Abstract
Objective:
To examine the biochemical and histopathological effects of ischemia/reperfusion (I/R) injury in a ruptured abdominal aortic aneurysm (RAAA) model in rats, and to investigate the potential protective role of resveratrol.
Methods:
Thirty-two male Sprague-Dawley rats were randomly assigned into four groups—control, I/R, sham (I/R + solvent/dimethyl sulfoxide), and I/R + resveratrol. The control group underwent midline laparotomy only. In the other groups, infrarenal vascular clamps were attached following 60-min shock to the abdominal aorta. Ischemia was applied for 60 min followed by reperfusion for 120 min. In the I/R + resveratrol group, intraperitoneal 10 mg/kg resveratrol was administered 15 min prior to ischemia and immediately before reperfusion. The I/R + dimethyl sulfoxide group received dimethyl sulfoxide, and the I/R group was given saline solution. All animals were sacrificed by exsanguination from the carotid artery at the end of the experiment. In addition to histopathological examination of the rat kidney tissues, malondialdehyde, glutathione, catalase, and nitric oxide levels were also investigated.
Results:
A decrease in glutathione, catalase and nitric oxide levels, together with increases in malondialdehyde levels, numbers of apoptotic renal tubular cells, caspase-3 levels, and tubular necrosis scores, were observed in the IR and I/R + dimethyl sulfoxide groups. In contrast, resveratrol increased glutathione, catalase and nitric oxide levels in renal tissues exposed to I/R, while reducing malondialdehyde levels, apoptotic renal tubular cell numbers, caspase-3 levels, and tubular necrosis scores.
Conclusion:
Our findings suggest that resveratrol can be effective against I/R-related acute kidney damage developing during RAAA surgery by reducing oxidative stress and apoptosis.
Keywords
Introduction
Ruptured abdominal aortic aneurysm (RAAA) is responsible for 1–2% of deaths in men over 65. 1 Population-based studies report that 90% of RAAAs are associated with mortality. However, different vascular surgery centers report mortality rates between 40% and 70%. 2 Despite advances in endovascular aneurysm repair techniques and perioperative patient management, no dramatic improvement has been achieved in RAAA patient mortality rates, in contrast to elective (non-ruptured) abdominal aortic aneurysm surgery. 2,3 Rapid surgery is the optimal method of treatment. Lower torso ischemia caused by cross-clamping of the abdominal aorta during surgical repair and hemorrhagic shock constitute the basis of “two-hit” ischemia/reperfusion (I/R) injury. 2 The synergic effect of these two events results in systemic inflammatory response syndrome (SIRS) characterized by increased microvascular permeability and neutrophil sequestration. The process begins with SIRS and provokes multiple organ failure and mortality with injury to distant organs such as the kidney. 1 –3
The anaerobic metabolic process during ischemia triggers irreversible damage by impairing cell membrane functions. Reperfusion enhances tissue oxygenation, but also leads to an inflammatory process that results in further tissue damage with the formation of reactive oxygen species (ROS). ROS cause lipid peroxidation by reacting with unsaturated fatty acids in the cell membrane.
2,4
Malondialdehyde (MDA) emerges with damage to unsaturated fatty acids caused by ROS, and is a widely employed marker for indicating oxygen-mediated tissue injury and lipid peroxidation.
2
Glutathione peroxidase (GSH) and catalase (CAT) are important endogenous antioxidant enzymes that protect cells against ROS damage and are used as markers of antioxidant activity. An increase in the levels of these enzymes is indicative of the severity of the oxidative stress occurring.
5
–7
Nitric oxide (NO), concerning whose role in I/R various suggestions have been made, is synthesized by the enzyme nitric oxide synthase (NOS) from
Postoperative treatment of RAAA patients in the intensive care unit is directed toward establishing hemodynamic stability and correcting acid base imbalances. However, there is still no specific treatment aimed at preventing frequent reperfusion injury and multiple organ failure, and at reducing mortality rates. 2 The present study investigated the potential protective effects of resveratrol against renal damage caused by RAAA surgery using biochemical and immunohistochemical analysis. The aim of the study was to offer a new perspective toward therapeutic methods that may be developed in the future.
Material and methods
Animals
This study was performed following receipt of approval from our university animal experiments ethical committee (no. 2017-24). Thirty-two male Sprague Dawley rats 3–4 months in age and weighing 257 ± 38 g were used. All rats were housed in the university animal application center at 22 ± 2°C room temperature and 55–60% humidity in a 12-h light:12-h dark cycle until the day of study. Rats were fasted for 12 h and allowed only access to water before the experiment. All animals received humane care according to the criteria outlined in the “Guide for the Care and Use of Laboratory Animals” prepared by the National Academy of Sciences and published by the National Institutes of Health.
Experimental groups
The RAAA model employed was based on that described in previous studies. 3,9,10 Rats were randomly assigned into four groups of eight animals each—control, ischemia/reperfusion (I/R), sham (I/R + solvent/dimethyl sulfoxide), and I/R + resveratrol. No procedure was performed on the control group other than aortic exploration. The other groups were exposed to 60-min shock, followed by ischemia for 60 min and reperfusion for 120 min.
Aortic clamping technique
Anesthesia was applied in such a manner as to preserve spontaneous respiration. Intraperitoneal 50 mg/kg ketamine hydrochloride (Ketalar®, Eczacıbaşı Parke-Davis, Istanbul, Turkey) and 10 mg/kg xylazine hydrochloride (Alfazyne®, Alfasan International B.V. Woerden, the Netherlands) were administered for anesthesia. Anesthesia was maintained with occasional ketamine administration as required. Anesthetized rats were immobilized in the supine position. The right internal jugular vein was located with surgical exploration and was cannulated for fluid and blood replacement. The left common carotid artery was then explored surgically for mean arterial pressure (MAP) monitoring and was cannulated with a 22G intracath. The arterial line was monitored using a three-way tap and a transducer set, and MAP was monitored throughout the experiment. A heat lamp was used to maintain the animals’ body temperature. Rats were given 3 mL/kg/h saline solution (0.9% NaCl) to compensate for insensible fluid losses.
Blood was gradually withdrawn from the carotid artery cannula into a plastic injector containing 500 IU heparin (Nevparin, 5000 U/mL, Mustafa Nevzat, Turkey) in all groups apart from the control group. Hemorrhagic shock was thus established by maintaining MAP at ≤50 mm Hg over 60 min, and aneurysm rupture was thus simulated. The blood collected was stored at room temperature. Once the shock stage had been completed, the abdominal region scheduled for surgery was shaved and sterilized with povidone-iodine solution. Laparotomy was performed with a midline incision in all groups. Rats in the control group received anesthesia for 4 h with no other surgical procedure being performed, after which the laparotomy incision was closed with 5/0 prolene sutures. In the groups exposed to I/R, the abdominal aorta was explored, and anticoagulation was established before placement of vascular clamps by the intravenous administration of 250 IU heparin. After 2 min, lower torso ischemia was induced with the attachment of bulldog clamps to the abdominal aorta from the infrarenal and iliac bifurcation levels. Following placement of the clamps, half of the blood previously collected and stored at room temperature was gradually returned using the internal jugular vein cannula. The surgical aortic repair stage was simulated with a period of ischemia beginning with the placement of the vascular clamps and lasting 60 min. At the end of the ischemia stage, all the remaining blood was immediately returned without removing the clamps. Following removal of the bulldog clamps, the laparotomy incision was closed with 5/0 prolene sutures, and the reperfusion stage was maintained for 120 min. MAP was kept at 100 mmHg with the administration of saline solution infusion when required. 3,10 Animals in the I/R + resveratrol group received 10 mg/kg intraperitoneal resveratrol 15 min prior to ischemia and immediately before reperfusion. 9 Animals in the I/R + dimethyl sulfoxide group received an equal quantity of dimethyl sulfoxide administered in the same manner, while those in the I/R received the same quantity of saline solution in the same manner. All rats were sacrificed by exsanguination from the carotid artery at the conclusion of the experiment.
Biochemical analysis
Tissue homogenization
A 20 mM pH 7.4 buffer solution consisting of 1 L sodium phosphate + 140 mM potassium chloride was first made ready. Next, 1 mL of this solution was added to 0.1 g of tissue. This mixture was then subjected to homogenization, after which all homogenates were centrifuged for 20 min at 2500 rpm at 4°C. The reactives were then raised to room temperature (18–25°C). MDA, GSH, CAT, and NO levels were investigated from the resulting supernatants. 11 –15
Determination of MDA levels
MDA levels were determined in our university laboratory by means of the enzyme-linked immunosorbent assay (ELISA) method (YL Biont Rat MDA kit, catalog no. YLA0029RA).
Test principle
This kit employed ELISA, based on biotin double antibody sandwich technology, to determine rat MDA levels. MDA was first added to the kit wells, these being pre-coated with MDA monoclonal antibody, and then incubated. Biotinylated anti-MDA antibodies were added, and these then combined with streptavidin-HRP to produce an immune complex. All unbound enzymes were eliminated after incubation and washing, and substrates A and B were then added. The solution first turned blue and subsequently yellow due to the presence of acid. The final shade of the solution was positively correlated with the amount of rat MDA present. 11
Determination of GSH levels
GSH concentrations in specimens were also measured in the same laboratory by means of ELISA (YL Biont Rat GSH kit, catalog no. YLA0121RA).
Test principle
This kit employed ELISA, based on biotin double antibody sandwich, technology to determine concentrations of rat GSH. Initially, GSH was added to wells which were already coated with GSH monoclonal antibody. Following incubation, biotinylated anti-GSH antibodies were added, and these combined with streptavidin-HRP, yielding an immune complex. Any remaining unbound enzymes were removed after incubation and washing, prior to the addition of substrates A and B. The acid turned the resulting solution first blue and subsequently yellow. The shade of the resulting solution was positively correlated with the concentration of rat GSH. 11
Determination of CAT levels
CO levels were determined in our university laboratory using ELISA (YL Biont Rat CAT kit, catalog no. YLA0123RA).
Test principle
This kit also employed ELISA based on biotin double antibody sandwich technology to determine concentrations of rat CAT. Briefly, rat CAT was added to wells containing a pre-coating of rat CAT monoclonal antibody. Following incubation, biotinylated anti-CAT antibodies were added, and these then combined with streptavidin-HRP, yielding an immune complex. After incubation and washing, any remaining unbound enzymes were removed, and substrates A and B were added. The acid turned the solution first blue and then yellow. The shade of the solution corresponded to the concentration of rat CAT. 11
Determination of NO levels
NO levels in specimens were also measured in the same laboratory using ELISA with a commercial nitrate/nitrite colorimetric assay kit (Cayman kit, catalog no. 780001).
Test principle
Total nitrite/nitrate concentrations can be accurately and simply measured with Cayman’s nitrate/nitrite colorimetric assay kits in a permits accurate and convenient measurement of total nitrate/nitrite two-stage process. In the first stage, nitrate was converted to nitrite by means of nitrate reductase. In the second step, the Griess reagents that convert nitrite into a deep purple azo compound were added. Photometric measurement of the absorbance resulting from this azo chromophore permitted a highly accurate calculation of NO2 − concentrations. 12 –15
Histopathological analysis
Kidney tissues with a volume of 1.5 cm3 were trimmed and fixed for 36 h in neutral formalin (Sigma-Aldrcih, Saint Louis, MO, USA). Following fixation, tissues were subjected to routine histological procedures. Tissues were passed through increasing ethanol series (Merck GmbH, Darmstadt, Germany) with a tissue processor (Shendon Citadel 2000, UK) for dehydration. They were then cleared through two series of xylol solution (Merck GmbH, Darmstadt, Germany), and finally embedded in soft paraffin (42–44°C, Merck GmbH, Darmstadt, Germany) and hard paraffin (52–54°C, Merck GmbH, Darmstadt, Germany). Kidney tissue samples were then placed into tissue embedding cassettes (Isolab GmbH, Germany) using a tissue embedding device (Leica, EG1150, Germany). Paraffin-embedded tissue sections 3–4 µm in thickness were taken using a rotary microtome (Leica RM2525, Germany) and placed onto poly-
Immunohistochemical (IHC) analysis
Anti-Cleaved Caspase-3 antibody (Rabbit polyclonal, ab2302, Abcam, UK) was used as the primary antibody for detecting apoptotic cells, together with secondary antibodies appropriate to the primary antibodies (Goat Anti-Rabbit IgG H&L (HRP), ab205718, Abcam, UK). Sections 2–3 µm in thickness were then taken from the kidney tissue paraffin blocks using a rotary microtome (Leica RM2525, Germany) and placed onto positively charged slides (Patolab, PRC). Sections were prepared with a closed and fully automatic system with a Leica Bond Max IHC stainer device (Leica microsystem, Melbourne, Australia) in accordance with the instructions of the manufacturer of the antibody kits. Sections were then counter-stained with Harris’ hematoxylin (Merck GmbH, Darmstadt, Germany).
Semi-quantitative analysis
Histopathological findings in H&E-stained sections were scored as shown in Table 1 using a previously described tubular necrosis scoring system. 16 Immunopositive cells to which cleaved caspase-3 primary antibody was applied were scored as shown in Table 2. Semiquantitative analyses were performed by two histopathologists blinded to the study groups. Twenty different randomly selected areas on sections from each rat were scored using a ×20 magnifying lens.
Jeong Sung et al.’s tubular necrosis score modified classification.

Grading of caspase-3 staining positivity scores.

Statistical analysis
All the data acquired in this study were analyzed using SPSS 20.00 (IBM Corp. IL, Chicago, USA) software. Non-parametric data obtained from histopathological analysis were calculated as median and 25–75% interquartile range values. Intergroup differences were analyzed using the Kruskal Wallis and Tamhane T2 tests. p values ≤0.05 were regarded as statistically significant.
Results
Biochemical analysis
An increase was determined in MDA levels in the IR and I/R + dimethyl sulfoxide groups compared to the control group (Table 3, p ≤ 0.05). In addition, kidney tissue MDA levels decreased significantly in the I/R + resveratrol group compared to the IR and I/R + dimethyl sulfoxide groups (Table 3, p ≤ 0.05).
Biochemical analysis results.

MDA: Malondialdehyde, GSH: Glutathione peroxidase, CAT: Catalase, NO: Nitric Oxide, I/R: Ischemia/Reperfusion, DMSO: Dimethyl sulfoxide, Res: Resveratrol.
GSH and CAT levels in kidney tissue decreased significantly in the IR and I/R + dimethyl sulfoxide groups compared to the control group (Table 3, p ≤ 0.05). In contrast, GSH and CAT levels in the I/R + resveratrol group were significantly higher than in the IR and I/R + dimethyl sulfoxide groups (Table 3, p ≤ 0.05).
NO levels in kidney tissue decreased in the IR and I/R + dimethyl sulfoxide groups compared to the control group (Table 3, p ≤ 0.05), but were significantly higher in the I/R + resveratrol group compared to the I/R and I/R + dimethyl sulfoxide groups (Table 3, p ≤ 0.05).
Urea and creatinine levels were significantly increased in IR and I/R + dimethyl sulfoxide groups compared to the control group (Table 3, p ≤ 0.05). On the other hand, urea and creatinine levels were significantly decreased in I/R + resveratrol group compared to IR and I/R + dimethyl sulfoxide groups (Table 3, p ≤ 0.05).
Histopathological analysis
A normal renal corpuscle and tubules were observed in control group sections (Figure 1(A) to (B); Table 4; TNS: 1.00 (0–1)). In contrast, widespread tubular necrosis and vascular congestion were observed in the I/R + dimethyl sulfoxide and I/R groups. Impaired brush border structures were present in the proximal tubules, with numerous vacuolizations in tubular epithelial cells (Figure 1(C) to (F); Table 4; TNS: 8 (8–8); TNS: 8 (7–9), respectively). On the other hand, we observed a decrease in vacuolization in tubular epithelial cells and necrotic tubules in the I/R + resveratrol group (Figure 1(G) and (H); Table 4; TNS: 3 (3–3)).

The representative light microscopic image of kidney tissue. H&E staining Proximal tubule (pt). Distal tubule (dt). Glomerule (g). Brush margin (arrow). (A) (×20) and (B) (×40): Control group section demonstrated that normal glomerular (g), proximal tubule (pt) and distal tubule (dt) histology (TNS score: 1 (0–1)). (C) (×20) and (D) (×40): Sections from the sham group show numerous necrotic tubule (arrow head). In addition, vacualation (spiral arrow) and loss of brush margins (tailed arrow) in the epithelial cells. Vascular congestion (arrowhead) can be seen in intertubular area (C) (TNS score: 8 (7–9)). (E) (×20) and (F) (×40): Sections from the I/R group numerous atypical proximal tubules with loss of brush margins (arrow) can also be seen. Vascular congestion (c) can be seen in interstitial spaces (tailed arrow) (TNS score: 8 (8–8)). (G) (×20) and (H) (×40): RES treatment group sections show typical glomerules (g), proximal tubules (pt) and distale tubules (dt) (TNS score: 3 (3–3)).
Tubular necrosis score (TNS) analysis results (median (25–75% interquartile range).

I/R: Ischemia/Reperfusion; DMSO: Dimethyl sulfoxide; Res: Resveratrol.
c p = 0.838 Compared with the I/R + DMSO group.
f p = 0.996 Compared with the I/R + DMSO group.
k p = 0.992 Compared with the I/R + DMSO group.
m p = 0.987 Compared with the I/R + DMSO group.
Kruskal Wallis–Tamhane’s T2 test.
Immunohistochemical analysis
Tubular epithelial cells from the control group exhibited a normal appearance (Figure 2(A), Table 5; IHC positivity score 0.00 (0–1)). However, we determined an increase in apoptotic tubular epithelial cells in the I/R + dimethyl sulfoxide group (Figure 2 (B), Table 5; IHC positivity score 2 (2–2)). Similarly, we observed a significant increase in cleaved caspase-3 positivity in tubular epithelial cells in the I/R group compared with the control group (Figure 2 (C), Table 5; IHC positivity score 3 (2–3)). We also determined a significant decrease in apoptotic tubular epithelial cells in the I/R + resveratrol group compared with the I/R + dimethyl sulfoxide and I/R groups (Figure 2 (D), Table 5; IHC positivity score 1 (1–1)).
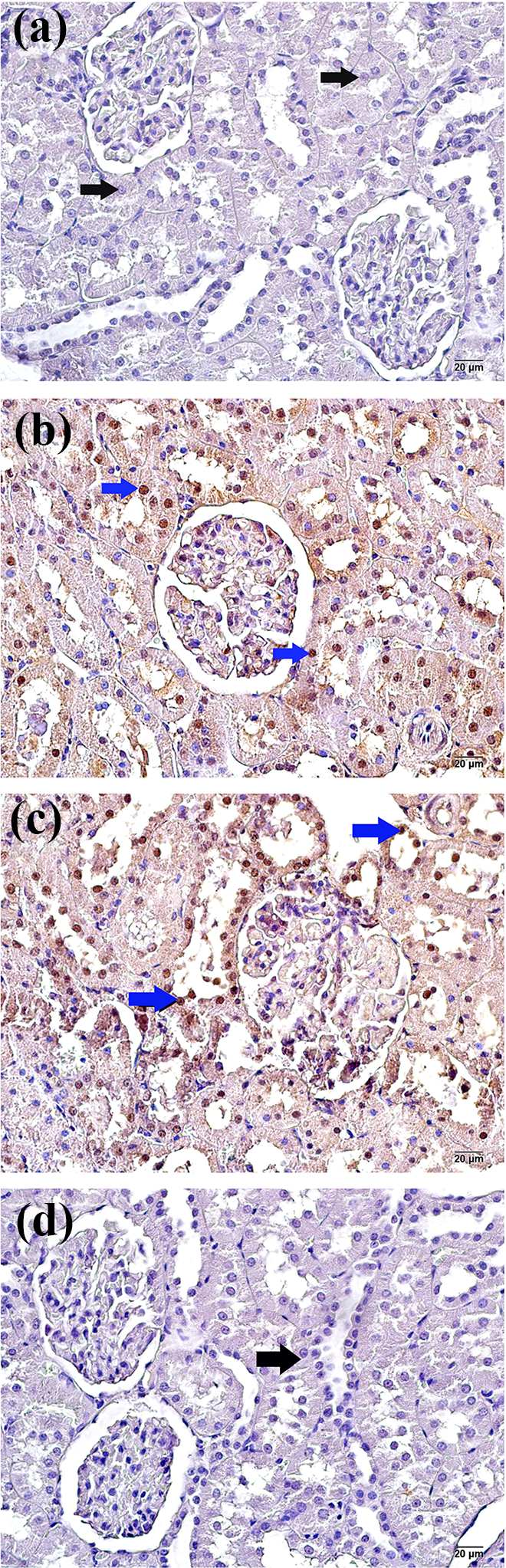
The representative light microscopic image of cleaved caspase-3 primary antibody incubed. (A) (×40): Sections from the control group show a normal structure in tubular epithelial cells (arrow) (cleaved caspase-3 positivity score: 0 (0–1)). (B) (×40): I/R+dimethyl sulfoxide group sections apoptotic epithelial cells (blue arrow) in the proximal and distal tubule (caspase-3 positivity score: 2 (2–2)). (C) (×40): I/R group section showing intense caspase-3 positivity in proximal and distal tubule epithelial cells (cleaved caspase-3 positivity score: 3 (2–3)). (D) (×40): RES treatment group sections showing a typical structure in proximal and distal tubule epithelial cells (arrow) (caspase-3 positivity score: 1 (1–1)).
IHC positive scores results (median 25–75% interquartile range).

IHC: Immune Histo Chemical, I/R: Ischemia/Reperfusion, DMSO: Dimethyl sulfoxide, Res: Resveratrol.
Kruskal Wallis–Tamhane’s T2 test.
Discussion
The prevalence of major complications and mortality during intensive care follow-up is higher after RAAA repair than with other interventions requiring laparotomy for surgical access. 17 In order for bleeding to be brought under control, surgical exploration must be completed as quickly as possible, and a cross-clamp must be applied to the abdominal aorta. However, aortic cross-clamp attachment induces SIRS development. Excessive bleeding, massive blood transfusion, acidosis, clotting problems, and reperfusion damage following removal of the cross-clamp in particular all make control of SIRS difficult in these patients by exacerbating the inflammatory process. 17,18 Complications developing after surgery generally derive from damage in organs distant from the site of the injury. Inflammatory mediators passing to distant organs through the circulation play an important role in damage by activating neutrophils present in these organs. Widespread activation of inflammatory mediators is the most critical stage on the pathway leading to multiple organ failure, the most common cause of death in intensive care patients. The death rate following the failure of one organ is approximately 30%, and every additional organ failure increases the risk still further. 17 The kidneys are essential organs whose functions are impaired due to I/R damage in patients undergoing RAAA repair, and are one component of multiple organ failure. 18
It is generally possible for cross-clamps to be attached to the infrarenal abdominal aorta during RAAA repair. However, the supra-renal abdominal aorta may also sometimes have to be clamped to control rapid bleeding during emergency surgery. This increases the possibility of acute kidney failure by preventing blood flow to the renal arteries. Cross-clamps have been attached to the supramesenteric abdominal aorta in some previous similar studies. 3,10 In the present study of the distant organ effects of I/R on the kidneys, the cross-clamp was attached at the infrarenal level. Our purpose in doing this was to prevent the biochemical and histopathological outcomes of localized ischemic damage in the kidney and other internal organs, particularly the intestines.
Although the mechanism involved in acute kidney injury developing following RAAA repair is not yet fully understood, studies have implicated lipid peroxidation and oxidative damage associated with ROS production. 19 –21 The principal sources of ROS that form during reperfusion are activated neutrophils, mitochondrial electron transport chain leakage, and the xanthine/xanthine oxidase system. 2,19 Toklu et al. reported that I/R caused by clamping of the abdominal aorta leads to a significant proportion of oxidative damage with lipid peroxidation in bladder tissue, and also increases MDA and reduces GSH levels. 9 I/R studies involving clamping of the renal artery have reported an increase in MDA levels in renal tissue, consumption of the antioxidant pool through a decrease in GSH and CAT levels, and neutrophil infiltration accompanying oxidative tissue damage. 5,7,20,21 To the best of our knowledge, GSH levels in the kidneys have not been investigated in previous studies involving an abdominal aorta clamping model in rats. Consist with previous results, I/R resulting from aortic cross-clamp attachment increased MDA levels in kidney tissue and lowered GSH and CAT levels.
The role of NO during I/R injury is controversial. Some studies have reported that kidney injury during I/R is associated with abnormally low NO levels. ROS may lead to an increase in renal vascular resistance by compromising the integrity of the endothelium permitting NO production. In addition, NO has been reported to reduce leukocyte adhesion, neutrophil infiltration, and the formation of inflammatory mediators during I/R. 7,12 In their study involving renal artery clamping, Chander et al. observed that in addition to causing histopathological changes in kidney tissue, I/R injury significantly lowered GSH, CAT and NO levels. Some other studies have reported that NO released from activated leukocytes at the beginning of I/R facilitates peroxynitrite production by binding to superoxide molecules. Peroxynitrite possesses high toxicity and triggers the peroxidation of lipids in the cell membrane and apoptosis. 5,6 One study involving clamping of the renal artery reported that MDA and NO increased in renal tissue, while GSH and CAT decreased. 5 In addition, NO has been reported to be involved in the inflammatory process and to mediate cell damage developing during I/R. 5,6 The present study determined a decrease in NO levels in addition to antioxidant enzymes in renal tissue subjected to I/R.
Gulmen et al. observed focal glomerular necrosis, dilatation in Bowman’s capsule and tubules, degeneration in the tubular epithelium, and necrosis in kidney tissue following induction of I/R with infrarenal aortic clamping. 22 Mohey et al. reported that I/R resulted in morphological changes in rats such as glomerular damage, detachment of the basement membrane from glomeruli, loss of brush border, tubular dilatation, atrophy, and neutrophil accumulation. 21 Şener et al. showed that I/R increased levels of the proinflammatory cytokine TNF-α and neutrophil infiltration in kidney tissue. They particularly reported that TNF-α was associated with glomerular cell infiltration, fibrin accumulation, and endothelial damage, and also induced renal cell apoptosis and kidney failure. 20 Similarly, in the present study, we observed widespread tubular necrosis, vascular congestion, impairment of brush border structures in proximal tubules, and numerous vacuolizations in tubular epithelium cells in kidney tissues following I/R.
Caspase-3 is an important enzyme involved in inflammation and apoptosis. 23 Studies have shown that caspase-3 activity, involved in the irreversible terminal stage of apoptosis, increases with I/R and that I/R leads to apoptosis in renal tubular epithelial cells. 23 –25 Yang et al. showed that caspase-3, an apoptosis marker, increased in both renal tubular epithelial cells and in microvascular endothelial cells in the early stages of acute kidney damage induced with a renal artery I/R model. 24 Adali et al. reported increased caspase-3 in renal tubular epithelial cells following I/R induced by infrarenal abdominal aortic clamping, together with apoptosis, degenerative necrotic tubular epithelium, disorganized tubular structures, and epithelial separation from the basal membrane. 25 Similarly, in the present study, we observed a marked increase in apoptotic tubular epithelial cells and caspase-3 positivity in renal cells.
Studies have shown that resveratrol exhibits anti-inflammatory and anticarcinogenic effects in addition to lipid metabolism modulation. Its anti-inflammatory effect is associated with suppression of oxidation, inflammatory mediator expression, and leukocyte activation. Resveratrol prevents leukocyte adherence to the endothelium and leukocyte infiltration, and increases blood flow to organs by reducing plasma viscosity. 8,9,20,26 Although studies have investigated the antioxidant effects of resveratrol, to the best of our knowledge, no research has to date examined its effect on distant organ damage caused by abdominal aortic clamping. 8,9,20 Xia et al. reported that resveratrol increased the levels of antioxidant enzymes in the cardiovascular system and accelerated ROS detoxification. 8 Toklu et al. showed that resveratrol entirely eliminated I/R-related oxidative damage and neutrophil accumulation in bladder tissue, as well as increasing levels of such endogenous antioxidants as GSH and CAT and reducing those of MDA. 9 In their renal artery clamping study, Şener et al. showed that resveratrol protected the GSH pool in kidney tissue exposed to I/R and caused a decrease in MDA levels. Those authors reported that resveratrol entirely eliminated I/R-associated oxidative kidney damage and tissue neutrophil accumulation. They also emphasized that it ameliorated kidney function impairment by protecting living cells against oxidative injury. 20 Chander et al. observed that resveratrol ameliorated renal functions and morphological changes in kidney tissue exposed to I/R, and increased depleted antioxidant enzyme and NO levels. Those authors also reported that resveratrol exhibited its renoprotective effect by means of NO. 7 Similarly, in the present study, we observed that resveratrol raised GSH, CAT and NO levels that had been lowered by I/R in kidney tissue, while also reducing MDA elevation. In addition to the biochemical findings, we also determined decreased vacuolizations in tubular epithelial cells, apoptotic tubular epithelial cells, and necrotic tubules in the I/R + resveratrol group.
In addition to our findings, there are also a number of limitations to the present study. It is possible that dimethyl sulfoxide used as a solvent in the preparation stage for resveratrol administration to rats may have affected our results. However, every endeavor was made to prevent this by establishing a group in which dimethyl sulfoxide only was administered to healthy rats exposed to I/R. Additionally, evaluation of I/R damage with agents such as other antioxidant enzymes and proinflammatory cytokines in larger patient groups might further enhance the value of our findings.
To the best of our knowledge, this is the first study in the literature to investigate the effects of resveratrol on acute kidney injury in a RAAA model. In RAAA, the ischemic process in the kidneys begins with hemorrhagic shock that develops in the preoperative period. Applying a clamp to the abdominal aorta during surgery and removing th clamp after repair exacerbates I/R damage. Surgical treatment of many patients is completed quickly and quite successfully. Nevertheless, acute kidney failure in intensive care after RAAA repair, and subsequent multiple organ failure, remains an important problem for cardiovascular surgeons. The development of a specific treatment capable of reducing I/R damage inducing acute kidney failure will have a critical role to play in reducing mortality rates. Since the benefits of a treatment applied after the development of acute renal failure may be limited, it may be more useful to apply such a treatment before the clamp is installed. The findings of the present study show that resveratrol exhibited protective effects against I/R-induced kidney damage in rats. Resveratrol, a phenolic compound, reduced levels of MDA, the end product of lipid peroxidation, in kidney tissues exposed to I/R, and increased GSH, CAT and NO levels. It also significantly reduced histopathological changes associated with I/R-induced renal damage, apoptotic tubular epithelial cells, and caspase-3 positivity. These results suggest that resveratrol may in the future play a role in the treatment of kidney failure developing due to I/R after RAAA surgery.
Footnotes
Abbreviations
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All animals received humane care according to the criteria outlined in the “Guide for the Care and Use of Laboratory Animals” prepared by the National Academy of Sciences and published by the National Institutes of Health.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
