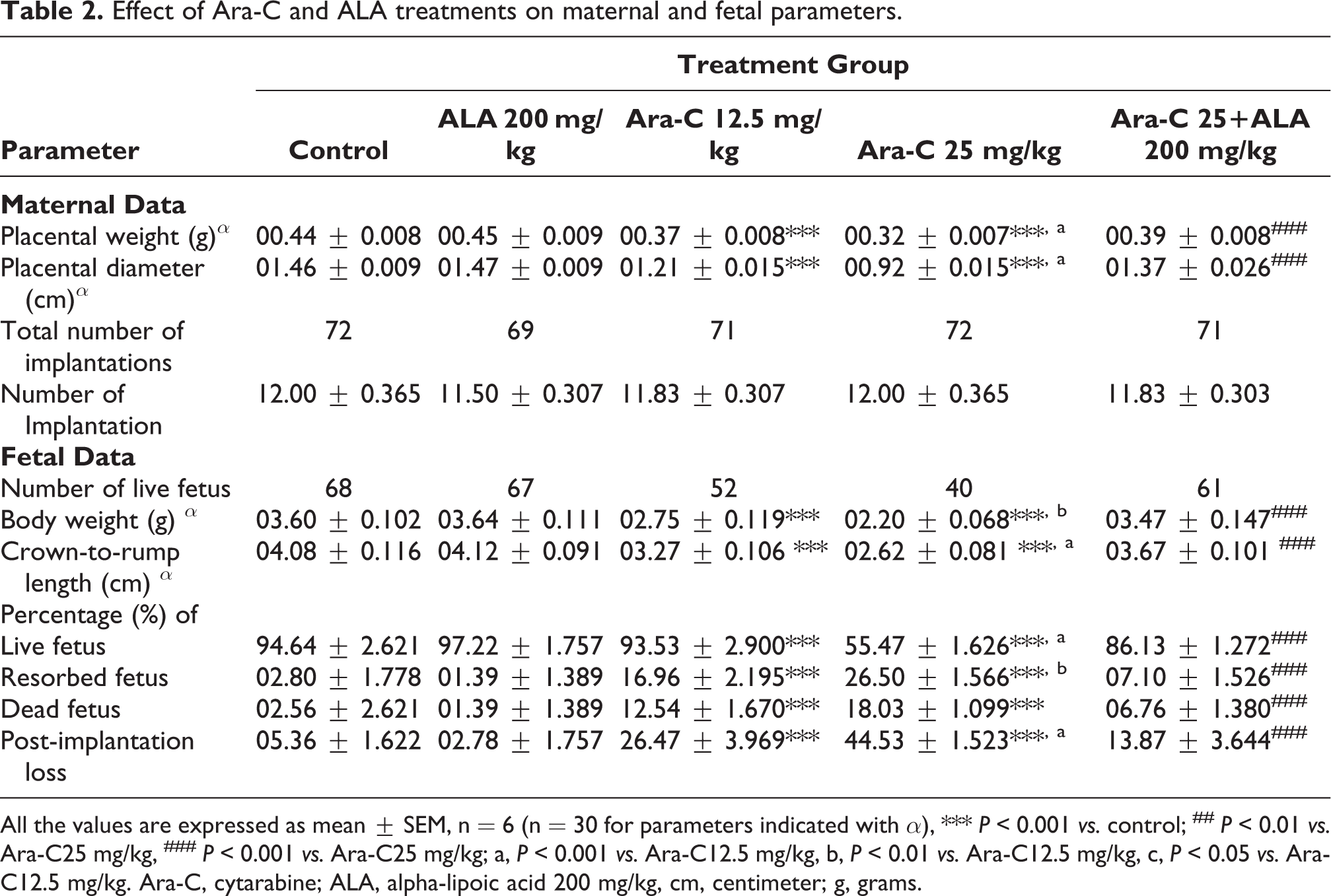
The dams of control and ALA groups found to have normal (symmetrical) distribution of implanted fetuses in uterine two horns (Figure 2) and placental weight (Figure 1f and Table 2). The dams treated with Ara-C had a significant and dose-dependent increase in number of embryonic resorbed sites in the uteri as compared to control group while the dams co-treated with ALA 200 mg/kg and Ara-C 25 mg/kg showing a significant reduction as compared to dams received Ara-C 25 mg/kg alone. There was no significant difference in number of implantation sites among groups (Table 2).
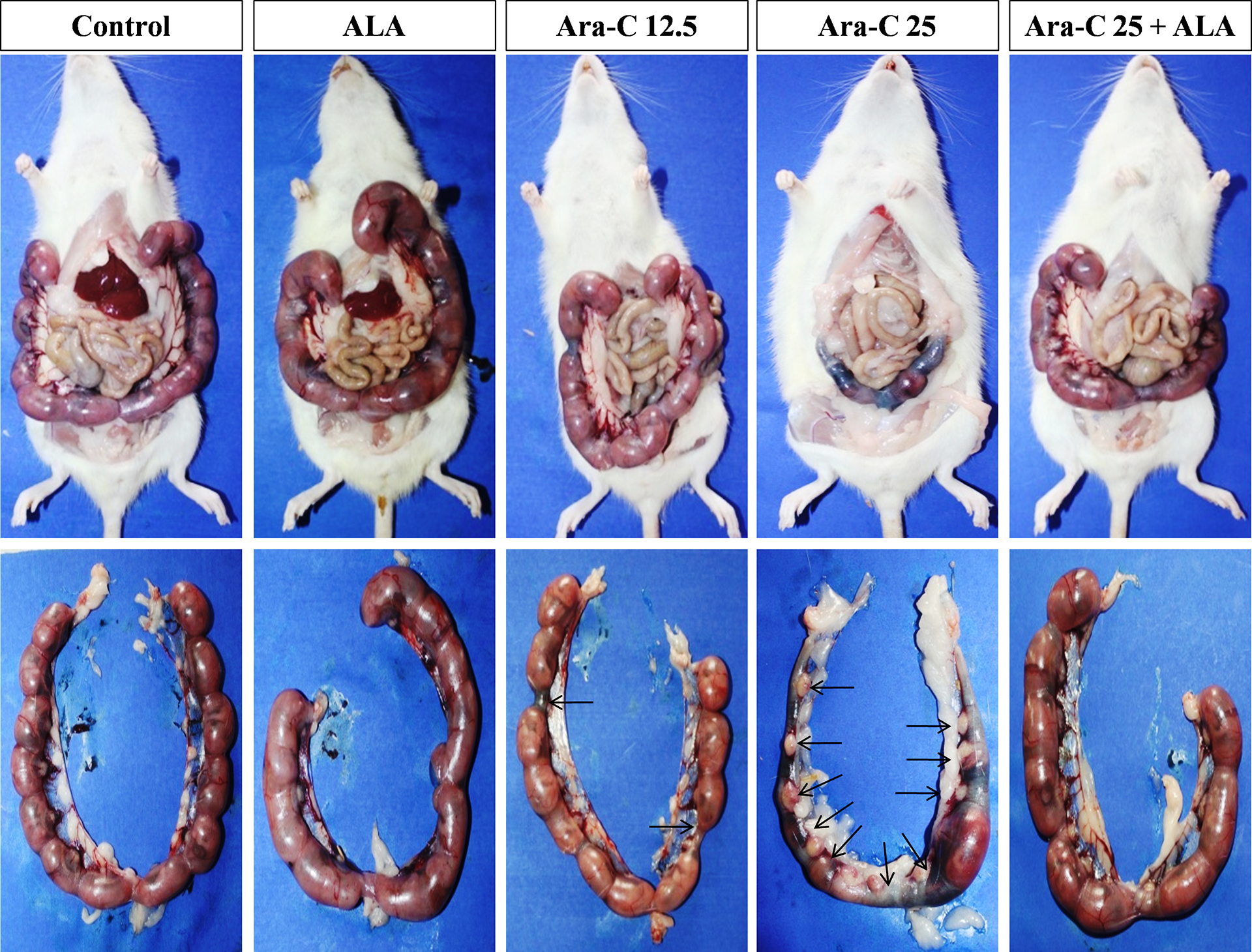
Representative photomicrographs of rat uterine horns reflecting the effect of Ara-C and ALA treatments on fetal resorptions. Black arrow pointing toward a fetal resorption site in the uterine horns.
Effect of Ara-C and ALA treatments on maternal and fetal parameters.
All the values are expressed as mean ± SEM, n = 6 (n = 30 for parameters indicated with α), *** P < 0.001 vs. control; ## P < 0.01 vs. Ara-C25 mg/kg, ### P < 0.001 vs. Ara-C25 mg/kg; a, P < 0.001 vs. Ara-C12.5 mg/kg, b, P < 0.01 vs. Ara-C12.5 mg/kg, c, P < 0.05 vs. Ara-C12.5 mg/kg. Ara-C, cytarabine; ALA, alpha-lipoic acid 200 mg/kg, cm, centimeter; g, grams.
Further, the dams treated with Ara-C have shown a dose-dependent and significant reduction in placental GSH, SOD, GPx, CAT levels and increase in MDA levels as compared to control rats. Interestingly, dams co-treated with ALA 200 mg/kg and Ara-C 25 mg/kg have shown a significant increase in placental GSH, SOD, GPx, CAT levels and decrease in MDA levels as compared to dams received Ara-C 25 mg/kg alone (Figure 4).
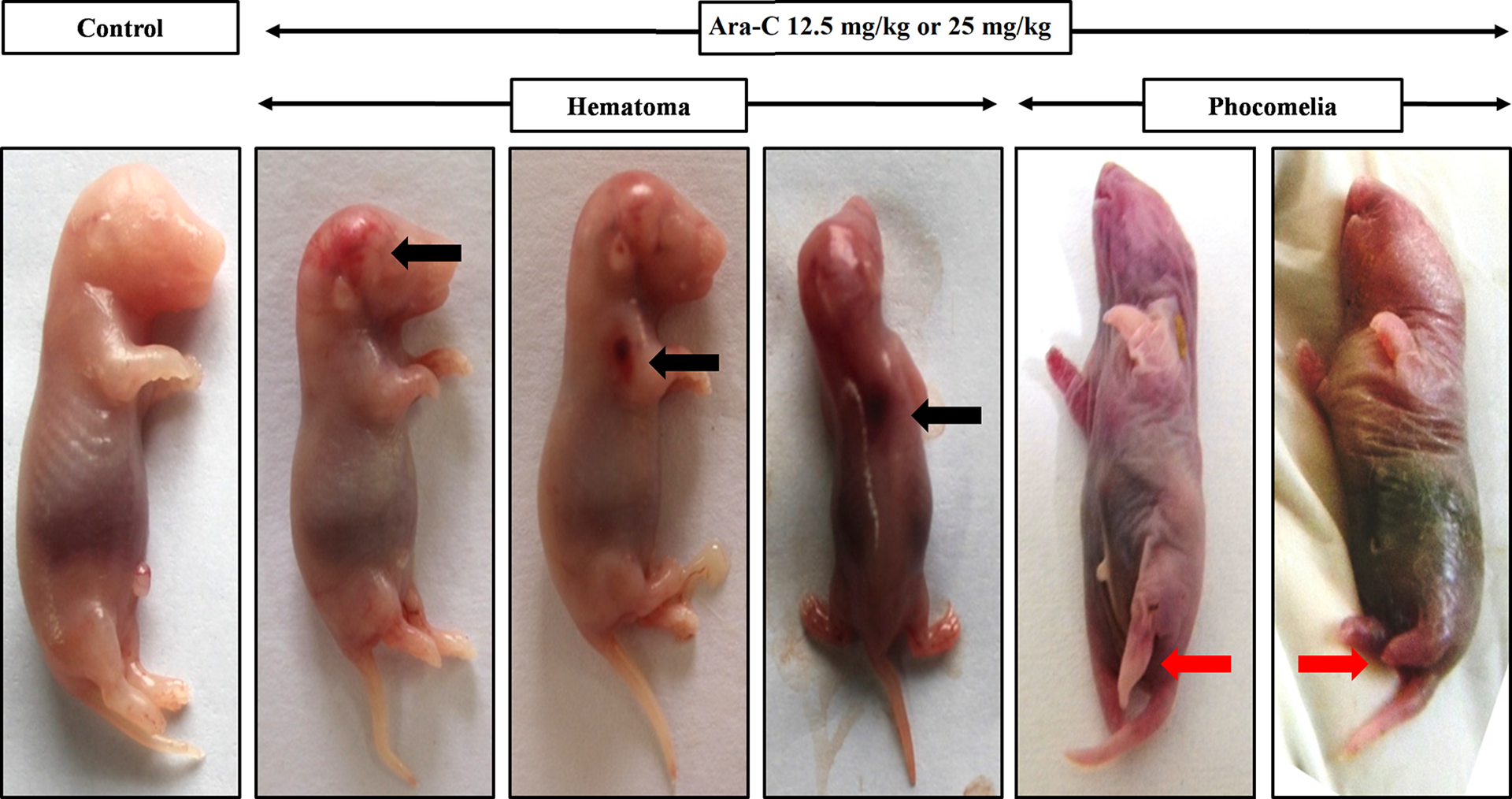
Representative photomicrographs of rat fetus of dams received Ara-C indicating hematoma and phocomelia on GD21. Black arrow indicates hematoma and red arrow indicates phocomelia.

Effect of Ara-C and ALA treatments on oxidative stress markers (a) MDA, (b) SOD, (c) GSH, (D) GSH-Px, and (e) Catalase. All the values are expressed as mean ± SEM, (n = 10), *** P < 0.001 vs. control, ** P < 0.01 vs. control; ### P < 0.001 vs. Ara-C25 mg/kg; a P < 0.001 vs. Ara-C12.5 mg/kg, b P < 0.01 vs. Ara-C12.5 mg/kg, c P < 0.05 vs. Ara-C12.5 mg/kg. Ara-C, cytarabine; ALA, alpha-lipoic acid 200 mg/kg, GD, gestation day; g, grams. MDA, malondialdehyde; SOD, superoxide dismutase; GSH, glutathione; GSH-Px, glutathione peroxidase.
Fetal toxicity
Growth retardation and developmental toxicity
The fetus of control and ALA groups found to have normal weight, and length (crown to rump). However, there was a significant and dose-dependent growth retardation represented by a reduction in fetal body weight and length in fetuses of Ara-C treated groups when compared to the fetuses of control group. Further, a significant increase in fetal body weight, and body length was observed in fetus of the dams co-treated with ALA 200 mg/kg and Ara-C 25 mg/kg as compared to fetus of dams received Ara-C 25 mg/kg alone (Table 2).
The dams treated with Ara-C shown a dose-dependent and significant increase in the incidence of resorbed fetuses, dead fetuses, and fetus with hematoma (Figure 3) and limb abnormalities (phocomelia, adactyly (Figure 3), oligodactyly, brachydactyly, and shortness of limbs) as compared to control group. However, co-treatment of dams with ALA 200 mg/kg with Ara-C 25 mg/kg resulted in a significant reduction in incidence of resorbed fetuses, dead fetuses, and fetus with hematoma and limb abnormalities as compared to dams received Ara-C 25 mg/kg alone (Table 2).
Fetal oxidative stress
In-utero exposure to Ara-C resulted in a dose-dependent and significant increase in fetal MDA levels and decrease in fetal GSH, SOD, GPx, CAT levels as compared to the control fetuses. Interestingly, fetuses of dams co-treated with ALA 200 mg/kg and Ara-C 25 mg/kg have shown a significant increase in GSH, SOD, GPx, CAT levels and decrease in MDA levels as compared to the fetuses of dams received Ara-C 25 mg/kg alone (Figure 4).
Skeletal anomalies
The fetuses of control group and ALA group have shown normal skeletal system development as evidenced from the ARAB double staining (Figure 5). The shape, size, and extent of ossification of all the bones found to be up to the level expected to be at GD21. The fetuses of the dams treated with Ara-C have shown a clear impairment in the development of the skeletal system as compared to fetuses of control group (Figure 5a). Ossification in bones of skull (Figure 5b–c), vertebrae (Figure 7a), sternum (Figure 6a), ribcage (Figure 5a), scapula (Figure 6b), pelvic girdle (Figure 7b), fore limbs (Figure 6b) and hind limbs (Figure 6c) of fetuses of Ara-C treated dams was incomplete or missing as evidenced from reduced intensity or absence of Alizarin staining. The intensity of alizarin red staining was less in the skull bones of the fetuses of dams treated with Ara-C clearly indicating the impaired ossification as compared to control fetus. Further, the fetuses of dams co-treated with ALA 200 mg/kg and Ara-C 25 mg/kg have shown increase in the intensity of Alizarin red staining indicating improved ossification in skull bones as compared to fetuses of dams received Ara-C 25 mg/kg alone. The fetuses that belongs to dams treated with Ara-C have shown less ossification in clavicle, humerus, radius, ulna, femur, fibula and tibia and reduced number of ossification centers in sternum, (Figure 6a), and centrum and arches of sacral and caudal vertebrae pelvic bones (Figure 7b), and metacarpals and metatarsals (Figure 7c). Interestingly, the fetuses of dams co-treated with ALA 200 mg/kg and Ara-C 25 mg/kg have shown a significant increase in the number of ossification centers and percent of ossification as compared to fetuses of dams received Ara-C 25 mg/kg alone (Table 3). In the ossification process, the bilateral ossification centers in centrum of vertebrae and sternabrae fuse to form a single ossification center and hence they were considered as a single ossification center. Therefore, the bilateral ossification centers or a dumbbell-shaped structure in vertebrae or sternum of the fetuses of dams treated with Ara-C (Figure 6a and Figure 7a) clearly indicates the impaired ossification.

Representative photomicrographs of the skeleton (a), lateral (b) and ventral view (c) of the skull of the rat fetus at GD21 stained with Alizarin S red and Alcian blue. The intensity of alizarin red staining was less in the skull bones of the fetuses of dams treated with Ara-C clearly indicating the impaired ossification as compared to control fetus. Further, the fetuses of dams received ALA200 mg/kg and Ara-C25 mg/kg have shown increase in the intensity of alizarin red staining indicating improved ossification in skull bones as compared to fetuses of dams received Ara-C25 mg/kg alone. At, atlas; Bs, basisphenoid; Bo, basioccipital; C-P canal, craniopharyngeal canal; Eo, exoccipital; F, frontal; Hy, hyoid; Ip, interparietal; M, maxilla; Mn, mandible; N, nasal; P, parietal; Pa, palatine; Pa foramen, palatine foramen; pre-M, premaxilla; Pt, pterigoid; S, squamosal; So, supraoccipital; T, tympanic; Ty B, tympanic bulla; Zy, zygomatic.

Representative photomicrographs of (a) sternum, (b) forelimb and (c) hind limb of the rat fetus at GD21 stained with Alizarin S red and Alcian blue. The ossification in the sternum, forelimb and hind limb was less in the fetuses of dams treated with Ara-C as compared to control fetus. The fetuses of dams received ALA200 mg/kg and Ara-C25 mg/kg have shown improved ossification in sternum, forelimb and hind limb as compared to fetuses of dams received Ara-C25 mg/kg alone. C, carpals; F, femur; Fi, fibula, H, humerus; Mn, manubrium; Mc, metacarpals; Mt, metatarsals; Ph, phalanges; R, Ribs; Sc, scapula; Ta, tarsals; T, tibia; St, sternabrae; XP, xiphoid process. The bilateral ossification centers in sternum are considered as a single ossification center.

Representative photomicrographs of (a) thoracic vertebrae, (b) pelvic girdle, sacral and caudal vertebrae, (c) metacarpals and (d) metatarsals of the of the rat fetus at GD21 stained with Alizarin S red and Alcian blue showing number of ossification centers. The number of ossification centers in pelvic girdle, sacral and caudal vertebrae, metacarpals and metatarsals of the fetuses of dams treated with Ara-C were less as compared to control fetus. Further, the fetuses of dams received ALA200 mg/kg and Ara-C25 mg/kg have shown increase in number of ossification centers in above mentioned bones as compared to fetuses of dams received Ara-C25 mg/kg alone. Ar, arch of the vertebrae; C, centrum of the vertebrae; Cv, caudal vertebrae; Il, ilium; Is, ischium; P, pubis; R, rib; Sv, sacral vertebrae. The bilateral ossification centers or dumbbell-shaped structures in centrum of vertebrae are considered as a single ossification center.
Effect of Ara-C and ALA treatments on number of ossification centers and percentage ossification in the fetuses on GD21.

All the values are expressed as mean ± SEM, n = 30, *** P < 0.001 vs. control; ### P < 0.001 vs. Ara-C25 mg/kg; a, P < 0.001 vs. Ara-C12.5 mg/kg; b, P < 0.01 vs. Ara-C12.5 mg/kg; c, P < 0.05 vs. Ara-C12.5 mg/kg. Ara-C, cytarabine; ALA, alpha-lipoic acid 200 mg/kg; GD, gestation day; g, grams.
The manubrium and xiphoid process of the sternum and portions of some of the ribs that are articulating with sternum in fetuses of dams treated normal saline and ALA alone have shown some extent of ossification indicated by Alizarin staining. The ossification was absent in the manubrium and xiphoid process of the sternum and portions of some of the ribs that are articulating with sternum in fetuses of dams treated Ara-C while same was present in fetus of dams co-administrated with ALA and Ara-C.
Bone mineral analysis
The bone calcium and phosphorus content in the fetuses of dams exposed to Ara-C was significantly low as compared to the fetus of control dams whereas the fetus of dams co-treated with ALA 200 mg/kg and Ara-C 25 mg/kg have shown a significant increase in bone calcium and phosphorus content as compared to fetuses of dams received Ara-C 25 mg/kg alone (Table 4).
Effect of Ara-C and ALA treatments on the fetal limb abnormalities, and calcium and phosphorous content in the bone at GD21.
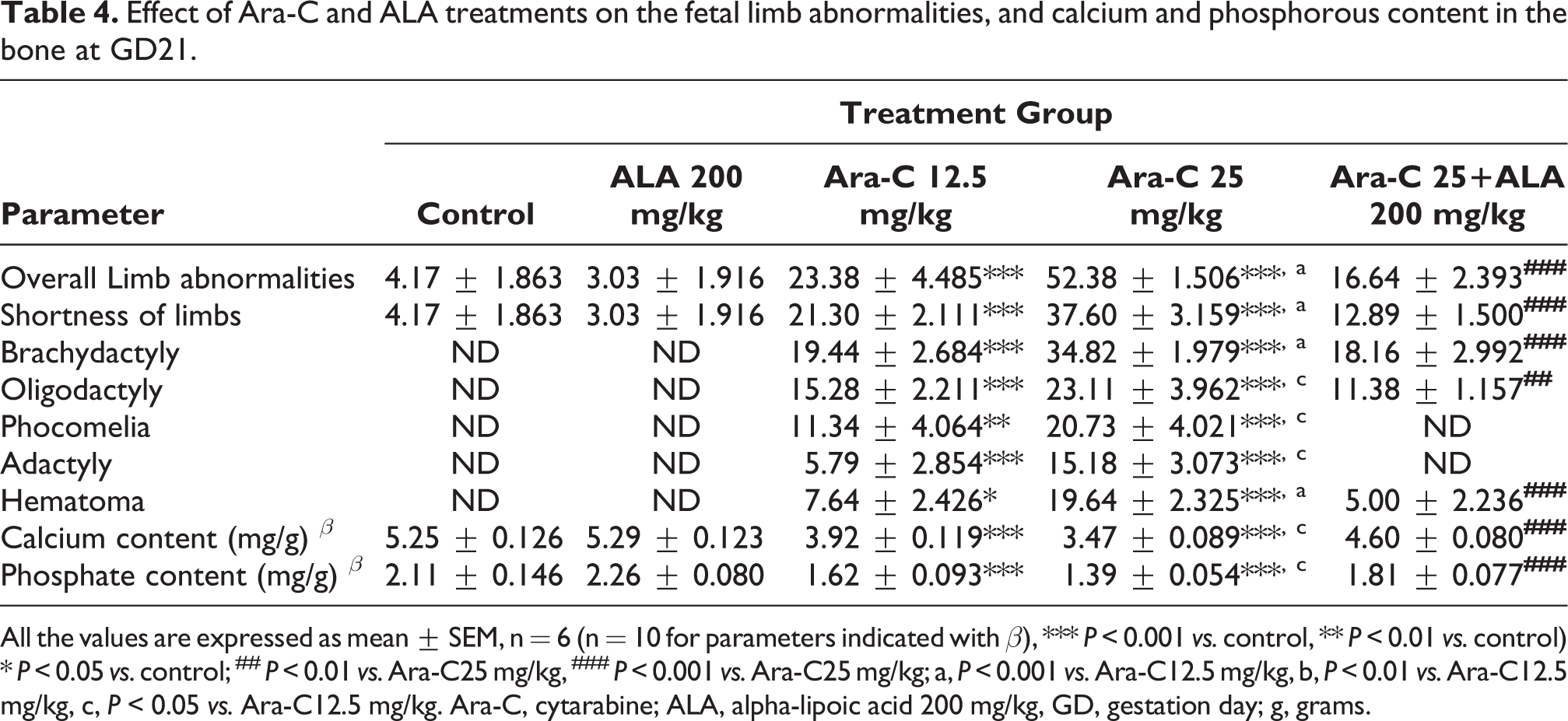
All the values are expressed as mean ± SEM, n = 6 (n = 10 for parameters indicated with β), *** P < 0.001 vs. control, ** P < 0.01 vs. control) * P < 0.05 vs. control; ## P < 0.01 vs. Ara-C25 mg/kg, ### P < 0.001 vs. Ara-C25 mg/kg; a, P < 0.001 vs. Ara-C12.5 mg/kg, b, P < 0.01 vs. Ara-C12.5 mg/kg, c, P < 0.05 vs. Ara-C12.5 mg/kg. Ara-C, cytarabine; ALA, alpha-lipoic acid 200 mg/kg, GD, gestation day; g, grams.
Discussion
The results of present study unveiled the potential of ALA supplementation in alleviating the Ara-C induced developmental toxicity in rats. To the best of our knowledge, this is the first report to portray the role of natural antioxidants in diminishing the teratogenic effects of Ara-C in rodents. Our study demonstrated that ALA supplementation rescues the toxic insult imposed on pregnant mother rat and fetus there by reduces various developmental anomalies including impairment in skeletal system development associated with in-utero exposure to Ara-C.
Any dysfunction and damage to the placenta adversely affect the maintenance of pregnancy and fetal growth and cause developmental anomalies including intrauterine growth restriction which may leads stillbirth, preterm birth, miscarriage, perinatal mortality, and various developmental anomalies in fetus. 75 –78 Ara-C by virtue of its mechanism causes DNA damage and inhibits cell division in rapidly proliferating cells at S-phase. Further, it produces OS which also contributes to its cytotoxic potential. 16,17,79,80 In study of Yamauchi et al. Ara-C inhibited cell proliferation and increased apoptosis in labyrinth zone of the placenta through p53 involvement and caused hypoplacia in placenta that resulted impairment in placental development. 17 In line to several previous reports, our study also showed that the prenatal Ara-C exposure resulted in both maternal and fetal toxicity in rats in a dose-dependent manner. The significant reduction in food intake, body weight gain, and placental weight, and increase in OS markers in placenta in dams treated with Ara-C clearly indicating the maternal toxicity. Elevated OS leads to DNA damage and oxidation of lipids, proteins, and polysaccharides which cause disruption of the pathways involved in maintenance of pregnancy and embryogenesis which ultimately contributes to congenital anomalies. Further, OS is considered as one of the main mechanisms of teratogenic action which affects ossification. 81,82
In our study, Ara-C induced oxidative damage was clearly indicated by a dose-dependent elevation in MDA levels, and reduction in GSH, GSH-Px, CAT and SOD levels in placenta of the dams which had in-utero Ara-C exposure. Ara-C induced OS and its cyctostatic action in placenta could have contributed to maternal toxicity reflected by reduction in food intake, body weight gain, and placental weight. Further, it can be attributed that these changes may at least in part contribute to the development of fetotoxicity. ALA administration along with Ara-C reduced the maternal toxicity caused by Ara-C indicating its potential in offering protection against Ara-C induced developmental toxicity. ALA protective role in ameliorating the pregnancy-related disorders or chemical-induced developmental toxicity has been demonstrated in several studies. 46,83,84 In few clinical studies, ALA vaginal administration demonstrated its capability in healing subchorionic hematomas in subjects with threatened miscarriage 85,86 and reducing premature or preterm delivery. 87 Further, in a recent experimental study, ALA proved to contrast preterm delivery induced by mifepristone and PGE2 in rats. 44
Further, fetal tissue is more vulnerable to oxidative damage due to rapid cell proliferation. 81 Ara-C is known to cause fetotoxicity which includes fetal resorption, still birth, weight reduction, and various external and internal developmental anomalies. The developmental anomalies reported with Ara-C use includes cleft palate, cleft lip, micrognathia, kinky tail, hydrocephalus, encephalocele, club foot, phocomelia, hemimelia, and defects in axial and appendicular skeletal system. The most common congenital anomalies with use of Ara-C are digit anomalies (Table 1). In our study, we found a dose-dependent increase in the incidence of fetal resorption, death, weight reduction, crown-to-rump length, hematoma, phocomelia, oligodactyly, and incomplete ossification in the bones of fetuses obtained from dams treated with Ara-C at dose levels 12.5 mg/kg and 25 mg/kg. The reduction in fetal weight and crown-to-rump and incomplete ossification of the bones and reduced number of ossification centers in the fetal skeleton can be taken as the indicators of fetal growth retardation. 88,89
The bilateral ossification centers or a dumbbell-shaped structure in vertebrae or sternum of the fetuses of dams treated with Ara-C (Figure 6a and Figure 7a) clearly indicates the impaired ossification. 90
In agreement with previous reports, Ara-C prenatal treatment in our study also demonstrated fetal growth retardation as reveled by above said indicators. Further, the elevated OS is believed to implicate in pathological process of bone related disorders including bone loss through perturbing the orchestrated action of osteoclasts, osteoblasts and osteocytes. OS fuels the apoptosis of osteoblasts and osteocytes, and promotes osteoclastogenesis and curtails the mineralization and osteogenesis. 91 Ritter and colleagues reported that Ara-C administration causes extensive cell death in developing bone and cause impairment in bone growth. 23 Moreover, several experimental studies demonstrated the prenatal Ara-C exposure-induced developmental anomalies in fetal bones including incomplete ossification, impaired mineralization and growth (Table 1). In our study, we observed a significant elevation OS marker in fetuses that are challenged with maternal Ara-C exposure. Further, the incomplete ossification in both axial and appendicular system and an apparent reduction in number of ossification centers in sternum, carpals, tarsals, vertebrae, pelvic bones clearly indicating impaired skeletal system development with in-utero exposure to Ara-C. The reduced calcium and phosphorus content in bones on GD21 is clearly indicating the impaired mineralization in fetuses of Ara-C treated dams. These results are in agreement with previous reports. 20,34,35 We postulate that the Ara-C induced impairment of cellular proliferation, induction of apoptosis and intrusion of mineralization may be accountable for defects in skeletal development in fetus.
The antioxidant supplementation in pregnancy is gaining attention in recent years to improve the pregnancy-related disorders or drug-/chemical-induced pregnancy disorders. 59 ALA, a naturally occurring antioxidant is commonly found in mitochondria of all the cells. ALA acts as a co-factor several dehydrogenase enzymes present in mitochondria play and essential role in mitochondrial metabolism. It is required by cells for their growth, mitochondrial activity, and harmonization of energy metabolism. 92 ALA along with its metabolite DHLA is regarded as double antioxidant and this redox couple has capability to scavenge wide variety of a ROS, chelate transition metals, and regenerate other endogenous antioxidants like vitamins C, vitamins E and GSH. Further, due to its capability to curb OS in all the parts of body i.e. in both hydrophilic and hydrophobic environment it is regarded as universal antioxidant. 7,93 Copious literature is emphasizing that ALA with its pleotropic effects offers therapeutic benefits in various pathological conditions including amelioration of drug-induced toxicities. 43,93 In our study, we observed that ALA administration at 200 mg/kg dose level reduced the Ara-C-induced developmental toxicity in rats. ALA reduced both maternal and fetal OS. As mentioned previously, OS is implicated in the pathogenesis of several pregnancy-related disorders and drug- or chemical-induced developmental anomalies. The use of ALA in pregnancy found to be very promising in improving pregnancy outcomes and have no adverse effects on pregnancy. 40,44,45 Further, ALA protective role in ameliorating the pregnancy-related disorders like subchorionic hematomas, miscarriage, and preterm delivery 44,85 –87 or chemical- or disease-induced developmental toxicity has been demonstrated in several studies. 47,84,94 Co-administration of ALA showed a significant reduction in OS markers, the incidence of fetal resorption, death, hematoma and increase in weight reduction, crown-to-rump length as compared to group received Ara-C only and is clearly indicating its protective effect. Further, supplementation with ALA resulted in improved % ossification in both axial and appendicular system, and mineralization and increased the number of ossification centers in sternum, carpals, tarsals, vertebrae, pelvic bones of fetuses as compared to fetuses of dams received Ara-C alone. ALA reported to prevent bone loss induced by oxidative stress, 53 diabetes, 46,94 inflammation, 50 radiation, 54 β-adrenergic signaling, 95 periapical bone loss, 96 periodontitis, 97 fracture, 98 starvation-induced bone remodeling, 99 ovariectomy or menopausal, 49,57,100,101 high-fat diet, 58 glucocorticoids, 56,102 collagen-induced arthritis, 55,103 H2O2/Tumor Necrosis Factor-Alpha(TNF-α) 48 receptor activator of nuclear factor kappa beta (RANKL)/TNF-α, 52 cadmium, 104 nano-zinc oxide, 105 and lead 51 and improve bone formation. Further, ALA reduces developmental anomalies in fetal skeletal system caused by diabetes. 46,94 Therefore, the results of our study are corroborating the existing literature and asserting that ALA supplementation could reduce the bone loss and improve bone formation there by minimize developmental anomalies in skeletal system.
The limitation of this study was the lack of molecular level mechanisms involved in protection offered by ALA against Ara-C induced developmental toxicity. However, the studies that explored the Ara-C induced developmental toxicity have not investigated the pathomechanisms implicated at molecular level. Based on the results of our study and evidence from existing literature, we postulate that the protection offered by ALA against Ara-C induced developmental toxicity was mediated, at least in part, through inhibition of the oxidative damage, apoptosis and modulating various messengers and regulators that are involved pathological mechanisms implicated in Ara-C induced developmental toxicity.
Conclusion
Prenatal exposure to Ara-C causes a dose-dependent developmental toxicity in rats through oxidative damage and inhibition of cell proliferation. Co-administration of ALA at a dose of 200 mg/kg ameliorates the Ara-C induced developmental toxicity. The results of this study uncovered the potential of ALA in alleviating chemotherapy-induced congenital anomalies. However, future experimental and clinical studies are required to explore in depth molecular mechanisms that are responsible for protective effect of ALA against disease-, drug- or chemical-induced developmental toxicities.
Footnotes
Acknowledgments
We would like to acknowledge Bhaskar Medical College, Hyderabad, and Bhaskar Pharmacy College, Hyderabad for providing facilities and also Vivo Bio Tech Ltd, Pregnapur, Telangana for the support in technological aspects.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
