Abstract
Oxidative stress is considered a key hallmark of preeclampsia, which causes the dysregulation of trophoblast cells, and it contributes to the pathogenesis of preeclampsia. Emerging evidence has suggested bromodomain-containing protein 4 (BRD4) as a key regulator of oxidative stress in multiple cell types. However, whether BRD4 participates in regulating oxidative stress in trophoblast cells remains undetermined. The current study was designed to explore the potential function of BRD4 in the regulation of oxidative stress in trophoblast cells. Our data revealed that BRD4 expression was elevated in trophoblast cells stimulated with hydrogen peroxide. Exposure to hydrogen peroxide caused marked decreases in the levels of proliferation and invasion but promoted apoptosis and the production of ROS in trophoblast cells. Knockdown of BRD4, or treatment with a BRD4 inhibitor, markedly increased the levels of cell proliferation and invasion and decreased apoptosis and ROS production following the hydrogen peroxide challenge. Further data indicated that suppression of BRD4 markedly decreased the expression levels of Keap1, but increased the nuclear expression of Nrf2 and enhanced Nrf2-mediated transcriptional activity. BRD4 inhibition-mediated protective effects were markedly reversed by Keap1 overexpression or Nrf2 inhibition. Overall, these results demonstrated that BRD4 inhibition attenuated hydrogen peroxide-induced oxidative stress injury in trophoblast cells by enhancing Nrf2 activation via the downregulation of Keap1. Our study highlights the potential importance of the BRD4/Keap1/Nrf2 axis in the modulation of the oxidative stress response in trophoblast cells. Targeted inhibition of BRD4 may offer new opportunities for the development of innovative therapeutic approaches to treat preeclampsia.
Introduction
Preeclampsia is a severe and life-threatening complication of pregnancy and is a leading cause of maternal and neonatal death. 1,2 Preeclampsia, characterized by hypertension and proteinuria, usually occurs during the second or third trimester of gestation and affects approximately 3–8% of pregnant women globally. 1,2 Multiple risk factors contribute to the initiation and development of preeclampsia; however, the precise molecular mechanisms involved in the initiation of the condition are poorly understood. 3 Notably, oxidative stress within the placenta has been suggested as a crucial intermediate step in the pathogenesis of preeclampsia. 4,5 Placental oxidative stress promotes the abnormal functioning of trophoblast cells and results in decreases in proliferation and invasion. This leads to impaired embryo implantation and altered fetal development, which is related to the development of preeclampsia. 6,7 Therefore, elucidating the mechanisms involved in oxidative stress injury in trophoblasts may provide researchers with a better understanding of the molecular pathogenesis of the development of preeclampsia.
Bromodomain-containing protein 4 (BRD4) is a member of the bromodomain and extra-terminal domain (BET) protein family and exerts key effects on a wide range of cellular processes. 8 –10 BRD4 is the best characterized BET protein and plays a pivotal role in the regulation of gene expression by interacting with a number of transcription factors. 11,12 Currently, a growing body of evidence has been collected that has demonstrated that BRD4 modulates a variety of biological functions, and its dysregulation contributes to the pathogenesis of multiple diseases. 13 –16 Interestingly, BRD4 has emerged as a crucial regulator of oxidative stress in various pathological processes. 17 –20 BRD4 inhibition has been shown to exert cytoprotective effects against oxidative stress induced by various stimuli. 21 –23 Thus, BRD4 serves as an attractive target for overcoming oxidative stress.
Nuclear factor erythroid-derived 2-like 2 (Nrf2) is a type of basic leucine zipper transcription factor that has an essential role in the maintenance of redox homeostasis in cells. 24 Under quiescent conditions, Nrf2 binds to its repressor, kelch-like ECH-associated protein 1 (Keap1), in order to promote its sequestration in the cytoplasm for proteasomal degradation. 25 When the oxidative stress response is triggered, Keap1 is inactivated and Nrf2 is liberated from Keap1-mediated repression. This leads to the stabilization of Nrf2 and its nuclear translocation. 25 In the nucleus, Nrf2 binds to antioxidant response element (ARE) sites in promoter regions of target genes in order to initiate the transcription of a number of cytoprotective enzymes, which strengthens reactive oxygen species (ROS) scavenging. 25 Notably, recent evidence has demonstrated that Nrf2 plays a key role in the pathogenesis of preeclampsia. 26,27 Accumulating evidence has shown that activation of Nrf2 signaling is capable of protecting trophoblast cells from experiencing oxidative stress injury, which provides insight into the potential therapeutic role for Nrf2 in the treatment of preeclampsia. 28 –30
Emerging evidence has revealed that BRD4 acts as a key regulator of oxidative stress in various cell types. However, whether BRD4 participates in the modulation of oxidative stress in trophoblast cells remains unclear. In the current study, we aimed to determine the potential role and molecular mechanism of BRD4 in the regulation of oxidative stress in trophoblast cells.
Materials and methods
Cell culture
The human, first-trimester, extravillous trophoblast cell line, HTR8/SVneo, was purchased from BeNa Culture Collection (BNCC, Kunshan, China). HTR8/SVneo cells were cultured in Dulbecco’s Modified Eagle Medium with Nutrient Mixture F-12 (DMEM/F12; Thermo Fisher Scientific, Waltham, MA, USA) that was supplemented with 10% fetal bovine serum (FBS). To maintain cell growth, cells were placed into a 5% CO2-containing atmosphere at 37°C.
Hydrogen peroxide (H2O2) exposure
For the induction of oxidative stress injury, HTR8/SVneo cells were exposed to various concentrations (100, 250, and 500 μM) of H2O2 (Merck KGaA, Darmstadt, Germany) for 24 h.
RNA isolation and real-time quantitative polymerase chain reaction (RT-qPCR) assays
To detect the mRNA expression of BRD4, real-time quantitative polymerase chain reaction (RT-qPCR) was performed as previously described. 31 To analyze the expression of target genes, the housekeeping gene GAPDH was used as an internal control. Data were calculated in accordance with the 2−ΔΔCt method. The primer sequences used in this experiment were as follows: BRD4, forward: 5′-GAGCTACCCACAGAAGAAACC-3′ and reverse: 5′-GAGTCGATGCTTGAGTTGTGTT-3′; GAPDH, forward: 5′-GGAGCGAGATCCCTCCAAAAT-3′ and reverse: 5′-GGCTGTTGTCATACTTCTCATGG-3′.
Protein extraction and Western blot
To determine the levels of proteins, Western blot was performed as previously described. 32 Primary antibodies against BRD4, Keap1, Nrf2, Histone H3, and GAPDH (Abcam, Cambridge, UK) were utilized in the experiments. Proteins were developed by a horseradish peroxidase-conjugated secondary antibody (Abcam) and ECL Plus Western Blotting Substrate (Thermo Fisher Scientific, Waltham, MA, USA). The intensity of each protein band was measured using Image-Pro Plus 6.0 software (MediaCybernetics, Rockville, MD, USA).
Cell transfection
The siRNA sequences targeting BRD4 were synthesized using GenePharma software (Shanghai, China). The CDS region of BRD4 or Keap1 was subcloned into a pcDNA3.1 plasmid to construct either BRD4 or Keap1 expression plasmids. The transfection of siRNA or/and plasmids was achieved using Lipofectamine 3000 Transfection Reagent (Thermo Fisher Scientific, Waltham, MA, USA) in compliance with the manufacturer’s instructions.
Cell proliferation assay
To measure the proliferation of HTR8/SVneo cells, cell counting kit-8 (CCK-8) assay was carried out using a CCK-8 kit (Dojindo Molecular Technologies, Tokyo, Japan) according to a previously described method. 33 Cell proliferation was determined by measuring the absorbance at 450 nm with a microplate spectrometer (Bio-Rad, Hercules, CA, USA).
Transwell invasion assay
To assess the capacity of HTR8/SVneo cells, transwell invasion assay was performed as previously described. 33 Invading cells were conventionally fixed, stained, and counted using an inverted microscope.
Flow cytometric apoptosis assay
To evaluate the apoptotic rate of HTR8/SVneo cells, flow cytometric apoptosis assay was carried out using an Annexin V-FITC/PI Apoptosis Detection Kit (Beyotime, Shanghai, China) according to a previously described method. 34 The cell apoptosis rate was analyzed using a FACSCalibur system (BD Biosciences, San Jose, CA, USA).
Reactive oxygen species (ROS) assay
To measure the levels of intracellular ROS, ROS assay was performed using an ROS Assay Kit (Beyotime, Shanghai, China) as per a previously described method. 35 The ROS levels were determined by measuring the fluorescence intensity using flow cytometry.
Luciferase reporter assay
To monitor the transcription activity of Nrf2/ARE, luciferase reporter assay was carried out using pARE-luc reporter plasmids (Beyotime, Shanghai, China) according to a previously described method. 36 Luciferase activities within cells were measured using the Dual-Lumi Luciferase Reporter Gene Assay Kit (Beyotime).
Statistical analysis
Experimental data were expressed in the form of means ± standard deviation (SD). Statistical analyses were performed using GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA). Statistical differences were assessed using one-way analysis of variance (ANOVA) followed by the Tukey’s post hoc test. When p < 0.05, differences were deemed statistically significant.
Results
BRD4 expression in trophoblast cells following H2O2 exposure
To determine the relevance of BRD4 in the regulation of oxidative stress in trophoblast cells, we first examined changes in BRD4 expression in trophoblast cells in response to H2O2 exposure using an in vitro model for studying oxidative stress injury in trophoblast cells. Our data demonstrated that BRD4 mRNA expression was markedly elevated in a dose-dependent manner in response to H2O2 treatment in HTR8/SVneo cells (Figure 1A). In accordance with these data, BRD4 protein expression was also shown to increase in a dose-dependent manner in HTR8/SVneo cells following H2O2 exposure (Figure 1B). Taken together, these data indicated that BRD4 expression was elevated in response to oxidative stress in trophoblast cells.

The effect of H2O2 exposure on BRD4 expression in HTR8/SVneo cells. HTR8/SVneo cells were exposed to various doses of H2O2 (100, 250, and 500 μM) and cultured for 24 h before subsequent detection of mRNA and protein expression levels. (A) Relative mRNA expression of BRD4 was determined using an RT-qPCR assay. (B, C) Protein expression of BRD4 was determined via Western blot. *p < 0.05.
Effect of BRD4 knockdown on H2O2 exposure-induced injury in trophoblast cells
To explore the function of BRD4 in the regulation of H2O2-induced oxidative stress injury in trophoblast cells, the effects of BRD4 loss-of-function were assessed using siRNA-mediated gene silencing. Transfection of siRNA targeting BRD4 markedly decreased the expression of BRD4 in H2O2-exposed trophoblast cells (Figure 2A–C). These data revealed that exposure to H2O2 markedly decreased the proliferative and invasive capacities of HTR8/SVneo cells (Figure 2D and E). Notably, knockdown of BRD4 significantly rescued the observed decreases in proliferation and invasion in H2O2-exposed HTR8/SVneo cells (Figure 2D and E). In addition, H2O2 treatment promoted apoptosis and ROS production in HTR8/SVneo cells. These effects were markedly attenuated when BRD4 was knocked down (Figure 2F and G). Overall, these results suggested that knockdown of BRD4 alleviated H2O2-induced oxidative stress injury in trophoblast cells.

BRD4 silencing protects trophoblast cells from H2O2-induced oxidative stress injury. HTR8/SVneo cells were transfected with NC siRNA or BRD4 siRNA for 48 h and then exposed to H2O2 (500 μM) for another 24 h. (A) BRD4 mRNA expression was analyzed via RT-qPCR, and (B, C) BRD4 protein expression was determined via Western blot (**p < 0.01 compared with the control group or the H2O2 + NC siRNA group). (D) Cell proliferation was assessed using a CCK-8 cell proliferation assay (**p < 0.01 compared with the control group or the H2O2 + NC siRNA group). (E) Cell invasion was evaluated using a Transwell invasion assay (*p < 0.05 compared with the control group or the H2O2 + NC siRNA group). (F) Cell apoptosis was measured using the Annexin V-FITC/PI apoptosis assay (**p < 0.01 compared with the control group or the H2O2 + NC siRNA group). (G) ROS levels were monitored via a DCFH-DA probe staining assay (***p < 0.001 compared with the control group or the H2O2 + NC siRNA group).
Effect of BRD4 inhibitor on H2O2 exposure-induced injury in trophoblast cells
To confirm that BRD4 inhibition exerted protective effects against oxidative stress injury, we investigated the effect of treatment with the BRD4 inhibitor, JQ1, on H2O2-induced oxidative stress injury in trophoblast cells. As expected, treatment with JQ1 markedly enhanced the proliferative and invasive capacity of HTR8/SVneo cells exposed to H2O2 (Figure 3A and B). Moreover, H2O2 exposure-induced apoptosis and ROS production in HTR8/SVneo cells were attenuated post-JQ1 treatment (Figure 3C and D). These data confirmed that BRD4 inhibition exerted protective effects against oxidative stress injury in trophoblast cells.

Treatment with the BRD4 inhibitor, JQ1, alleviated H2O2-induced oxidative stress injury in trophoblast cells. HTR8/SVneo cells were cultured with 5 μM of JQ1 for 48 h and were subsequently exposed to 500 μM of H2O2 for another 24 h. (A) Cell proliferation was assessed using a CCK-8 cell proliferation assay (**p < 0.01 compared with the control group or the H2O2 + vehicle group). (B) Cell invasion was evaluated via the Transwell invasion assay (*p < 0.05 compared with the control group or the H2O2 + vehicle group). (C) Cell apoptosis was measured using an Annexin V-FITC/PI apoptosis assay (**p < 0.01 compared with the control group or the H2O2 + vehicle group). (D) ROS levels were monitored using a DCFH-DA probe staining assay (***p < 0.001 compared with the control group or the H2O2 + vehicle group).
Effect of BRD4 overexpression on H2O2 exposure-induced injury in trophoblast cells
Considering that BRD4 inhibition protected trophoblast cells from oxidative stress injury, we speculated that BRD4 overexpression may exacerbate oxidative stress injury in trophoblast cells. To assess the effects of BRD4 overexpression, we transfected HTR8/SVneo cells with a BRD4 expression vector. The upregulation of BRD4 in H2O2-induced HTR8/SVneo cells was confirmed by Western blot (Figure 4A and B). As expected, BRD4 overexpression further decreased the proliferative and invasive capacities of HTR8/SVneo cells exposed to H2O2 (Figure 4C and D). Moreover, BRD4 overexpression markedly exacerbated H2O2-induced apoptosis and ROS production in HTR8/SVneo cells (Figure 4E and F). Overall, these findings suggested that upregulation of BRD4 exacerbated H2O2-induced oxidative stress injury in trophoblast cells.
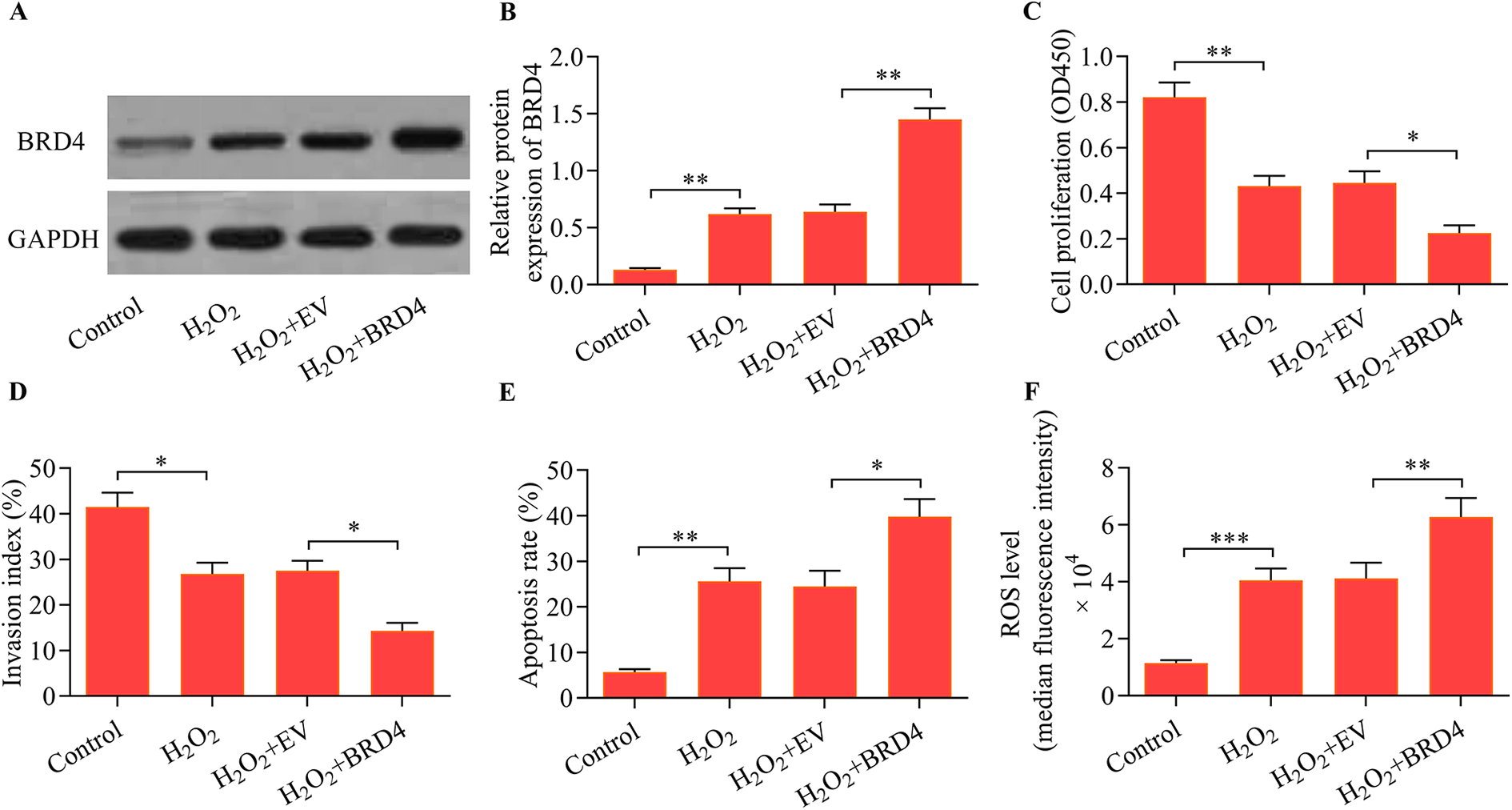
BRD4 overexpression exacerbated H2O2-induced oxidative stress injury in trophoblast cells. HTR8/SVneo cells were transfected with either an empty vector (EV) or BRD4 expression vector for 48 h followed by H2O2 exposure (500 μM) for another 24 h. (A, B) BRD4 protein expression was determined by Western blot (**p < 0.01 compared with the control group or the H2O2 + EV group). (C) Cell proliferation was examined using a CCK-8 cell proliferation assay (**p < 0.01 compared with the control group; *p < 0.05 compared with the H2O2 + EV group). (D) Cell invasion was assessed via the Transwell invasion assay (*p < 0.05 compared with the control group or the H2O2 + EV group). (E) Cell apoptosis was monitored using an Annexin V-FITC/PI apoptosis assay (**p < 0.01 compared with the control group; *p < 0.05 compared with the H2O2 + EV group). (F) ROS levels were measured using a DCFH-DA probe staining assay (***p < 0.001 compared with the control group; **p < 0.01 compared with the H2O2 + EV group).
Effect of BRD4 inhibition on Keap1/Nrf2 axis in trophoblast cells following H2O2 exposure
To uncover the molecular basis of BRD4 inhibition-mediated protection against oxidative stress injury in trophoblast cells, we investigated the regulatory effect of BRD4 inhibition on Nrf2 signaling. These data revealed that knockdown of BRD4 markedly decreased the expression of the Keap1 protein but promoted the nuclear expression of Nrf2 (Figure 5A and B). Moreover, BRD4 knockdown significantly enhanced the transcriptional activity of Nrf2/ARE (Figure 5C). Notably, similar data were obtained by using the BRD4 inhibitor, JQ1 (Figure 5D–F). Collectively, these results indicated that BRD4 inhibition enhanced Nrf2 activation in trophoblast cells following oxidative stress.
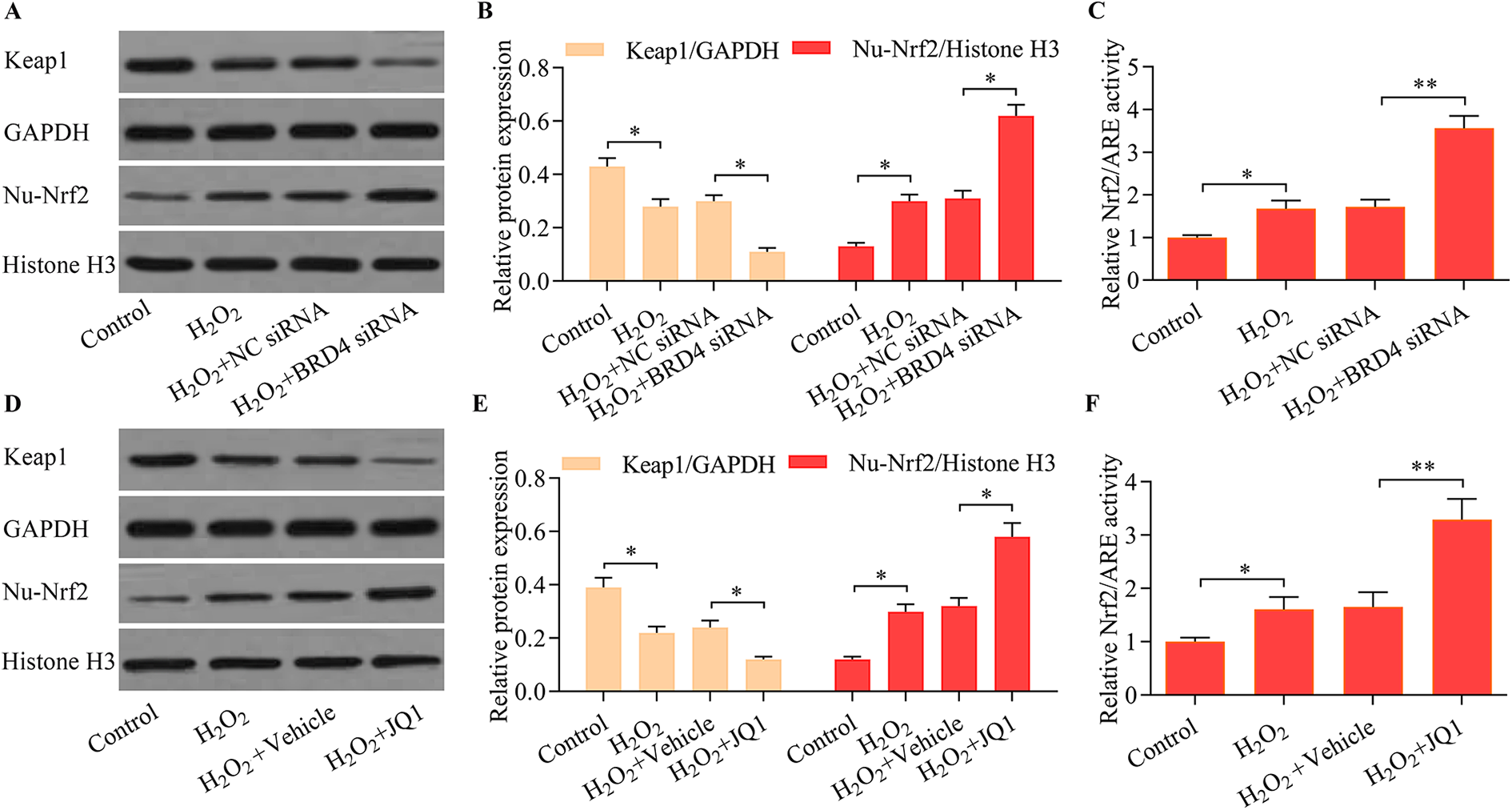
BRD4 inhibition enhanced Nrf2 activation and Keap1 downregulation. HTR8/SVneo cells were transfected with NC siRNA or BRD4 siRNA for 48 h and then exposed to H2O2 (500 μM) for another 24 h. (A, B) The effect of BRD4 knockdown on Keap1 and nuclear Nrf2 expression was determined using Western blot (*p < 0.05 compared with the control group or the H2O2 + NC siRNA group). (C) The effect of BRD4 knockdown on Nrf2/ARE-mediated transcriptional activity was monitored using a luciferase reporter assay (*p < 0.05 compared with the control group or the H2O2 + NC siRNA group). HTR8/SVneo cells were cultured with 5 μM of JQ1 for 48 h and were subsequently exposed to 500 μM of H2O2 for another 24 h. (D, F) The effect of the BRD4 inhibitor, JQ1, on Keap1 and nuclear Nrf2 expression was assessed via Western blot (*p < 0.05 compared with the control group or the H2O2 + vehicle group). (E) The effect of the BRD4 inhibitor, JQ1, on Nrf2/ARE-mediated transcriptional activity was monitored using a luciferase reporter assay (*p < 0.05 compared with the control group; **p < 0.01 compared with the H2O2 + vehicle group).
Effect of Keap1 overexpression on BRD4 knockdown-mediated effect in trophoblast cells following H2O2 exposure
To confirm whether BRD4 inhibition enhances Nrf2 activation via downregulation of Keap1, we assessed whether the overexpression of Keap1 could rescue the effects of BRD4 knockdown. We found that transfection of the Keap1 expression vector markedly restored Keap1 expression in HTR8/SVneo cells transfected with BRD4 siRNA (Figure 6A and B). As expected, Keap1 upregulation significantly abrogated the BRD4 knockdown-mediated Nrf2 activation (Figure 6C). Moreover, Keap1 upregulation markedly reversed the observed BRD4 knockdown-mediated anti-oxidative stress injury (Figure 6D–G). These data confirmed that BRD4 knockdown enhanced Nrf2 activation via the downregulation of Keap1.

Keap1 overexpression reversed BRD4 knockdown-mediated anti-oxidative stress injury in trophoblast cells. HTR8/SVneo cells were cotransfected with BRD4 siRNA and a Keap1 expression vector for 48 h followed by H2O2 exposure (500 μM) for another 24 h, and (A, B) Keap1 protein expression was determined via Western blot (*p < 0.05 compared with the control group or the H2O2 + NC siRNA group; **p < 0.01 compared with the H2O2 + BRD4 siRNA + EV group). (C) Nrf2 activation was monitored using a luciferase reporter assay (*p < 0.05 compared with the control group; **p < 0.01 compared with the H2O2 + NC siRNA group or the H2O2 + BRD4 siRNA + EV group). (D) Cell proliferation was examined via a CCK-8 cell proliferation assay (**p < 0.01 compared with the control group or the H2O2 + NC siRNA group or the H2O2 + BRD4 siRNA + EV group). (E) Cell invasion was assessed using a Transwell invasion assay (*p < 0.05 compared with the control group or the H2O2 + NC siRNA group or the H2O2 + BRD4 siRNA + EV group). (F) Cell apoptosis was monitored using an Annexin V-FITC/PI apoptosis assay (**p < 0.01 compared with the control group or the H2O2 + NC siRNA group; *p < 0.05 compared with the H2O2 + BRD4 siRNA + EV group). (G) ROS levels were measured using a DCFH-DA probe staining assay (***p < 0.001 compared with the control group or the H2O2 + NC siRNA group or the H2O2 + BRD4 siRNA + EV group).
Effect of Nrf2 inhibition on BRD4 knockdown-mediated effect in trophoblast cells following H2O2 exposure
To confirm that BRD4 knockdown protected trophoblast cells from oxidative stress injury by enhancing Nrf2 activation, we assessed the effect of Nrf2 inhibition on BRD4 knockdown-mediated anti-oxidative stress injury. We found that treatment with the Nrf2 inhibitor, ML385, markedly decreased the Nrf2 activation that was induced via BRD4 knockdown in H2O2-induced trophoblast cells (Figure 7A). As expected, the protective effect evoked by BRD4 knockdown on H2O2-induced oxidative stress injury in trophoblast cells was markedly reversed via Nrf2 inhibition (Figure 7B–E). Overall, these data indicated that BRD4 knockdown had a protective effect on oxidatively stressed trophoblast cells, which functioned by enhancing Nrf2 activation.

Nrf2 inhibition blocked BRD4 knockdown-mediated anti-oxidative stress injury in trophoblast cells. HTR8/SVneo cells were transfected with BRD4 siRNA and incubated for 48 h in the presence of 2 μM ML385, followed by H2O2 exposure (500 μM) for another 24 h. (A) Nrf2 activation was monitored using a luciferase reporter assay (*p < 0.05 compared with the control group; **p < 0.01 compared with the H2O2 + NC siRNA group or the H2O2 + BRD4 siRNA + vehicle). (B) Cell proliferation was examined via a CCK-8 cell proliferation assay (**p < 0.01 compared with the control group or the H2O2 + NC siRNA group or the H2O2 + BRD4 siRNA + vehicle). (C) Cell invasion was assessed via a Transwell invasion assay (*p < 0.05 compared with the control group or the H2O2 + NC siRNA group or the H2O2 + BRD4 siRNA + vehicle). (D) Cell apoptosis was monitored using an Annexin V-FITC/PI apoptosis assay (**p < 0.01 compared with the control group or the H2O2 + NC siRNA group; *p < 0.05 compared with H2O2 + BRD4 siRNA + vehicle). (E) ROS levels were measured via a DCFH-DA probe staining assay (***p < 0.01 compared with the control group or the H2O2 + NC siRNA group or H2O2 + BRD4 siRNA + vehicle). (F) A schematic model of the cellular response to oxidative stress injury by BRD4/Keap1/Nrf2/ARE signaling in trophoblast cells.
Discussion
The current study was the first to reveal a pivotal role of BRD4 in the regulation of oxidative stress in trophoblast cells. Our data showed that BRD4 expression was elevated in response to H2O2-induced increases in oxidative stress. Functional experiments revealed that BRD4 inhibition attenuated H2O2-induced oxidative stress injury in trophoblast cells, whereas BRD4 overexpression exacerbated H2O2-induced oxidative stress injury in trophoblast cells. Moreover, we elucidated that the underlying molecular mechanism of the BRD4 inhibition-mediated protective effects was associated with the enhancement of Nrf2/ARE activation via the downregulation of Keap1 (Figure 7F). Overall, our study suggests that the BRD4/Keap1/Nrf2/ARE axis may play a key role in regulating oxidative stress injury in trophoblast cells.
Recent studies have indicated that BRD4 is a key regulator of oxidative stress in multiple cell types experiencing various pathological stimuli. Researchers have shown that knockdown of BRD4 markedly decreases the apoptosis and ROS production induced by H2O2 in prostate cancer cells and chondrocytes. 19,22 Furthermore, other reports have indicated that BRD4 inhibition attenuates ischemia/reperfusion injury-associated effects of oxidative stress in multiple organs. 21,23 BRD4 blockage alleviates hypertrophy, fibrosis, and inflammation of the heart through the suppression of ROS production. 37 Moreover, BRD4 inhibition represses high-glucose-induced apoptosis and ROS generation in podocytes, suggesting the involvement of BRD4 in diabetic nephropathy. 38 These findings indicate that BRD4 inhibition exerts considerable cytoprotective functions via the downregulation of oxidative stress.
The involvement of BRD4 in the regulation of oxidative stress in trophoblast cells has not been previously shown. However, an increasing number of studies have found that exposure to H2O2 results in decreased cellular proliferation and invasion and leads to increased levels of apoptosis and ROS production in trophoblast cells, which cause oxidative stress injury. 39 –42 In this study, we utilized H2O2-stimulated trophoblast cells to study the role of BRD4 in the regulation of oxidative stress injury in trophoblast cells. Interestingly, we found that BRD4 inhibition significantly rescued the decreased proliferative and invasive effects promoted by H2O2 exposure in trophoblast cells and attenuated H2O2-induced apoptosis and ROS production. Furthermore, BRD4 upregulation produced the opposite effect, indicating that BRD4 participates in the modulation of oxidative stress in trophoblast cells. Our study confirms that BRD4 inhibition functions to combat oxidative stress in trophoblast cells.
Notably, a recent study demonstrated that BRD4 expression was enhanced in the placentas of pregnant women with preeclampsia. 43 This study revealed that BRD4 inhibition markedly downregulated the expression of proinflammatory cytokines in inflammatory-induced trophoblast cells, suggesting a key role for BRD4 in the propagation of inflammation in the pathophysiology of preeclampsia. 43 Coincidentally, our study also suggested the involvement of BRD4 in preeclampsia. Our findings demonstrated that BRD4 contributed to the modulation of oxidative stress in trophoblast cells. Therefore, our work suggests that BRD4 may participate in the regulation of oxidative stress in preeclampsia.
It has been reported that the antioxidant function of BRD4 inhibition is related to the activation of Nrf2 signaling. 44 –46 Overexpression of BRD4 is associated with high levels of Keap1 expression in prostate cancer, and BRD4 inhibition decreases Keap1 expression, which leads to the activation of Nrf2. 19 In acute myelocytic leukemia, it has been shown that BRD4 directly regulates the transcription of Keap1. 47 Therefore, BRD4 inhibition promotes Nrf2 activation and increases the expression of anti-oxidant genes. 47 Moreover, the regulatory effect of BRD4 on the Keap1/Nrf2 axis has been confirmed in oligodendrocyte precursor cells and podocytes. 21,38 In this study, we also demonstrated that BRD4 participates in the regulation of the Keap1/Nrf2 axis in trophoblast cells. Our data showed that BRD4 inhibition decreased the expression of Keap1 and increased Nrf2 nuclear translocation, which lead to enhanced activation of Nrf2/ARE-mediated transcriptional activity. Keap1 overexpression or Nrf2 inhibition apparently reversed BRD4 inhibition-mediated protective effects against oxidative stress injury in trophoblast cells. Considering that Nrf2 activation provides beneficial effects for preeclampsia by targeting BRD4 to stimulate Nrf2 activation, it may represent an attractive approach for use in the treatment of preeclampsia.
In conclusion, the current findings demonstrate that BRD4 inhibition protects trophoblast cells from oxidative stress injury by enhancing Nrf2/ARE activation via the downregulation of Keap1. These data indicate a novel regulatory mechanism used for protection against oxidative stress by BRD4-mediated Keap1/Nrf2 signaling in trophoblast cells. The BRD4/Keap1/Nrf2 regulatory axis likely influences the oxidative stress involved in preeclampsia. However, further work using animal models will be required to confirm the regulatory role of BRD4 on oxidative stress in preeclampsia in vivo.
Footnotes
Author contributions
Yiqing Wu: designed the work, performed the experiments, and drafted the article. Yang Mi: performed the experiments. Fan Zhang: collected and interpreted the data. Yimin Cheng: collected and interpreted the data. Xiaoling Wu: designed the work, and revised the article. All authors have approved of the final version of this article for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 81702578).
