Abstract
Despite wide application of sodium nitrite (SN) as food additive, it exhibits considerable side effects on various body organs at high dose or chronic exposure. The aim of this study was to test whether Glycyrrhizic acid (GA) could ameliorate SN-induced toxicity in lung and submandibular salivary gland (SMG). A sample size of 30 adult male albino rats was randomly allocated into 3 groups. Group 1 served as control group. Rats were treated orally with 80 mg/kg of SN in group 2 or SN preceded by (15 mg/kg) GA in group 3. Lung & SMG tissues were used for oxidative stress assessment, examination of histopathological changes, fibrosis (MTC, TGF-β and α-SMA) and inflammation (TNF-α, IL-1β and CD-68). Concurrent administration of GA ameliorated pulmonary and salivary SN-induced toxicity via restoring the antioxidant defense mechanisms with reduction of MDA levels. GA reduced the key regulators of fibrosis TGF-β and α-SMA and collagen deposition. In addition to reduction of inflammatory cytokine (TNF-α, IL-1β) and macrophages recruitments, GA amended both pulmonary and salivary morphological changes. The present study proposed GA as a promising natural herb with antioxidant, anti-inflammatory and antifibrotic effects against pulmonary and salivary SN-induced toxicity.
Introduction
Sodium nitrite (SN) is an inorganic salt widely used in food industry to give processed meat their characteristic flavor and color, to prevent their spoilage and to delay oxidative rancidity. 1 Apart from the benefits offered by SN, it was reported to exhibit organ toxicity in animals and human at high dose or chronic exposure. 2,3 SN reacts with hemoglobin to produce methaemoglobin with compromised oxygen carrying capacity. Consequently, acute exposure to SN in fatal methemoglobinemia. 4 Single acute exposure to SN resulted in intestinal damage in rats. 1 On chronic exposure, SN is accompanied by mutagenicity, carcinogenicity, endocrine disturbance, nervous system damage and respiratory dysfunction. By reaction with diet’s amines and amides, SN is converted to nitrosamines and nitrosamides, respectively which trigger oxidative stress, inflammation, and tumorigenesis. 5 Furthermore, these harmful chemical products can be formed from SN under acidic medium in stomach or under high temperature during charring or overcooking meat. In vitro 6 and in vivo 7 studies reported production of highly carcinogenic N-nitrosocompounds from reaction of nitrite with secondary amines and N-alkyl amides at low pH. Currently, exposure to and/or consumption of SN beyond the non-toxic recommended daily intake is alarmingly increasing, especially in high-risk categories, as infants and children. This is attributed to industrial growth, improper agricultural practices, and uncontrolled consumption of processed food. 8 Therefore, development of therapeutic strategies to halt SN intoxication is of great demand.
Recently, therapeutic application of natural dietary compounds to forestall unfavorable signaling mechanisms triggered by toxins in various body organs is widely used trend in clinical medicine. Glycyrrhizic acid (GA) is the chief constituent of licorice root Glycyrrhiza glabra. It is a triterpenoid saponin traditionally used for treatment of jaundice, bronchitis, and gastritis. Previous studies reported multiple beneficial pharmacological effects for GA including anti-inflammatory, immunity boosting, anti-tumor and anti-fibrotic activities, anti-cariogenic and even antiviral against SARS−Coronavirus. 9 –11 We previously reported protective effect of thymoquinone against SN-induced nephrotoxicity. 3 Among various organs adversely affected by SN, Kadry and Ali 12 and Al-Rasheed et al. 13 reported severe pulmonary injury in SN intoxicated rats. However, whether SN possesses toxic effect on submandibular salivary gland (SMG) has not previously reported.
Intoxicated SMG would directly affect general wellbeing as result of xerostomia, possible rampant caries and decreased immunity of oral cavity. Recently, an expanding prospective of pulmonary salivary gland-type tumors as well as metastasis of certain pulmonary carcinoma into SMG has been linking SMG to precancerous and neoplastic changes in lungs. 14,15 Moreover, both lung and SMG are lately proposed responsible for transmission and replication of Andes Hantavirus (ANDV) that caused 40% fatality rate in human. 16 Herein, we aimed to evaluate the role of oxidative stress, inflammation, and fibrosis in the toxic effect of SN on lung and SMG and to highlight the potential protective role of GA, as an attempt to ameliorate the side effects of inevitable SN exposure on such important organs.
Material and methods
Experimental design
Thirty male Sprague Dawley rats weighing 100–150 g were housed in specific plastic cages with wire mesh lids. All rats were pre-acclimatized to laboratory conditions for one-week pre-experimentation. Rats were kept in regular light cycle of 12/12 h, with 50% relative humidity and free access to food and water. All study procedures were approved by ethical committee of Faculty of Medicine Mansoura University (No: R.20.07.938) and complied to the ARRIVE guidelines of Animal Research. All rats were randomly and equally allocated using the Research Randomizer software (https://www.randomizer.org/) into 3 groups.
Euthanasia and sample collection
All rats were anesthetized then euthanized by overdose of halothane. 20 Both SMG and lung specimens were collected and processed for further analysis. The sublingual gland was separated from submandibular-sublingual gland complex before processing.
Determination of levels of malondialdehyde (MDA), glutathione (GSH) and total antioxidant capacity (TAC) in lung and SMG tissue
The left halves of lung and SMG were washed with ice cold phosphate-buffered saline (pH = 7.4), then butylated hydroxytoluene and proteolysis inhibitor were added to prevent the proteolysis and oxidation of the samples. After homogenization and centrifugation, total protein content in the supernatants was measured. To evaluate endogenous intoxication of lung and SMG tissues, levels of malondialdehyde (MDA) as a pro-oxidant parameter was determined with the MDA Assay Kit (Biodiagnostic Company, Cairo, Egypt) according to manufacturer’s instructions. Basically, following the addition of thiobarbituric acid, absorbance of formed chromophore was measured at 532 nm. Reduced glutathione (GSH), a cysteine carrier, detoxifies lipid peroxidation products and carcinogens. As a major intracellular antioxidant, levels of GSH were measured (Biodiagnostic Company, Cairo, Egypt) spectrophotometrically to detect a yellow anion that resulted from reduction of 5,5/-dithiobis-(2 nitrobenzoic acid) (DTNB) by GSH at 412 nm. 3 Further, to assess the oxidative stress intensity of lung and SMG against SN induced toxicity, TAC (Biodiagnostic Company, Cairo, Egypt) was assayed reflecting the efficiency of enzymatic and non-enzymatic antioxidant defense mechanisms.
Measurement of pro-inflammatory and fibrotic markers in lung and SMG tissue
Based on enzyme-linked immuno-sorbent assay (ELISA), the levels of biochemical parameters in both lung and SMG homogenates were measured using rat transforming growth factor-beta (TGF-β), Interleukin 1-beta (IL-1β) and Tumor necrosis factor-alpha (TNF-α) specific ELISA kits (eBioscience, Inc., San Diego, CA, USA) according manufacturer’s instructions.
Histological, special and immunohistochemical (IHC) stains
The right halves of specimens were fixed, processed into 5 µm paraffin sections, and stained with Hematoxylin and Eosin (H&E), 21 Masson’s Trichrome (MTC) stain to detect collagen fibers, and Periodic Acid Schiff (PAS) to evaluate the presence and alteration of polysaccharides in the acinar cells, basement membrane and goblet cells. 22 For immunostaining, the sections were deparaffinized, blocked with 5% serum for 30 minutes at 20°C and processed for antigen retrieval by heat mediation in a citrate buffer at 20°C for 45 minutes. Then, sections were incubated for 24 hours with primary CD-68 antibody, (ab125212, 1:100) or α-smooth muscle actin (CBL171, Millipore, USA; 1:100) using appropriate secondary antibodies. Negative controls were stained by substituting the primary antibodies by phosphate buffered salin. 23
Evaluation and scoring of special & IHC staining
Assessment was performed by double-blinded calibrated examiners to measure the number of positively stained CD68 cells and to threshold the percentage of positive immunoreactive area of α-SMA and MTC to the total area of each section. An intraclass correlation coefficient was conducted to assess reliability of scores (supplementary file). The score was calculated as follow: 0 → 100% negative expression, and 1 → (>0%–<10%) very mild expression, 2 → (>10%–<30%) mild expression, 3→ (>30%–<50%) moderate and 4 → (>50%) intense positive expression.
Statistical analysis
Statistical package for social sciences (SPSS) (Version 25; Inc, Chicago, IL, USA) was used. Qualitative data was tested for normality using Shapiro-Wilk test. Parametric data were tested by One Way ANOVA (analysis of variance) followed by Tuckey post-hoc test for multiple comparisons. Kruskal–Wallis test followed by post-hoc Dunn’s were used for comparing non-parametric data. P value <0.05 was considered statistically significant. The statistical tests were based on a type 1 error value of 5% (α = 0.05) and on a power of 0.85 sample size, using G*Power 3.1.9.4 program.
Results
Effect of GA administration on SN-induced oxidative stress
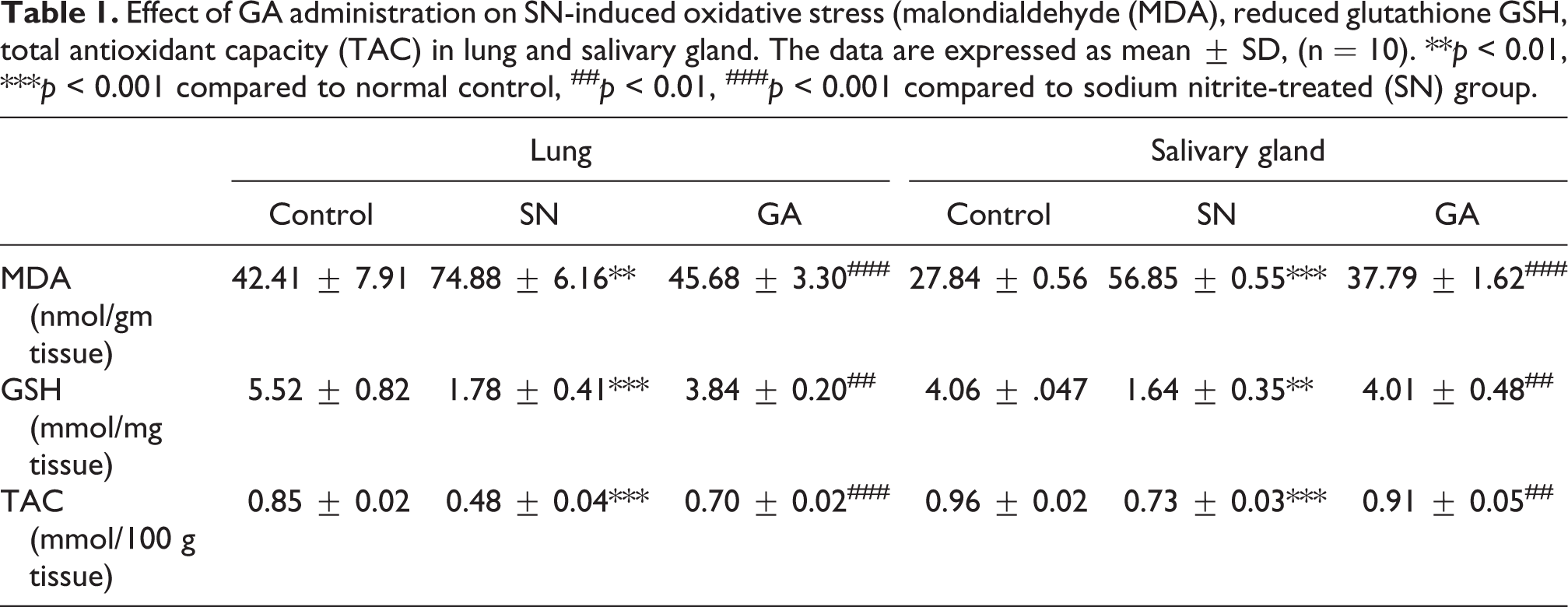
Pulmonary homogenates from SN group showed a significant higher MDA level (P = 0.003), a significant lower GSH (P < 0.001) and TAC levels (P < 0.001) compared to control group. GA administration significantly decreased MDA levels (p = 0.006) and restored GSH (P = 0.0016) and TAC levels (P = 0.0004) compared to SN group. Salivary intoxicated SN samples showed significantly higher MDA levels (P < 0.001) and diminished GSH and TAC (P < 0.01, P < 0.001) compared to control group. GA treatment restored levels of both GSH, and TAC compared to SN group (P = 0.0024, 0.0032) respectively and reduced MDA levels compared with SN treated samples (P < 0.001) (Table 1).
Effect of GA administration on SN-induced oxidative stress (malondialdehyde (MDA), reduced glutathione GSH, total antioxidant capacity (TAC) in lung and salivary gland. The data are expressed as mean ± SD, (n = 10). **p < 0.01, ***p < 0.001 compared to normal control, ##p < 0.01, ###p < 0.001 compared to sodium nitrite-treated (SN) group.
Effect of GA administration on SN-induced inflammation
SN treatment markedly increased TNF-α and IL-1β levels in both lung and SMG homogenates compared to control group (p < 0.001). However, GA administration suppressed SN-induced TNF-α and IL-1β up-regulation in both pulmonary (p = 0.032, p < 0.001) and SMG (p = 0.011, p < 0.001) homogenates, respectively (Table 2).
Effect of GA administration on SN-induced inflammatory and fibrotic cytokines (tumor necrosis factor-α (TNF-α), tumor growth factor-β (TGB-β), interleukin-1β (IL-1 β). The data are expressed as median and interquartile range (IQR) for TNF-α and TGF-β and as mean ± SD for IL-1β, (n = 10). ***p < 0.001 compared to normal control, ###p < 0.001 compared to sodium nitrite- treated (SN) group.

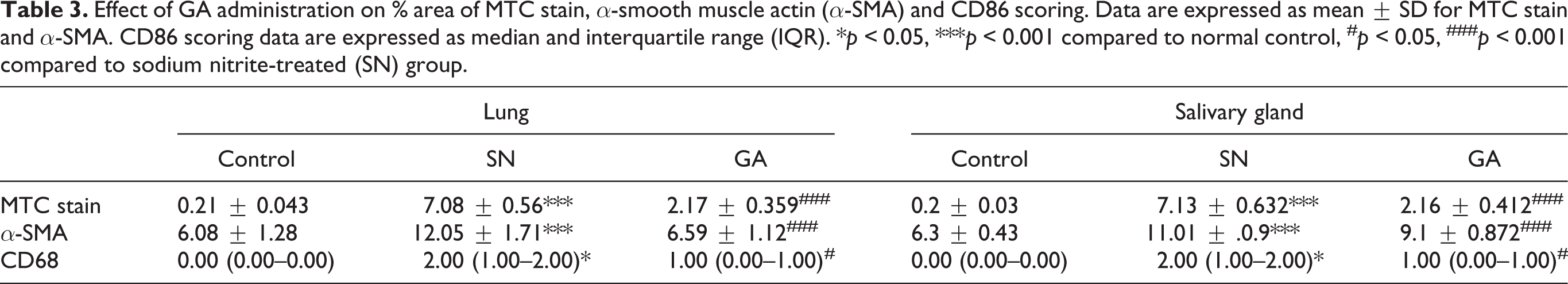
Further, testing CD68 as a marker of macrophage infiltration showed increased bright brown positive IHC staining, reflecting macrophage infiltration into peribronchial, perivascular and interstitial tissue in pulmonary sections from SN treated group. Nevertheless, macrophages were markedly decreased in pulmonary sections from GA group (Table 3, Figure 1A). Similarly, SMG sections from SN group showed positive reaction especially around the highly vacuolated ducts and morphologically distorted acini, whereas GA treated group showed less brown stained cells (Table 3, Figure 1B).
Effect of GA administration on % area of MTC stain, α-smooth muscle actin (α-SMA) and CD86 scoring. Data are expressed as mean ± SD for MTC stain and α-SMA. CD86 scoring data are expressed as median and interquartile range (IQR). *p < 0.05, ***p < 0.001 compared to normal control, #p < 0.05, ###p < 0.001 compared to sodium nitrite-treated (SN) group.

Effect of GA administration on SN-induced inflammation. A) Microscopic pictures of CD68 immunostained lung sections showing, negative staining in control group. In contrast, lung of SN group showed increased expression of bright brown positively stained macrophages infiltrating into peribronchial, perivascular and interstitial tissue (arrows). While in GA group the numbers of infiltrating macrophages were markedly decreased (arrows). (X200, bar 50). B) Microscopic picture of CD68 immunostained SMG sections. Control group showed very minimal reaction interstitially (arrow), while the SN group showed positive reaction especially around the highly vacuolated ducts (arrow) and even intra-acinar in the morphologically distorted acini (arrow head), while GA group showed a lesser number of positively stained macrophages (arrow). (X400, bar 25).

Effect of GA administration on SN-induced fibrosis
Our results revealed increased TGF-β levels in lung and SMG homogenate from SN treated group compared to control group (p < 0.001). In contrast, significant decrease in TGF-β levels was observed in pulmonary and SMG homogenate in GA treated group (p = 0.011) compared to SN treated group (Table 2).
Additionally, pulmonary sections from SN group showed increased bright brown α-SMA positive staining in peribronchial, perivascular and interstitial tissue associated with areas of lesion. However, the immunostaining of α-SMA was markedly decreased in GA group compared to SN group (Table 3, Figure 2A). Likewise, normal sections of SMG showed positive α-SMA reaction, where it binds to microfilament bundles in the myoepithelial cells of the acinar cells and intralobular ducts. It represents the degree of myoepithelial cell proliferation, in the form of brown deposits in their cytoplasm. Positive expression of α-SMA increased in SN treated group with increased number of processes around the ducts and acini. GA group showed lesser degree of α-SMA reaction (Table 3, Figure 2B).

Effect of GA administration on SN-induced fibrosis. A) Microscopic pictures of α-SMA immunostained lung sections showing normal bright brown positive staining in wall of blood vessels, bronchi, and bronchioles (blue arrows) in Control group. In contrast, SN group lung sections showed increased expression of α-SMA in peribronchial, perivascular and interstitial tissue (yellow arrows) associated with areas of lesion. Amount of α-SMA positive staining in peribronchial, perivascular and interstitial tissue (yellow arrows) was markedly decreased in GA group. (X100, bar 100). B) Microscopic picture of SMG sections stained with α-SMA showing normal brown staining of myoepithelial cells mainly around the acini and interlobular the ducts in Control group. In contrast there was an increase in positive expression of α-SMA with increased number of processes around the ducts (arrow) and acini (arrow heads) in SN group. While there was a decreased α-SMA staining (arrow) in GA group. (X400, bar 25).
Histopathological changes
The control group of both lung (Figure 3A) and SMG (Figure 3B) showed almost normal morphology indicating proper experimental conditions. In SN group, pulmonary sections showed peribronchial lymphoid hyperplasia and degeneration in alveolar epithelium, while in SMG, the duct system seemed to be more affected by SN toxicity than the acini. Loss of acinar structures, granular convoluted duct changes were also observed. Moreover, widening and thickening of interstitial spaces and tissues were marked in both lung and SMG sections. Thickening and fibrosis were much confirmed with the MTC stain results in both lung (Table 3, Figure 4A) and SMG sections (Table 3, Figure 4B) from SN group. Fibrous deposition in both lung (Table 3, Figure 4A

A) Microscopic pictures of H&E stained lung sections showing normal alveoli, bronchioles, interstitial tissue, and blood vessels in Control group. In contrast, lung of SN group showed perivascular leukocytic cells aggregation (black arrows), peribronchial lymphoid hyperplasia (green arrow) and degeneration in alveolar epithelium (blue arrows). Marked improvements were noticed in GA group including mild thickening of interstitial tissue (yellow arrows), mild congestion and few perivascular leukocytic cells aggregation. (X100, bar 100 and X400, bar 50). B) Microscopic picture of H&E stained SMG sections. Normal group showed acini with narrow lumen bordering pyramidal cells with and basal nuclei (arrows), interlobular ducts with tall columnar cells and basal nuclei (arrow heads). SN group showed loss of normal duct structures (arrows), thickening of interlobular duct wall with collapse of the lumen (curved arrows) with hyalinized homogenous areas (arrow heads), nuclear changes (n) in both duct and acini and congested blood vessels (*). SN group also showed loss of parenchymal tissue and wide interstitial spaces (arrows). While GA group revealed considerable restoration of normal glandular architecture and regeneration of parenchymal including GCD and connective tissue components. Some ductal (arrows) and nuclear (n) changes were also observed. GCT; granular convoluted tubules, A; acini. (X400, bar 25).


A) Microscopic pictures of MTC stained lung sections showing no fibrous tissue deposition in interstitial tissue in Control group. In contrast, lung of SN group showed blue stained fibrous tissue deposition peribronchial, perivascular and in interstitial tissue associated with areas of lesion (arrows). Fibrous deposition was minimal to absent peribronchial, perivascular and in interstitial tissue (arrows) in GA group. (X100, bar 100 and X400, bar 50). B) Microscopic picture of rat SMG stained with MTC showing negative staining except for some fibers surrounding the ducts in Control group. In contrast there was a thickened periductal fibrous connective tissue (arrow) and fibrous thickening of the wall of congested blood vessels (arrow heads) in addition to some interacinar fibrous deposition SN group, while there was a decrease in amount and thickness of fibrous tissues (arrow) in GA group. (X100, bar 100 and X400, bar 25).

PAS stain revealed normal lung structure, continuous epithelial lining of bronchioles with no goblet cells in normal control group. In contrast, marked increase in number of goblet cells in epithelial lining of bronchioles with deposition of pink stained mucus admixed with leukocytes in their lumens were observed in SN treated group. Goblet cells were markedly decreased to absent in bronchial epithelial lining of GA treated group (Figure 5A). In addition, PAS stained SMG sections showed continuous basement membrane surrounding acini and the secretory granules in acinar cells in control group. SN treated group showed loss of continuity of epithelial lining surrounding duct system, while GA treated group almost regained normal basement membrane (Figure 5B).

A) Microscopic pictures of PAS stained lung sections showing normal epithelial lining of bronchioles with no goblet cells (blue arrows) in Control group. In contrast, lung of SN group showed marked increase in number of goblet cells in epithelial lining of bronchioles (yellow arrows) with deposition of pink stained mucus admixed with leukocytes in their lumens (black arrows). Goblet cells markedly decreased to absent in epithelial lining of bronchioles (yellow arrows) in GA group. (X100, bar 100 and X400, bar 50). B) Microscopic picture of PAS stained SMG sections showing positive staining of basement membrane (BM) delaminating the acini and ducts (arrow) in Control group (arrow) while discontinuities of the epithelial lining of interlobular ducts (arrow) were observed in SN group. GA group showed almost restoration and less destructed BM (arrow). (X400, bar 25).
Discussion
Despite the importance of food additives for maintenance and/or improvement of food quality, current consumption of great amounts of these additives with reported predisposition to their toxic hazards has prompted the scientific community to oversee this issue. SN is widely used in food technology. Moreover, increased exposure to high levels of SN in contaminated drinking water has been reported. 24 This inevitable vulnerability to SN toxicity raises the importance of delineating its possible harmful effects on various body organs as well as detection of appropriate protective strategies. 25 In the present study, we reported SN-induced toxicity on lung and SMG with protective effect of concomitant GA administration. This inference was based on the findings that GA treatment combated SN-induced oxidative stress, inflammation, and fibrosis.
Multiple mechanisms have been proposed for SN induced toxicity, among them oxidative stress is a crucial player. It was reported that administration of SN was accompanied by disruption of redox homeostasis in various body organs. This was manifested as increased free radical production, lipid peroxidation and reduced endogenous antioxidant defense species responsible for quenching of produced free radicals. On the other hand, natural antioxidants have been shown to preserve liable organs against SN-induced oxidant/antioxidant imbalance. 3,8,26 In this context, our results demonstrated increased MDA levels in lung and SMG concomitant with compromised antioxidant capacity in SN treated rats. Whereas GA administration improved redox status, attenuated lipid peroxidation, and restored total antioxidant capacity both in lung and salivary gland. Consistent with our finding, GA has been shown to be neuroprotective 27 and cardioprotective 28 via antioxidant mechanism. Moreover, Zhao et al. 29 demonstrated that GA attenuated oxidative stress in a model of sepsis-induced acute lung injury.
Stimulation of inflammatory response featured by recruitment of macrophages concomitant with up-regulated production of inflammatory cytokines was outlined as a cardinal mechanism in SN-induced organ injury. 3,30 –32 In line, we found increased macrophage infiltration accompanied by increased TNF-α and IL-1β levels in lung and SMG of SN intoxicated rats. In addition, PAS staining demonstrated increased goblet cells, mucus, and leukocytes in epithelial lining of bronchioles and discontinuity of epithelial lining of SMG ducts. However, GA administration reversed inflammatory status triggered by SN. Our results were in agreement with previous finding that reported anti-inflammatory capacity of GA in various inflammatory conditions 33,34 including lung inflammation both in vivo 35 and in vitro. 36
Persistent uncontrolled inflammation leads ultimately to scar tissue formation and fibrosis. Inflammatory cytokines produced by recruited inflammatory cells, mainly TNF-α and IL-1β, activate TGF-β a crucial player in over synthesis and accumulation of extracellular matrix proteins during fibrosis, resulting in tissue repair. Many reports revealed that TGF-β mediates lung and SMG fibrosis via activation of fibroblasts, induction of α-SMA and enhancement of ECM synthesis. Therefore, TGF-β represents a central fibrotic protein that can be targeted for inhibition of fibrotic events in both lung and SMG fibrosis. 37,38 SN toxicity is accompanied by stimulation of TGF- β signaling with consequent triggering of the fibrotic processes in various organs including lung. 13 In agreement, our data indicated increased expression of TGF-β, α-SMA and collagen in pulmonary and salivary gland tissue of SN treated rats. This fibrotic condition was markedly relieved by GA administration. Similarly, GA anti-fibrotic capability was previously demonstrated in experimental models of liver 39 and lung 40 fibrosis.
Conclusion
In conclusion, our study presented protective capacity of GA against pulmonary and salivary gland toxicity induced by SN in rats. This was mediated via multiple mechanisms including restoration of oxidant/antioxidant balance, mitigation of inflammatory status and suppression of fibrosis in rat lung and salivary gland (Figure 6).

Graphical abstract.
Supplemental material
Supplemental Material, Supplementary-data-1 - Glycyrrhizic acid ameliorates sodium nitrite-induced lung and salivary gland toxicity: Impact on oxidative stress, inflammation and fibrosis
Supplemental Material, Supplementary-data-1 for Glycyrrhizic acid ameliorates sodium nitrite-induced lung and salivary gland toxicity: Impact on oxidative stress, inflammation and fibrosis by Amira M Elsherbini, Nadia M Maysarah, Mohamed El-Sherbiny, Mohammed MH Al-Gayyar and Nehal M Elsherbiny in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
