Abstract
Alzheimer disease (AD) is a chronic neurodegenerative disease with multi-pathways pathogenesis. Sildenafil is a selective phosphodiesterase-5 inhibitor with a potential benefit in the treatment of AD. This study investigated the possible mechanisms underlying the effect of sildenafil in AD with emphasis on vascular endothelial growth factor (VEGF), and vascular cell adhesion molecule-1 (VCAM-1). Twenty-four adult male rats were classified into four groups; control group: received vehicles, sildenafil–control: received sildenafil (15 mg/kg/day, p.o.), AD group received Aluminum (25 mg/kg/day, p.o.), AD-treated group: received sildenafil (15 mg/kg/day, p.o.) for 6 weeks. AD was assessed by memory performance test and confirmed by histopathological examination and immunostaining of, neurogenesis marker nestin and α-synuclein. The levels of VEGF-A, VCAM-1, oxidative stress markers and TNF-α in brain tissue were evaluated. AD rats showed histopathological evidences of AD; along with increased latency time in the memory test. There was a decrease in VEGF-A, and an increase in VCAM-1, TNF-α, and oxidative stress markers. Immunohistochemical study showed a significant increase in α-synuclein and a significant decrease in nestin expressions in brain tissues. Sildenafil administration ameliorated the histopathological changes and decreased latency time. Such effect was associated with a decrease in VCAM-1, TNF-α and oxidative stress as well as an increase in VEGF-A. Sildenafil caused a significant increase in nestin and a decrease in α-synuclein immunostaining. These findings suggested a protective effect of sildenafil via modulation of VEGF-A, and VCAM-1.
Introduction
Alzheimer’s disease (AD) is a progressive age-related neurodegenerative disease that represents nearly 70% of dementia cases worldwide. In developed countries, it is predicted that by 2025; AD patients will increase in numbers several times if compared to 1980. 1
Although the exact pathogenesis of AD is not so clear, a neurotoxic β-Amyloid (Aβ) peptide accumulation is considered one of the most important pathogenesis. Such accumulation is either due to Aluminum (AL) exposure or to reduction in cerebral blood flow with a decrease in its clearance from the brain. 2
The sequence of events in AD started from deposition of amyloid filaments and hyperphosphorylated tau along with abundant accumulation of α-synuclein, leading to neuronal degeneration and significant synapses loss. 3,4
Several interacting endothelial cell mediators including nitric oxide (NO), vascular endothelial growth factor (VEGF), and vascular cell adhesion molecule-1 (VCAM-1) were implicated in several neuronal effects including cell proliferation, angiogenesis, inflammation, and neurological impact on glial and neuronal cells. 5,6 AD was associated with increased vascular inflammation, as evidenced by elevated expression and localization of inflammatory markers such as VCAM-1 and Tumor necrosis factor-α (TNF-α) surrounding AD-plaques. 7
Previous studies reported that decline in VEGF-A was correlated with the aggravation of AD pathology indicated by the high level of α-synuclein. In turn, overexpression of α-synuclein caused further decline in VEGF-A, indicating that the extent of decline of VEGF-A is correlated with the level of expression of α-synuclein. These findings raise the possibility of a vicious cycle in the accumulation of α-synuclein leads to cerebral hypo-perfusion, which accelerates the progression of AD. 8
Nitric oxide/cyclic guanosine monophosphate/phosphodiesterase (NO/cGMP/PDE) pathway plays a crucial role in AD. 9 Interestingly, both NO donors and cGMP analogs counteract the Aβ-induced-brain injury. 10 One of the most effective ways to up regulate the NO pathway is increasing cGMP levels by PDE 5 inhibition. PDE is expressed in multiple brain regions associated with cognitive functions such as the hippocampus, cortex and cerebellum. 11
Sildenafil is a specific PDE 5 inhibitor that activates NO/cGMP pathway and is commonly prescribed in treatment of erectile dysfunction. It has been reported that sildenafil enhanced the learning and memory functions by raising hippocampal cGMP levels without an evident effect on the male reproductive system. 12 –14 To the best of our knowledge, the effect of sildenafil on VEGA-A and VCAM-1 in AD model was not addressed. This study aimed at investigating the possible mechanisms underlying the effect of sildenafil on AD pathophysiology with emphasis on VEGF and VCAM-1.
Materials and methods
Chemicals and kits
Aluminum chloride hexahydrate (AlCl3.6H2O) and sildenafil citrate were purchased from Lab Chem Inc and Pfizer Co., respectively. They were dissolved freshly before use in saline and distilled water, respectively. 15,16 TNF-α enzyme-linked immunosorbent assay (ELISA) kit was purchased from Sigma-Aldrich Co. (St. Louis, MO, USA), Catalog No: E-EL-R0019. VCAM-1 and VEGF-A ELISA kits were purchased from Elabscience Biotechnology Inc (USA), Catalog No: E-EL-R1061 and E-EL-R2603 respectively. cGMP ELISA kit was purchased from Wuhan Fine Biological Technology Co., (Wuhan, Hubei, China), Catalog No: ER0831. The ready to use anti-α-synuclein and anti-nestin antibodies were purchased from Thermo Fisher Scientific Inc./Lab Vision (Fremont, CA, USA).
Animals
Total 24 male albino rats weighing 250 ± 30 g, aged 8–10 weeks, were obtained from the National Research Center (Dokki, Giza, Egypt), housed in metallic cages (42 × 26 cm), at a temperature of 23 ± 0.5°C, under a 12/12-h light/dark cycle (lights on from 8:00 AM to 8:00 PM), and left to acclimatize to the laboratory environment for 1 week prior to the experiment. Animals were freely accessed to standard food and water. All experimental procedures were complied with the ARRIVE guidelines and carried out in accordance with the U.K. Animals Act, 1986 and EU Directive. The study protocol was approved by the Faculty of Medicine, Minia University Council; approval No. (147:1/2019).
Experimental design
Animals were divided into four groups (six rats in each group): Control group: received vehicles (p.o.). Sildenafil group: received sildenafil (15 mg/kg/day, p.o.). AD group: received Aluminum (AL) (25 mg/kg/day, p.o.). 17 Sildenafil-treated group: co-administered with AL (25 mg/kg) plus sildenafil citrate (15 mg/kg). Rats were treated for 6 weeks, the duration of the study. 18
Assessment of memory performance
Morris water maze (MWM) was used to assess the spatial memory. Rats were tested at the end of the experiment. The water maze was a circular pool (diameter 1.2 m) filled with water maintained at 20°C. Rats were trained (learning sessions) for three consecutive days (eight trials per day) swimming to a raised platform (visible-platform). The animals were placed into the water facing the water pool wall from four different quadrant sites. In the MWM test the rats were subjected to find the hidden platform within 120 s as they were trained during learning procedures. The latency time to reach the hidden platform was recorded. 19
Sample collection and storage
At the end of the study, rats were weighed and then sacrificed under anesthesia with urethane hydrochloride (1 g/kg i.p.). The brain tissues were rapidly removed from the skulls and dissected into different parts. Parts of the cortex and hippocampus regions were removed for histopathological and immunohistochemical studies. Other parts were homogenized in ice-cold phosphate buffer (0.01 M, pH 7.4; 20% w/v). The homogenate was centrifuged at 5000 rpm for 15 minutes and the supernatant was aliquoted into several microcentrifuge tubes and were kept at −80°C until use to determine values of biochemical parameters.
Biochemical measurements
Assessment of brain tissue oxidative stress parameters
Brain tissue content of reduced glutathione (GSH) was assessed by spectrophotometry as previously described. 20 The method was based on the fact that the sulfhydryl group of GSH interacts with 5,5′-dithio-bis-2-nitrobenzoicacid, Ellman’s reagent, that produce a yellow colored 5-thio-2-ni-trobenzoic acid. It was measured at 405 nm using Beckman DU-64 UV/VIS spectrophotometer.
Superoxide dismutase (SOD) activity was evaluated chemically as previously described by Marklund and Marklund, 21 that based on the fact that SOD has the ability to inhibit the pyrogallol autoxidation. One unit of SOD is equal to the enzyme level which can inhibit 50% of pyrogallol autoxidation; SOD activity was detected at 420 nm.
Brain tissue malondialdehyde (MDA) content was detected biochemically, using spectrophotometry depending on the reaction between thiobarbituric acid and MDA as previously described 22 which measures the thiobarbituric acid reactive substances concentration, sometimes referred to as MDA concentration. This method measures the cardiac MDA which is the breakdown product of brain tissue lipid peroxides.
The end products of NO oxidation; nitrite and nitrate (NOx) were assessed as an indicator for synthesis of NO. NOx was measured after the reduction of nitrate to nitrite by copperized cadmium granules in glycine buffer at pH 9.7 (2.5 to 3.0 g of Cd granules for a 4 ml reaction mixture). Quantitation of nitrite was based on the Griess reaction, in which a chromophore with a strong absorbance detected at 540 nm is formed by reaction of nitrite with a mixture of naphthyl ethylene diamine and sulfanilamide. 23
Measurement of TNF-α, in brain tissue
TNF-α brain tissue level was evaluated by using an ELISA kit according to the manufacturer’s instructions.
Measurement of VCAM-1, VEGF-A in brain tissue
A monoclonal anti-rat antibody for VCAM-1 was precoated onto 96-well plates. Samples and standard were added to the wells followed by addition of biotinylated antibody to each well. Avidin-Horseradish Peroxidase (HRP) conjugate was added followed by a substrate solution which produced a blue color that changed to a yellow color after adding acidic stop solution. The density of the color was monitored at 450 nm immediately.
VEGF-A brain tissue level was evaluated by using a sandwich ELISA kit. The micro ELISA plate has been pre-coated with anti-rat antibodies for VEGF-A. Standards and samples were added to the plate wells and combined with the specific antibody. Then a biotinylated detection antibody and Avidin-HRP conjugate were added successively to each micro plate well and incubated. The substrate solution was added to each well which subsequently appeared blue in color. By adding the stop solution the color turns yellow. The optical density was measured spectrophotometrically at a wavelength of 450 nm.
Measurement of cGMP in brain tissue
The brain tissue level of cGMP was assessed using an ELISA kit according to the instructions of the manufacturer’s.
Histological studies of brain tissue
At the end of the experiment animals were decapitated under anesthesia and their brain removed for histological assessments. First, the brains were fixed in 10% formalin and later processed for embedding with paraffin, after which serial sections of 6 µm of thickness was prepared for staining of cerebral cortex and hippocampus cells. 24 The pathological changes were viewed under light microscope after staining with haematoxylin and eosin (H&E).
Immuno-histopathological studies of brain tissue
Immunohistochemical staining was performed by using avidin-biotin peroxidase technique for mouse monoclonal anti-nestin antibody (Abcam, ab22035, UK) (1:100), and a sheep polyclonal anti-α-synuclein antibody (Abcam, ab6162, UK) (1:200). Sections were counterstained with hematoxylin, dehydrated, cleared and mounted. The reaction appeared as brownish cytoplasmic granules. Positive controls were done on control brain sections. Negative control was done after omitting the primary antibody.
Image capture
H&E and immunohistochemical sections were photographed using high-resolution color digital camera mounted on a BX51 microscope (Olympus, Japan); connected to a computer programmed with LC micro application software. It was carried out in the light microscopic unit of Histology and Cell Biology Department, Faculty of Medicine, Minia University. Images were saved as jpg formatting and printed.
Morphometric measurements
All morphometric values were measured in randomly chosen five fields/sections in five sections for every rat in each group at magnification ×40.
The area percentage of α-synuclein positive cells were calculated using the image J program analysis software (Image J 1.48V, Wayne Rasband National Institutes of Health, USA) to measure the color intensity. The number of apoptotic cells and nestin-positive cells were also counted.
Statistical analyses
The results are expressed as the mean ± S.E.M. Statistical significance was determined using Graph Pad 7 Software (San Diego, Calif., USA). One-way ANOVA followed by Tukey’s test was used for multiple comparisons. The results were considered significant when p < 0.05. Normal distribution of the quantitative variables was tested first by Shapiro–Wilk test, normally distributed; (p more than 0.05).
Results
Effect of sildenafil on memory performance
In AD rats, there was a significant increase in latency time in comparison to the control group. Sildenafil-treated rats showed a significant shortening in latency time compared to the Alzheimer group (Figure 1).

Effect of sildenafil on memory performance. Data are presented as mean ± S.E.M (n = 6 rats). Results are considered significantly different; p < 0.0001. aSignificant difference compared to control, bsignificant difference compared to AD group. SIL: sildenafil; AD: Alzheimer’s disease.
Effect of sildenafil on oxidative stress parameters
As compared to the control group, AD caused a significant decrease in brain tissue GSH level and SOD activity, however; it increased the MDA and NOx levels. Administration of sildenafil to AD induced rats significantly increased both GSH and SOD and attenuated both MDA and NOx levels in comparison to the AD group (Table 1).
Effect of sildenafil on oxidative stress parameters in brain tissue.

SOD: super oxide dismutase; GSH: reduced glutathione; MDA: manoldialdehyde; NOx: total nitrite/nitrate; SIL: sildenafil; AD: Alzheimer’s disease
Values are presented as mean ± S.E.M (n = 6 rats). Results are considered significantly different when p < 0.05.
a Significant difference compared to control.
b Significant difference compared to AD group.
Effect of sildenafil on TNF-α, in brain tissues
There was a significant increase in brain tissue content of the inflammatory mediators; TNF-α in AD group when compared with a control group. Administration of sildenafil to AD rats significantly decreased in brain tissue content of TNF-α in comparison to the AD group (Table 2).
Effect of sildenafil on TNF-α, VCAM-1, VEGF-A and cGMP in brain tissues.

SIL: sildenafil; AD: Alzheimer’s disease.
Values are presented as mean ± S.E.M (n = 6 rats). Results are considered significantly different when p < 0.05.
a Significant difference compared to control.
b Significant difference compared to AD group.
Effect of sildenafil on VCAM-1and VEGF-A in brain tissues
There was a significant increase in brain tissue content of VCAM-1in AD group when compared with control group. Administration of sildenafil to AD rats significantly decreased in brain tissue content of VCAM-1 in comparison to the AD group (Table 2).
In comparison with the control group, AD caused a significant decrease in brain tissue levels of VEGF-A. Administration of sildenafil to AD rats significantly increased its level in comparison to the AD group (Table 2).
Effect of sildenafil on cGMP, in brain tissues
In comparison with the control group, AD caused a significant decrease in brain tissue levels of cGMP. Administration of sildenafil to AD rats significantly increased cGMP in comparison to the AD group (Table 2).
Histopathological study
H&E sections of the control group showed a normal histological structure of the cerebral cortex and hippocampus, with normal appearance of a neuron; having large vesicular nucleus and prominent nucleolus, also, some endothelial lined blood vessels and microglial cells appear in the field (Figures 2(A); and (B) and 3(A) and (B)).

Representative photomicrograph of the cerebral cortex histopathology. In figure (A and B), control group and sildenafil group; a normal histological appearance of nerve cells with vesicular nuclei and prominent nucleolus (arrow) is present. Endothelial cells lined blood vessels (arrowhead) and some microglial cells could be observed (asterisk). In figure (C) of AD; apparent gliosis (arrow) with central amyloid plaque (*) is found. Some degenerated nerve cells are present (arrowhead). In figure (D), sildenafil-treated group, few apoptotic cells with pyknotic nuclei are observed (arrowhead). Many cells with normal appearance of vesicular nucleus and prominent nucleolus reappear in the field (arrow). H&E scale bar 50 um.
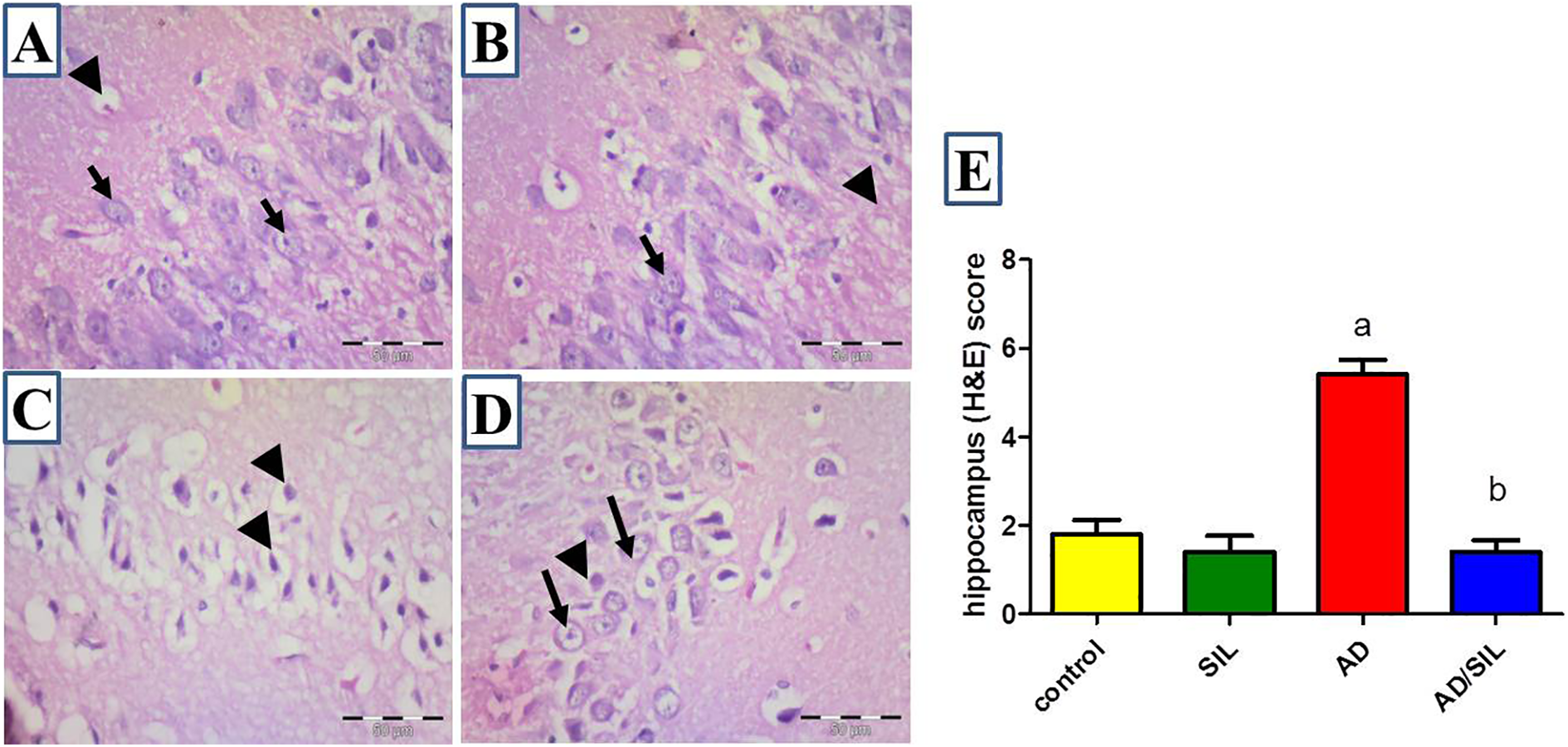
Representative photomicrograph of the hippocampus histopathology. In figure (A and B), control group and sildenafil group; normal histological arrangement of hippocampal nerve cells with vesicular nuclei and prominent nucleolus (arrow) is present. Endothelial cells lined blood vessels (arrowhead) could be observed. In figure (C) of AD; abnormal arrangement of the hippocampal cells is present. Many shrunken cells with darkly stained nuclei (arrowhead) are scattered in the field. In figure (D), sildenafil-treated group; few shrunken cells with pyknotic nuclei (arrowhead) are observed. Many cells with normal appearance of vesicular nucleus and prominent nucleolus (arrow) reappear in the field. Notice the small size of regenerating cells. H&E scale bar 50 um. In figure (E): Scoring of the histopathological changes. Values are presented as mean ± S.E.M (n = 6 rats). Data are presented as mean ± S.E.M. a,bSignificant difference from control group, AD group respectively; p < 0.0001. AD: Alzheimer’s disease; SIL: sildenafil.
In AD group, cerebral cortex showed degenerated neurons and some aggregated neuroglia cells surrounding structure of less eosinophilic areas of amyloid degeneration (Figure 2(C)).
The hippocampus showed distorted histological arrangement of the neurons with the appearance of many apoptotic cells having darkly stained nuclei (Figure 3(C)).
The sildenafil-treated AD group showed that the number of apoptotic neurons significantly decreased in the cerebral cortex and hippocampus with reappearance of normal neuron; having large vesicular nucleus and prominent nucleolus. No aggregation of neuroglial cells could be observed in the cerebral cortex (Figures 2(D) and 3(D)). The previously mentioned results matched with the enhancement of the histopathological finding score (Figures 2(E) and 3(E)).
Immuno-histopathological study for anti-α-synuclein and anti-nestin antibodies
The control group showed a low expression of α-synuclein detected in the cerebral cortex, hippocampus and neuropil (Figure 4(A), (E), and (I)), in sildenafil group nearly the same finding of low expression of α-synuclein in the cerebral cortex, hippocampus and neuropil (Figure 4(B), (F), and (J)) as control group.

Representative photomicrograph of immunostaining of alpha synuclein expression in the brain. In figures (A), (E) and (I), low expression of alpha synuclein protein in the cerebral cortex, hippocampus and neuropil of control group respectively is present. In figures (B), (F) and (J), expression of α-synuclein protein in the cerebral cortex, hippocampus and neuropil of sildenafil group nearly the same as control group. In figures (C), (G) and (K), there is an increase in the deposition of α-synuclein protein in the nerve cells of cerebral cortex (B) (arrow), hippocampus (E) (arrowhead) and neuropil (H) (*) of AD group. In figures (D), (H) and (L), expression level of α-synuclein nearly back to normal in nerve cells of cerebral cortex, hippocampus, and neuropil of sildenafil-treated group. Figure (M), the scoring of α-synuclein protein expression. Values are presented as mean ± S.E.M (n = 6 rats). Data are presented as mean ± S.E.M. a,bSignificant difference from control group, AD group respectively; p < 0.0001. Anti-α-synuclein immune staining scale bar 50 um. AD: Alzheimer’s disease; SIL: sildenafil.
In AD group, the expression of anti-α-synuclein was significantly increased in all studied brains areas. In the cerebral cortex, there was a highly positive cytoplasmic expression in the neuron, while in the hippocampus the expression was mainly extracellular in the form of brown granular color. The neuropil showed aggregation of brown color surrounding the nerve endings (Figure 4(C), (G) and (K)).
Sildenafil-treated AD group showed a significant down regulation in α-synuclein expression in the cerebral cortex, hippocampus and neuropil. The level of expression nearly returned to its normal level (Figure 4(D), (H) and (L)).
For anti-nestin antibody; immuno-histopathological study of the control and sildenafil groups showed that the dentate gyrus of adult brain hippocampus has a positive expression for anti-nestin antibody in some cells (Figure 5(A) and (B)). In AD group, the dentate gyrus of the hippocampus showed a significant decrease in the number of nestin-positive cells (Figure 5(C)). While, in sildenafil-treated AD group a significant increase in the number of immuno-positive cells was present. Also, new immature cells appear in the hylus as well (Figure 5(D)).
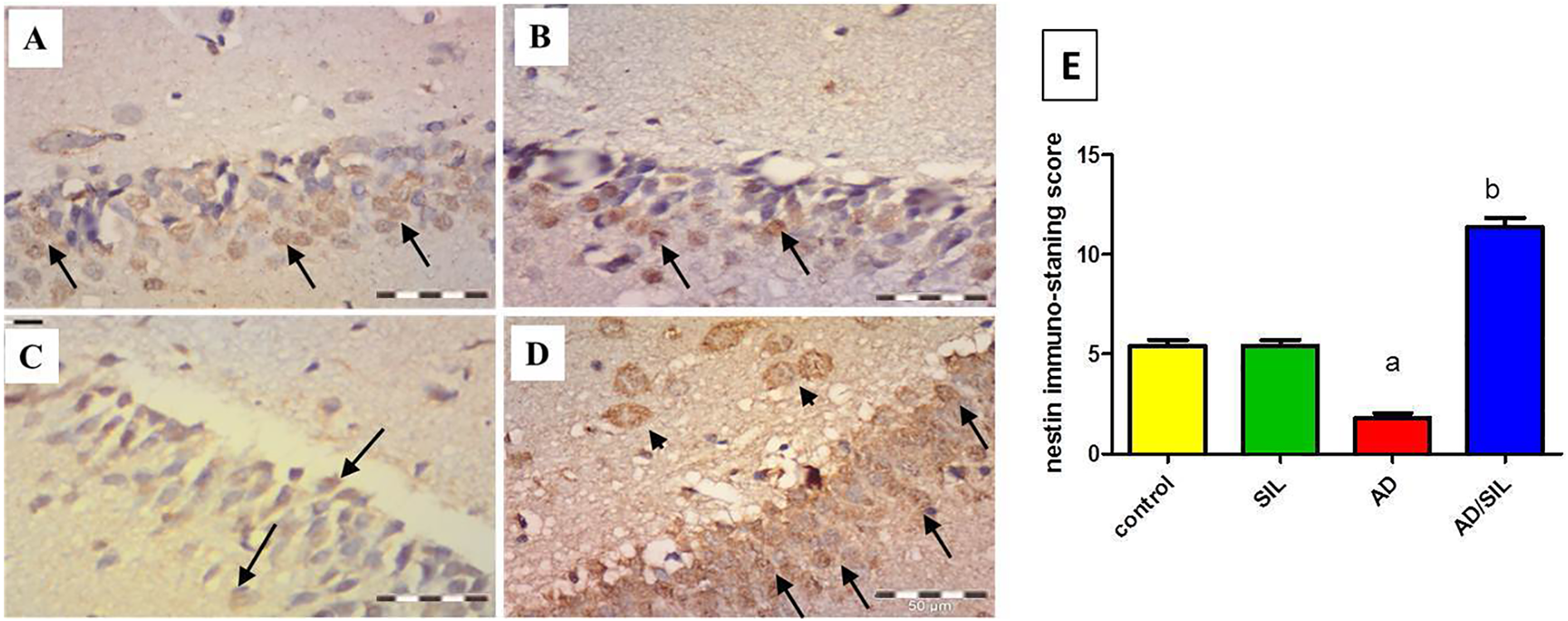
Representative photomicrograph of nestin immunostaning in the dentate gyrus hippocampus. In figure (A and B), some immune positive cells in the dentate gyrus of hippocampus of control and sildenafil groups (arrow). In figure (C), few nestin immune positive cells in the dentate gyrus of hippocampus of AD (arrow). In figure (D), many nestin immune positive cells in the dentate gyrus of hippocampus of sildenafil-treated group (arrow). Apparent immune positive cells in the hilus (arrow head) are observed. Figure (D), scoring of the nestin immune positive cells. Values are presented as mean ± S.E.M (n = 6 rats). Data are presented as mean ± S.E.M. a,bSignificant difference from control group, AD group respectively; p < 0.0001. Anti nestin immune staining scale bar 50 um. AD: Alzheimer’s disease; SIL: sildenafil.
Discussion
Aluminum-induced AD is a widely used animal-model for studying the pathogenesis of AD. 25,26 To date, there is no effective therapeutic intervention in the treatment of AD. 27
The present study is a trial to re-purpose the previously used drugs to be a new approach to cut the pathophysiology of AD.
In the current present study AL-induced AD rats showed an increase in the time spent in target quadrant in MWM test, reflecting an impairment in the learning ability and the spatial memory, as AL interferes with downstream effector molecules involved in memory potentiation. 28 Such findings were matching the study of Ali et al. 29
Memory impairment encountered in AD rats, was associated with evidence of an oxidative stress indicated by an increase in MDA and NO along with a decrease in SOD and GSH. The role of oxidative stress in AD has been previously reported. The oxidative stress may either directly damage the brain cells or release inflammatory cytokines that initiate inflammation. 30
NO is water and lipid soluble molecule that plays diverse functions in normal and pathological states including regulation of blood flow and modulation of oxidative stress. 31 NO activates guanylyl cyclase enzyme that converts GTP to cGMP. cGMP has the central role in NO signaling and in regulation of physiologic responses. 32
PDEs superfamily is cyclic nucleotide that degrades cGMP to 5′-GMP which is its relative inactive isomer. At this point, the NO message may be interrupted. 33 NO, in oxidative stress and inflammatory conditions, can react with superoxide radical (O2–) to produce reactive oxygen species and reactive nitrogen species. Therefore, NO can damage the cell of its origin and cause a variety of diseases including AD. 34
On the other hand, it was also reported that inflammatory diseases like AD there is up-regulation of PDEs that results in degrading cGMP to its inactive isomer, 5′-GMP. As a result, the cell is getting short of bioactive NO and responses daespite of upregulation of NOS and guanylyl cyclase. 33
In the present study, the brain tissue level of NO was significantly increased; however, the cGMP level was decreased in the AD group compared to the control group. This finding implies high production of iNOS-derived NO which exerted the nitrostive effect with little physiological effect mainfested by the decreased cGMP level that may be secondary to upregulation of PDEs.
As sildenafil is a specific and strong inhibitor of PDE 5 12 that prevents degradation of cGMP, 13 it significantly decreased the brain tissue NO and increased the cGMP levels which implies that sildenafil decreased the iNOS-derived NO formation but preserved the physiological NO-derived cGMP from the degradation by PDE 5.
In consistence with the previous studies, the present data reported increase in the inflammatory mediator, TNF-α. TNF-α is an immunomodulatory and proinflammatory cytokine implicated in neuro-inflammation and neuronal damage, It has been well established that elevated TNF-α level is critically involved in the hippocampus-dependent cognitive deficit, which was also proved in our study by a significant increase in TNF-α expression in rats with AD in comparison to the control group. Previous studies reported that TNF-α level is increased in AD patients and that inhibition of TNF-α is proposed to treat AD patients. 35,36
In this study, oxidative stress markers and TNF-α were ameliorated in sildenafil-treated rats, reflecting its antioxidant and anti-inflammatory effect, matching with previous studies reported that cGMP accumulation, via PDE 5 inhibtion by sildenafil, inhibited the neuronal inflammation, by blocking the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and decreased the level of proinflammatory cytokines of IFN-γ, TNF-α, IL-1β, IL-2 and cycloxygenase-2. 37,38
Based on the universal effect of vascular dysfunction and inflammation in cognitive decline and neurodegeneration observed in AD patients, 39 the present study reported the effect of sildenafil on VCAM-1 and VEGF-A production and their relation to α-synuclein deposition and nestin expression.
The data of the current experiment reported a significant decrease in VEGF-A in the brain of AD rats along with increased in immunostaining of α-synuclein in the cerebral cortex and hippocampus, matching the previous studies that reported overexpression of α-synuclein caused a decline in VEGF-A level. Such decline was correlated with the level of α-synuclein in brain tissue. 8 Furthermore, patients with AD showed low levels of VEGF-A in serum and brain tissues. 40
α-Synuclein plays a role in the neuropathological lesions of not only AD of Parkinson disease and amyotrophic lateral sclerosis as well. The proposed mechanism of spreading of pathology resulted from intra- and intercellular propagation of pathological seeds of tau, Aβ, α-synuclein following what is called “a prion-like mechanism.” The term “prion-like” means unknown mechanisms in which toxic misfolded monomers or multimers initiate an autocatalytic cycle of pathological misfolding and aggregation. 41
Deger et al. 4 mentioned that the formation of α-synuclein aggregates plays a role in the development of neurodegenerative disorders in AD. As α-synuclein level was significantly high in the cerebrospinal fluids in patients with AD, α-synuclein was considered an early pathophysiological event in the development of AD. 42,43
In the present study, sildenafil increased VEGF-A content and decreased immunoexpression of α-synuclein in brain areas. Increased VEGF-A is linked to neovascularization and increased tissue perfusion. Moreover, sildenafil increased cGMP levels in the current study. Such effects may represent important mediators of the perfusion of brain tissue leading to marked improvement in the neurodegenerative changes. 44 Such observations are consistent with previous studies. 45
Cell adhesion molecule, VCAM-1 is involved in several neurological diseases including AD. Its expression is regulated in brain micro vessel endothelial cells by cytokines such as interleukins and TNF-α. 46 It has been demonstrated that VCAM-1 was not only engaged in trans-endothelial migration of leukocytes but also served as signal transducers that initiated endothelial signaling and influence the progression of neuroinflammation. Increased level of VCAM-1 which was evident in our study has also been proposed as an indicator of inflammatory conditions in the CNS including AD with a positive correlation between increased levels of brain VCAM-1 and AD severity. 47,48 It has been reported that sildenafil could reduce VCAM-1 level, 9 Such effect was attributed to its effect on NO pathway, carrying a potential beneficial effect in the treatment of the neuro inflammatory /demyelinating disorders.
Nestin is a neural stem cell marker expressed in immature neurons; these cells have become the focus of many studies as they are an ideal therapeutic target in the therapeutic development for neurological pathologies. 49
In the present work, the immunohistochemical study showed a decrease in the number of nestin immune-positive neural stem cells in dentate gyrus of the hippocampus in AD rats and sildenafil treatment increased nestin expression, matching other studies reporting that sildenafil increased the number of nestin positive neural stem cells and their neuronal and oligodendrocyte types in the ischemic brain of middle-aged mice. 50
Taken together, the results of the present study suggested that sildenafil, through preventing cGMP degradation by modulating VEGF-A and VCAM-1 along with its antioxidant and anti-inflammatory effects mitigated AD in rats. Such effect of sildenfail was associated with the modulation of α-synuclein and nestin.
Conclusion
Administration of sildenafil could be a key factor in recovering cognitive dysfunction in AD patients by VEGF-A and VCAM-1 modulation; along with its antioxidant and anti-inflammatory impacts with modulation of the α-synuclein and nestin brain tissue expression.
Supplemental Material
Supplemental Material, highlights - Sildenafil ameliorates Alzheimer disease via the modulation of vascular endothelial growth factor and vascular cell adhesion molecule-1 in rats
Supplemental Material, highlights for Sildenafil ameliorates Alzheimer disease via the modulation of vascular endothelial growth factor and vascular cell adhesion molecule-1 in rats by MA Ibrahim, MASA Haleem, SA AbdelWahab and AM Abdel-Aziz in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
