Abstract
Nephrotoxicity is a dose-limiting side effect of long-term use of tenofovir, a reverse transcriptase inhibitor that is used for the treatment of HIV infection and chronic hepatitis B infection. Identifying an agent that prevents tenofovir disoproxil fumarate (TDF)-induced renal injury can lead to its better tolerance, and a more effective treatment can be achieved. The present study is aimed at investigating whether melatonin, a potent antioxidant and anti-inflammatory agent, protects against TDF nephrotoxicity in rats and to determine its cellular targets. Rats were divided into groups and treated as follows. Group I (control): Rats in this group (n = 6) received sterile water only by gavage for 35 days. Group II: Rats (n = 6) in this group received 600 mg/kg body weight TDF in sterile water by gavage for 35 days. Group III: Rats (n = 6) in this group received once daily 20 mg/kg bodyweight melatonin i.p. 2 h before the administration of 600 mg/kg body weight TDF in sterile water by gavage for 35 days. Group IV: Rats were pretreated daily with 20 mg/kg body weight melatonin i.p. 2 h before the administration of sterile water by gavage. All the rats were sacrificed on the 36th day, after overnight fast. Melatonin pretreatment protected the rats against TDF nephrotoxicity both histologically and biochemically. Biochemically, melatonin pretreatment attenuated TDF-induced, oxidative stress, nitrosative stress, mitochondrial pathway of apoptosis, PARP overactivation and preserved proximal tubular function (p < 0.01). This suggests that melatonin may be useful in ameliorating TDF nephrotoxicity.
Introduction
Tenofovir, derived from its oral prodrug Tenofovir disoproxil fumarate (TDF) is currently the only nucleotide analog reverse transcriptase inhibitor that is approved by the FDA, USA, for the treatment of HIV infection. 1 It is also used for chronic hepatitis B treatment. Although it has strong clinical efficacy and generally well-tolerated, TDF can lead to severe renal toxic effects, especially in patients with risk factors for kidney disease. 2 Severe cases of renal tubular toxicity have been reported in patients with TDF exposure and it is reported that approximately 15% of patients treated with tenofovir develop renal tubular dysfunction. 3 –5 The main site of TDF toxicity is the proximal tubule, and patients can develop Fanconi syndrome or acute kidney injury. 6 Several human and animal studies haves shown that TDF damages specifically renal proximal tubular mitochondria. 7,8 Continuous tubular damage may promote tubular cell death resulting in interstitial fibrosis, decreased glomerular filtration and ultimately renal failure. 9 Prevention of TDF nephrotoxicity is necessary to avoid irreversible renal damage. Identifying an agent that prevents/minimizes TDF-induced renal injury can lead to better tolerance to TDF, and hence a more effective treatment can be achieved in HIV-infected patients.
We have been actively involved in investigating the mechanism of TDF nephrotoxicity using rat model. We have demonstrated that several mechanism are involved in TDF nephrotoxicity. These include increased oxidative stress, 10 depletion of antioxidant enzymes, 11 iNOS induction and nitrosative stress, 11 mitochondrial damage, 12 apoptosis, 13 upregulation of NFkB inflammatory pathway, and neutrophil infiltration. 14
The present study was undertaken to 1.investigate whether pretreatment with melatonin can prevent/ attenuate TDF nephrotoxicity in rats and 2. determine its mechanism of action.
Melatonin (MT) was chosen for our study due to the following reasons. Melatonin is a lipophilic pineal hormone with multiple pleiotropic effects. It can cross cell membranes easily as it is lipophilic. 15 It protects cells from free radical induced injury by several mechanisms that include scavenging of free radicals including hydrogen peroxide, peroxynitrite anion, and superoxide anion, 16,17 increasing the levels of antioxidant enzymes, 18,19 preventing mitochondrial damage, 20,21 inhibiting apoptotic cell death 22,23 and myeloperoxidase (MPO) activity, a marker of neutrophil infiltration. 24 Melatonin has been shown to be protective in various nephrotoxic models such as cisplatin, gentamicin and mercury. 25 –27
Materials and methods
Materials
Tenofovir disoproxil fumarate (TDF), melatonin, (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid)-HEPES, hydrogen peroxide, sucrose, bovine serum albumin (BSA), sodium dodecyl sulfate(SDS), mannitol and ethylene glycol-bis(2-aminoethyl ether)-N, N, N′, N′-tetraacetic acid (EGTA) were purchased from Sigma (St Louis, Missouri, USA). The primary mouse monoclonal iNOS, nitrotyrosine, nitrocysteine, caspase 9, caspase 3, cyt c, HSP70, HO-1, PARP-1 antibodies and A/G Plus-agarose protein were obtained from Santa Cruz (Mumbai Maharashtra, India). Secondary antibodies (antirabbit and anti-mouse immunoglobulin G conjugated with horseradish peroxidase (HRP)) were obtained from Pierce Biotechnology (Rockford, California, USA). Protease inhibitor cocktail was obtained from Sigma-Aldrich. Polyvinylidene difluoride (PVDF) membrane (pore size 0.45 mm) was obtained from Millipore (Bengaluru, Karnataka, India). The Super Sensitive Polymer/HRP/DAB kit was obtained from BioGenex (Chennai, Tamil Nadu, India). ECL Dualvue Western blotting markers were purchased from Amersham (UK). West Dura chemiluminescent substrate for developing Western blots was purchased from Thermo Scientific (Waltham, California, USA. Transferase biotin-dUTP nick end-labeling (TUNEL) assay cell death kit was obtained from Roche Diagnostics (Germany). All other chemicals used were of analytical grade
Animals and treatments
Adult male Wistar rats (175–225 gm) were used for the study. They were housed in standard rat cages. The animals were acclimatized to laboratory conditions (23°C, 12h/12 h light/dark, 50% humidity, ad libitum access to food and water) for 2 weeks prior to experimentation. The experiments done were approved by the institutional animal ethics committee (IAEC), Christian Medical College (FWA 00002210), Vellore, Tamil Nadu and were done in accordance with the guidelines of the Committee for the Purpose of Control and Supervision of Experimentation on Animals (CPCSEA), Government of India.
Dose of TDF and melatonin
Rat model of TDF nephrotoxicity was standardized in our laboratory described by Biesecker et al, with modifications. 28 TDF at a dose of 600 mg/kg body weight per day orally for 5 weeks resulted in severe damage to the mitochondria of proximal tubules and Fanconi syndrome as seen in humans. 8 For the present study, the dose of 20 mg/kg body weight per day of melatonin was chosen based on a recent study, which showed that intraperitoneal (i.p.) administration of 20 mg/kg body weight per day of melatonin protects rats against diquat-induced renal damage. 29
Experimental design
Rats were randomly assigned to four groups and were treated as follows. Group I (control): Rats in this group (n = 6) received sterile water only by gavage for 35 days. Group II: Rats (n = 6) in this group received 600 mg/kg body weight TDF in sterile water by gavage for 35 days. Group III: Rats (n = 6) in this group received once daily 20 mg/kg bodyweight melatonin i.p. 2 h before the administration of 600 mg/kg body weight TDF in sterile water by gavage for 35 days. Group IV: Rats were pretreated daily with 20 mg/kg body weight melatonin i.p. 2 h before the administration of sterile water by gavage.
All the animals were killed the 36th day, after overnight fast.
Mortality checks, clinical observations, and body weights
All animals were observed daily for any clinical signs of toxicity, morbidity, and mortality throughout the experimental period. The body weight of each rat was also monitored on a weekly basis until the end of treatment.
Collection of blood, urine and kidneys
Twenty-four hours before sacrifice, the rats were placed individually in metabolic cages, and urine was collected for biochemical analysis. On the 36th day, after overnight fast, blood samples were collected from the rats under halothane anesthesia, by cardiac puncture into tubes and allowed to clot at room temperature. Thereafter, serum was separated by centrifugation at 1200 g for 15 min at 4°C for clinical chemistry. Then the animals were sacrificed by over dose of halothane anesthesia. The abdomen was opened by midline incision and kidney was dissected out carefully and cleaned off the extraneous tissue and weighed. Half of left kidney was cut in cross-section and fixed in 10% buffered formalin for light microscopy, and the remaining half was snap-frozen using liquid nitrogen at −70°C until use for biochemical studies.
Serum clinical chemistry
Serum was separated out and used for the estimation of phosphate, potassium, bicarbonate, and uric acid by standard spectrophotometric methods.
Urinalysis
Urine samples were centrifuged to remove suspended material, and the supernatants were used for the estimation of bicarbonate, phosphate, potassium, and uric acid by standard spectrophotometric methods.
Morphological examination of the kidney (light microscopy)
Kidney tissue was fixed in 10% buffered formaldehyde and embedded in paraffin. The slices were embedded in paraffin and then sectioned. Four-micrometer thick paraffin sections were stained with hematoxylin and eosin for light microscopic examination using the conventional protocol. A minimum of eight fields for each kidney section were examined and assigned for severity of changes by an observer blinded to the treatments of the animals. Tubular injury was graded on a semi quantitative scale as described by Ramesh and Reeves. 30 Tubular injury was defined as tubular epithelial necrosis, cast formation, tubular dilatation, and the loss of the brush border. Tubular injury was scored by grading the percentage of affected tubules under 10 randomly selected, non-overlapping fields (magnification, 200 X) as follows: 0, 0%; 1, <10%; 2, 11–25%; 3, 26–45%; 4, 46–75%; and 5, 76–100%. To score injured tubules, whole tubule numbers per field were considered as the standard under a magnification of 200 X. The grading percentage was calculated in each field as follows
TUNEL assay for the detection of apoptosis
Kidney tissues was fixed in 10% neutral buffered formalin, embedded in paraffin wax. The tissue sections (5 μm) were mounted on a slide, dewaxed and rehydrated in graded alcohol solutions. DNA fragmentation for apoptosis was detected by the modified TUNEL method as described by Tunçdemir et al. 31 and following the manufacturer’s instructions. TUNEL staining was performed using an apoptosis detection kit (Roche Diagnostics GmbH, Mannheim, Germany). Brown nuclear labeling were considered as TUNEL-positive. Staining was evaluated using Olympus light microscope under 400X magnification.
Immunohistochemistry
Immunohistochemistry was done as described by Cuzzocrea et al. 32 4-mm thick sections were obtained from paraffin-embedded kidney fixed in 10% formalin. The sections were permeabilized with 0.1% Triton X-100 in Tris-buffered saline for 15 min, after deparaffinization. 3% hydrogen peroxide was used to quench endogenous peroxidases. The universal protein blocking agent was applied over the sections after the buffer wash. The respective primary antibody was applied over the sections and incubated overnight. The bound primary antibody was detected by the addition of secondary antibody conjugated with HRP polymer and 3, 3-diaminobenzidine substrate. Later, the slides were counterstained with Harris hematoxylin and mounted. Primary antibody dilution for IHC, iNOS (1:250), PARP (1:100), Nitrotyrosine (1:250), HO-1 (1:200), HSP 70 (1:100), Caspase 3 (1: 250), Caspase 9 (1:200), Cytochrome C (1:100), and Nitrocysteine (1:100).
IHC scoring method
The strength of staining was based on color and texture features. A strength score used was in the range of 0–3. 33 The IHC staining was graded as 0 negative staining, 1 for weak staining, 2 for intermediate and 3 for strong.
Western blot procedure
Western blot was carried out using standard procedure. Proteins were first separated by electrophoresis and then electroblotted onto PVDF membranes at 90 V for 3 h. A commercially available protein molecular weight marker was also electrophoresed on the polyacrylamide gel and electroblotted onto the PVDF membrane along with the samples. The primary antibodies were used at the following dilutions: anti iNOS (1:1000), anti-nitrotyrosine (1:1000) and anti-caspase 3 (1:1000). Incubation with antibodies against the primary antibody and beta-actin was followed by incubation with peroxidase-labeled antibodies (1:1000).A commercially available S protein-HRP 133 (1 in 2500) conjugate (required for visualization of the molecular weight markers) was also incubated along with secondary antibody. The secondary antibody and the S protein were tagged with a peroxidase that acted on a chemiluminescent substrate. The bands obtained were visualized using ECL West Dura substrate, and their intensity was quantitated using a chemiluminescent imaging system (FluorChem TM SP, Alpha Innotech, San Jose, California, USA).
Real-time polymerase chain reaction (PCR) of iNOS, and caspase 3 genes
Total RNA was isolated from intestinal tissue using TRIzol reagent (Invitrogen, United States) according to the manufacturer’s instructions. Equal amounts of RNA were used to synthesize first strand cDNA (Promega, United States), and quantitative real-time polymerase chain reaction (RT-PCR) was performed on an ABI PRISM 7300 PCR System (Applied Biosystems, United States) using Syber Green I GoTaq® qPCR Master Mix (Promega, United States). PCR was performed as: one cycle at 95°C for 5 min, followed by 40 cycles of 95°C for 15 s, 58°C for 20 s and 72°C for 30 s. Data for each gene of interest was normalized relative to the internal reference gene β actin. 34 This was done by subtracting the Ct values of beta-actin in each sample from that of the gene of interest. The value obtained was referred to as the ΔCt value. The relative fold-change in the gene of interest was determined according to the comparative Ct method using the following formula: Relative fold-change = 2^- (ΔCt) .The PCR primer sequences are shown below.
Rat Beta-Actin Forward primer: 5′-GGACTTCGAGCAGGAGA-TGG-3′ Reverse primer: 5′-GCACCGTGTTGGCGTAGAGG-3′
Caspase-3 Forward primer: 5′-AATTCAAGGGACGGGTCATG-3′ Reverse primer: 5´-GCTTGTGCGCGTACAGTTTC-3′
Rat iNOS Forward primer: 5′ GACCAAACTGTGTGCCTGGA 3′ Reverse primer: 5′ TACTCTGAGGGCTGACACAAGG 3′
Caspase 3 activity determination
Preparation of kidney homogenate
The kidney tissue obtained was washed in ice-cold saline, decapsulated and minced into small pieces using a pair of sharp scissors and immediately homogenized (10% w/v)in the homogenization buffer containing 0.05 MHEPES and 125 mM potassium chloride pH 7.4 using a Potter–Elvehjem homogenizer at 5000 × g for 3 min(10–15 strokes). The homogenates were centrifuged at 7500 × g for 10 min. The supernatant was used for the measurement of caspase 3 activity. The activity of caspase3 was determined using a commercial kit (Abcam, Cambridge, UK) according to the manufacturer’s instruction. This assay is based on the detection of cleavage of substrate DEVED-AFC (AFC: 7-amino4-trifluoromethyl coumarin). DEVD-AFC emits blue light (λ max = 400 nm); upon cleavage of the substrate by CPP32 or related caspases, free AFC emits a yellow–green fluorescence (λ max = 505 nm), which was quantified using a fluorescence microplate reader.
Statistical analysis
Data were analyzed using the Statistical Package for the Social Sciences (SPSS) software package, version 16. All parameters were studied in a minimum of six animals for statistical validity. The statistical significance of differences among control and experimental rats were evaluated by one-way ANOVA or two-way ANOVA followed by a Bonferroni’s post hoc test. P-values <0.05 were considered to be statistically significant.
Results
Melatonin pretreatment ameliorated TDF-induced renal damage
The H &E staining results of kidneys of control rats and TDF-treated rats is shown in Figure 1. Sections of the kidneys from the control group showed normal histological structure of the glomeruli and renal tubules in the cortex and normal tubules in the medulla. In the TDF-treated rat kidney cortex, the proximal convoluted tubules were distorted and their lining epithelium was destroyed. Interstitial edema and cytoplasmic vacuolations were also observed. Isolated proximal tubules showed some detachment of cells and congestion. Some glomeruli showed atrophy. In the medulla, there was destruction of the lining epithelium of the loops of Henle and the collecting duct. Pretreatment with melatonin (MT + TDF) attenuated TDF-induced renal damage. There was comparatively less damage to the tubules and glomerulus in the melatonin pretreated rats as compared with rats treated with TDF alone. The cortex of MT pretreated rats showed near normal morphology although some tubules showed mild epithelial desquamation and edema. However, there was no vacuolization or necrosis. The glomeruli appeared normal. Pretreatment with MT reduced TDF-induced damage to the medulla. Most of the tubules appeared normal. Few tubules showed dilatation. Treatment with 20 mg/kg body wt. melatonin alone had no significant toxic effect on the renal histology and the features were comparable with that of control rats. Scoring: Control-0, TDF- 3+, MT + TDF 1+, MT 0.

Representative images of the effect of melatonin pretreatment on renal histology. (A). Cortex of control rats showing normal architecture, (H&E) magnification ×100. (B) In the cortex of TDF-treated rats, the convoluted tubules are distorted and their lining epithelium is destroyed in proximal tubules (green arrow), destruction of the epithelial cells of distal convoluted tubules (blue arrow), dilation of proximal convoluted tubules (red arrow) and some glomeruli are shrunken (Black arrow). (C) Cortex of 20 mg Melatonin + TDF-treated rat. Black arrow indicates the normal glomerulus [G]. The damage was very much less as compared with the group treated with TDF alone. There was mild dilation of the proximal tubules (Blue arrow), and distal convoluted tubules (Red arrow) (D) Cortex of 20 mg Melatonin treated rat—Normal architecture. (E) medulla of control rat showing normal architecture (F) Medulla of TDF-treated rat showing destruction of the lining epithelium of the loops of Henle and the convoluted tubules (yellow arrow) in medulla, (Hematoxylin and Eosin) magnification ×100. (G) Medulla of 20 mg melatonin + TDF-treated rat—Normal architecture, Hematoxylin and Eosin, magnification ×100. (H) Medulla of kidney 20 mg Melatonin treated rat—Normal architecture, Hematoxylin and Eosin, magnification ×100.
Melatonin pretreatment partially attenuated TDF-induced tubular dysfunction
Proximal tubular function was impaired in TDF-treated rats, as evidenced by increased urinary bicarbonate, phosphate, potassium and uric acid excretion and decreased serum potassium and phosphate (Table 1). MT pretreatment completely restored the serum bicarbonate levels and serum uric acid level to normal values.
Effect of melatonin pretreatment on serum levels of potassium, phosphate, and urine levels of potassium, phosphate, bicarbonate, and uric acid.

Values are represented as Mean ± SD, n = number of rats. * P < 0.05, ** P < 0.01 vs. control.
Melatonin pretreatment attenuated TDF-induced renal nitrosative stress
Melatonin pretreatment attenuated TDF-induced increased iNOS protein and mRNA expression
The results are shown in Figure 2. Basal iNOS immunostaining was seen both in cortical and medullar structures of kidney taken from control rats. The expression of iNOS was prominent in the proximal tubules and distal tubules (A&B). In TDF-treated rats, extensive and prominent iNOS immunoreactivity was noted in the medulla, whereas iNOS immunostaining in the cortex was mild. In the cortex, iNOS immunoreactivity was seen in the glomerulus, and few distal tubules (DT) and proximal tubules (PT). In the medulla, strong and uniform iNOS immunostaining was seen in the MTAL and collecting duct cells as well as in the endothelium of the vasa recta (C&D). Melatonin pretreatment decreased iNOS immunostaining in both cortex and medulla (E &F). Melatonin alone treated rats revealed minimum staining for iNOS in both cortex and medulla [G &H]. IHC scores: control cortex and medulla, 1+; TDF cortex and medulla, 2+, MT+TDF- 1+, MT-0

Representative Immunohistochemical images of the effect of melatonin pretreatment on iNOS expression. Basal iNOS immunostaining was seen both in cortical and medullar structures of kidney taken from control rats (A &B). In TDF-treated rats, extensive and prominent iNOS immunoreactivity was noted in the medulla, whereas iNOS immunostaining in the cortex was sparse (C & D). Reduced positive immunostaining for iNOS in the cortex (E) and medulla (F) of melatonin + TDF-treated rat kidneys. The cortex and medulla of melatonin alone treated rats showing negligible staining for iNOS (G & H). Brown color indicates immunopositivity. N = 5 rats for each group. Black arrows indicate G-glomerulus, PT-proximal convoluted tubule, DT-distal convoluted tubule, HL-Henle’s loop. CT-collecting tubule. (Scale Bar 20 μm).
With respect to iNOS protein expression by western blot, TDF treatment increased iNOS expression significantly as compared with control MT pretreatment partially decreased TDF-induced increase in iNOS protein expression (Figure 3A &B). With respect to iNOS mRNA expression, TDF treatment resulted in nearly twofold increase. Pretreatment with melatonin partially prevented TDF-induced increased mRNA expression (Figure 3 C).

Melatonin pretreatment prevented TDF-induced increased nitrotyrosine (NT) and nitrocysteine contents in the kidney
Basal NT was observed in the kidneys of control rats (Figure 4A). Nitrotyrosine expression was increased in the cortex and medulla of TDF-treated rats. Similar to iNOS staining, the staining for NT was more intense in the medulla as compared with cortex. In the cortex, nitrotyrosine was localized to the apical region of proximal tubular cells, glomerulus, and distal convoluted tubules. In the medulla, the collecting duct and loop of Henle stained intensely for NT. Pretreatment with melatonin reduced the NT levels. IHC scores: control cortex and medulla, 0; TDF cortex and medulla, 2+. MT + TDF- 1+, MT-0.

Western Blot analysis is shown in Figure 4B and C. NT protein level was significantly higher in the TDF-treated rats as compared with control. Pretreatment with melatonin partially reduced the NT levels.
Immunolocalization of nitrocysteine showed negligible expression in control (Figure 5). In the TDF-treated rat kidneys, the staining for nitrocysteine was more intense in the medulla as compared with cortex. In the cortex, nitrocysteine was localized to the apical region of proximal tubular cells, glomerulus, and distal convoluted tubules. In the medulla, the collecting duct and LH stained intensely for nitrocysteine. Melatonin pretreatment minimized TDF-induced increased nitrocysteine expression IHC scores: control cortex and medulla, 0; TDF cortex and medulla, 3+. MT + TDF- 1+, MT-0
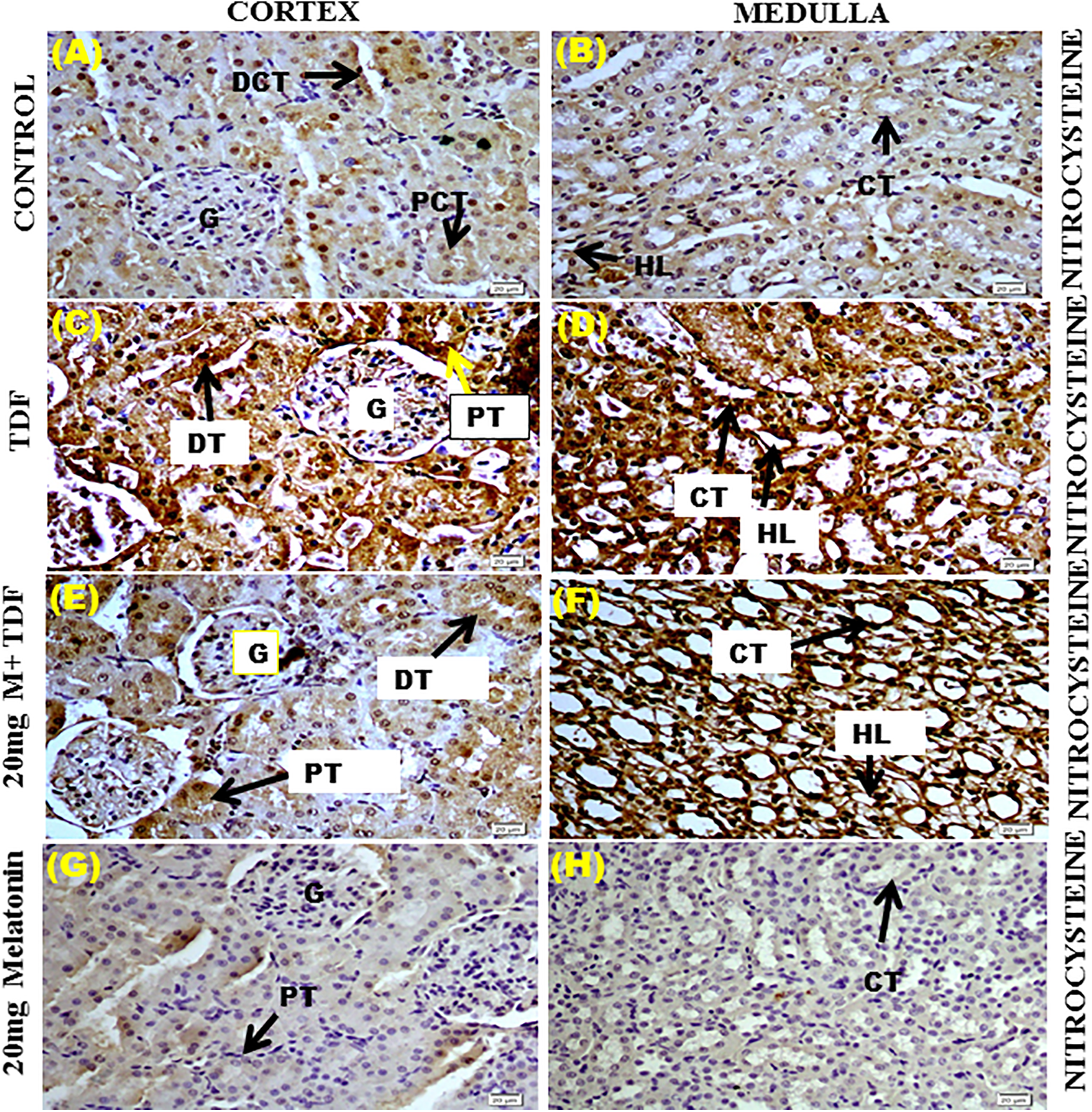
Representative immunohistochemical images of the effect of melatonin pretreatment on nitrocysteine expression
Melatonin pretreatment reduced TDF-induced renal cell apoptosis (TUNEL assay)
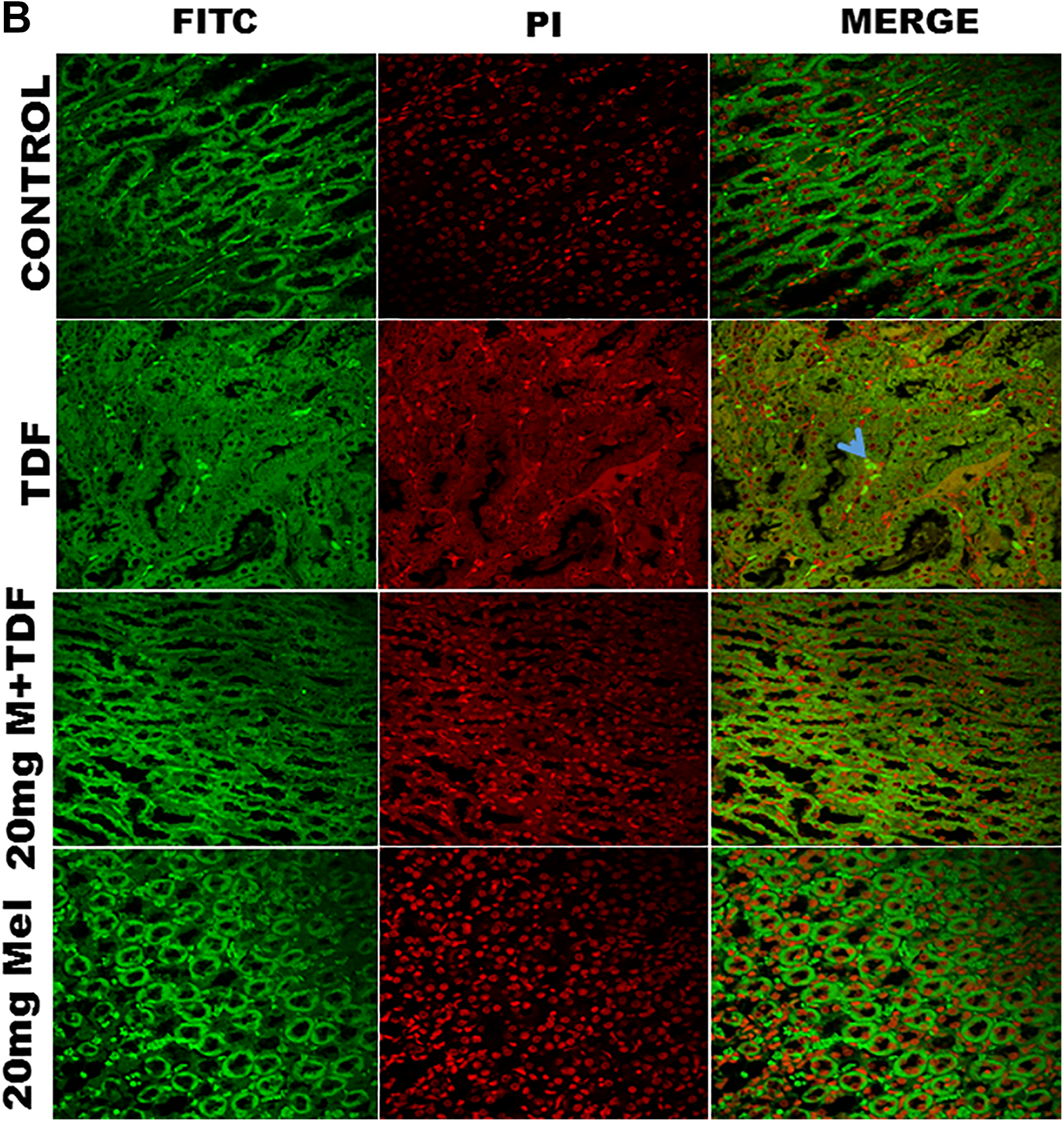
The number of apoptotic cells in rat renal tissues was evaluated by TUNEL assay in the cortex and medulla (Figure 6.A & B). Kidneys of control rats showed few apoptotic cells in both glomeruli and tubuli. Kidneys from the rats with TDF treatment showed an increase in the number of TUNEL-positive nuclei in both tubular cells as well as glomerular cells, indicating extensive apoptosis in both cell types. Melatonin pretreatment reduced the number of apoptotic cells.

(A) Effect of melatonin pretreatment on apoptosis induced by TDF in kidney cortex. Apoptosis was evaluated by TUNEL staining using terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate (TdT) nick end-labeling (A–D). Nuclei were counter stained with propidium iodide (PI) (E–H). Thus, red indicates propidium iodide (PI) stained nuclei, TUNEL-positive nuclei stain green and when red and green nuclei overlap they appear yellow. Arrows indicate TUNEL-positive cells. Scale bars = 60 μm.
Melatonin pretreatment reduced TDF-induced increased expression of apoptotic proteins, cyt c, caspase 3 and 9
After mPTP opening, cytochrome c is released, activating caspase 3 and promoting cell death. Immunohistochemical analysis showed a cytoplasmic punctate staining of cytochrome c and diffuse light cytochrome c staining and nuclear reactivity in the TDF-treated rat kidneys, compared with the control group, in which there was no staining (Figure 7). Horseradish peroxidase-based immunohistochemistry is not sensitive enough to determine mitochondrial content of cytochrome c; it can only detect diffuse cytosolic and nuclear cytochrome c-containing areas.

Representative images of Immunohistochemical localization of cyt. c in kidney tissue.
Caspase 3 and caspase 9 immunopositive staining was observed in the same TUNEL-positive areas in the TDF-treated rat kidney. The immunostaining of caspase 9 was less dense than caspase 3.
Immunohistochemistry showed that caspase 9 expression was virtually absent in the glomerulus and tubuli of control rats (Figure 8). In the TDF-treated rats, cell nuclei of both glomerular and tubular cells stained moderately for caspase 9. Pretreatment with melatonin almost completely prevented TDF-induced increase in caspase 9 protein expression, IHC scores: control cortex and medulla, 0; TDF cortex and medulla, 2+. MT + TDF- 1+, MT alone -0/1+.
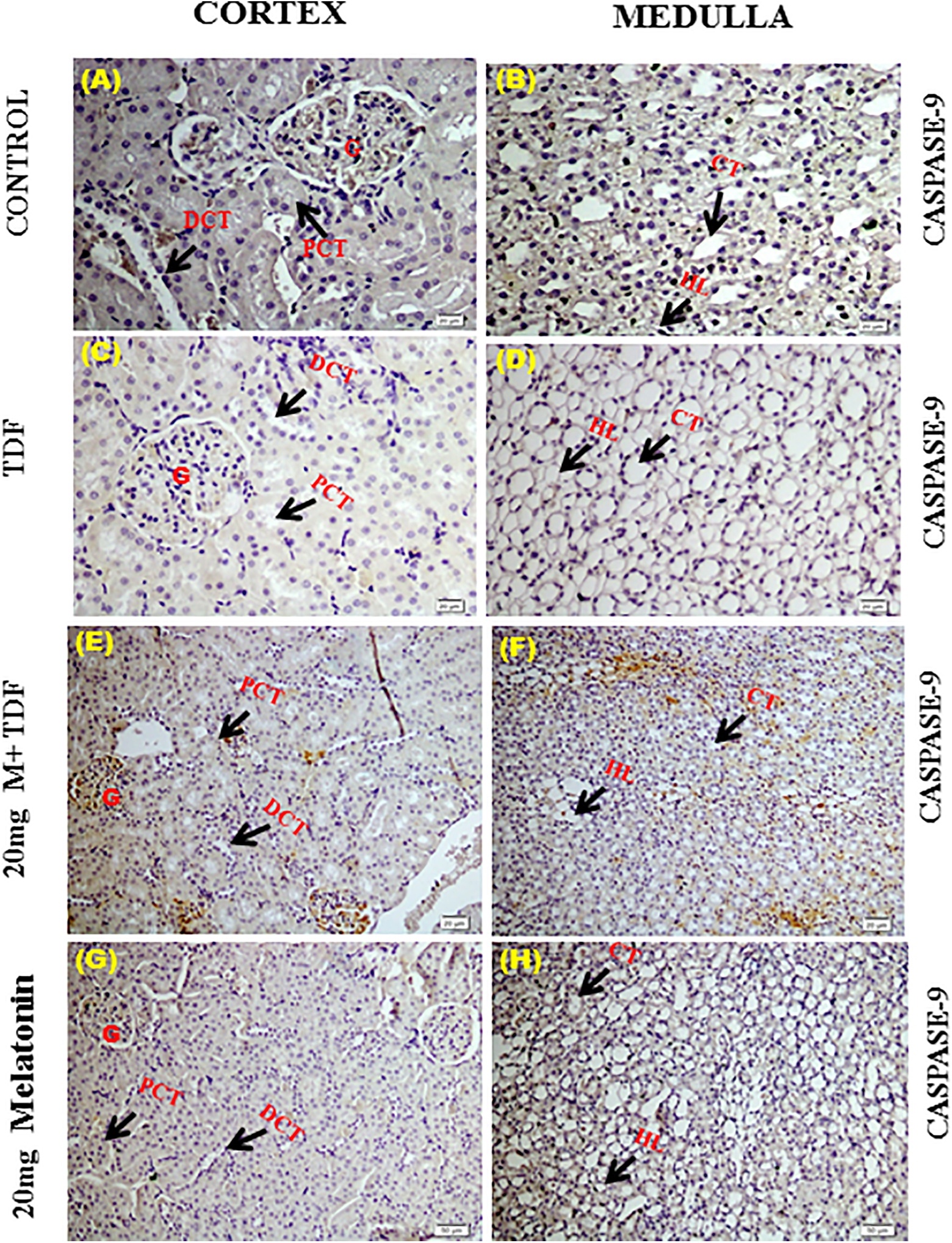
Representative images of Immunohistochemical localization of caspase-9 in kidney. (A)&(B) Control-Negative expression of caspase-9 in the cortex and medulla. (C)&(D) TDF-Mild expression of caspase-9 in the cortex region, intense staining in the medulla in TDF group. (E)&(F) M + TDF-Negligible expression of Caspase-9 in the cortex and moderate expression medulla in 20 mg melatonin + TDF-treated rats.
As cleaved caspase 3 is a well-characterized cell apoptotic marker, the protein level of cleaved caspase 3 was detected by immunohistochemistry and Western blot technique. No detectable caspase 3 immunostaining was observed in both cortical and medullar structures of kidney taken from the control rats (Figure 9A). In the TDF-treated rats, cell nuclei of both glomerular and tubular cells stained intensely for caspase 3. In the renal cortex of TDF-treated animals, diffuse and strong caspase 3 staining was seen in the proximal and distal tubules. The staining was mainly distributed in the basal part of the tubular cells where numerous mitochondria are distributed. The HL and vasa recta in outer medulla from TDF-treated rat showed marked increase in the expression of caspase 3. Pretreatment with melatonin almost completely prevented TDF-induced increase in caspase 3 protein expression, IHC scores: control cortex and medulla, 0; TDF cortex and medulla, 3+. MT + TDF- 0, MT-0

Representative images of immunohistochemical localization of caspase-3 in kidney. (A)&(B) Control-Negative expression of caspase-3 in the cortex and medulla.(C)&(D) TDF-Increased expression of caspase-3 in the glomerulus (G), mild expression in the proximal tubular cells (PCT), distal tubules (DCT) of the cortex region, intense staining in the loop of Henle (HL) and moderate expression in the collecting tubule (CT) of the medulla in TDF group. (E)&(F) M + TDF-Negligible expression of Caspase-3 in glomerulus (G), moderate expression in the proximal (PCT) and distal tubules (DCT) of the cortex, Henle’s loop (HL) and collecting tubules (CT) of the medulla in 20 mg melatonin + TDF-treated rats
Western blot analysis also revealed marked increase in caspase 3 protein levels in TDF-treated rats, which was significantly decreased upon melatonin pretreatment (Figure 9 B &C).
As a biochemical marker of apoptosis, we measured caspase activity (Figure 10A). With respect to caspase 3 activity more than twofold increase was observed in TDF-treated rats as compared with control. Melatonin pretreatment partially but significantly reduced TDF-induced increased caspase 3 activity.
TDF treatment increased caspase 3 mRNA expression as compared with control. Melatonin pretreatment had no significant effect on TDF-induced increase in caspase 3 mRNA expression (Figure 10B).

Effect of melatonin pretreatment on caspase-3 activity, n = 6 in each group. Data represents mean ±SD. * P < 0.05, ** p < 0.01 vs. control, # p < 0.05 vs. TDF. B. Relative expression of caspase 3 mRNA in kidneys of rats, n = 5 in each group. Data represents mean ± SD. *indicates p < 0.05 vs. control values, # p < 0.05 vs. TDF group.
Melatonin pretreatment on TDF-induced PARP over activation
Glomerulus and tubules of the control rat kidneys showed mild staining for PARP. In TDF-treated rat kidney cortex, the glomerulus and convoluted tubules stained intensely for PARP. In the medulla, the collecting tubules and HL also stained intensely for PARP (Figure 11). Melatonin pretreatment decreased the intensity of the stain, it was less as compared with TDF groups IHC scores: control cortex and medulla, 0; TDF cortex and medulla, 3 +; MT+TDF 1+, melatonin alone treated group showed negligible expression 0/1+.

Immunohistochemical localization of PARP-1 in kidney tissue. (A)&(B) Control-Negligible–mild expression of PARP in the cortex and medulla. (C)&(D) TDF-Focal increased expression of PARP stain in the glomerulus (G) and strong positivity in proximal convoluted tubules (PCT), distal convoluted tubules (DCT) of the cortex, and in the loop of Henle (HL) and collecting tubule (CT) of the medulla in TDF group. (E)&(F) M + TDF-Moderate expression of PARP in the cortex and medulla in 20 mg melatonin + TDF-treated rats. (G)& (H) M-Negligible expression of PARP in the cortex and medulla in 20 mg Melatonin alone treated rats. Scale Bars = 20μm.
Melatonin pretreatment restored the expression of oxidative stress proteins HSP70 and HO-1
In the control rats, moderate to high expression of HSP 70 was seen indicating a physiological role for HSP70 in the kidney (Figure 12). Moderate expression was seen in the glomerulus, increased expression was seen in the proximal convoluted tubules and distal convoluted tubules of cortex region, diffused expression in the region of Henle’s loop and collecting tubules of control rats (A & B). In the TDF-treated rats, decreased expression of HSP70 in the glomerulus, focal positivity in the proximal convoluted tubules and distal convoluted tubules, of the cortex region, low intensity of stain in the loop of Henle, collecting tubule of the medulla of TDF-treated rat(C & D). Melatonin pretreatment almost completely restored HSP70 expression. IHC score control 3+, TDF 1+, TDF + MT- 3+ and MT alone -3+/2+.

Representative immunohistomicrographs of HSP 70 protein expression in the kidneys. (A) & (B) Moderate expression of HSP 70 in the cortex and diffused expression in the medulla of control rats. (C) & (D) Decreased expression of HSP70 in the glomerulus (G), focal positivity in the proximal convoluted tubules (PCT) and distal convoluted tubules, (DCT) of the cortex region, low intensity of stain in the loop of Henle, collecting tubule (HL, CT) of the medulla of TDF-treated rat. (E) & (F) Moderate expression of HSP 70 in the cortex and diffused expression in the medulla of MT + TDF-treated rats. G&H. Moderate expression of HSP 70 in cortex and, diffused expression in the medulla of MT alone treated rats. Scale Bars = 50 μm.
With respect to HO-1 expression, mild expression of HO-1 was seen in the glomerulus, proximal convoluted tubule, distal convoluted tubules of cortex and loop of Henle and collecting tubules in control rats (Figure 13) indicating a physiological role for HO-1 in the kidney. In the TDF-treated rats, negligible expression of HO-1 was seen in the glomerulus, proximal convoluted tubules, distal convoluted tubules of the cortex, in the loop of Henle and collecting tubule of the medulla of TDF-treated rats (C & D). Melatonin pretreatment almost completely restored HO-1 expression. IHC score control 1+, TDF 0, TDF + MT- 1+ and MT alone -1+.

Representative immunohistomicrographs of HO-1 protein expression in the kidneys. (A) & (B) Mild expression of HO-1 in the kidneys of control rats. (C) & (D) Negative expression of HO-1 in the glomerulus (G), proximal convoluted tubules (PCT), distal convoluted tubules (DCT) of the cortex, and in the loop of Henle (HL) and collecting tubule (CT) of the medulla of TDF-treated rats. (E) & (F) Mild expression of HO-1 in the kidneys of MT + TDF-treated rats. (G) & (H) Mild expression of HO-1 in the kidneys of MT alone treated rats. Scale Bars = 20 μm.
Discussion
Several human and animal studies have shown that TDF damages the kidneys, specifically the renal proximal tubular mitochondria. 7,8 As persistent tubular injury may promote tubular cell loss, interstitial fibrosis and subsequent decreased glomerular filtration and renal failure, prevention of TDF tubulopathy is vital to avoid irreversible tubulointerstitial damage. Identification of therapeutic agents or prophylactic agents to treat or prevent TDF nephrotoxicity can lead to better tolerance to TDF, and a more effective treatment can be achieved in HIV-infected patients. The present study was undertaken to investigate whether pretreatment with MT can prevent/attenuate TDF nephrotoxicity in rats and to determine the cellular pathway that it targets.
We first examined whether melatonin pretreatment protects against TDF-induced histological changes in the kidney and proximal tubular function. Pretreatment with melatonin attenuated TDF-induced renal damage. MT pretreatment completely restored proximal tubular function in the TDF-treated rats. These findings suggest that MT is effective in preventing TDF-induced renal damage in rats. Melatonin has been shown to be effective in protecting against severe free radical-mediated nephrotoxicity in a variety of conditions, including, chemotherapy, and ischemia-reperfusion injury acute renal failure caused by mercuric chloride, and gentamicin and cisplatin. 25 –27
During cellular stress, iNOS is induced resulting in the production of nitric oxide (NO). 35 The cytotoxic potential of NO is a consequence of its ability to form peroxynitrite, 36 which is a potent nitrating and oxidizing agent. Peroxynitrite readily reacts with proteins to form 3-nitrotyrosine, 36 or nitrosothiols/nitrocysteine, 37 which can either lead to inactivation or activation of the proteins. 37,38 Peroxynitrite production is usually measured by estimating 3-nitrotyrosine levels (3-NT). 39 3-NT is considered as the footprint of peroxynitrite production, and its presence in tissues is taken as a diagnostic marker for exposure to peroxynitrite. 40 Therefore, we investigated iNOS protein expression by IHC and western blot and mRNA expression by RT CR, protein tyrosine nitration and nitrocysteine. TDF treatment increased iNOS expression by IHC and western blot and mRNA expression. In control rats, minimal staining for nitrotyrosine was seen in the epithelial cells of proximal tubules in agreement with the findings of Bian et al. 41 TDF treatment resulted in increased 3 NT level both immunohistochemical method and immunoblot thereby providing evidence for peroxynitrite formation. MT pretreatment significantly attenuated TDF-induced iNOS protein and mRNA expression, protein tyrosine nitration and cysteine nitrosylation. The results show that melatonin pretreatment attenuates TDF-induced nitrosative stress.
Peroxynitrite can damage the cells by the oxidation of free thiols-cysteine residues. 39 Dysregulated (diminished or excessive) S-nitrosylation has been implicated in several pathophysiological conditions. 39 In the present study, we observed a massive increase in nitrocysteine staining in kidneys of TDF-treated rat. This suggest that S-nitrosylation may play a role in TDF nephrotoxicity. S-nitrosylation is modulated by the cellular redox status; its formation is dependent on the state of redox equilibrium and is prevented by high levels of antioxidants. 42 NO has been found to block cell death after GSH depletion by preserving the redox status of mitochondrial protein thiols, by a mechanism that involves S-nitrosylation of mitochondrial protein thiols. 43 This may represent an endogenous protective mechanism for the mammalian cell against nitrosative/oxidative stress when intracellular thiols or other redox constituents have decreased significantly. We have previously shown that TDF treatment results in the depletion of renal reduced GSH and protein thiol. 11 This may explain the massive increase in nitrocysteine levels in the TDF-treated rat kidneys. Melatonin has been shown to be a powerful antioxidative and antinitrosative agents. 44,45 Melatonin has been shown to be effective in protecting against iNOS-mediated nephrotoxicity in a variety of conditions, including ischemia-reperfusion injury, 46 and acute renal failure caused by mercuric chloride, 27 gentamicin. 26
Apoptosis is a physiological or pathological process that occurs by a complex signaling cascade and caspase activation plays a central role in this process. The TUNEL technique is commonly used for detection of apoptosis the tissue sections. Caspase activation is a specific indicator of apoptosis and it can be detected immunohistochemically. 47 Therefore, we used active caspase-3 immunolabeling also to confirm the results of the TUNEL technique.
Cytochrome c is best known as an indicator of apoptosis in any organ/tissue and is released during mitochondrial damage. During apoptosis, cytochrome c is released from the mitochondria into the cytosol. Cytochrome c activates the apoptotic protease activating factor-1 (Apaf-1). Subsequently, Apaf-1 and cytochrome c bind to procaspase-9 and activate it. After activation, caspase-9 activates the effector caspase 3. We observed immunostaining of caspase-3 and caspase-9 in the same TUNEL-positive areas in TDF-treated group, which we consider evidence for mitochondria-related apoptosis. Melatonin pretreatment reduced the number of apoptotic cells. Pretreatment with melatonin almost completely prevented TDF-induced increase in caspase 9 protein expression and caspase 3 protein expression. Melatonin pretreatment partially but significantly reduced TDF-induced increased caspase 3 activity. These findings suggest that the protective effect of MT may be mediated by its ability to inhibit apoptosis. MT treatment has been shown to prevent against cisplatin, gentamicin and docetaxel induced kidney injury by inhibiting apoptosis. 25,26,48
PARP-1 (poly (ADP-ribose) polymerase-1) is a Zn-finger nuclear protein that is needed for DNA repair upon free radial induced damage. PARP-1 activated by single-strand DNA breaks, ROS, peroxynitrite or disruption of mitochondrial membrane potential. 49 –54 In response to inflammatory stress, PARP-1 induces the transcription of iNOS gene. The subsequent production of nitric oxide and peroxynitrite triggers DNA-strand breaks that activate PARP-1. The basal enzymatic activity of PARP1 is very low, but it is stimulated dramatically under conditions of cellular stress. 55,56 While moderate PARP activity protects cellular genome integrity; excessive PARP activation can lead to cell death from ATP depletion. 57 The over activation of PARP leads to NAD+ and ATP depletion and, finally, cell death. 58 Thus, oxidative and nitrosative stress and/or peroxynitrite-induced DNA damage is a potent trigger of PARP-1 activation. PARP-1 activation is also involved in the activation of various pro-inflammatory transcription factors including nuclear factor kB (NF-kB). 59 Accordingly in our earlier studies we have demonstrated increased oxidative stress, nitrosative stress, mitochondrial damage, PARP-1 overactivation and NFkB activation in TDF-treated rat kidneys. In the present study we have demonstrated that MT pretreatment prevents TDF-induced PARP overactivation. MT has been shown to PARP activation and prevent cellular injury. 60 –62 MT has been shown to ameliorate renal injury by inhibiting NFkB upregulation. MT has been shown to protect against various model of renal injury including cisplatin, gentamicin and ureteral obstruction through inhibition of NkB inflammatory pathway. 26,63 –67
Heat shock proteins (HSPs), also known as stress proteins/molecular chaperones, play an important role in protecting cellular processes from toxic injury, by several mechanisms including preserving the structure of normal proteins and repairing or removing damaged ones. 68 In the present study, we observed decrease expression of HSP70 in the kidneys of TDF treated rats. This may be due to consequence of increased oxidative stress induced by TDF. Hsp70 has been previously reported to result in cellular protection against biological stress in vitro and in vivo. 69 –72 It was also reported that HSP70 could attenuate cisplatin-induced nephrotoxicity and the protective effects of HSP70 are associated with decreased apoptosis. 73 Several studies demonstrated that HSP70 s are induced in tubular epithelial cells following renal ischemia/reperfusion 74,75 and toxic injury. 76,77 HSP70 has been shown to prevent renal damage and restore renal function. 78 Hsp70 expression has previously been shown to have beneficial effect on renal injury through the inhibition of inflammation and reduced expression of inflammatory cytokines. 79 Bidmon et al. showed that Hsp70 expression was associated with repair of the proximal tubule following renal ischemia. 80 In the present study, TDF groups showed a marked decrease in immunoreactivity against HSP70 in the kidneys of rats. MT-treated rats showed increased positive immunoreactivity against HSP70, which confirms its anti-inflammatory and anti-oxidant potential in our study.
HO-1, a ubiquitously expressed microsomal enzyme is an important protecting factor in both inflammatory and non-inflammatory renal damage. HO-1 is induced by heat, cytokines, and reactive oxygen species. 81 In human cells, HO-1 is involved mainly in the protection against the adverse effects of oxidative stress. 82 Overexpression of HO-1 has been shown to attenuate pathological processes, such as peritubular capillary (PTC) loss, tubular apoptosis, and proliferation of myofibroblasts. 83 The protective role of HO-1 has been demonstrated in ischemic and toxic kidney damage, rejection of kidney transplant, obstructive nephropathy, 83 and diabetic nephropathy. 84 In the present study MT pretreatment restored TDF-induced decrease in HO-1 expression in the kidneys of rats.
In humans and rodents, three melatonin receptors (MT1, MT2, and MT3) have been shown to be expressed in various extrapineal tissues including brain and retina, kidney tubules, cardiovascular system, liver and gallbladder, intestine, immune cells, adipocytes, prostate and breast epithelial cells, ovary/granulosa cells, myometrium, and skin. 85
The presence of high-affinity melatonin receptors, MT1 and MT3 in the kidneys has been confirmed. 86 The MT1 receptor (belonging to the GPCR/G protein coupled receptor subgroup)is coupled to G proteins, and its effect is a decrease in intracellular cAMP, and consequently a decrease in protein kinase A activity and CREB (cyclic-AMP response element-binding) phosphorylation and an increase in intracellular concentration of calcium ions. 87,88 MT1 receptors are present in mitochondrial membranes and is suggested to play a role in the inhibition of cytochrome c release and apoptosis. 89,90 MT3, Quinone reductase 2 enzyme/ QR2 is involved in prevention from oxidative stress by inhibiting the electron transfer reactions of quinones. 91
The safety and efficacy of melatonin have been extensively investigated in several clinical trials that documented its potential therapeutic usefulness in variety of disease conditions including renal damage. 92 Pretreatment of melatonin has also been found to ameliorate the iron and erythropoietin-induced oxidative stress in anemic patients with end-stage renal failure. 93 A double-blind placebo controlled clinical trial has revealed that concomitant administration of melatonin and zinc acetate ameliorate the poorly controlled microalbuminuria induced renal failure in type 2 diabetes mellitus patients treated with biguanides. 94 Melatonin appears to be promising in the management of chronic kidney disease. 95 Considering the nephroprotective benefits of melatonin, there is an ongoing randomized, double-blind, placebo controlled clinical trial to test the nephroprotective potential of melatonin in polymyxin B induced nephrotoxicity. 91 In addition, phases 2 and 3 clinical trials are under way in order to improve quality of life in patients with CKD and ESRD who are undergoing dialysis and have sleep disturbances (ClinicalTrials.gov; NCT01922999, NCT00698360, NCT00388661).
Conclusion
In the present study, we have demonstrated that melatonin treatment is effective in attenuating TDF nephrotoxicity in rats by targeting multiple cellular pathways including upregulation of oxidative stress proteins, nitrosative stress, mitochondrial apoptosis, and PARP overactivation. In our earlier studies, we have demonstrated that MT pretreatment prevented TDF induce renal damage by inhibiting oxidative stress, MPO activity and downregulation of NFkB inflammatory pathway. These suggest pleiotropic effects of MT in preventing TDF-induced renal damage (Figure 14). The renoprotective findings of melatonin in clinical settings might open a new window into therapeutic strategies for subjects with TDF nephrotoxicity. If melatonin is shown to be protective against TDF nephrotoxicity in clinical trials, melatonin would be an excellent adjunctive therapy against TDF nephrotoxicity as it is natural, inexpensive, widely available, and has minimal side effects

Graphical representation of the possible cascades or pathways involved in the renoprotective potential of melatonin against tenofovir-induced nephrotoxicity.
Footnotes
Acknowledgments
We would like to thank the Centre for Scientific and Industrial Research (CSIR), New Delhi for the financial support. Ms. Hemalatha Ramamoorthy is a Senior Research Fellow on the project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

