Abstract
Colistin methanesulfonate (CMS), a clinical form of colistin, is widely used as a last-line treatment for multidrug-resistant (MDR) gram-negative bacterial infections in critically ill patients presenting a considerably high mortality rate. However, nephrotoxicity is considered to be a critical adverse effect that limits CMS’s clinical use. Alpha-lipoic acid (ALA) is a strong antioxidant that is effective in preventing nephrotoxicity in many models. The aim of this study was to investigate ALA’s ability to protect against nephrotoxicity induced by colistin in rats. Male Wistar albino rats were randomly divided into four groups. Group 1 was the control group (Control; n = 6), in which isotonic saline was administered to the rats. Group 2 was the ALA group (ALA; n = 6) in which rats received 100 mg/kg ALA. Groups 3 was the CMS (CMS; n = 7) in which 450.000 IU/kg/day of CMS was administered to the rats. Groups 4 was the CMS + ALA group (n = 6), in which rats were injected with 100 mg/kg of ALA 30 min before administration of CMS. All injections were performed intraperitoneally at 1, 4, 7, and 10 days. Urine was collected by using a metabolic cage for 24 h after each administration. The rats were euthanized under ether anesthesia after 24 h of the last administration. Blood and kidney samples then were collected for histological and biochemical analysis. ALA pretreatment could reverse the effects of colistin-induced nephrotoxicity, partly through its suppressing effect on Nox4 and caspase-3, which in turn results in its antioxidant and antiapoptotic effect. Therefore, ALA may be an effective strategy for the management of colistin nephrotoxicity.
Introduction
Colistin, also called as polymyxin E, is an old antibiotic with a cationic polypeptide structure. It has bactericidal and antiendotoxicin effects against multidrug-resistant (MDR) gram-negative bacterial infections resulting from Pseudomonas aeruginosa, Klebsiella pneumoniae and Acinetobacter baumannii. 1 It has actually been commercially available since 1950 but was withdrawn in 1970s due to its high rates of nephrotoxicity. 2 In the past decade, the lack of newly discovered antibiotics have resulted in the reintroduction of colistin as a last-line treatment for MDR infections, which result in a considerably high mortality rate in critically ill patients. 3,4 Colistin is administrated parenterally as colistin methanesulfonate (CMS), which is an inactive prodrug. Then, CMS is converted to colistin. 5 Nephrotoxicity has been reported to occur in 45% to 55% of patients treated with the currently recommended CMS dosage regimens. Although the precise mechanism underlying colistin-induced nephrotoxicity is not clearly understood at present, it is reported to be associated with the total dose of CMS and the duration of treatment. 2 Thus, nephrotoxicity is considered to be a critical, dose-limiting adverse effect that affects the clinical use of CMS.
It is well established that a small fraction of the dose of colistin is excreted in the urine after being filtered out by the glomeruli. The majority of the filtered colistin is highly susceptible to tubular reabsorption, resulting in an accumulation in the tubular cells, thereby causing renal tubular damage. Moreover, previous studies demonstrate that oxidative stress played a major role in colistin-induced nephrotoxicity. 6,7 In the kidney, renal tubular cells are especially vulnerable to oxidative stress since they are rich in mitochondria, which are the main sites of intracellular reactive oxygen species (ROS) production via the respiratory chain and NADPH oxidase (Nox). 8,9 The Nox family has seven members: Nox1–5, Duox1 and Duox2. They transfer electrons from NADPH to generate ROS. Among them, Nox4 has been reported to be constitutively active in the presence of the its docking subunit, p22phox, in the mitochondria of the kidney cortex and plasma membrane. The upregulation of Nox4 was reported to play a critical role in renal oxidative stress, which resulted in kidney injury. 10 Also, it has been demonstrated that the inhibition of Nox4 genetically and pharmacologically in the experimental colistin nephrotoxicity model resulted in attenuation of kidney injury. 11 Therefore, an antioxidant that can suppress Nox4 may be able to manage oxidative kidney injuries induced by colistin.
Alpha-lipoic acid (ALA), also called thioctic acid, is a strong antioxidant that is both water and fat soluble. It has several functions including being a free radical scavenger, a metal chelator and a regenerator of endogenous antioxidants. It is also a clinically applicable drug, especially in patients with diabetic neuropathy. 12,13 No significant adverse effects have been reported with high doses of ALA administration. 14 Moreover, it was reported that ALA had a favorable action against drug-induced nephrotoxicity in the experimental models. 15 –20 Thus, our study was designed to determine whether ALA can exhibit a protective effect against colistin-induced nephrotoxicity in rats.
Materials and methods
Animals
Experimental protocol
This study was performed with the approval of the Animal Care and Use Committee of Dokuz Eylul University. Twenty-five adult, male, albino Wistar rats were used, and all animals were kept in a standard controlled environment (22 ± 1°C, 40–70% humidity with a 12 h light/dark cycle). Rats had free access to food and water during the experiment.
The pilot study was first performed to determine the appropriate colistin dose and duration since there is a considerable heterogeneity regarding the use of colistin dose and duration, which is partly due to the differences in terms of species, activity and behavior among the animals of the experiment. 7 Based on previous studies, 300.000 IU/kg/day 21,22 and 450.000 IU/kg/day of CMS 23,24 were injected intraperitoneally every day for 7 and then 10 days. Our histomorphological findings demonstrate that 450.000 IU/kg/day of CMS for 10 days led to severe kidney damage, which allowed for the protective effect of ALA to be tested. 1 mg of CMS equals 12.500 IU of CMS, and this corresponds to 0.41 mg of colistin. 7 Thus, the dosage of 36 mg/kg/day used for this study corresponds to the CMS dose rather than that of colistin.
Briefly, rats were divided randomly into four groups. Group 1 was the control group (Control; n = 6) in which isotonic saline was administered to the rats. Group 2 was the ALA group (ALA; n = 6) in which rats received 100 mg/kg of ALA (Thioctacid T flacon, MEDA Pharma, GMbH & CoKG, Hamburg, Germany). Groups 3 was the CMS group (CMS; n = 7), in which rats were injected with 450.000 IU/kg/day of CMS (Colimycin®, Koçak Farma, Istanbul, Turkey). Group 4 was the CMS + ALA group (n = 6) in which rats received 100 mg/kg of ALA 30 min before the administration of CMS. The dosage of ALA used in this study was reported to prevent marked nephrotoxicity due to other nephrotoxic drugs such as cisplatin, 15,17,18 chloroquine 16 and iron sucrose. 19 All injections were performed intraperitoneally and adjusted according to the body weight of each animal in the study. Urine was collected by metabolic cage within 24 h of each administration. Rats were euthanized under ether anesthesia 24 h after receiving their last dose. Blood samples were collected from the abdominal aorta for biochemical assessment. The kidneys were collected for histological and biochemical evaluation.
Histomorphological analyses
Hematoxylin–eosin (H-E) staining was performed on paraffin blocks of the kidney tissues. Then, tubule brush border loss, tubule atrophy, tubule dilatation, interstitial inflammation, cast formation, apoptotic and necrotic cells were evaluated by using an image analyzer (Olympus BX-51, Tokyo, Japan) to assess tubular injury and scored semi-quantitatively as follows: 0 = normal, 1 = mild, 2 = moderate and 3 = severe, respectively meaning that no tubules, 5–25% of tubules, 26–50% of tubules and more than 50% of tubules were damaged. 25
For apoptosis evaluation, active caspase-3 protein expression was evaluated immunohistochemically. After performing, deparaffinization, rehydration and blocking, all sections were subjected to an active caspase-3 antibody (1:100, AB3623, Millipore, Temecula, CA) and secondary antibody. Then, DAB staining (Roche Diagnostics, Mannheim, Germany) and Mayer hematoxylin counterstaining were performed. Finally, active caspase-3 positive staining in the sections were scored semi-quantitatively as follows: 0 = no immunoreactivity, 1 = little, 2 = moderate, and 3 = strong positive staining. 26
Biochemical analyses
The level of kidney injury molecule-1 (KIM-1) in the urine, which is considered to be a strong biomarker for tubular injury, levels were evaluated with an ELISA kit (Boster Biological Technology, Wuhan, China). The results were presented as pg/mL. Also, serum creatinine, urine microalbumin and urine creatinine levels were analyzed with an autoanalyzer (Beckman Coulter, AU-5800, Brea, CA, USA) at the Central Laboratory of Dokuz Eylul University Hospital. Data were presented as µg/dL, mg/dL, and mg/dL, respectively.
Preparation of kidney tissue homogenates for biochemical analyses
Kidney tissues were homogenized using a TissueLyser (TissueLyser II, Qiagen, UK) in a homogenate buffer, with a 50 mM potassium phosphate buffer, pH 7.8, which included 1 µg/mL of leupeptin, 1 µg/mL of phenylmethylsulfonyl fluoride (PMSF), 1 µg/mL of pepstatin, 1 µg/mL of aprotinin and 1 µg/mL of sodium orthovanadate. The homogenates were centrifuged at 2500 × g for 10 min to obtain the supernatant for malondialdehyde (MDA) analysis. Then, further centrifugation was performed at 11,000 × g for 15 min to collect the supernatants for superoxide dismutase (SOD), total oxidant status (TOS), tumor necrosis-alpha (TNF-α) and p38 MAPK protein expression analysis. For gene expression analyses, kidney tissue homogenization was performed in Trizol reagent (Invitrogen, USA).
Oxidative stress parameters in kidney tissues
Kidney MDA level
Kidney MDA levels were evaluated for lipid peroxidation by using the high-performance liquid chromatography (HPLC) method. 27 The principle of this method is based on the formation of thiobarbituric acid (TBA)-MDA complex. After injection of that MDA-TBA complex onto a column (Nucleosil 100-5, 250-4.6 mm; Macherey-Nagel Inc., Düren, Germany) of a HPLC system (Shimadzu VP Series, Japan), the fluorescence intensity was measured at 515 nm excitation and 553 nm emission. MDA levels were expressed as μmol/mg protein.
The kidney TOS levels were determined for evaluation of the total oxidant status by using a colorimetric kit (Rel Assay Kit Diagnostics, Turkey). In this method, the oxidation of the ferrous ion-chelator complex to ferric ions by oxidants that exist in the sample was monitored spectrophotometrically at 530 nm. 28 Hydrogen peroxide (H2O2) was used as a standard to construct a calibration curve. The results are expressed as µmol H2O2 equivalent/g protein.
To assess antioxidant defense, SOD-activity levels in the kidney were analyzed using the colorimetric kit (Biovision, Milpitas, USA). In this method, superoxide anion generated by xanthine oxidase reduced water-soluble tetrazolium (WST-1) salt to WST-1 formazan. Inhibition of that reaction by SOD in the sample was monitored spectrophotometrically at 450 nm. Data were presented as U/mg protein.
Kidney TNF-α level
The TNF-α level, which is a proinflammatory cytokine, in the kidney was measured with an ELISA kit (Boster Biological Technology, Wuhan, China). The results were expressed as pg/mg protein.
Quantitative real-time PCR
The total RNA in the kidney homogenate, which was prepared in Trizol reagent, was isolated from the aqueous phase described previously. 29 Then, it was converted to cDNA by a reverse transcription kit (Easy Script Plus, ABM, BC, Canada). Quantitative real-time PCR was performed with a SYBR Green PCR Master Mix (Applied Biosystems, Warrington, UK) by using the Lightcycler 1.5 Instrument (Roche Diagnostics, Germany). The primers are as follows: Nox4, 5′-GTG AAC GCC CTG AAC TTC TC-3′ (forward) and 5′-TTC TGG GAT CCT CAT TCT GG-3′ (reverse); p22phox 5′-TGT TGC AGG AGT GCT CAT CTG TCT-3′ (forward) and 5′-AGG ACA GCC CGG ACG TAG TAA TTT-3′ (reverse); KIM-1, 5′-ACT CCT GCA GAC TGG AAT GG3′ (forward) and 5′-CAA AGC TCA GAG AGC CCA TC-3′ (reverse). β-actin was used for the normalization of mRNA expressions of each gene, which were designed as 5′-TGC AGA AGG AGA TTA CTG CC-3′ (forward) and 5′-CGC AGC TCA GTA ACA GTC C-3′ (reverse). The 2−ΔΔ Ct method was used to calculate the relative expression levels of mRNAs. 30
Western blot
Kidney homogenates (80 µg protein/well) were run on 12% SDS-polyacrylamide gels as previously described. 31 Then, they were transferred to a polyvinylidene difluoride (PVDF) membrane (Millipore, MA, USA). Blocking was performed with 5% fat dry milk. The membrane was then subjected to the primary phosphorylated p38 (p-p38) MAPK antibody (Thr180/Tyr182, Cell Signaling, Danvers, MA, USA). After washing, it was incubated with a secondary antibody (Santa Cruz Biotechnology, Inc.). Visualization of the bands was carried out with ECL reagent (Millipore, Bedford, USA). Then, the same membrane was subjected to the primary p38 MAPK antibody (1:1000, Cell Signaling, Danvers, MA, USA), followed by incubation with a stripping buffer (Thermo, Rockford, IL, USA). The band densities were evaluated by densitometric analysis. The results were presented as a ratio of p-p38 to p38 MAPK.
Protein assay
Bicinchoninic acid (BCA) assay (Sigma, Germany) was used to measure protein levels in the kidney homogenate. 32
Statistical analysis
All statistical analyses were performed by using SPSS statistical software (IBM Corp., Armonk, USA). Kruskal–Wallis and Mann–Whitney U tests were used for comparing differences between multiple independent groups and two independent groups, respectively. The Spearman test was used for the correlation analysis. All values are expressed as mean ± SEM. P values less than 0.05 were considered to be statistically significant.
Results
Kidney histology
As shown in Figure 1, renal tubule injury scores indicated that CMS administration significantly increased renal tubule injury as compared to the control group (p = 0.0006). However, ALA attenuated that injury in the CMS + ALA group (p = 0.0122).

Histomorphological damage in the kidney of the experimental groups. (a) The representative images of H-E staining in the kidney tissue of the experimental groups (×20), (b) Scores of renal histomorphological damage. (⇨) and (☆) indicate red blood cell extravasation and mononuclear cell infiltration in the renal tubules, respectively. Control (n = 6), physiological saline was administrated to rats; ALA (n = 6), 100 mg/kg ALA was given intraperitoneally to rats; CMS (n = 7), 450.000 IU/kg was injected intraperitoneally; CMS + ALA (n = 6), 450.000 IU/kg was subsequently followed by 100 mg/kg ALA administration. Data are expressed as mean ± SEM. *p < 0.05 vs control; **p < 0.05 vs CMS.
Urine and serum renal function parameters
Urine KIM-1 levels were found to be significantly increased in the CMS group on days 1, 4 and 7 (1656.17 ± 250.67, 1356.96 ± 244.54 and 500.10 ± 136.34 pg/mL, respectively) as compared to the control group (277.17 ± 52.27, 222.61 ± 76.50 and 182.21 ± 17.57 34 pg/mL, respectively) (p = 0.003, p = 0.004 and p = 0.003, respectively). However, ALA pretreatment resulted in a significant reduction in urine KIM-1 levels in the CMS + ALA group on days 1 and 4 (237.32 ± 65.89 and 598.9 ± 221.76, respectively) (p = 0.003 and p = 0.006, respectively) (Figure 2(a)). On the other hand, there were no significant differences in the ratio of urine microalbumin to creatinine (Figure (2b)) nor in serum creatinine levels at the end of the experiment, on day 10 (Figure (2c)).

Urine and serum renal function parameters of the experimental groups. (a) Urine KIM-1 level, (b) the ratio of urine microalbumin to urine creatinine, (c) serum creatinine level. Control (n = 6), physiological saline was administrated to rats; ALA (n = 6), 100 mg/kg ALA was given intraperitoneally to rats; CMS (n = 7), 450.000 IU/kg was injected intraperitoneally; CMS + ALA (n = 6), 450.000 IU/kg was subsequently followed by 100 mg/kg ALA administration. Data are expressed as mean ± SEM. *p < 0.05 vs control; **p < 0.05 vs CMS.
Oxidant-antioxidant status in the kidney
The MDA level of the CMS group was increased significantly as compared to the control group (0.503 ± 0. 067 vs 0.866 ± 0.033 μmol/mg protein) (p = 0.023). There was a decrease in the level of MDA in the CMS + ALA group (0.795 ± 0.173 μmol/mg protein); however, the difference did not reach statistical significance (p > 0.05) (Figure 3(a)). Additionally, TOS levels in the kidney were found to be significantly higher in the CMS group (1.263 ± 0.094 µmol H2O2 equivalent/g protein) as compared to the control group (0.976 ± 0.077 µmol H2O2 equivalent/g protein) (p = 0.030). However, there was no significant difference between the CMS + ALA group (1.305 ± 0.150 µmol H2O2 equivalent/g protein) and the CMS group (p > 0.05) (Figure 3(b)). On the other hand, there were differences in the SOD-activity level, but these differences were not significant (p > 0.05) (Figure 3(c)).
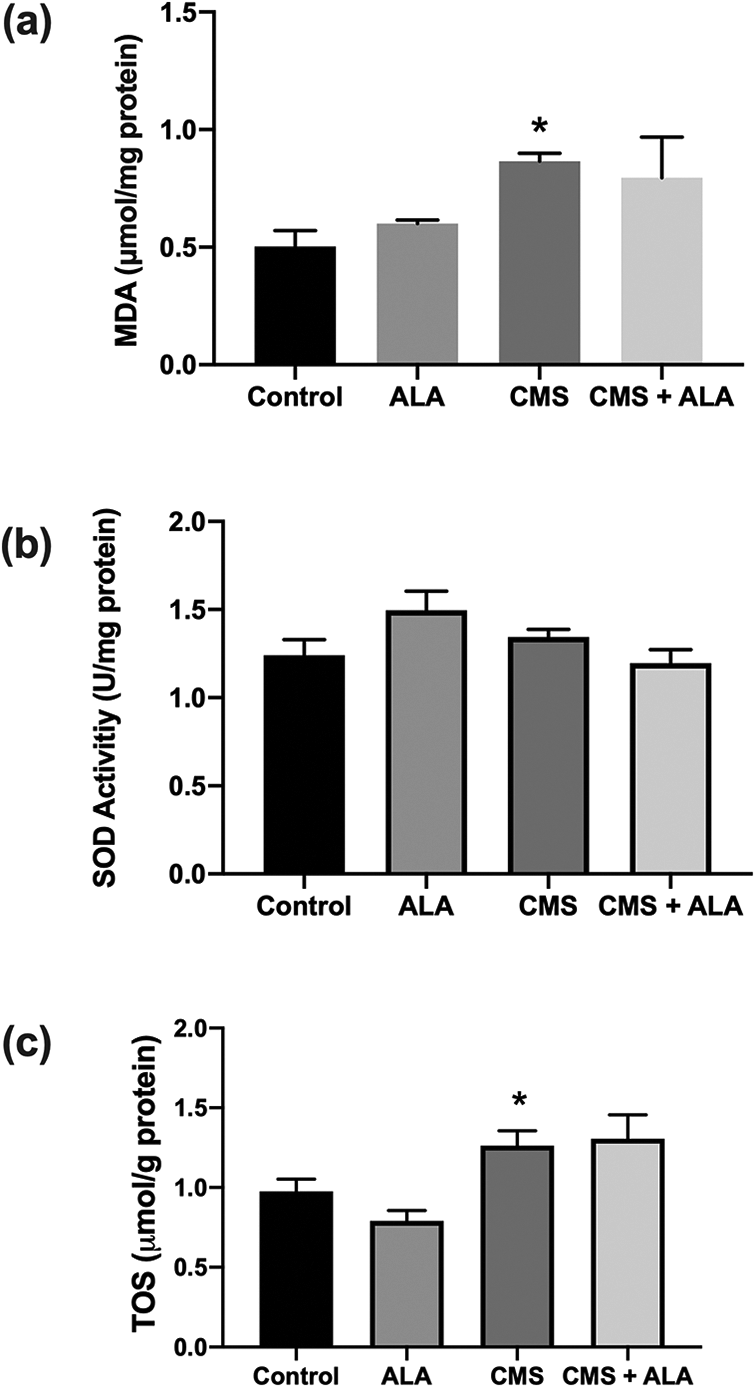
The oxidant-antioxidant status in the kidney of the experimental groups. (a) MDA level, (b) TOS level, (c) SOD-activity level. Control (n = 6), physiological saline was administrated to rats; ALA (n = 6), 100 mg/kg ALA was given intraperitoneally to rats; CMS (n = 7), 450.000 IU/kg was injected intraperitoneally; CMS + ALA (n = 6), 450.000 IU/kg was subsequently followed by 100 mg/kg ALA administration. Data are expressed as mean ± SEM. *p < 0.05 vs control.
mRNA expressions of Nox4 and p22phox in the kidney
The mRNA expressions of Nox4 and p22phox, which are the subunits of Nox, were analyzed to assess the antioxidant effect of ALA on the Nox, which is a major enzymatic reactive oxygen species (ROS) source. Compared with the control, Nox4 and p22phox mRNA expressions in the CMS group were found to have increased significantly by 2.94 and 19.88-fold (p = 0.006 and p = 0.004; respectively). However, ALA significantly suppressed mRNA expressions by 1.40 and 5.40-fold (p = 0.008 and p = 0.015, respectively) (Figures 4(a) and 4(b), respectively).

mRNA expressions of the subunits of NADPH oxidase in the kidney of the experimental groups. (a) Nox4 mRNA expression, (b) p22phox mRNA expression. Control (n = 6), physiological saline was administrated to rats; ALA (n = 6), 100 mg/kg ALA was given intraperitoneally to rats; CMS (n = 7), 450.000 IU/kg was injected intraperitoneally; CMS + ALA (n = 6), 450.000 IU/kg was subsequently followed by 100 mg/kg ALA administration. Data are expressed as mean ± SEM. *p < 0.05 vs control; **p < 0.05 vs CMS.
Kidney TNF-α level
As demonstrated in Figure 5, the TNF-α levels in the CMS group increased significantly (602.96 ± 36.94 pg/mg protein) as compared to the control group (409.57
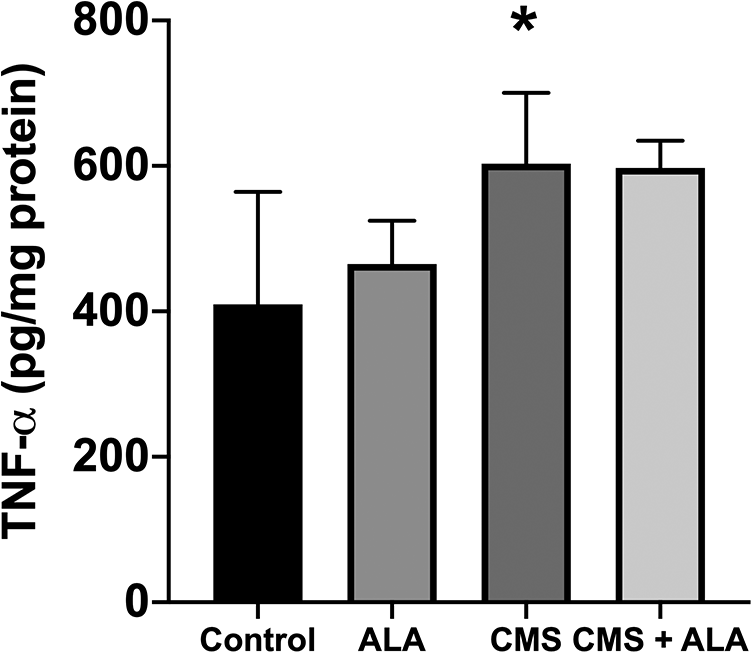
TNF-α level in the kidney of the experimental groups. Control (n = 6), physiological saline was administrated to rats; ALA (n = 6), 100 mg/kg ALA was given intraperitoneally to rats; CMS (n = 7), 450.000 IU/kg was injected intraperitoneally; CMS + ALA (n = 6), 450.000 IU/kg was subsequently followed by 100 mg/kg ALA administration. Data are expressed as mean ± SEM. *p < 0.05 vs control.
Kidney active caspase-3 protein expression
Active caspase-3 positive cells, which is a marker for apoptosis, increased significantly in the kidney tissue of the CMS group as compared to the control (p < 0.001). However, ALA significantly reduced the active caspase-3 positive cells (p = 0.002) (Figures 6 (a) and 6(b)). Additionally, active caspase-3 positive cells were correlated positively with both Nox4 and p22phox (r = 0.59, p = 0.003).

Active caspase-3 protein expression in the kidney of the experimental groups. (a) The representative images of the active immunoreactivity. Arrows (⇨) indicate anti-active caspase-3 immun-positive cells, (b) the scores of active caspase-3 immun-positive cells in the kidneys of experimental groups. Control (n = 6), physiological saline was administrated to rats; ALA (n = 6), 100 mg/kg ALA was given intraperitoneally to rats; CMS (n = 7), 450.000 IU/kg was injected intraperitoneally; CMS + ALA (n = 6), 450.000 IU/kg was subsequently followed by 100 mg/kg ALA administration. Data are expressed as mean ± SEM. *p < 0.05 vs control; **p < 0.05 vs CMS.
mRNA expression of KIM-1 in the kidney
KIM-1 mRNA expression in the kidney was found to have significantly increased by 3.78-fold in the CMS group (p = 0.0317). However, ALA significantly downregulated its mRNA expression by 0.95-fold (p = 0.015) (Figure 7). Additionally, KIM-1 mRNA expression was positively correlated with Nox4 (r = 0.59, p < 0.001).
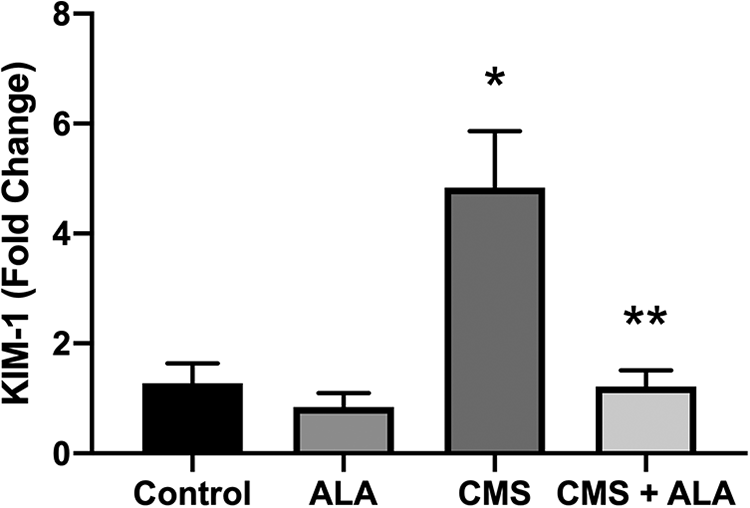
KIM-1 mRNA expression in the kidney of the experimental groups. Control (n = 6), physiological saline was administrated to rats; ALA (n = 6), 100 mg/kg ALA was given intraperitoneally to rats; CMS (n = 7), 450.000 IU/kg was injected intraperitoneally; CMS + ALA (n = 6), 450.000 IU/kg was subsequently followed by 100 mg/kg ALA administration. Data are expressed as mean ± SEM. *p < 0.05 vs control; **p < 0.05 vs CMS.
p38 MAPK signaling
As shown in Figure 8, the activation of p38 MAPK, which was manifested in its phosphorylated form, significantly induced in the CMS group as compared to the control (p = 0.002). Although, there was a decrease in the protein expression of the p-p38 MAPK in the CMS + ALA group, the difference did not reach statistical significance (p > 0.05).
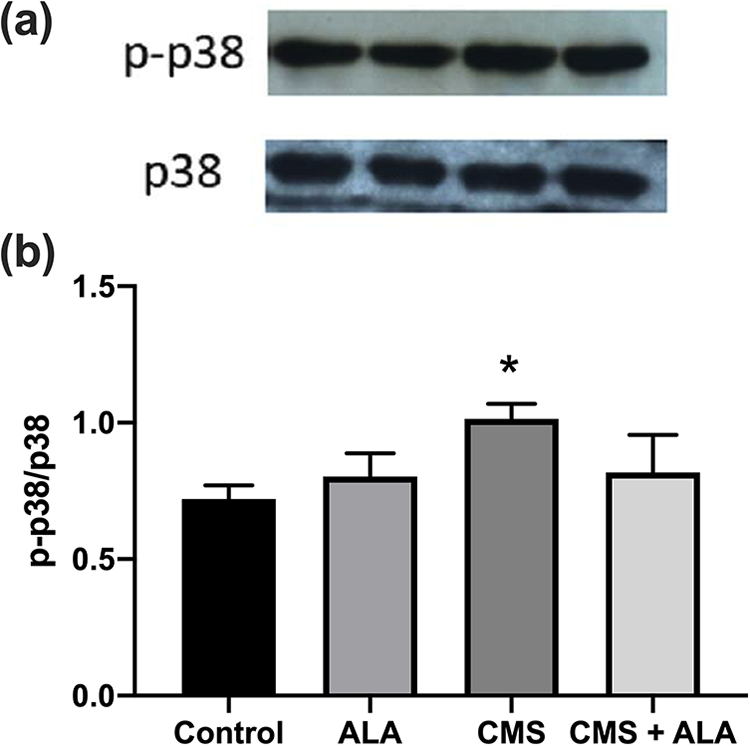
Protein expressions of p38 MAPK signaling pathway in the kidneys of the experimental groups. (a) Representative Western blot image, (b) densitometric analysis. Control (n = 6), physiological saline was administrated to rats; ALA (n = 6), 100 mg/kg ALA was given intraperitoneally to rats; CMS (n = 7), 450.000 IU/kg was injected intraperitoneally; CMS + ALA (n = 6), 450.000 IU/kg was subsequently followed by 100 mg/kg ALA administration. Data are expressed as mean ± SEM. *p < 0.05 vs control.
Discussion
In this study, we demonstrate that ALA’s have a protective effect against colistin-induced nephrotoxicity in a rat model for the first time. Its renoprotective effect was demonstrated by the reduced oxidative stress and renal tubular apoptosis, which was at least partially associated with the suppression of Nox4 and active caspase-3 during colistin treatment.
In this study, the administration of CMS, which is a clinical form of colistin, led to the significant impairment of morphological changes in the kidneys of rats, including loss of brush border and tubul dilatation as reported in previous studies. 23,24 However, the administration of ALA at a dose of 100 mg/kg/day followed by CMS resulted in a significant improvement in renal tubule architecture as compared with the group treated only with CMS. Under consideration of the safety profile of ALA 14 and its ability to protect from marked nephrotoxicity caused by other nephrotoxic drugs, 15 –19 ALA was used only at a dosage of 100 mg/kg/day. Additionally, the improved histological findings by ALA in this study were also confirmed by monitoring the urinary excretion of KIM-1. In fact, KIM-1 is a phosphatidyl serine receptor expressing physiologically at very low levels on the apical surface of proximal tubular cells. However, it is overexpressed upon acute tubular injury and plays a function in the phagocytosis of apoptotic and necrotic bodies that prevent intratubular obstruction. 33 After its disintegration by metallopeptidase, the ectodomain of KIM-1 is released into the urine. Urinary KIM-1 has been proven to be specific to proximal damage because it exhibits superior accuracy and sensitivity to other renal function parameters, such as serum creatinine, which has some limitations depending on age, body mass, sex and nutrition. 6 In this context, the urine KIM-1 levels were monitored on days 1, 4, 7 and 10 in this study. The significant increase in the urine KIM-1 level was detected on the first day at the highest level, and it remained significantly high until days 4 and 7. However, ALA administration resulted in a significant reduction of the urine KIM-1 level on days 1 and 4. In contrast, the ratio of urine microalbumin to urine creatinine excretion and serum creatinine did not exhibit significant changes, which were consistent with previous studies. 23,24 All of these findings emphasized that KIM-1 was a more reliable biomarker than serum creatinine. Moreover, the higher kidney KIM-1 mRNA expression differentiated significantly in the CMS-treated group from either the control or ALA administrated CMS groups. However, a dose-dependent study of ALA, which determines the appropriate dose and its impact on the tubular transport mechanisms of colistin will provide precise information for its renoprotective action against colistin-induced nephrotoxicity.
As previously mentioned, increased ROS is considered to be an important mechanism in colistin-induced nephrotoxicity, which can lead to cell membrane damage and cellular apoptosis. 6 In this context, we compared the extent of oxidative stress among the groups that were previously first analyzed, and it was found that there were significant increases in the levels of MDA and SOD activity within the CMS-treated group as compared to the control groups, which were in keeping with previous studies. 23,24,34 –39 However, ALA administration did not significantly affect their levels. These findings were also parallel to the TOS level, which was analyzed to evaluate the total oxidant status, including other species such as reactive nitrogen species and ROS. In respect to these parameters, conflicting findings also exist in the literature, which report no significant changes in the MDA, 40 TOS levels, 41 and SOD-activity levels of CMS-treated groups as compared to the control group. 21,22 Also, it should be remembered that the CMS dose used and the duration of those studies were different than those used in this study, which was also reported that there was a heterogeneity among the experimental protocols of colistin nephrotoxicity. 7 Furthermore, ALA’s effect on oxidative stress and the antioxidant system should not be restricted to MDA and SOD parameters. Therefore, oxidant-antioxidant status requires additional evaluation with other oxidative stress parameters that are more sensitive than MDA, such as protein carbonyl and conjugated dienes, and other antioxidants, such as glutathione peroxidase, catalase and GSH.
Moreover, Nox4 is known as a major enzymatic source for generations of ROS, which has been shown to be related to development of many pathological conditions in the kidney. 10 Also, it has been revealed that Nox4 activation was responsible for colistin-induced nephrotoxicity. 11 To date, there is no information on the effect of ALA on the regulation of Nox4 in the treatment of a colistin-induced kidney injury. This study clearly demonstrated that ALA markedly decreased the Nox4 and p22phox mRNA expressions, which are the subunits of NADPH oxidase required for its constitutive activation. This finding provided another confirmation of ALA’s antioxidant action in addition to its known actions, indicating that ALA could directly suppress the expression of Nox4 on the gene level. Therefore, the insignificance effect of ALA on increased MDA and TOS levels seems partially related to ROS that is most likely generated by other oxidases, such as xanthine oxidase and mitochondrial enzymes, except for Nox4 in the kidney. Additionally, it has been reported that Nox4 inhibition directly reduced colistin-induced activation of caspase-3, which is a key mediator for tubular apoptosis. 11 In this regard, we also determined that activation of Nox4 and caspase-3 were significantly together with increased KIM-1 expression during colistin treatment, thus suggesting that there might be relationship among them. We also demonstrate for the first time that ALA markedly reduces active caspase-3 protein expression during CMS treatment, which also reflects to improved histopathological findings and decreased KIM-1 in the ALA administrated of CMS group. These findings also serve as an in vivo evaluation of the hypothesis that ALA has a renoprotective effect against colistin-induced injury, confirming the strong connection between oxidative stress and renal tubular apoptosis in colistin-induced nephrotoxicity, which was also demonstrated by previous studies. 11,34 –37
Furthermore, p38 MAPK signaling is well established as a major pathway vulnerable to increased ROS production. 42,43 It has also been reported that p38 MAPK can mediate renal cell apoptosis and inflammation in both colistin-induced kidney injury 11,34 and other drug-induced kidney injuries such as those resulting from gentamycin, 44 vancomycin 45 and iron sucrose use. 19 However, the impact of ALA on p38 MAPK signaling during colistin administration has not yet been investigated. In this study, p38 MAPK activation increased significantly after CMS administration, which reflects p-p38 MAPK protein expression, which was in accordance with previous studies. 11,34 However, a significant effect of ALA on the activation of p38 MAPK was not detected. In contrast to this finding, ALA has been shown to affect p38 MAPK activation during iron-sucrose-induced kidney injury, which was another drug-induced kidney injury experimental model. 19 Additionally, this study determined that ALA had no effect on the increased mRNA expression of TNF-α as induced by colistin. TNF-α is known as the most important proinflammatory cytokine produced in the proximal tubular epithelial cells, and it is regulated by p38 MAPK signaling. In fact, there is a link between oxidative stress and inflammation, which can induce one another. 46 Furthermore, this study shows that oxidative stress caused by colistin might play an important role in the induction of the mRNA expression of TNF-α via activation of the inflammatory response in accordance with previous studies. 37 –39 On the other hand, the insignificant effect of ALA on TNF-α levels induced by colistin was similar to p38 MAPK signaling finding. It might be supposed that those findings were complementary to each other, which indicate an ineffective role of ALA on inflammation during colistin administration. However, there are several studies demonstrating the anti-inflammatory role of ALA in drug-induced kidney injury models, such as caused by cisplatin 17 and iron sucrose. 19 Taken collectively, our findings indicate that other related signaling pathways may play a part in the renoprotective role of ALA. Thus, further studies are needed to clarify these mechanisms.
In conclusion, this study first demonstrates that ALA pretreatment could attenuate colistin-induced nephrotoxicity in rats, despite the lack of its effective role on inflammation. This seems to be at least partially related to its suppressant effect on Nox4 and active caspase-3, which in turn result in its antioxidant and antiapoptotic effect. The precise mechanisms of the renoprotective effect of ALA, however, require further study. Nevertheless, ALA may be an effective strategy for the management of nephrotoxicity and reducing its related mortality in patients treated with colistin.
Footnotes
Author’s note
Ayse Kocak is now affiliated with Kütahya Health Science University, Tavşanlı Vocational School of Health Services, Kütahya.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported financially by Dokuz Eylül University Research Project Administration and carried out by Dokuz Eylül University Medical School Research Laboratory (R-LAB).
