Abstract
Endemic goitre and associated iodine deficiency disorders (IDDs) are a major concern in public health even in the period of post salt iodization in many regions. Among others the consumption of cyanogenic plants found responsible for the persistence of such diseases. Bamboo shoots (BS) is one such cyanogenic plant food that caused disruption of certain thyroid hormone synthesizing regulatory element as has already been reported in our earlier study. In this investigation the possible mechanism of thyrocytes disruption along with interruption of thyroid hormone biosynthesis by BS has been worked out. Commonly consumed BS, Bambusa Balcooa Roxb (BBR) water extract was analysed by GC MS; three doses below IC50 were administered to thyrocytes in culture with and without iodine. Expressions of thyroglobulin (Tg), pendrin (PDS) and monocarboxylate transporter 8 (MCT8) were evaluated in thyrocytes with cell cycle analysis, reactive oxygen species (ROS) generation, DNA oxidation and apoptotic regulation through Bax, Bcl-2 and p53. Phytochemical analysis of BBR extract revealed the presence of precursors and metabolic end products of cyanogenic glycosides. Dose dependent decrease in expression of Tg and PDS with concomitant decrease in gene expression of these with MCT8 were observed. Increased ROS, DNA oxidation and associated imbalance were found through increased Bax and p53 with decreased Bcl-2 that perturbed thyrocytes cell cycle. Cyanogenic constituents of BBR generates ROS associated oxidative changes in thyrocytes with DNA damage and oxidation and cell cycle disruption followed by inhibition of thyroid hormone synthesizing regulatory elements; addition of extra iodine showed partial prevention.
Introduction
Thyroid gland is responsible for production of the vital metabolic hormones thyroxine (T4) and tri-iodothyronine (T3). Besides iodine the functioning of the thyroid gland is affected by many environmental factors present in food and water. 1 An understanding of the interactions of the environmental agents containing antithyroidal constituents with thyroid and its dysfunction would provide insight to the prevailing thyroid disorders. Information regarding changes at the molecular and cellular level caused by such agents will help to mitigate long standing thyroid disorders affecting 4.2 million people in India alone. 1
Bamboo shoots (BS), a plant containing dietary goitrogens of cyanogenic origin is eaten quite popularly in the South East Asian regions and as an almost regular food item in the North Eastern states of India. 2 Several tribes as well as most inhabitants of this region consume this plant as a staple food in a variety of preparation like soups, curries, pickles and with meat. 3 Presently, BS has gained a worldwide popularity in a variety of different forms including chips and a thickener. It has been researched for its nutritive properties and its high content of fibres, vitamins and the like compounds have made it popular as a functional food. 4
The goitrogenic constituents of BS, cyanogenic glycosides, glucosinolates and thiocyanate are known to interfere with thyroid hormone synthesis on regular consumption. 5 The goitrogen content of cyanogenic plants including BS that grows in different geographical regions could perhaps vary due to ecological variations (soil type, temperature, rainfall, genetic variation, etc.) but that has not been studied widely. The presence of cyanogenic constituents in BS having antithyroidal properties, however, is well established. 2 –8 There are a few studies indicating differences in hydrogen cyanide (HCN) content in BS which is the mother constituent of cyanogenic compounds. 9,10 The amount of goitrogenic constituents in BS that has been used in this study and some other forms of the same geographical region has been reported to contain about 650 mg/kg wet weight. 5 –7 It has been reported in previous in vivo studies that intake of BS inhibits thyroid peroxidase (TPO) – the most important regulatory enzyme of thyroid hormone synthesis in rats and causes endemic goitre in children. 8 It has been reported that after exposure of Bambusa Balcooa Roxb (BBR) on thyrocytes cellular hydrogen peroxide (H2O2) concentration was increased while the antioxidants levels were decreased. Consequently nitric oxide (NO) and lipid peroxide (LPO) levels were enhanced which in turn caused DNA and cellular damage as evident from lactate dehydrogenase levels along with the decrease in mRNA and protein expressions of sodium iodide symporter (NIS), TPO and Paired box gene 8 (PAX8). 7
In this study the effects of BS on expression of other thyroid hormone synthesizing regulatory elements thyroglobulin (Tg) and pendrin (PDS) along with the mRNA expression of those with monocarboxylate transporter 8 (MCT8) that transports T3 and T4 outside thyroid follicular cell into circulation were studied. Cell cycle analysis and apoptotic control through Bcl-2 Associated X Protein (Bax), B-cell lymphoma 2 (Bcl-2) and tumour suppressor protein 53 (p53) levels have also been investigated. The generation of reactive oxygen species after exposure of BBR extract was further studied with DNA oxidation in thyrocytes. The overall observations would sketch out the possible mechanism of BS mediated antithyroidal action in thyrocytes.
Materials and methods
Chemicals and reagents
All reagents used in this study were of molecular biology grade and were purchased from SRL India and Sigma Aldrich, USA. Monoclonal antibodies to anti-pendrin, anti-GAPDH was purchased from biorbyt technologies and anti-thyroglobulin and secondary antibodies were purchased from Santa Cruz Biotechnologies (CA, USA). RNA extraction Kit and SYBR green were purchased from Invitrogen (Thermofischer Scientific, USA). Bax and Bcl-2 ELISA kit was purchased from Novus Biologicals. p53 ELISA kit was purchased from RayBiotech. 8-hydroxy-2’-deoxyguanosine (8-OHdG) DNA oxidation kit was procured from JaICA, Japan; Propidium Iodide (PI)/RNAse solution from BD Pharmingen. Reactive Oxygen Species detection kit was purchased from Cell Biolabs, Inc.
Collection, extraction and estimation of dietary goitrogens in bamboo shoots (Bambusa balcooa Roxb)
Voucher specimen of bamboo shoots collected from Manipur, India during monsoon season; it was submitted to Botanical Survey of India (BSI), Howrah, West Bengal, India bearing number PHY/CU/BS/27/2018. It was acknowledged to be Bambusa balcooa Roxb (BBR). BBR was then sun dried and extracted in ultra-pure water (1 kg raw Bambusa balcooa Roxb in 1litre) and applied in doses to thyrocytes in culture.
Gas chromatography mass spectrometry (GC/MS) analysis of BBR extract
Ten milligram sample of BBR extract was dissolved in methanol (MeOH) and water (H2O) of High Performance Liquid Chromatography (HPLC) grade respectively. Then 100 µl of the dissolved BBR extract was evaporated to dryness in microcentrifuge tubes. The obtained residue was dissolved further in 10 µl of methoxyamine hydrochloride (20 mg/ml in pyridine) and subsequently shaken for 90 mins at 30°C. The mixture was derivatised with 90 µl of N-Methyl-N-(trimethylsilyl)-trifluoroacetamide (MSTFA) to increase its volatility to get a better chromatographic separation of metabolites at 37°C for 30 mins. FAME markers (Fatty Acid Methyl Esters markers) [a mixture of internal Retention Index (RI) markers] were prepared using C8, C10, C12, C14, C16, C18, C20, C22, C24 and C26 fatty acid methyl esters of linear chain length prepared in Chloroform (HPLC) at a concentration of 0.8 mg/ml (C8–C16) and 0.4 mg/ml (C18–C26) was added. 11
Chromatographic conditions
GC/MS analysis was carried out following the method of Kind et al. 11 with little modification. 12 DB-5MS capillary column [Agilent J &W; GC Columns (USA), length 30 m + 10 m Duraguard, diameter 0.250 mm narrowbore, film 0.25 µm] was used. An injection was given in sandwich mode with fast plunger speed without viscosity delay or dwell time. The analysis was performed under the following temperature programme: oven ramp 60°C (1 min hold time) to 325°C at 10°C/min; 10 mins hold before cool down, 37.5 mins run time. The injection temperature was set at 250°C, the MS transfer line at 290°C, and the ion source at 230°C. Helium was used as the carrier gas with a constant flow rate of 2.5 ml/min (carrier linear velocity 57.95 cm/s).
One microlitre of sample was injected via split less mode onto the GC column. Prior to analysis the method was calibrated with the FAME standards available with the Fiehn GC/MS Metabolomics library (2008) (Agilent Chem Station, Agilent Technologies Inc., Wilmington, USA). Identification of the metabolites was done by comparing the fragmentation patterns of mass spectra and retention times (RT) with entries of mass spectra and retention time in Agilent Fiehn Metabolomics Library using Agilent Retention Time Locking (RTL) method. Fiehn retention indices were also compared. AMDIS (Automated Mass spectral Deconvolution and Identification System) was used to deconvolute GC/MS analysis and to identify the chromatographic peaks. Identification of some metabolites was further validated by comparing the retention time (RT), retention index (RI) and mass spectra (MS) of standard compounds available in laboratory.
Animals
Female adult rats of Wistar strain weighing ∼120–140 g (purchased from registered animal breeders, Kolkata) were acclimatized under laboratory conditions. Food and water was given ad libitum and animals were maintained at 12 h light and dark cycle. 4 All animal experiments were performed adhering to the guidelines of the Institutional Animal Ethical Committee (IAEC), Department of Physiology, University of Calcutta, Kolkata, India (approval number: IAEC/IV/AC/Proposal/AC-01/2017, dated: 15/03/2017). The animals were sacrificed on day of experiment by cervical dislocation for collection of thyroid gland.
Cell culture
Single cell suspensions of thyroid glands (excised aseptically from rats) were made by passing the cell population through a nylon mesh with 60 µm pore size in the media RPMI 1640. Lysis of red blood cells (RBCs) was obtained by incubating the cells in 0.83% Ammonium Chloride solution for 10 mins at 4°C. Thyroid follicular cells were categorized by Wright-Giemsa staining and viability was measured to be >95%. Isolated cells were then plated in 35 mM plates at 106 cells/ml, in RPMI 1640 in triplicate (for each experiment) in presence of 100 U/ml streptomycin and 50 U/ml penicillin and 750 μg/ml amphotericin and 550 IU/ml gentamycin and in different experimental conditions for 48 h at 37°C in a CO2 incubator. 13 Iodine at 5 µm/ml 14 and thyroid stimulating hormone (TSH) at 1 mU/ml 15 was added as per experimental condition.
Assessment of inhibitory concentration (IC50) via cytotoxicity (MTT) assay and treatment groups
Thyroid follicles obtained from rats were cultured for 24 hours (h) before treatment. The effect of the treatment of different doses of BBR extracts (1 ng/µl to 100 ng/µl) on the viability of cells via Trypan blue exclusion test was evaluated. In this test, viable cells had clear cytoplasm, while dead cells stained blue. Inhibitory concentration (IC50) was calculated from the percentage of the number of viable cells divided by the number of total cells and multiplying by 100 or % viable cells. After determination of IC50, three doses of BBR extract below IC50 that is 3 ng/μl, 6 ng/µl and 12 ng/µl were applied to thyroid cells in culture for subsequent experiments. Thyroid cells were incubated with iodine at normal levels (5 μM/ml) in certain cases. The treatment groups were as follows: CON: RPMI 1640 media only DI (D1): 3 ng/µl BBR extract DII (D2): 6 ng/µl BBR extract DIII (D3): 12 ng/µl BBR extract I: 5 μm/ml KI DI + I (D1 + I): 3 ng/µl BBR and 5 μm/ml KI DII + I (D2 + I): 6 ng/µl BBR and 5 μm/ml KI DIII + I (D3 + I): 12 ng/µl BBR and 5 μm/ml KI
Western blot
Whole cell lysates were prepared by washing cells in PBS and followed by lysis in ice cold buffer (50 mM Tris-HCl, 150 mM NaCl, 1 mM EGTA, 1 mM EDTA, 20 mM NaF, 100 mM Na3VO4, 0.5% NP 40, 1% Triton X-100, pH 7.4) with freshly added protease inhibitor cocktail (Sigma Aldrich, USA). The lysate was centrifuged at 11,100 rpm for 20 mins; the supernatant was collected, aliquoted and stored at −80°C.
10% SDS polyacrylamide gel was used for electrophoresis of proteins. The gel was transferred onto a PVDF membrane using a semi-dry transfer apparatus, and the membrane was blocked in 5% bovine serum albumin for 1–2 h at room temperature. The membrane was washed with TBST (50 mM/L Tris-HCl, pH 7.6, 150 mM/L NaCl and 0.1% Tween 20). The membrane was then incubated with primary antibodies (1:2500) overnight at 4°C. The membrane was washed again and incubated with appropriate secondary antibodies (1:3000) for 4 h. Blots were developed using NBT/BCIP (1:1) dye in the dark. In analogous experiments, immunoblotting of beta actin was done to confirm equal loading. The blots were analysed by Image J software. 16
RNA extraction and real time PCR
RNA extraction minikit (Invitrogen) was used to extract total RNA following the manufacturer’s instructions. Real-Time PCR amplifications were performed using Platinum SYBR Green 135 qPCR Super Mix-UDG (Life Technologies, Carlsbad, CA). Two micrograms of total RNA was utilized for reversed transcription. One microlitre of the product reaction was then diluted using a Real-Time PCR buffer. The reaction conditions consisted of one step at 50°C for 2 mins and 95°C for 5 mins, followed by 40 cycles of three steps: 20 s at 95°C for denaturation, 60 s at 60°C for annealing and 20 s at 72°C for extension. The threshold cycle (Ct) was recorded in triplicate for each sample. Relative changes in gene expression (Gene-specific primers sequences are described in the supplementary section) were calculated using the 2−ΔΔCt method and GAPDH was used as internal control (to normalize target genes). 17
Cell cycle analysis
Cultured cells were fixed with chilled ethanol. The cells were then washed and incubated with PI/RNAse. Cell cycle phase distribution was studied using a flowcytometer. 18
Measurement of Bax, Bcl-2 and p53 levels
Bax and bcl-2 levels were measured using kits as per manufacturer’s protocol. Levels of p53 were also measured using kit from Raybiotech as per manufacturer’s protocol.
Measurement of reactive oxygen species
Reactive Oxygen Species was measured colorimetrically using OxiSelect ROS Assay Kit as per manufacturer’s protocol.
Measurement of DNA oxidation via 8-hydroxy-2′-deoxyguanosine (8-OHdG) levels
8-hydroxy-2′-deoxyguanosine (8-OHdG) levels were assayed using kit as per manufacturer’s protocol.
Statistical analysis
All values are expressed as mean ± SD. Significant differences between the groups were determined with Graphpad prism using one-way analysis of variance (ANOVA) and the group means were compared by using Dunnett’s test and P/α values lower than 0.05 (p < 0.05) were considered significant.
Results
GC MS analysis for characterization of phytochemicals in BBR extract
Gas chromatography and mass spectrometry study of BBR water extract revealed the presence of different organic acids, fatty acids, sugar alcohols along with precursor and catabolic products of cyanogenic glycosides along with isothiocyanates. Detected compounds have been represented in Table 1 and total ion chromatogram has been represented in Figure 1.
Metabolites identified by gas chromatography mass spectrometry (GCMS) in BBR aqueous extract.
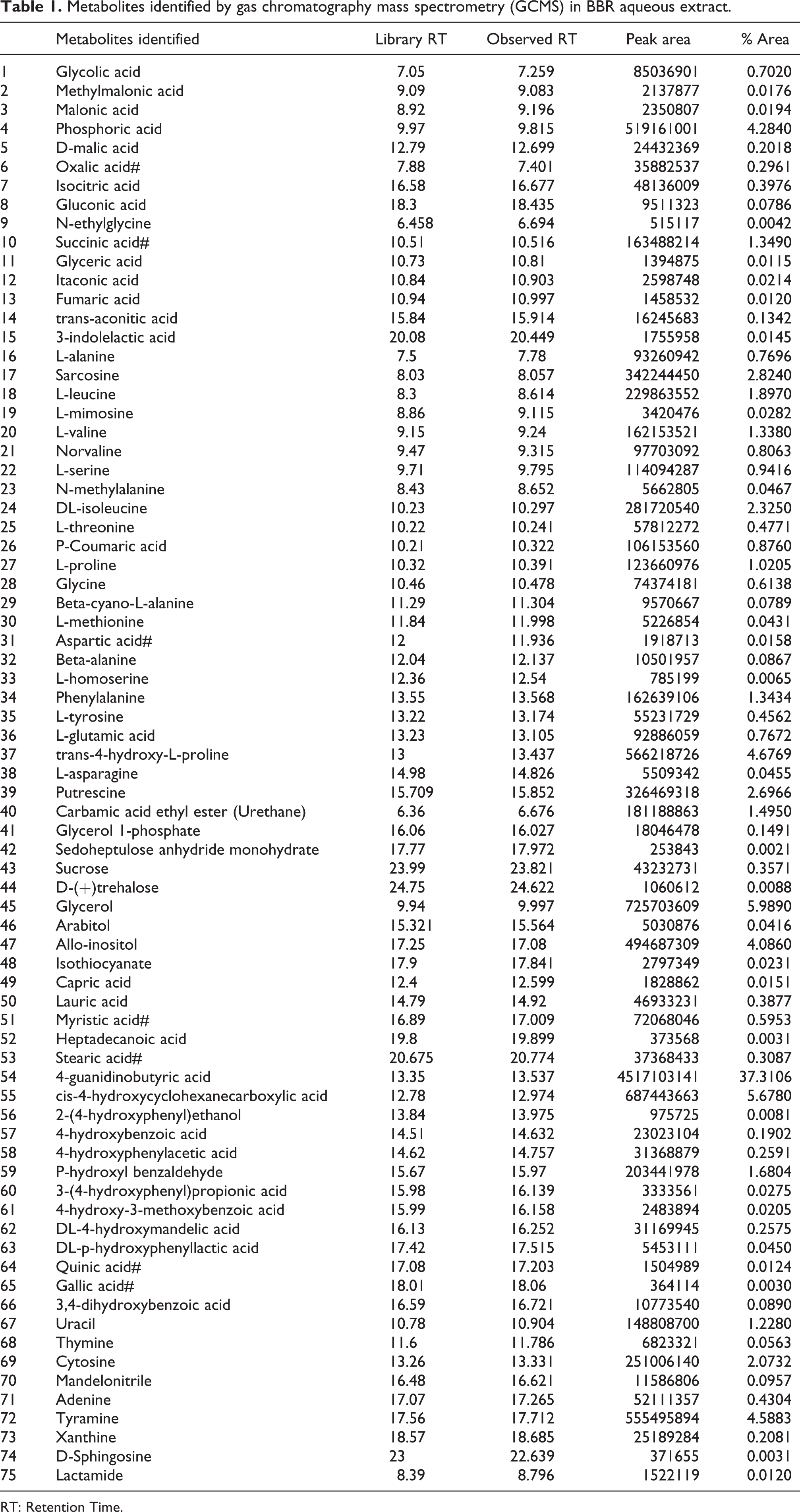
RT: Retention Time.

Total ion chromatogram (TIC) of Bambusa Balcooa Roxb (BBR) water extract by gas chromatography mass spectrometry with precursors and end products of cyanogenic glycosides marked. (1) P-Coumaric acid, (2) L-tyrosine, (3) Isothiocyanate, (4) P-hydroxyl benzaldehyde, (5) Mandelonitrile.
Cytotoxicity and IC50 of BBR extract on thyroid cell
On exposure to BBR extract there was a dose-dependent increase in cell death in the thyroid cells. IC50 was determined to be 24 ng/µl of antithyroidal substances (cyanogenic glycosides, glucosinolates and thiocyanate) in bamboo shoots.
BBR extract modulates Thyroglobulin (Tg) TG and pendrin (PDS) protein expressions
Thyroglobulin (Tg) and pendrin (PDS) expressions were decreased dose-dependently on exposure to BBR extract however, improved partially on iodine supplementation (Figure 2).

Effect of different concentrations of BBR extract on thyroglobulin (Tg) and pendrin (PDS) expression with and without extra iodine in relation to control. A and B (I) Immunoblot. A and B (II) Densitometric analysis. Data is represented as mean ± SD, n = 3, values as tested by ANOVA (P < 0.05) followed by Dunnett’s test. ‘*’ represents significant difference between control and all treated groups (only BBR extract and BBR extract with iodine treated groups), ‘#’ represents significant difference between only BBR extract and BBR extract with iodine treated groups.
BBR extract regulates the expression of genes involved in thyroid hormone synthesis
Subsequently, mRNA levels of these Tg and PDS genes were further analysed by Real Time Quantitative PCR (RT-qPCR). Expression of PDS at the transcription level was decreased significantly only after the highest dose of BBR extract while the expression of Tg were decreased significantly and dose dependently. Monocarboxylate transporter 8 (MCT 8) transports T3 and T4, its expression also decreased dose dependently on exposure to BBR extract. Addition of extra iodine likely improved expressions of those genes to a certain extent (Figure 3).

Effects of different doses of BBR extract along with and without extra iodine on mRNA expression of (A) thyroglobulin (Tg), (B) pendrin (PDS) and (C) MCT 8 as estimated by real time PCR. Data is represented as mean ± SD, n = 3, values as tested by ANOVA (P < 0.05) followed by Dunnett’s test. ‘*’ represents significant difference between control and all treated groups (only BBR extract and BBR extract with iodine treated groups), ‘#’ represents significant difference between only BBR extract and BBR extract with iodine treated groups.
BBR extract causes disruptions in cell cycle of thyroid cells
A significant dose dependent increase in the percentage of cells in the sub G1 phase was observed, while cells remained more or less constant in G0/G1 phase of the cell cycle as compared to control. Addition of extra iodine improved such changes to a certain extent; while the S phase and G2/M phases of the cell cycle were decreased dose dependently but significantly, however, recovered to an extent after addition of extra iodine (Figure 4). The FACS results indicate increased DNA fragmentation and an imbalance of cell death and proliferation.
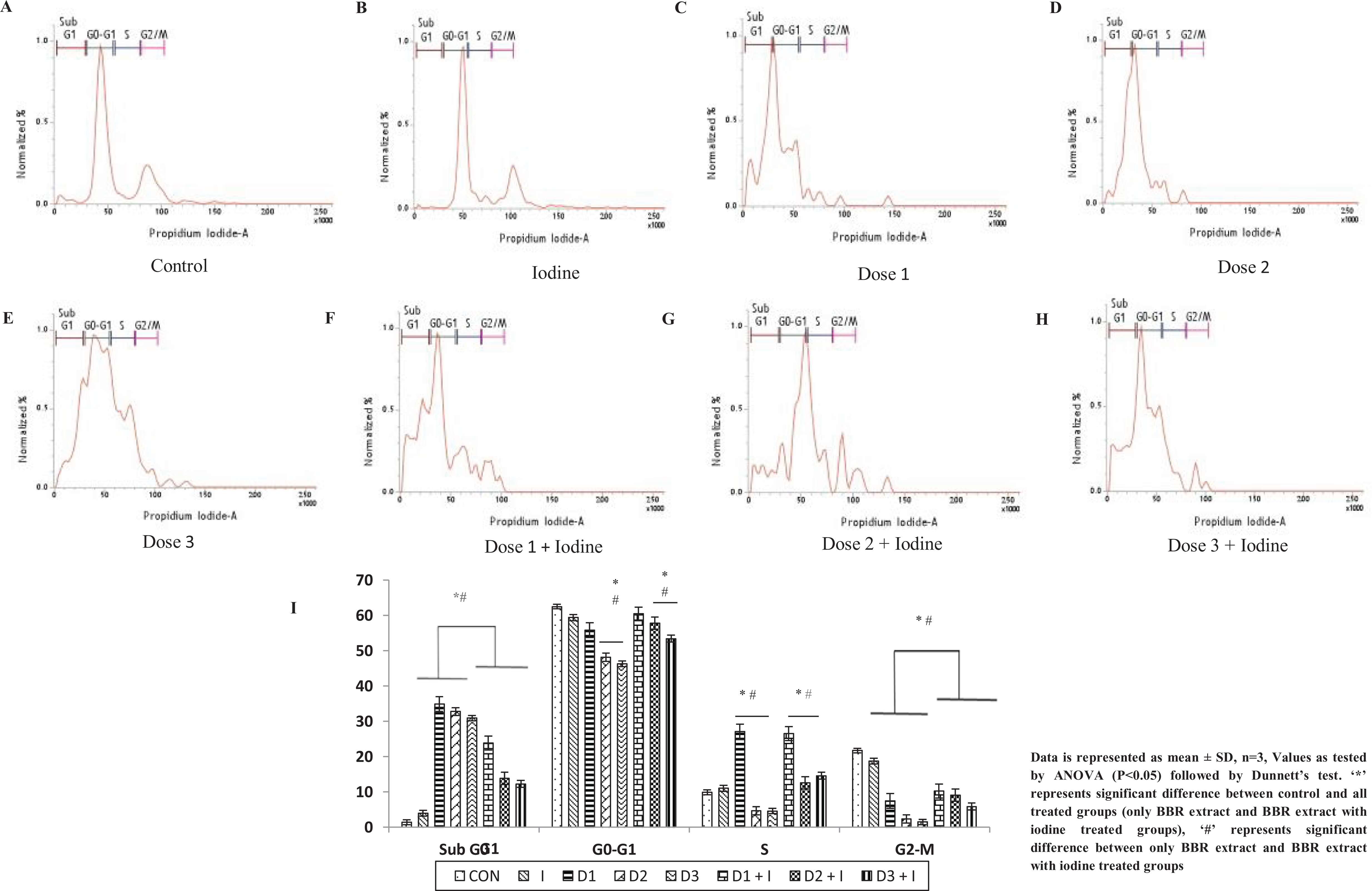
Effect of different doses of BBR Extract with and without extra iodine on cell cycle analysis of thyrocytes. (A–H) Histogram representing cell cycle analysis of thyrocytes under different doses of BBR Extract with and without extra iodine. (I) Graphical representation of cell number in each stage under influence of BBR extract.
BBR extract exposure in thyroid cells alters Bax, Bcl-2 and p53 levels
On exposure to BBR extract, pro apoptotic Bax levels were increased, while anti-apoptotic Bcl-2 levels were decreased dose dependently, while p53, which is an important cell cycle regulator involved in apoptosis was found increased significantly and dose dependently; on addition of extra iodine, partial amelioration was observed (Figure 5).

Effects of different doses of BBR extract along with and without extra iodine on levels of (A) Bax, (B) bcl-2 and (C) p53. Data is represented as mean ± SD, n = 3, values as tested by ANOVA (P < 0.05) followed by Dunnett’s test. ‘*’ represents significant difference between control and all treated groups (only BBR extract and BBR extract with iodine treated groups), ‘#’ represents significant difference between only BBR extract and BBR extract with iodine treated groups.
BBR extract increases ROS and causes DNA oxidation in thyrocytes
Reactive oxygen species (ROS) were measured colorimetrically, and the levels were found to increase dose dependently. 8-hydroxy-2′-deoxyguanosine (8-OHdG) is one of the major products of DNA oxidation and concentration of 8-OHdG within a cell is considered as a measure of oxidative stress. 8-OHdG levels were increased dose dependently under the influence of BBR extract, which, however were ameliorated partially on addition of extra iodine similarly to that of ROS levels (Figure 6).

Effects of different doses of BBR extract along with and without extra iodine on levels of (A) 8-OhdG and (B) reactive oxygen species (ROS). Data is represented as mean ± SD, n = 3, values as tested by ANOVA (P < 0.05) followed by Dunnett’s test. ‘*’ represents significant difference between control and all treated groups (only BBR extract and BBR extract with iodine treated groups), ‘#’ represents significant difference between only BBR extract and BBR extract with iodine treated groups.
Discussions
In our earlier study the activities of thyroid hormone synthesizing enzymes sodium potassium ATPase (Na-K-ATPase), TPO and deiodinase were decreased while the expression of proteins as NIS, TPO and PAX8 were also down regulated in vitro under the influence of BBR extract dose dependently. The effect of the extract on other important enzymes and regulatory factors related with thyroid hormone synthesis like Thyroglobulin (Tg), Pendrin (PDS), Monocarboxylate Transporter 8 (MCT8), however, remain explored. 7 In this study the activities of the latter have been worked out to understand the possible pathway by which these potent antithyroidal cyanogenic compounds promote disruption of thyroid cells at cellular and molecular level.
Cyanogenic glycosides in their intact structures are not attainable in the extract of BBR but GCMS analysis revealed the presence of their precursors (p-coumaric acid, tyrosine) and metabolic products (isothiocyanate, mandelonitrile and p-hydroxybenzaldehyde) in the extract similar to our earlier study. 19 Families of organic acids, fatty acids, sugar alcohols were detected in the extract. Three doses below IC50 of the BBR extract were added to thyrocytes in culture to study the changes in certain regulatory elements and cellular milieu as Tg, PDS and MCT8.
Thyroglobulin (Tg) is a major protein of the colloid unique to the thyroid follicular luminal component required for synthesis of T3 and T4 as it is an indispensable substrate for organification and coupling of the iodinated tyrosines. 20 The expression of Tg decreased dose dependently on exposure to BBR extract. Decrease in thyroglobulin expression is indicative of less organification as Tg is the essential substrate for the step. It is also the major protein component of the colloid in the thyroid follicular lumen. The decrease in this protein, thus, indicates a decrease in the bulk of thyroid hormone synthesis as a whole leading to a hypothyroid state. 21 Pendrin (PDS), another transporter protein is localized at the apical surface of thyrocytes, in contrast to the basolateral side where NIS is present. It has been associated with iodide efflux. 22 The expression of PDS also decreased dose dependently after exposure to BBR extract and this decrease can also be associated with impaired efflux of organic iodine from the cytosol to follicular lumen. Decreased PDS expressions are associated with hypothyroidism as well as thyroid carcinomas. 23 In association with their protein expressions, gene expressions of Tg, PDS and MCT8 were also examined. Accordingly gene expressions of Tg also decreased markedly in concurrence with that of the protein expressions which signifies impairment of thyroid hormone synthesis at the organification step both at transcriptional and translational level. PDS expression however decreased significantly under the highest dose of BBR extract as used in the study indicating the impact of regular consumption of BS leading to impairment of thyroid hormone biosynthesis. MCT8 is a specific thyroid hormone transporter required for transport of active thyroid hormones outside the thyroid follicular cells 24 found to decrease dose dependently. T3 is more prominently transported via the MCT8, thus, a decrease in its level indicates less availability of the most bioactive hormone in circulation rather their site of actions. Amelioration in expressions of these regulatory proteins was observed on addition of extra iodine in the media both at the transcriptional and translational levels to a certain degree. Iodine being utilized by Tg and transported by PDS predictably negated the effect of BBR extract partially. It has been reported that the expression of NIS and thyroid peroxidase (TPO) were decreased both at the transcriptional and translational level. 7 This study further showed that almost all the steps of thyroid hormone biosynthesis are disrupted by BBR extract. Cyanogenic glycosides through its metabolic end products thiocyanate, isothiocyanates are known to inhibit the activity of certain enzymes as TPO, deiodinase, and others. 7 This study further reveals the inhibition at both transcriptional and translational status of all the thyroid hormone synthesizing regulatory elements after exposure of BBR extract is first of its kind.
Reactive Oxygen Species (ROS) was increased dose dependently after exposure to BBR in corroboration to previous study where increased hydrogen peroxide levels have altered pro-oxidant and antioxidant status of thyrocytes. DNA oxidation levels were also increased dose dependently which is similar to that of previously observed DNA damage. 7 Increase in ROS and DNA damage goes hand in hand to alter oxidant state of cells and bring about changes in the regular cell cycle. 25
BBR extract altered the normal pattern of cell cycle of thyrocytes as observed in the study. The increase in the sub G1 phase cell percentage is indicative of DNA fragmentation and damage due to apoptosis. 26 G0/G1 phase remained significantly unaltered from that of control in initial dose and recovered to almost control percent with iodine addition, indicating that cell proliferation had not stopped. In S phase and G2/M phase cell percentage had decreased indicating the imbalance between the cell death and proliferation that could be due to disruptions in replication or mitosis. 27
Regulation of apoptosis is an essential link in the pathogenesis of many diseases, including thyroid disorders, auto immunity and others. 28 In this study the levels of pro-apoptotic Bax and Bak and antiapoptotic Bcl-2 proteins in thyrocytes were assayed along with p53 level to understand the apoptotic regulation mediated by BBR extract on thyrocytes. A balance between cell death and proliferation is crucial for goitre formation. 29 An imbalance at this point due to DNA damage and perturbed oxidant status might shift the cellular equilibrium towards pathogenesis including impaired synthesis of thyroid hormone and goitre involution. In this study Bax levels increased dose dependently while in Bak levels showed no such increase; however, Bcl-2 levels decreased markedly and p53 increased consequently. All these observations are in line with that of DNA oxidation and damage along with altered redox potential of the cells. 30 The results indicate that the thyrocytes might have lost their normal apoptotic control after the exposure of BBR extract. This loss in apoptotic control results in cellular death and proliferation imbalance as apoptosis is in turn known to induce proliferation and proliferation also apoptosis. The close associations of proliferations and apoptosis can be bi-directional events and indeed apoptosis is non-automatously induced by proliferating cells. 31 Hyperplasia of thyroid follicular cells due to cellular proliferation is considered as hallmark of hypothyroidism and goitre development. The hyperplasia that is observed in such conditions is likely apoptosis induced hyperplasia, which takes places in such a competitive cellular interaction such that the slow growing cells are eliminated by fast growing cells. 32 Presence of iodine improved the situations by shifting this imbalance towards a more balanced state due to its association with regulatory elements at each step of thyroid hormone synthesis. These observations were consistent with those obtained from cell cycle analysis where the imbalance in the early cell death and proliferation and their self-interactions observed plausibly.
Endemic goitre is one of the clinical manifestations of biochemical hypothyroidism caused by iodine deficiency or interruptions of iodine metabolisms in thyroid by dietary goitrogens from cyanogenic glycosides. Endemic goitre is thus prevalent in many regions where the people commonly consume BS rich in cyanogenic glycosides as in North East India. This study reveals that the goitrogenic/antithyroidal constituents present in BS disrupt cellular and molecular milieu of thyrocytes interrupting thyroid hormone synthesis leading to biochemical hypothyroidism followed by apoptotic imbalance of thyrocytes resembling goitre.
Conclusions
The study reveals that the cyanogenic constituents of BBR extract disrupt the major regulatory elements of thyroid hormone biosynthetic pathway as well as transport of hormones after synthesis due to changes in the cellular milieu and functioning of thyrocytes by increased ROS generation, cell cycle disruption, DNA oxidation and imbalance in apoptotic regulation. Extra iodine ameliorates the goitrogenic effects of BBR on thyroid function to an extent. The persistence of residual goitre in the effective post iodization phase for the consumption of BS as reported in our earlier studies might be for the molecular mechanisms as described.
Supplemental material
Supplementary - Possible mechanism of bamboo shoots (Bambusa balcooa) induced thyroid disruption – An in vitro study
Supplementary for Possible mechanism of bamboo shoots (Bambusa balcooa) induced thyroid disruption – An in vitro study by D Sarkar, AK Chandra, S Chattopadyay, M Biswas, S Das, LH Singh and I Ray in Human & Experimental Toxicology
Footnotes
Acknowledgements
The authors are thankful Dr. Ritesh Tiwari and Mr. Sayantan Bose for help with flowcytometry.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding received from Department of Biotechnology, Government of India (Sanction Number BT/471/NE/TBP/2013). Author Deotima Sarkar received fellowship from Department of Science and Technology, Government of India.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
