Abstract
Environmental chemicals and drugs can induce cardiotoxicity, mainly by generating free radicals. Reactive oxygen species play a critical role in the pathogenesis of cardiac tissue injury. This highlights a need for prevention of cardiotoxicity by scavenging free radicals. Melatonin has been shown to act as a protector against various conditions in which free radicals cause molecular and tissue injury. Some of the mechanisms by which melatonin operates as a free radical scavenger and antioxidant have been identified. The importance of endogenous melatonin in cardiovascular health and the benefits of melatonin supplementation in different cardiac pathophysiological disorders have been shown in a variety of model systems. Melatonin continues to attract attention for its potential therapeutic value for cardiovascular toxicity. The therapeutic potential of melatonin in treatment of cardiotoxicities caused by various chemicals along with suggested molecular mechanisms of action for melatonin is reviewed.
Introduction
Cardiovascular diseases are major causes of disability and mortality globally. 1,2 Complications from free radical influence of cardiac physiology include atherosclerosis, hypertension, and ischemia/reperfusion (I/R). Reactive oxygen species (ROS) play a vital role in the pathogenesis of cardiac tissue damage. A major source of ROS in cardiomyocytes during ischemia and early reperfusion may be the result of a dysfunction in the electron transport chain (ETC) in the mitochondria. 2 Beside their therapeutic value, drugs such as doxorubicin (DXR), cyclosporine A (CsA), and isoproterenol have been reported to induce cardiotoxicity. 3 –5 Chemicals like aluminum phosphide and lead have also been reported to cause cardiac tissue damage. 6,7 Treatments to reduce or prevent the cardiotoxicity caused by such drugs and chemicals have not always been successful; therefore, new medications and a better understanding of the pathophysiology are needed.
Melatonin (73-31-4), N-acetyl-5-methoxytrypamine, was discovered in 1958 in the bovine pineal gland. 8 Melatonin is also produced in cardiac tissue. 9,10 Melatonin is best known for its effect on circadian physiology. Melatonin also has been evaluated as a protector against various conditions in which free radicals cause tissue damage. 9 –11 Its anticancer activity may be due to activation of p53 tumor suppressor pathway. Melatonin has been suggested to have the potential to decrease genetic alterations after exposure to DNA-damaging agents. 12 It also has been suggested that melatonin may be beneficial in attenuating molecular injury and cellular death. 13
Melatonin has been reported to show beneficial effects in cardiovascular diseases including hypertension, I/R injury, atherosclerosis, hypertrophic cardiomyopathy, QT prolongation (torsade de point), and in cardiotoxicity induced by drugs and environmental chemicals.
2,9,10
It has been reported that melatonin and its metabolites can influence the cardiovascular system by their antioxidant action.
2,10,11
The ability of melatonin to inhibit the pro-oxidative enzyme, nitric oxide synthase (NOS), is apparently mediated by a tertiary metabolite.
14
The capability of melatonin and/or its metabolites to prevent or at least reduce free radical toxicity in cardiac tissue suggests the possibility of melatonin limiting oxidative stress and/or inflammation within cardiac cells.
2,11
Moreover, melatonin could inhibit QT prolongation and reduce the KCNH2 gene expression level.
15
Therefore, melatonin may be a promising therapeutic agent against arrhythmia related to an abnormality in the human hERG (the human Ether-à-go-go-Related Gene), a gene (
Some of the mechanisms by which melatonin may function as a free radical scavenger and as an antioxidant include its influence on cardiovascular pathophysiology through both receptor-mediated and receptor-independent mechanisms. 13 The melatonin membrane receptors are present throughout the vascular system including the heart. 10 These receptors are associated with both the vasoconstrictor and/or vasodilator effects of melatonin. 13 According to Reiter and Tan, there are numerous nuclear binding sites for melatonin. 9 Melatonin has been reported to interact with nuclear receptors and intracellular proteins such as calmodulin, or tubulin-associated proteins. Melatonin has also been shown to have both a direct and an indirect antioxidant effect. 16
Several studies have reported that high doses of exogenous melatonin can affect mitochondrial energy homeostasis and that melatonin can neutralize the reduced activity and expression of the complex electron transport chain, which normally leads to ATP production. Moreover, melatonin has been shown to promote gene expression and the activity of various antioxidant enzymes such as glutathione peroxidase (GPx), superoxide dismutase (SOD), glutathione reductase (GRx), and catalase (CAT). 6
This review provides a literature update on the potential protective effect of melatonin against cardiotoxicity induced by various chemicals, including metals, environmental pollutants, oxidative agents, and anticancer drugs.
Aluminium phosphide
Aluminium phosphide (AlP), an inexpensive insecticide, affects all stages of insect development with no untoward changes in seed viability or quality. AlP interacts with hydrochloric acid in the gastrointestinal system or with atmospheric moisture, releasing the active phosphine. AlP, which is readily available as a fumigant, is highly toxic and is a major, though under-reported, environmental and public health problem. It has been reported as a cause of suicidal deaths. Its high mortality rate is the result of cardiac dysfunction due to the death of cardiomyocytes. Death results from profound shock, myocarditis, and multi-organ failure. The mechanism by which AlP causes cardiomyopathy is inhibition of mitochondrial cytochrome c oxidase in cardiomyocytes resulting in a reduction of ATP production and an increased ROS generation. 6,17 –19
Phosphine at a dose of 16.7 mg/kg in rats can trigger apoptosis pathways in different tissues including heart, kidney, and liver. It has been demonstrated that AlP influences mitochondrial membrane fluidity, resulting in mitochondrial swelling and outer membrane rupture, leading to the release of pro-apoptotic factors such as cytochrome c that in turn activates caspases to induce cell death. 6 Melatonin can inhibit mitochondrial permeability transition pore (mPTP) opening directly, preventing or reducing apoptosis. Melatonin also has been reported to inhibit the release of cytochrome c from mitochondria, reducing or even preventing caspase activation and apoptosis. Furthermore, this endogenous hormone can localize Bcl-2 intomitochondria, rendering Bax in an inactive form, again protecting cells from undergoing apoptosis. 6,17
Melatonin may have the potential to decrease AlP-induced cardiotoxicity related into mitochondria dysfunction. 6 However, further studies are required to determine the role, if any, of melatonin in such a protective mechanism. 6,18
Antracyclines
Daunorubicin
Daunorubicin (DNR) is the hydrochloride salt of an anthracycline antineoplastic antibiotic with therapeutic effects similar to doxorubicin. There are, however, severe daunorubicn-induced degenerative changes in cardiac tissue, including decreases in cell number with interstitial edema and cytoplasmic vacuolization. The acute alterations in the myocardium and chronic issues such as congestive heart failure are characteristics of DNR toxicity. Myocardial toxicity is manifested in its most severe form as potentially fatal congestive heart failure which may occur during therapy or months to years after termination of treatment. Special attention must be given to the potential cardic toxicity of DNR, particularly in infants and children. 20,21
DNR exhibits its cytotoxic activity through topoisomerase-mediated interaction with DNA, thereby inhibiting DNA replication and repair and RNA and protein syntheses. Metabolism of the anthracyclines can stimulate oxygen-centered ROS generation. DNR also inhibits activation of respiratory chain enzymes, leading to cellular necrosis. In addition, these radicals can damage cellular membranes due to lipid peroxidation. 20,21
DNR was administered to rats (10 mg/kg/day, single dose or 3 mg/kg weekly for 3 weeks) and melatonin was injected subcutaneously before and after administration of the drug at a dose of 10 mg/kg. Cardiac muscle injury was reduced in rats treated with melatonin in comparison with the rats given only DNR. In addition, lipid peroxidation markers such as MDA and 4-HDA were elevated following 3 mg/kg treatment with DNR for 3 weeks. Administration of melatonin before DNR prevented the increase. These results suggested antioxidants, including several antioxidative enzymes, were involved. 21
Doxorubicin
Anthracyclines such as doxorubicin (DXR) are some of the more potent and generally prescribed chemotherapeutic drugs. The use of this antineoplastic agent, however, is restricted by multiple and serious side effects, one of which is cardiotoxicity. DXR-induced cardiotoxicity may be acute or early to late-onset, all resulting in heart failure. 22
Treatment of rats with 6 mg/kg/day melatonin for 2 weeks prevented the cardiotoxic effects of DXR (12 mg/kg). Cardiac function was enhanced in rats treated with melatonin and DXR as indicated by the elevation in cardiac output, total work performance, and stroke volume in comparison with rats that received only DXR. A significant decline in total body weight and in heart weight was observed in DXR-treated rats both of which were alleviated in rats treated with the combination of DXR and melatonin. 23 Dwaich et al. reported that DXR significantly lowered arterial pressure and left ventricular fractional shortening; these cardiomyopathies were ameliorated with the melatonin treatment. 24
Apoptosis is a typical hallmark in the sequela of DXR-induced heart failure. In a model of acute DXR-induced cardiotoxicity in rats, cleaved caspase-3 level, an apoptotic cell death marker, was significantly expressed in cardiac tissue. However, in the DXR and melatonin treated rats expression of caspase-3 cleavage in cardiac tissue was noticeably diminished. These results are in agreement with other research which indicated that caspase-3 activation is elevated during DXR-induced heart failure. 25 –28 Moreover, melatonin protected the heart from myocardial ischemia by down regulating the cleaved caspase-3 expression in this rat model. 28,29
DXR specifically targets and accumulates in the mitochondria at concentrations significantly higher than in the plasma. 30,31 Elevation of ROS generation leads to irreversible myocardial oxidative damage and generates a bioenergetic crisis culminating in cardiomyocyte death. 31,32 A review by Reiter and colleagues supports the classification of melatonin as a mitochondrial-targeted antioxidant because of its high degree of accumulation in these organelles. 33 The elevated concentration of melatonin in mitochondria is attributed to facilitation of peptide transporters 1 and 2 which are localized in the mitochondrial membrane. 34 A recent study showed that melatonin decreased the cellular oxidative stress induced by DXR through both direct and indirect pathways. A possible mechanism for these effects is interaction with calmodulin, which includes inhibition of the nuclear RAR-related orphan receptor alpha (RORa) receptor, epigenetic mechanisms, and modulating function or expression of nuclear factor erythroid 2–related factor 2 (Nrf2), which is a regulator of antioxidant proteins. 35
Mitochondrial dysfunction, a distinct feature of DXR-induced cardiotoxicity, is indicated by elevated levels of PTEN-induced putative kinase 1 (Pink1) and Parkinson protein (PARKIN protein) as potential factors for mitophagic events. The Pink1 protein level was remarkably reduced in heart tissue of DXR and melatonin treated rats but remained high in the DXR only treated rats. Additionally, the PARKIN protein level was enhanced following DXR treatment; however, only a marginal elevation of the PARKIN level occurred in animals treated with both DXR and melatonin. 25
In cardiac cells, mitochondrial fission and fusion are crucial processes that should be in balance. DXR interrupts mitochondrial fusion while favoring mitochondrial fission as demonstrated by a lower amount of mitofusin protein (Mfn1 and Mfn2) expression as well as an elevated mitochondrial fission protein (Drp1 and hFis1) expression. Pretreatment with melatonin inverted the change in expression of mitochondrial fission and fusion proteins, favoring mitochondrial fusion during DXR-induced myocardial damage. 22
Govender and coworkers showed that pretreatment with melatonin induced an elevation in cellular ATP levels and prevented the bioenergetics failure induced by DXR. It was suggested by Govender and his colleagues that melatonin maintained cellular ATP levels by reducing electron leakage from the ETC by modifying the uncoupling protein activity and thereby reducing molecular injury to the ETC. 22,25
It has been shown that sirtuin 1 is a critical upstream regulator of PGC-1α signaling. Sirtuin 1 activates the mitochondrial antioxidant defense system in cardic tissue by preventing a rise in mitochondrial ROS production caused by DXR. Furthermore, the anti-apoptotic and antioxidant effects of melatonin in rat heart tissue treated with DXR are likely allied with the upregulation of sirtuin 1 during DXR-induced cardiotoxicity. 25 The peroxisome proliferator-activated receptor-gamma coactivator-1alpha (PGC-1α) is an important regulator in energy metabolism and mitochondrial biogenesis. Melatonin has been shown to improve the PGC-1α expression that was reduced in DXR treated rat heart tissue. 25,36 –38 These mechanisms are outlined in Figure 1.
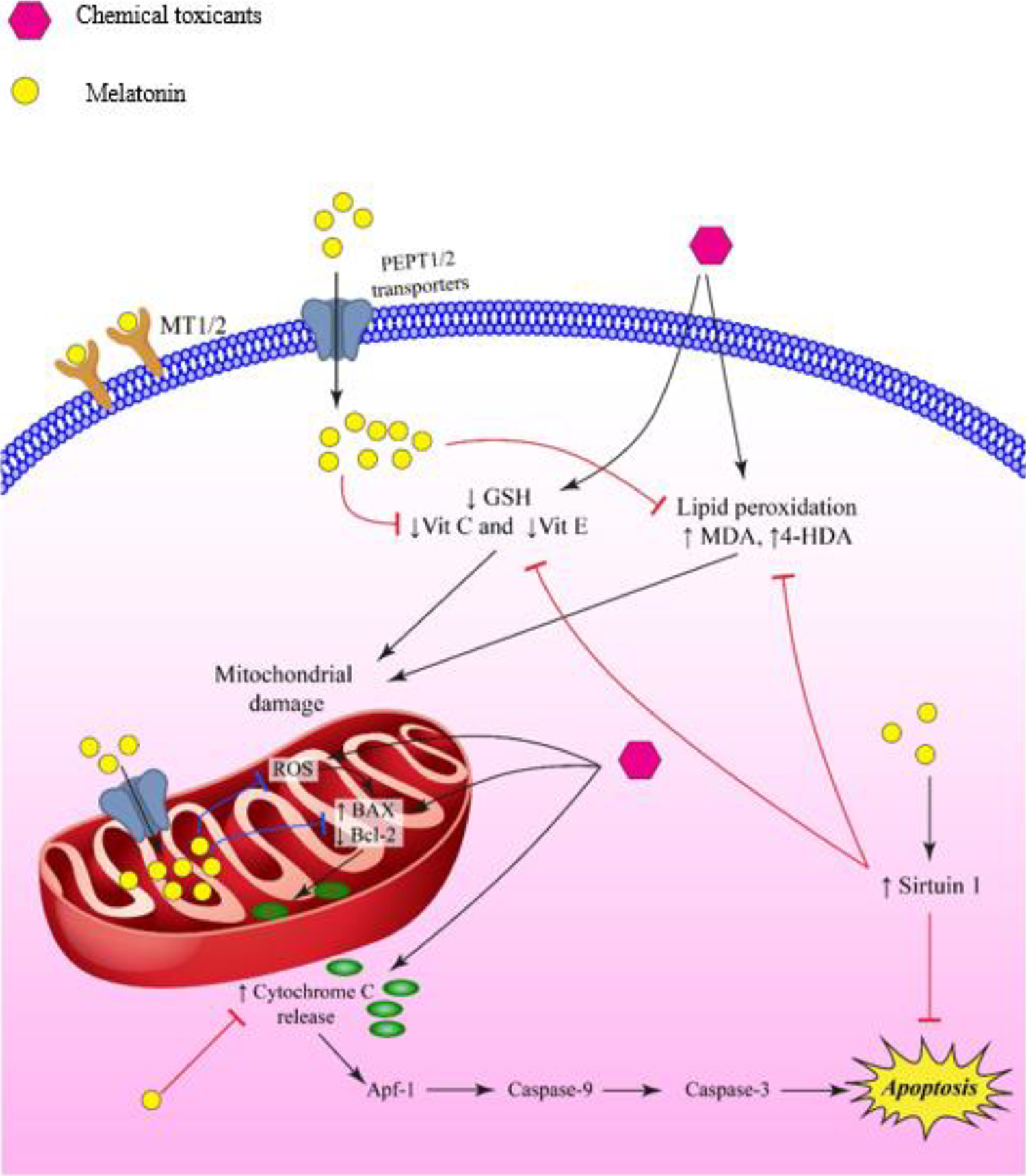
The melatonin molecule enters the cell through human peptide transporters 1 and 2 (PEPT1/2). It inhibits the decrease in GSH, Vit C, and Vit E levels which results in prevention of myocardial cell damage. Melatonin also protects cell from lipid peroxidation by inhibiting the elevation of MDA and 4HDA levels. Melatonin enters mitochondria through peptide transporters and via inhibition of MPTP opening, decreases the release of cytochrome c and caspase activation, localizing Bcl-2, and elevating sirtuin 1, thus inhibiting apoptosis. Melatonin accumulation in mitochondria decreases free radical generation. Melatonin prevents heart failure thorough impeding of mitochondrial membrane bound ATPase enzymes and Beta tubulin level. 4-HDA, 4-hydroxyalkenals; Apaf-1, Apoptotic protease activating factor-1; Bax, Bcl-2-associated X protein; Bcl-2, B-cell lymphoma 2; GSH, Glutathione; MDA, Malondialdehyde; MT1/2, Melatonin receptor 1/2; PEPT1/2, human peptide transporter 1 and 2; ROS, Reactive oxygen species; Vit C, Vitamin C; Vit E, Vitamin E.
In consideration of the protective role of melatonin in mitochondrial physiology in the heart during chemotherapy, clinicians may be concerned about melatonin having a similar protective effect in the mitochondria of cancer cells thereby improving cancer cell survival. However, there are considerable data supporting the tissue specificity of melatonin’s actions on the mitochondria in cancer cells and its opposite effect on the mitochondria in healthy cells. 25
Epirubicin
Epirubicin is another anthracyclines which is effective in the treatment of hematological malignancies and solid tumors. However, its clinical use also is limited due to its cardiotoxicity. Epirubicin can cause irreversible cardiac damage resulting in congestive heart failure and cardiomyopathy including myofibrillar loss and dilatation of the sarcoplasmic reticulum. 39,40
Guven et al. evaluated the protective effect of melatonin against the myocardial toxicity of epirubicin in rats. They administered a single dose of epirubicin (10 mg/kg, i.p.) on day 1 and at the same time, 200 mg/kg/day i.p. melatonin for 10 days. The degree of cardiac muscle cell alterations was measured histologically and biochemically. In this experiment, the immunohistochemistry showed dense laminin and fibronectin labeling in samples from the epirubicin-treated group; this immunoreactivity was weaker in samples from animals treated with both melatonin and epirubicin. 39
Furthermore, mitochondrial degeneration and swelling of the myofibrillary structure occurred; intracytoplasmic vacuolization also was observed in samples taken from the epirubicin-treated group. The administration of melatonin diminished the cellular injury and mitochondrial degeneration and fewer vacuoles were observed in samples from hearts of these animals. Since NO rapidly degrades to nitrate (NO3 −) and nitrite (NO2 −) in aqueous solution, the serum nitrite and nitrate levels were considered as an index of NO production. Rats treated with epirubicin had higher nitrite/nitrate levels compared with control animals, potentially leading to nitrozative stress. Melatonin injection into epirubicin-treated animals attenuated their nitrite/nitrate concentrations significantly which suggested the efficacy of melatonin in suppressing the epirubicin-induced nitrozative stress. 39 These results indicated that melatonin partially protects against epirubicin-induced cardiotoxicity which might be due to the suppression of epirubicin-induced nitrozative stress.
Cyclophosphamide
Cyclophosphamide is an alkylating anticancer drug with strong cytotoxic and immunosuppressive activity. 11,41 It is used in the treatment of Hodgkin’s and non-Hodgkin’s lymphoma, leukemia, rheumatoid arthritis, and lupus erythematous. 41 Cyclophosphamide cardiotoxicity affects between 7–28% of such patients. Cyclophosphamide has been shown to enhance proinflammatory cytokines such as nuclear factor-kappa B (NF-κB), cyclooxygenase-2 (COX-2), tumor necrosis factor-alpha (TNF-α), and interleukin-1 beta (IL-1β). 41 Exposure of animals to cyclophosphamide lead to a decrease of GRx, GPx, and SOD activity. 41 It is known that antioxidants are beneficial in decreasing cyclophosphamide cardiotoxicity by elevation of GSH and SOD levels and reduction in proinflammatory cytokines. 41 Since melatonin has similar antioxidant effects, and is used in treatment of cyclophosphamide hemorrhagic cystitis, melatonin should be evaluated for its effectiveness in preventing or at least reducing cyclophosphamide cardiotoxicity. 11
Cyclosporine A
Cyclosporine A (CsA) is a potent immunosuppressor which is used in transplant surgery and for the treatment of several autoimmune diseases. However, major side effects of CsA such as nephrotoxicity, hepatotoxicity, neurotoxicity, and cardiovascular toxicity have limited its usage. 4,42 It has been suggested that reactive oxygen metabolites and oxidative stress have a role in CsA-induced pathologies. Several in vivo studies have demonstrated that CsA induces degenerative myocardial changes involving size, shape, and organization of the cardiomyocytes both in the atria and the ventricles as well as a decrease in cytochrome-c-oxidase activity. 4,10
The protective effect of melatonin (1 mg/kg/day, IP, 21 days) against cardiotoxicity induced by CsA (15 mg/kg/day, SC) was evaluated in rats. CsA treatment decreased body weight gain and elevated systolic blood pressure (SBP) compared with control rats. Administration of melatonin completely inhibited the negative effects of CsA on both body weight gain and SBP. Degenerative morphological changes of the cardiac tissue were demonstrated after CsA treatment, but noticeably reduced or eliminated by melatonin. CsA caused a marked reduction of antioxidant enzyme activities which also was inhibited by melatonin treatment. 4 Increase levels of heat shock protein 70 (Hsp70) expression are essential to promote cell survival following a variety of environmental stresses. Levels of Hsp 70 were enhanced after CsA treatment. These results confirm previous data showing a relationship between oxidative stress and CsA-induced cardiotoxicity. Melatonin partly reversed the oxidative damage and prevented elevation of Hsp70 expression. 4
In another study, 10 it was shown that CsA increased myocyte cell death, leading to reduced left ventricular mass. This enhancement of the apoptotic process was increased by CsA after 21 days of exposure as quantified by the TUNEL method. While the exact mechanism is not understood, it was suggested by the authors that a sudden increase in the permeability of the inner mitochondrial membrane caused this apoptosis enhancement. These mitochondrial processes have been shown to be prevented or at least reduced by melatonin. When melatonin was loaded onto solid lipid nanoparticles (SLN), a significant reduction of myocyte cell death was observed. In fact, the SLN form of melatonin (MTSLN) did not involve melatonin1/malatonin2 receptors but rather a cellular internalization pathway that generated a more effective antiapoptotic process than melatonin alone. MTSLN appears to be an excellent vehicle to motivate the antiapoptotic mechanism. 10
Epinephrine (Adrenaline)
Epinephrine, a catecholamine, is a hormone and a neurotransmitter produced by the adrenal glands. Epinephrine plays a major role in cardiovascular regulation and can be used as a drug in the treatment of various life-threatening situations such as shock. It is, in fact, the prototype sympathomimetic drug. Epinephrine is a potent activator of α adrenergic receptors and it also activates β1 and β2 receptors. It is added to local anesthetic solutions to decrease systemic absorption and prolong the duration of the anesthetic effect. Its toxicity is usually caused by iatrogenic errors. Adrenergic effects of epinephrine can result in myocardial hypoxia, the overload of calcium and an increase in ROS generation. In overdose, there is a rapid onset of agitation, hypertension, tachycardia, and dysrhythmias. When administered in sufficiently high dosages, it causes cardiotoxic effects which can lead to Takotsubo cardiomyopathy or myocardial necrosis. 43,44
In an ex vivo study, melatonin was effective in reducing the epinephrine toxicity in the isolated rat heart. Melatonin was dissolved in a perfusion solution (50 μmol/L) and hearts were continuously perfused throughout the experiment with melatonin plus epinephrine. From the 10th minute after injection of epinephrine, changes in function and arrhythmogenesis were evaluated until the end of the experiment in the 20th minute. Immediately after the epinephrine injection, strong positive inotropic and chronotropic effects were observed in all hearts of both experimental groups. There were not any noticeable differences in the heart rate and coronary flow between the melatonin group and the control group. Indexes of ventricular contraction and relaxation (+dP/dtmax and −dP/dtmax), however, were markedly different between the experimental groups, indicating better preservation of contraction and relaxation in hearts treated with melatonin. 44
Following epinephrine administration, different types of ventricular arrhythmias were observed in the hearts of both experimental groups. However, no significant differences in the arrhythmia score, incidence, and total duration of severe ventricular arrhythmias (VT, VF, SVF) were noticed between the melatonin group and the control group. A significantly decreased average level of the arrhythmia parameters measured showed a reduction in the electrical instability of the hearts in the melatonin treated group. 44
It also had been reported that melatonin prevents elevation of blood pressure, decreases sympathetic tone and oxidative stress and has a protective function against cardiac injury in long term use of epinephrine. Vanzan and Ravingerova have suggested that melatonin may be beneficial for management of cardiovascular pathological caused by epinephrine or as a useful co-treatment in cases where epinephrine is administered. 44
Isoproterenol
Catecholamines are part of the stress induced neurohumoral system which regulates cardiac output, thus regulating the perfusion rate of peripheral tissues. However, overproduction of catecholamines or administration of exogenous catecholamines can be harmful.
45
Isoproterenol, a synthetic catecholamine and beta-receptors agonist, induces myocardial injury, cardiac hypertrophy, left ventricular
In experiments conducted by Simko and his coworkers, rats were treated for 2 weeks. Groups included: 1] the group treated with melatonin (10 mg/kg/ day), 2] the group treated with isoproterenol (5 mg/kg/day i.p.), and 3] the group treated with both melatonin and isoproterenol. LV remodeling generated by isoproterenol was characterized by hypertrophy of both ventricles with increased hydroxyproline levels in the soluble and insoluble fraction of the collagenous proteins. Melatonin decreased the concentration of insoluble collagen and total collagen in the left ventricle. However, melatonin did not diminish the LV dysfunction. Melatonin failed to reduce the relative weight of either the left or right ventricle. The systolic blood pressure decline induced by isoproterenol was partially reversed by melatonin. Although hypertrophy of the LV was not diminished by melatonin, this molecule altered the LV structure in terms of the shift from a pathological left ventricular hypertrophy to a more physiological hypertrophy. Most importantly, melatonin decreased mortality and extended the average survival time in the rats. Tubulin proteins are important components of the cytoskeletal microtubule network and their alteration is part of the remodeling process in heart failure. The level of beta-tubulin was decreased in the left ventricular of the isoproterenol-treated rats, while melatonin administration partially prevented the beta-tubulin change. 45
In another study, Mukherjee et al. reported that melatonin (10 mg/kg/24 h) pretreatment enhanced the activity of metabolism-related enzymes, lowered the lipid peroxidation and pro-apoptotic processes of cardiac cells, and elevated the glutathione level, along with a decrease in cardiac injury biomarkers and an enhancement in morphology and cardiac function. 46 It also has been shown that melatonin reduced the tumor necrosis factor-alpha level and myeloperoxidase activity and increased the level of myocardial nitrite. These alterations were reported to be due to the protection of membrane bound ATPase enzymes suggesting that these mechanisms may be related to the improvement in isoproterenol-induced heart failure following melatonin administration. 5
Lead
Lead is an environmental contaminant that can cause oxidative stress and that can induce damage in different organs including cardiac tissue. The mechanism by which lead causes oxidative stress is not completely recognized. 7,47
The protective effect of melatonin (10 mg/kg, orally) against cardiotoxicity induced by lead acetate (15 mg/kg, i.p.) was evaluated in rats. The results showed that pretreatment of rats with melatonin decreased the concentration of lead in cardiac tissue suggesting that melatonin removed some of the lead from the cardiac tissue either by chelating it or by increasing clearance. Moreover in the same study, it was reported that significant damage to cardiac tissue with focal ischemia and a reduction of collagen in the heart which occurred after lead exposure was prevented or reduced by melatonin. 7
The level of two marker enzymes for organ damage, serum glutamic oxalacetic transaminase (SGOT) and lactate dehydrogenase 1 (LDH1), increased following exposure of rats to lead. The level of reduction of these two markers was diminished following administration of melatonin. Pro-oxidant enzymes such as xanthine oxidase (XO) and xanthine dehydrogenase (XDH) are involved in the production of ROS. Exposure to lead enhanced their activity which was prevented by melatonin. Additionally, it was observed that lead caused a reduction in the activity of cardiac pyruvate dehydrogenase (PDH) and several mitochondrial Kreb cycle enzymes such as ICDH, KGDH, and SDH. Melatonin restored the activity of these enzymes. 7
These results suggest that melatonin could be a potential factor against lead-induced oxidative stress and heart injury.
Dioxin: 2,3,7,8 Tetrachlorodibenzo-p-dioxin (TCDD)
Dioxins are environmental contaminants produced by engineering processes like incineration, manufacture of pesticides, and chlorine bleaching of paper. 2,3,7,8 Tetrachlorodibenzo-p-dioxin (TCDD), which is the most toxic member of the polychlorinated dibenzo-p-dioxins class of compounds, has been reported to cause severe adverse effects in animals such as immunotoxicity, carcinogenicity, and cardiotoxicity. Human exposure to TCDD has been associated with cardiovascular complication. 48,49 One mechanism by which TCDD causes cardiotoxicity is generation of ROS leading to an increase in TBARS levels and a decrease in SOD, GSH, and CAT in the heart. Melatonin ameliorates ROS generation by its antioxidant activity and also activates several antioxidative enzymes while decreasing lipid peroxidation. 49
Chronic heart failure is one of the disorders associated with wasting syndrome in humans. In a study conducted by Sarihan and colleagues, TCDD was administered to rats at a dose of 2 mg/kg orally for 45 days, resulting in loss of heart weight and body weight. There also was impairment in certain hemodynamic parameters such as MAP and BP after TCDD exposure. It was shown that administration of melatonin at a dose of 5 mg/kg/day (i.p.) for 45 days restored body weight loss and these hemodynamic disorders. Moreover, there was a significant reduction in PO2 saturation in rats exposed to TCDD. Melatonin supplementation significantly improved oxygen saturation compared to the TCDD exposed rats. In addition, disorder in cardiac rhythm and an elevation in block and duration of PR, QRS, and QT intervals were observed in TCDD exposed rats. Melatonin normalized heart rate and reversed the cardiac arrhythmias in the TCDD exposed rats. Histopathological changes as a result of TCDD exposure were significantly reduced after melatonin administration. 49
Sarihan and his colleagues have suggested that melatonin could be an effective agent against TCDD-induced cardiotoxicity. Further studies are required, however, to confirm the protective effects of melatonin in TCDD-induced cardiac injury. 49
Titanium dioxide (TiO2)
Titanium dioxide, also known as titania, is a natural substance used in sunscreens, paints, and as a food coloring agent. The clotting time of titanium dioxide films, in vitro, was longer than that of a low temperature isotropic pyrolytic carbon film (LTI-carbon), which has been used in artificial heart valves. Fewer platelets adhered to and were deformed on the TiO2 films compared to the LTI-carbon film. In an in vivo investigation, implanting either a LTI-carbon or titanium dioxide coated LTI-carbon film into the ventral aorta of a dog for 2 weeks showed that the amount of thrombus on the titanium dioxide coated LTI-carbon was about one-eighth to that of LTI-carbon. 50 Titanium dioxide has been formulated into nanoparticles (NP) to improve its hemocompatibility for use as a bactericidal agent in artificial heart valves. The evidence suggests that TiO2-NPs enter the cardiac tissue and has the potential to cause cardiotoxicity including inflammation, induction of chromatin condensation, nuclear fragmentation, caspase activation, and apoptosis. 51 –53
TiO2-NPs were administered to rats using two doses (600 and 1000 mg/kg/day) orally for 5 consecutive days. No noticeably influence was reported in either group on final body weight, heart weight or heart/body weight ratio. A significant increase in the serum levels of the cardiac function parameters, myoglobin, troponin T, and creatine kinase MB isozyme was observed in both treated groups. Oral administration of melatonin (200 mg/kg) or carnosine (200 mg/kg) for 3 weeks resulted in a significant reduction in these parameters compared to the TiO2-NPs treated rats. The decrease in serum creatine kinase was greater in the carnosine treated group than in the melatonin treated group; there were no differences in the myoglobin level or in troponin T in the carnosine and melatonin treated groups in comparison with rats exposed to the higher dose of TiO2-NPs. 52
Administration of TiO2-NPs at 600 and 1000 mg/kg/day for 5 days increased the levels of caspase-3 and Tail DNA (TDNA) and Tail Length (TL) which was more apparent in the high dosed rats. TiO2-NPs also elevated immunoglobulin G (IgG) serum level more significantly in rats treated with 1000 mg/kg/day. Oral intake of melatonin or carnosine reduced the elevation of these parameters with greater reduction in TDNA in the melatonin treated group. 52
In regard to the anti-inflammatory markers, a raise in serum levels of tumor necrosis factor-α (TNF-α), C reactive protein (CRP), and interleukin-6 (IL-6) occurred after oral administration of TiO2-NPs, regardless of dose. However, the increase was more pronounced in rats receiving the higher dose of TiO2-NPs. Oral administration of melatonin or carnosine caused a reduced level of these anti-inflammatory markers. 52
The results suggest that the severity of the cytotoxic effects of TiO2-NPs on cardiac tissue of rats was dose-dependent. The treatment with either melatonin or carnosine was effective in alleviating the deleterious impact of TiO2-NPs in rats exposed to low doses of the NPs. 52
Conclusion
Clinical and nonclinical evidence offers support for the potential beneficial effects of melatonin in certain types of drug/chemical induced cardiotoxicity. The findings reviewed in Table 1 indicate that melatonin is influential in cardiovascular physiology and in the reduction of damage to the myocardium. The useful effects of melatonin administration against cardiotoxic agents are due to its action against free radical scavenger activity, its indirect antioxidant properties, and its anti-inflammatory function. A better understanding of how melatonin interacts in agent-induced cardiotoxicity in patients is needed. Researchincluding mechanistic and clinical studies are necessary to sort out the potential usefulness of melatonin in cardiotoxicity.
Protective effects of melatonin against chemical-induced cardiotoxicity.

Footnotes
Abbreviations
Acknowledgment
The authors are thankful from Mashhad University of Medical Sciences.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
