Abstract
Bacteroides fragilis (BF) plays a critical role in developing and maintaining the mammalian immune system. We previously found that BF colonization could prevent inflammation and tumor formation in a germ-free (GF) colitis-associated colorectal cancer (CAC) mouse model. The role of Toll-like receptor 4 (TLR4) in CAC development has not been clearly elucidated in BF mono-colonized gnotobiotic mice. The wild-type (WT) and TLR4 knockout (T4K) germ-free mice were raised with or without BF colonization for 28 days (GF/WT, GF/T4K, BF/WT, and BF/T4K) and then CAC was induced under azoxymethane (AOM)/dextran sulfate sodium (DSS) administration. The results showed that tumor formation and tumor incidence were significantly inhibited in the BF/WT group compared to those observed in the GF/WT group. However, the tumor prevention effect was not observed in the BF/T4K group unlike in the BF/WT group. Moreover, the CAC histological severity of the BF/WT group was ameliorated, but more severe lesions were found in the GF/WT, GF/T4K, and BF/T4K groups. Immunohistochemistry showed decreased cell proliferation (PCNA, β-catenin) and inflammatory markers (iNOS) in the BF/WT group compared to those in the BF/T4K group. Taken together, BF mono-colonization of GF mice might prevent CAC via the TLR4 signal pathway.
Introduction
Colorectal cancer (CRC) is one of the most common cancers worldwide. According to the World Health Organization GLOBOCAN database, CRC is now the third most commonly diagnosed cancer in the world. In addition to the genetic background, many studies indicate that inflammation and gut microbiota composition are two important risk factors for CRC. 1 –3 Inflammatory bowel disease (IBD) is generally considered a major risk factor in the progression of colitis-associated colorectal cancer (CAC). Evidence is accumulating that dysbiosis of intestinal microbiota may greatly influence the pathogenesis of IBD and CRC in both animal and humans models. 4 –9 Bacteroides fragilis (BF) has been considered as a potential probiotic in recent years. Polysaccharide A (PSA) is an immunogenic capsular component of the NCTC 9343 BF strain, which is reported to promote mucosal immune development and suppress colitis in a CAC mouse model. 10,11
Toll-like receptors (TLRs) are a class of receptors that play a key role in defense against pathogens. TLR4 serves as the main mediator of responses to lipopolysaccharide (LPS), immediately regulating several pro-inflammatory cytokines. Some studies indicate that TLR4 activation might be related to colitis and CAC development. 12,13 In contrast, TLR4 deficiency in mice aggravates dextran sodium sulfate (DSS) induced intestinal injury 14 and B. fragilis could induce high TLR4 expression via outer membrane vesicles (OMV) to reduce the immune responses. 15 Therefore, the role of TLR4 in IBD and CAC still controversial. 13,16,17
The role of TLR4 in CAC development in B. fragilis mono-colonized gnotobiotic mice has not been revealed. Therefore, we hypothesized that a lack of TLR4 might influence tumor progression and be associated with prognosis. In order to directly evaluate the role of BF and TLR4 in CAC, we used mono-colonized gnotobiotic techniques to examine AOM/DSS-induced tumorigenesis and related pathogenesis in T4K mice.
Materials and methods
Animals
Germ-free (GF) male C57BL/6JNarl (wild-type, WT) (7–8 weeks old) were obtained from National Laboratory Animal Center (Taipei, Taiwan). The GF male B6.B10ScN-Tlr4lps-del/JthJ (TLR4 knockout, T4K) mice were purchased from the Jackson Laboratory (Bar Harbor, ME, USA). Mice were maintained in a vinyl isolator in a room kept at a constant temperature (21 ± 1°C) and humidity (55–65%) with a 12-h/12-h, light/dark schedule. Mice were fed a commercial diet (5010 LabDiet, Purina Mills, St Louis, MO, USA) and sterile water ad libitum. To confirm GF status, microbiological assays were performed every month by culturing the feces, bedding, and drinking water in thioglycollate medium (DIFCO, Detroit, MI, USA).
Ethics statement
All procedures were performed in an animal facility accredited by the Association for Assessment and Accreditation for Laboratory Animal Care, with the approval of the Institutional Animal Care and Use Committee at National Laboratory Animal Center who approved the mouse experiments with the approval number IACUC2012M04. The methods applied in this study were carried out in accordance with approved guidelines.
BF culture and mono-colonization in mice
BF strain NCTC 9343 was obtained from the Food Industry Research and Development Institute (Hsinchu, Taiwan). Bacterial cultures were grown overnight in thioglycollate medium at 37°C in an anaerobic incubator, collected by centrifugation (3 min at 2000×g), and washed three times with phosphate-buffered saline. Pellets were resuspended in 20 mL sterile saline. BF was introduced into mice by oral gavage with 5 × 107 colony-formation units in 0.5 mL saline. Control mice were treated with the same volume of sterile saline. After 28 days of colonization, the BF CFUs were determined by a plate count from stool samples and calculated per gram feces. Control mice were treated with the same volume of sterile saline. The BF mono-colonization protocol was followed by Chang et al. and Lee et al. study. 18,19
Induction of experimental CAC in mice
Here we outline the application of the Azoxymethane (AOM)/Dextran sodium sulfate (DSS) model of CAC. The AOM/DSS model is a powerful, reproducible, and relatively inexpensive initiation-promotion model that utilizes chemical induction of DNA damage followed by repeated cycles of colitis. 20 –23 In our experiment, the mice were divided into the following four groups for treatment: GF condition induced CAC model (GF/WT and GF/T4K) and BF condition induced CAC model (BF/WT and BF/T4K). All animals were housed in plastic cages in the isolator with free access to drinking water and diets. A colonic carcinogen AOM was purchased from Sigma Chemical Co. (St. Louis, MO). DSS (molecular weight = 40,000) was purchased from MP Biochemicals (Aurora, OH, USA). The GF mice and BF mice were given a single intraperitoneal injection of AOM at a dose of 10 mg/kg body weight (Day 0). One week later, 1% DSS was given in the drinking water for 5 days, replaced with normal drinking water for next 16 days, and the cycle was repeated three times. At week 16, mice were sacrificed (Figure 1).

BF mono-colonization and induction of CAC in the WT and T4K mice. Animals in the model initially received a single intraperitoneal injection of AOM (10 mg/kg). After the AOM administration, each of groups received 1% DSS in drinking water for 5 consecutive days. GF: germ-free; BF: Bacteroides fragilis; CAC: colitis-associated colorectal cancer; T4K: TLR4 knockout; AOM: Azoxymethane; DSS: dextran sodium sulfate.
Tumor incidence of CAC
Colons tissue were harvested, and tumor sizes were measured with a caliper. Colonic neoplasms were observed to calculate tumor incidence in each group. Tumor incidence was defined as the tumor numbers of mice /total living mice in each group. The tumor sizes were separated as large: ≥3 mm and small: ≤3 mm. The procedure is based on our previous study. 19
Histopathologic evaluation of CAC severity
At the end of the experiments, mice were sacrificed by 95% CO2 asphyxiation, and the distal segments of the colon were excised longitudinally along the mesenteric margin. Colorectal tissues were collected and fixed in 10% phosphate-buffered formalin, embedded in paraffin, sectioned at 4 μm, stained with hematoxylin–eosin (H&E), and examined by light microscopy. The histology score consisted of a composite scale that indicated the overall degree of inflammation and hyperplasia (0 = absence of significant inflammation or dysplasia, 1 = mild inflammation without significant dysplasia, 2 = moderate inflammation with hyperplasia, 3 = severe inflammation with adenoma, 4 = severe inflammation with adenocarcinoma). Histological score was determined using a BX51 Olympus microscope in a blinded reading by a senior veterinarian. The histopathologic evaluation was followed by Lee et al. study. 19
Immunohistochemistry of PCNA, β-catenin, Ly6G, and COX-2
The 4-µm formalin-fixed, paraffin-embedded colorectal sections were subjected to deparaffinization and hydration prior to quenching of endogenous peroxidase activity (3% H2O2 in methanol for 10 min). For antigen retrieval, slides were submerged in 10 mM citrate buffer (pH 6.0) until boiling. The sections were incubated overnight with the primary anti-PCNA mouse monoclonal antibody (Invitrogen Co., Camarillo, CA) at a 1:200 dilution, primary anti-β-catenin rabbit polyclonal antibody (Thermo Scientific Co., Kalamazoo, MI) at a 1:100 dilution, primary anti-COX-2 rabbit monoclonal antibody (Thermo Scientific Co., Kalamazoo, MI) at a 1:100 dilution and primary anti-iNOS rabbit polyclonal antibody (Santa Cruz Biotechnology, Dallas, TX) at a 1:100 dilution. The slides were subsequently treated with PictureTM HRP Polymer conjugate at room temperature for 20 min. HRP localization was visualized using a Single Solution aminoethyl carbazole (AEC) kit. The number of PCNA, COX2 and iNOS positive cells were determined by at least 1000 cells were counted in 400X microscopic fields. This method was similarly and based on Wong et al. and our previous reports. 24 The β-catenin staining intensity was graded using a scale of 0–3 as follows: degree 0 = negative reaction, 1 = weak positive signal, 2 = moderate positive signal and 3 = strong positive signal. This method was based on Gao et al. in 2014. 25 Quantification of immunohistochemically positive cells was performed using the open source Fiji (ImageJ) software (version 1.51).
Statistical analysis
All Statistical analyses were performed in GraphPad Prism 6.0 (GraphPad Software, San Diego, CA, USA). The results are presented as the mean ± SD. Differences between groups were analyzed using Mann–Whitney U test or one-way ANOVA analysis for multiple comparisons. P-value less than 0.05 was considered significant difference.
Results
Clinical observation
After colonization with BF for 28 days, we did not observe animal death, clinical signs of CRC, and adverse alterations in the WT and T4K mice. As shown in Table 1, we found that the cecum/body weight ratio of the BF/WT and BF/T4K groups were significantly lower than that of the GF/WT and GF/T4K groups (p < 0.05). There was no significant difference in body weight, cecum weight, or colon length between the groups. The spleen weight of the BF/T4K group was significantly lower than that of the GF/T4K group. Similar results were also found for the spleen/body weight ratio (Table 1).
General observation in WT and T4K mice under GF or BF condition.

Data are presented as mean ± SD. Values not sharing a common superscript letter (a, b) differ significantly at P < 0.05 through one-way ANOVA analysis. GF: germ-free; BF: Bacteroides fragilis; WT: wild type; T4K: TLR4 knockout; bw: body weight.
Gross findings and tumor incidence
Macroscopically nodular and polypoid-like tumor masses were observed in the distal colon. There were no metastases found in different organs including the lungs, liver, heart, spleen, or kidneys. Tumor incidence was significantly lower in the BF/WT group compared to the GF/WT, GF/T4K and BF/T4K groups. Interestingly, there were no significant differences between BF/T4K and GF/T4K (100% and 100%, respectively). Colonic tumors were induced in 100% of mice in the GF groups. However, different incidence rates were found in the BF/WT group (2/6, 33%) and BF/T4K group (6/6, 100%). The mean tumor number was also significantly lower in the BF/WT group than in the GF/WT, GF/T4K, and BF/T4K groups. In addition, the tumor size was smaller (<3 mm) and fewer in number in the BF/WT group. The number of large tumors (>3 mm) in the BF/WT group was also significantly decreased compared to that in the other groups (Table 2).
Tumor assessment under GF or BF condition in the WT and T4K mice.

Data are presented as mean ± SD. Values not sharing a common superscript letter (a, b) differ significantly at P < 0.05 through one-way ANOVA analysis.
Histological findings and CAC severity
In histopathological findings, the colonic tissues had benign hyperplasia and tubular adenoma in all groups. Multifocal adenoma lesions were observed in the GF/WT and GF/T4K groups (Figure 2). The histopathology of non-tumor mice (4/6) showed only simple hyperplasia, and a few focal adenomatous lesions in the remaining mice (2/6) of the BF/WT group. However, histopathology revealed mild to moderately differentiated tubular adenoma (low grade) in the BF/T4K group. These results indicated that the TLR4 defect may not improve colorectal tumor development in BF gnotobiotic mice. However, colorectal tumor development was inhibited in the BF/WT group when compared to the BF/T4K group (Figure 2).

Gross observation and histopathology of BF mono-colonization by AOM/DSS-induced colon carcinogenesis in the WT and T4K mice. (a) Gross finding of the distal colon. (b) H&E stain of distal colon sections. (A) GF/WT/AD, (B) GF/T4/AD, (C) BF/WT/AD, (D) BF/T4/AD. H&E, magnification ×100, scale bar: 100 μm. GF: germ-free; BF: Bacteroides fragilis; WT: wild type; T4K: TLR4 knockout; AOM: Azoxymethane; DSS: dextran sodium sulfate.
Immunohistochemistry
The cellular composition of colorectal tumorigenesis and inflammation was analyzed using immunohistochemistry staining. Expression levels and positive signals of proliferating cell nuclear antigen (PCNA), β-catenin, cyclooxygenase 2 (COX2), and inducible nitric oxide synthase (iNOS) were assessed in the lamina propria and submucosa. The number of PCNA-positive cells was significantly decreased in the colons of the BF/WT group compared to that in other groups (BF/WT: 193.3 ± 19.9 vs. GF/WT: 425.3 ± 43.1, GF/T4K: 581.3 ± 140.6, BF/T4K: 580.3 ± 180.8, p < 0.05) (Figure 3).
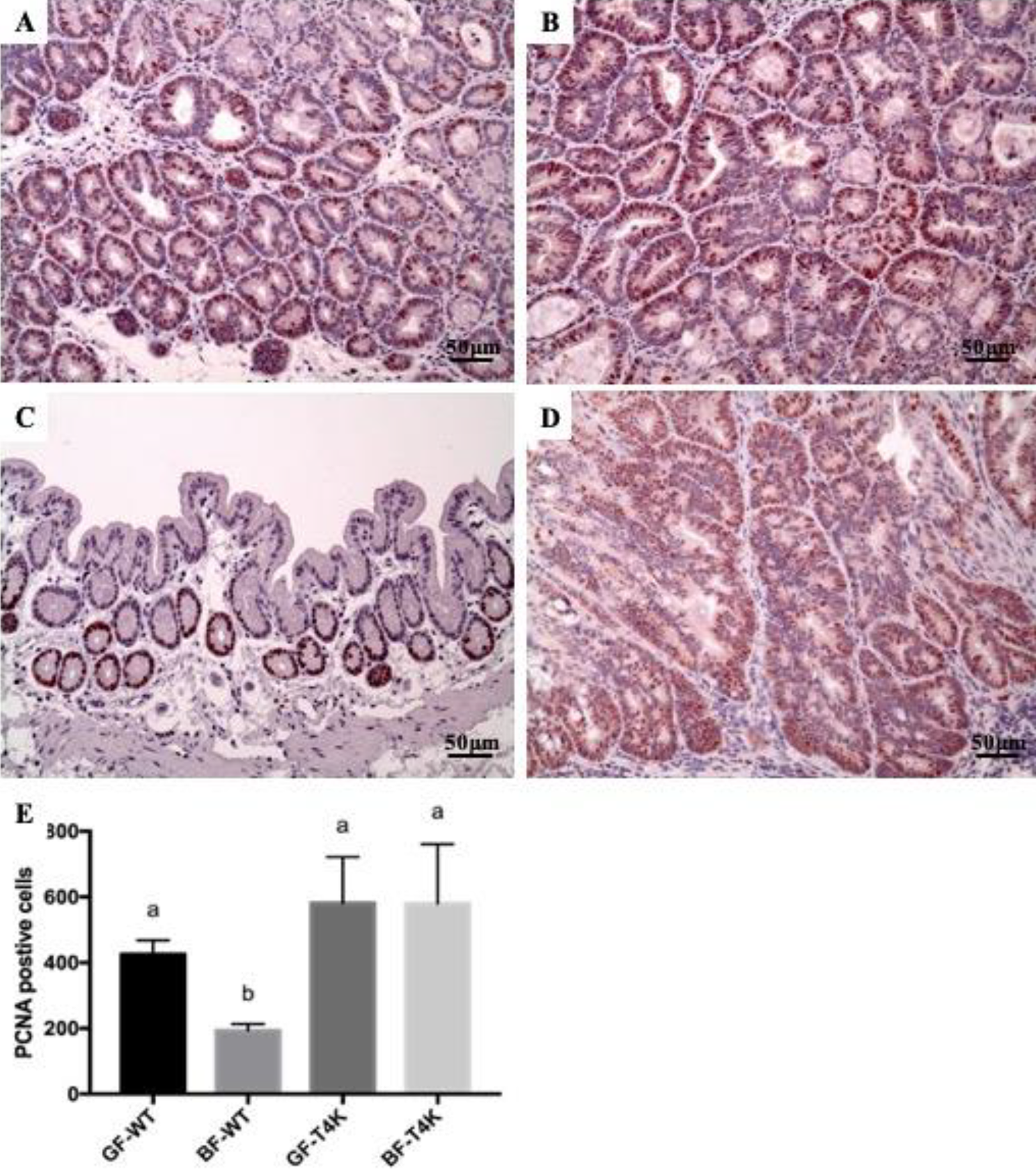
Immunohistochemistry staining for PCNA in colon tissue sections. (A) GF/WT, (B) GF/T4K, (C) BF/WT, (D) BF/T4K, (E) quantitative estimation of positive signal. Magnification ×200, scale bar: 50 μm. Data are presented as mean ± SD. Values not sharing a common superscript letter (a, b) differ significantly at P < 0.05 through one-way ANOVA analysis.
The β-catenin relative intensity was also significantly decreased in BF/WT mice compared to the GF/WT, GF/T4K and BF/T4K groups (BF/WT: 0.7 ± 0.6 vs. GF/WT: 3.0 ± 0, GF/T4K: 2.3 ± 0.6, BF/T4K: 2.3 ± 0.6, p < 0.05). In the BF/WT group, the β-catenin relative intensity was expressed in the mucosa epithelium membrane of the simple hyperplasia and dysplasia non-tumor tissue. However, the β-catenin relative intensity was expressed in the cytoplasm and nuclei of tumor tissue in the GF/WT, GF/T4K and BF/T4K groups (Figure 4).

Immunohistochemistry staining for β-catenin in colon tissue sections. (A) GF/WT, (B) GF/T4K, (C) BF/WT, (D) BF/T4K, (E) quantitative estimation of positive signal. Magnification ×200, scale bar: 50 μm. Data are presented as mean ± SD. Values not sharing a common superscript letter (a, b) differ significantly at P < 0.05 through one-way ANOVA analysis.
The number of iNOS-positive cells was decreased in the BF/WT group compared to that in the GF/WT group (BF/WT: 3.0 ± 1.0 vs. GF/WT: 49.7 ± 11.6, p < 0.05). The number of iNOS-positive cells in BF/T4K mice was significantly increased compared to BF/WT mice (BF/WT: 3.0 ± 1.0 vs. BF/T4K: 57.7 ± 16.7, p < 0.05). Interestingly, the number of iNOS-positive cells was increased in BF/T4K mice compared to that in GF/T4K mice (Figure 5).

Immunohistochemistry staining for iNOS (neutrophils) in colon tissue sections. (A) GF/WT, (B) GF/T4K, (C) BF/WT, (D) BF/T4K, (E) quantitative estimation of positive signal. Magnification ×200, scale bar: 50 μm. Data are presented as mean ± SD. Values not sharing a common superscript letter (a, b) differ significantly at P < 0.05 through one-way ANOVA analysis.
The number of COX2-positive inflammatory cells was not markedly different in any groups (Figure 6).

Immunohistochemistry staining for COX2 (macrophages) in colon tissue sections. (A) GF/WT, (B) GF/T4K, (C) BF/WT, (D) BF/T4K, (E) quantitative estimation of positive signal. Magnification ×200, scale bar: 50 μm. Data are presented as mean ± SD. Values not sharing a common superscript letter (a, b) differ significantly at P < 0.05 through one-way ANOVA analysis.
Discussion
The factors affecting CAC induction and progression include genetic background, diet, gut microbiota, inflammation, and innate immunity. 22,26,27 Innate immunity, particularly TLRs, plays an important role in intestinal homeostasis. In the present study, we demonstrated that the BF/TLR4 pathway plays an important role in a germ-free CAC mouse model.
The results showed that the cecum weight/body weight ratio was significantly decreased in the BF-inoculated groups. These data were consistent with our previous studies in BF gnotobiotic mice. 19,28 The cecum/body weight ratio was not significantly different between BF/WT and BF/T4K groups. Hence, we believe that the reduction of cecum was not affected with a defect of TLR4. Mazmanian and colleagues reported that BF is sufficient to correct several immunologic unbalanced statuses in GF mice. 29 For the reason, that the effect of TLR expression in the intestine might correlate with CAC. Lee et al. showed that the protective role of B. fragilis in CAC was associated with chemokine receptor CCR5 in the colon. This result was dependent on PSA production of B. fragilis and mediated by Toll-like receptor 2 (TLR2) signaling. 30 On the other hand, TLR4 overexpression was observed in the specific pathogen free (SPF) status in human and murine inflammation-associated colorectal neoplasia. 31 Another study has also shown the anti-CAC effects in TLR4 KO mice. 13 These studies have shown that TLR4 high expression in the SPF state might promote the progress of CAC. The anti-inflammatory and anti-tumorigenic effects against CAC in BF gnotobiotic micewere noted in our previous report. 19 Here, the anti-tumorigenic effect of the tumor mass in histological colonic adenoma was shown only in the BF/WT group rather than BF/T4K group. These findings suggest that effect of BF mono- inoculation against CAC might be mediated by TLR4 signaling.
The colon histopathological classification in the present study was defined with certainty as hyperplasia and adenoma. The result was slightly different in a previous study, which had 33% incidence of adenocarcinoma in GF/WT mice. 19 We suspect that the different results might be due to shortening experimental period from 27 weeks to 16 weeks. In the BF/T4K group, the tumor incidence was as high as in GF/WT and GF/T4K mice (100%). Interestingly, the mono-colonization of BF reduced the high tumor incidence in the WT mice (33.3%). These data showed that the BF/TLR4 cascade might be involved in the pathogenesis of colonic tumor formation. This result was similar to the finding that LPS induced high TLR4 expression could reduce DSS-induced mucosal injury. 32 These results support the conclusion that the LPS of Gram-negative commensal gut flora is necessary to ameliorate DSS-induced colitis. Consistently, an in vitro study showed that B. fragilis could induce high TLR4 expression via OMVs and significantly increase anti-inflammatory cytokines in the Caco-2 cell line. 15 Here, we found that TLR4 might play a protective role during B. fragilis mono-colonization. In contrast, when BF or TLR4 was deficient, the anti-tumor effect disappears. This phenomenon implies that TLR4 may decrease tumor incidence in the presence of the symbiotic bacteria B. fragilis.
We examined the immunoreactivities of paraffin-embedded sections associated with tumor pathogenesis and inflammation of colonic tissues. Kohno et al. indicated that colonic tumor development with AOM/DSS induction was positively correlated with higher cell proliferation and increased β-catenin immunoreactivity. 33 Here, PCNA and β-catenin were less expressed in BF/WT mice, suggesting that cell proliferation inhibition might be BF and TLR4-dependent. The reaction products of iNOS and COX-2, nitric oxide and prostaglandin E2, respectively, could contribute to colon tumorigenesis. 34 –36 Intestinal inflammation results in increased iNOS expression, which is thought to contribute to human CAC. 37 In addition, depletion of neutrophils in mice significantly decreased the severity of colon pathophysiological changes, including cancer incidence, and led to decreased expression of pro-inflammatory cytokines. 26 In our results, the iNOS (neutrophils) immunostaining was significantly decreased in BF/WT compared to BF/T4K. The iNOS results were similar to those of our previous CAC model. 19 The COX2 (macrophages)-positive cells were not significantly different in any group. According to above results, the anti-inflammatory effects of BF might correlate with TLR4-regulated neutrophil accumulation in CAC development. The tumor suppressive effect of BF may have many signal pathways involved. This suggests that the targets of BF might relate with reducing inflammation via IL-6/STAT3 or tumor proliferation via TNF/NF-κB pathway. However, further studies are required to elucidate how TLR4 interacts with the BF in colorectal carcinogenesis or CAC.
Taken together, our findings indicate that B. fragilis mono-colonization in GF mice might prevent CAC via TLR4 signaling. The protective effect of B. fragilis was dependent on TLR4. The mechanism may be different from the other studies in SPF condition. This study might provide a new understanding of the molecular mechanism of TLR4 and BF in CAC prevention.
Footnotes
Acknowledgments
The authors thank the Germfree & Gnotobiotic Section, Technical Services Division, National Laboratory Animal Center, National Applied Research Laboratories, Taiwan, for providing GF and gnotobiotic mice.
Author contributions
HLC and THC contributed equally to this work.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Ministry of Science and Technology (MOST 104-2311-B-492-001).
