Abstract
Graphene oxide (GO) has recently been considered one of the most promising carbon derivatives in nanotechnology. It has many excellent features such as tumor targeting ability, biocompatibility and low toxicity. Therefore, we conjugated docetaxel (DTX) to GO-PEG molecule and investigate its anticancer efficacy in prostate cancer cell line (DU-145). In order to obtain GO-PEG-DTX molecules, we conjugated the DTX via bonds to PEG chains pegylated to the GO surface. We also investigated the stability of GO-PEG-DTX in different biological fluids such as cell mediums, PBS and water in vitro conditions. GO-PEG-DTX has the highest zeta potential in water. In the current research SEM, UV-Vis, and FTIR analyses and zeta potential were utilized for the characterization of nano-sized GO-PEG-DTX. Anticancer efficacy of GO-PEG-DTX were then investigated in DU-145 prostate cancer cell line using MTT metod. The prostate cancer cells were treated by different concentrations of GO-PEG-DTX, GO-PEG, GO, and DTX (1–100 µg/ml) during 24, 48 and 72 h. The spectrophotometric analyzed values at 570 nm were recorded and analysed with Graphpad Prism7. IC50 growth inhibition values was determined. The data showed that the GO-PEG-DTX had a highly effective anticancer activity on prostate cancer cell lines after 24, 48 and 72 hours compared to other molecules. GO-PEG-DTX was found statistically significant in the DU-145 cell line (***p < 0.0001, **p < 0.001, and *p < 0.01). As a result, it can be said that PEGylated GO is an excellent nanocarrier system for the high anticancer activity of DTX. Loading of anticancer drugs using this type of graphene-based nano carrier and delivery to targeted tissues may find potential application in biomedicine.
Introduction
Cancer is a disease caused by uncontrolled or abnormal growth and proliferation of cells as a result of DNA damage in cells. 1 Nowadays, it is one of the most important problems in the world and in our country. According to international cancer research center data, it was reported that 7.6 million people out of 12.7 million cancer patients in the world died due to cancer and approximately 9.6 million died from 18.1 million cancer patients in 2018. It is estimated that cancer-related deaths will constitute 19.3 million in 2025. 2 Surgery, radiotherapy, and chemotherapy are the main treatment methods in cancer treatment. The development of the body’s resistance to the drugs used in chemotherapy, the inability of the drug to reach the targeted tumor region, and the destruction of normal cells, as well as cancer cells, have directed scientists to conduct research on this subject. 3,4
Docetaxel (DTX), which is commonly used in cancer treatment, is a chemotherapeutic drug approved by the American Food and Drug Administration (FDA) and exhibiting resistance independently of androgens and is clinically utilized for the treatment of cancers, such as breast cancer, metastatic prostate cancer, advanced stomach cancer, non-small cell lung cancer, and head and neck cancer. Drug resistance developing due to DTX use causes serious side effects. 5,6 Therefore, new nanotechnology-based approaches are promising in eliminating DTX resistance and side effects of the drug, which is applied in cancer treatment.
It is known that DTX, which is used as a chemotherapeutic agent in cancer treatment, affects tumor cells via apoptosis and also affects healthy cells. Recently, it has recently drawn attention that cancer drugs are coated with non-toxic and biocompatible polymers that are highly water-soluble, such as polyethylene glycol (PEG), polylactic acid (PLA), and poly(lactic acid-co-glycolic acid) (PLGA).
7
PEG represents a hydrophilic polymer with repeating ethylene ether groups. PEG is a cheap, versatile, non-toxic FDA-approved polymer with suitable pharmacokinetics and tissue distribution, which is used in many applications. The modification of the surface of nanoparticles with PEG (PEGylation) inhibits the agglomeration of nanoparticles in the liquid and also makes them protein-adsorption-resistant and increases their biocompatibility. Moreover, coating nanomaterials with PEG increases circulation time
Within the scope of this study, firstly, it was synthesized with GO-modified Hummers’ method, which is a nanocarrier system, and then modified with PEG, which is an FDA-approved polymer, and finally bound to the DTX drug delivery system, and GO-PEG-DTX (Figure 1) was synthesized. The characterization analyses of the synthesized nanocarrier-based drug were performed, and then stability analyses were conducted with zeta potential. Prostate cancer is a common disease, and there are many nanotechnological drug studies on this subject. 10,20 But, there are no studies in the literature that evaluates GO-PEG-DTX in prostate cancer. However, the assessment of the anticancer activity alone of GO-PEG-DTX may not be sufficient to understand its role in prostate cancer pathogenesis. Therefore, evaluation of the GO-PEG-DTX system is important for a better understanding of the role in prostate cancer. In this study, it was aimed to synthesize and characterize GO-PEG-DTX and to determine the anticancer activity of GO-PEG-DTX in prostate cancer cell line.

Scheme of GO-PEG carrier and loading of DTX.
Experimental
Materials and methods
Graphite flakes ( < 20 μm, 12.01 g/mol) were obtained from Sigma-Aldrich Co. Chemicals were utilized for the synthesis of GO. Furthermore, H2SO4 (98%) and H2O2 (30%) were acquired from Sigma-Aldrich Co. HCl (37%) was obtained from Merck, while KMnO4 was obtained from Tekkim Co.
Synthesis of graphene oxide (GO)
In the current research, the synthesis of GO was performed by employing the enhanced Hummers’ method suggested by Marcano et al. 21 In the above-mentioned method, firstly, a homogeneous solution was created as a result of mixing H2SO4 (360 mL) and H3PO4 (40 mL). Afterward, the addition of 3 g of graphite powder to the solution was performed, and it was stirred for a period of 6 h at a temperature of 80 °C by a magnetic stirrer. Then, the cooling of the mixture to room temperature was ensured. After the mixture was cooled, it was washed using purified water for the removal of the acid residue. The addition of the obtained semi-oxidized graphite to 250 mL of H2SO4 was carried out at a temperature of 0 °C for the purpose of full oxidation. Afterward, 18 g of KMnO4 was added to the solution in a gradual way, maintaining the temperature lower than 20°C, and the mixture was stirred at a temperature of 35 °C for a period of 4 h. Then, the addition of 2 L of distilled water to the solution was carried out, following which 40 mL of 30% H2O2 aqueous solution was added, and the mixture’s color altered to bright yellow with air bubbles. For the removal of metal impurities in the mixture, 0.1 M HCl solution was used to purify the mixture 4 times, and the final mixture was centrifuged with deionized water a number of times, and the resulting product was dried at a temperature of 60°C.
PEGylation of graphene oxide (GO-PEG)
With the aim of enhancing the stability of GO in solutions with a high content of salts or proteins, GO was modified with hydrophilic functional groups as a result of the conjugation of PEG groups onto GO. Firstly, carboxylic acid functional groups were introduced to GO, and the conversion of OH groups on GO sheets to COOH groups was performed by employing the method proposed by Sun et al. (2008). 15 Carboxylic acid-modified GO (GO-COOH) represented the final product. Secondly, PEG groups were introduced on GO sheets as a result of the reaction between the COOH groups of the GO-COOH and NH2 groups on the PEG-NH2 molecules. In short, the sonication of the modified GO (100 mL, *1 mg/mL) was carried out using 6-arm PEG-NH2 (300 mg) for a period of 5 min. Afterward, EDC.HCl (30 mg) was added, and the bath-sonication of the solution was performed for a period of 40 min at room temperature, following which a sufficient amount of EDC.HCl (80 mg) was added, and the mixture was stirred for a night. The removal of unbound PEG was carried out as a result of dialysis (14 kDa) against distilled water for 1 week. The solution represented the final product GO-PEG aqueous solution.
Characterization
GO and GO-PEG samples were pressed into potassium bromide pellets and studied with a Fourier transform infrared (Bruker: Tensor II) spectrophotometer in the interval of 3500–1000 cm−1. A scanning electron microscope (TESCAN MIRA3 XMU) was used to measure morphological and size characterization. The spectra of the specimens prepared in the interval between 200 and 600 nm were recorded by a UV-Vis spectrophotometer (UV-1280, Shimadzu, Japan) for the homogeneous dispersion of the specimens. A probe sonicator (Sonics & amp; materials INC, USA) was utilized at a power of 750 W power. The Malvern Zetasizer Nano Z was utilized with the aim of measuring the zeta potential values of the specimens.
Drug loading
The loading of the anticancer drug DTX onto the GO-PEG drug delivery system was performed simply as a result of mixing DTX to the GO-PEG solution, following which it was stirred for a night. The removal of the unbound DTX was carried out as a result of repeated washing at 9000 rpm at room temperature for a period of 20 min and its filtering through 100 KDa filter paper. The re-suspension of the obtained GO-PEG-DTX complex was performed, and it was kept at a temperature of 4°C. 4,15,22
In vitro anticancer assay
The cell viability of GO-PEG-DTX, GO-PEG, GO, and DTX was tested in DU-145 cells by employing the MTT (3-(4,5-dimethylthiazol2-yl)-2,5-diphenyltetrazolium bromide) analysis method. 23 DU-145 cells were grown in Dulbecco’s modified Eagle’s medium (DMEM). It was then planted in 96-well plates, and its incubation at a temperature of 37°C was carried out under 5% CO2 and 95% humid air, allowing the cells to hold. After the concentrations of 1–100 µg/ml of GO-PEG-DTX, GO-PEG, GO, and DTX were added to the cells, their incubation for a period of 24, 48, and 72 hours was ensured. Afterward, the MTT method was employed to determine the anti-cancer activity of GO-PEG-DTX, GO-PEG, GO, and DTX in cells.
Statistical analysis
SPSS (Statistical Package for Social Sciences, ver: 25.0) program was used to evaluate the data in the study. All experiments were run in triplicate and the results expressed as mean ± SEM. The data were analyzed using one-way ANOVA and differences were considered significant (***p < 0.0001, **p < 0.001, and *p < 0.01).
Results and discussion
Synthesis, functionalization, and characterization
The synthesis of graphene oxide nanoparticles was performed in a successful way by employing modified Hummer’s method. The conversion of the synthesized graphene oxide into nanographene oxide was carried out as a result of sonication. Various conjugates were made in a successful way using GO viz, GO-PEG, and GO-PEG-DTX.
In Figure 2, FTIR results are presented to determine the surface properties of GO and GO-PEG and to verify the performance of the reaction. FTIR measurements were used to confirm the conjugation of PEG onto GO in a successful manner. According to the data obtained after FTIR analysis, the OH (∼3400 cm−1), C=O (∼1720 cm−1), and C=C (∼1624 cm−1) functional groups are observed in GO. As can be seen from Figure 2, in comparison with GO, the peaks at –CH2– (∼2870 cm−1), –NH–CO– (∼1639 cm−1), and –C–O– (∼1100 cm−1) that emerged in the GO-PEG specimen were the signatures bond in PEG. The finding demonstrated that PEG was bound to GO in a successful manner.

FTIR spectra of GO and GO-PEG. The FTIR result shows that PEG is successfully bound to GO.
SEM images for the detailed morphological analysis of the GO-PEG-DTX system are presented in Figure 3 below. The GO sheets were determined to be considerably wrinkled and crinkled. The particles were detected to be 14.83 mm in width, as can be observed under extra high tension (EHT) of 10 kV. It is observed in the figures that a smooth homogenous structure is formed. Upon examining different sizes, it is observed in the last image that the drug has been homogenously bound to the delivery system, and there is no agglomeration.

SEM images of GO-PEG-DTX. In the last image, it is seen that the DTX is homogeneously bound to the GO-PEG.
The UV-vis absorbance spectra of the solutions prepared in GO, GO–PEG, and GO–PEG-DTX water are presented in Figure 4. The peak at 233 nm in the spectrum originates from the

UV-Vis absorbance spectra of GO, GO-PEG, and GO-PEG-DTX in the aqueous solution. The characteristic peaks are seen in the spectrum.
Zeta potential takes place among the essential analyses in deciding on the stability of drugs, colloidal dispersions. The sum of the electrical double layer repulsive and Van der Waals attractive forces determines the colloidal system’s stability. In order to ensure colloidal stability, the repulsive force should be higher than the attractive force between particles. There are two basic mechanisms influencing colloidal stability, one of which is polymeric repulsion and the other one is electrostatic repulsion. The degree of electrostatic repulsion between charged particles in the suspension is indicated by the magnitude of the zeta potential value. 25 According to ASTM, the stability of colloidal solutions with the zeta potential above ±40 mV is accepted to be very good. 26 In Figure 5, there are zeta potential values measured in the synthesized GO and GO-PEG-DTX water. In Figure 5, as a result of comparing the zeta potential value, it was determined that GO-PEG-DTX had, in general, better stability compared to GO in water. For the purpose of enhancing the stability of GO in fluids, the sonication of GO to small slices was performed, and 6-armed PEG was introduced onto GO sheets by the reaction between COOH groups of GO-COOH and NH2 groups on PEG-NH2 molecules.

Zeta potential of GO and GO-PEG-DTX dispersed in water. GO-PEG-DTX appears to have better stability compared to GO in water. It has been demonstrated that the pegylated nanocarrier based DTX can maintain its stability for a long time after dissolving in water. While DTX alone may lose its stability in water after a certain period of time, pegylated DTX can maintain its stability for a long time when using GO as a nano carrier.
The good dispersion of the GO-PEG-DTX synthesized in distilled water was ensured, and it remained stable for a few months. However, it aggregated in biological fluids with a high content of salts or proteins for a month, including EMEM, DMEM, F-12 K, PBS (Figure 6).

Zeta potential of GO-PEG-DTX dispersed in different fluids. GO-PEG-DTX has the highest stability in water.
As seen in Figure 6, GO-PEG-DTX with the highest zeta potential value is the medium, in which water is present. Since amino acids, carbohydrates, vitamins, and ions are present in the media, GO-PEG-DTX may have exhibited different behaviors in these liquids. The stability under biological conditions is significant for in vitro application in clinical terms.
In vitro anticancer assay
Compared to other drug carriers, GO, which is the oxidation derivative of graphene, has become the focus of interest in different fields, particularly in biological applications, in recent times.
27
–29
In the present study, after the synthesis of GO nanoparticles was performed, they were conjugated with PEG for the purpose of increasing biocompatibility and adding solubility to the drug. As is known, nanoparticles conjugated with PEG become more biocompatible. Deb et al. performed the hemolysis experiment in order to control the biocompatibility of GO and GO-PEG and, as a result, determined that hemolysis decreased after the conjugation of GO with PEG.
30
PEG is a polymer commonly used in biology due to its minimum toxicity, protein resistance, biocompatibility, and good solubility in water or other common solvents.
31
DTX, which is a water-insoluble anticancer drug, was loaded on GO coated with PEG to deliver it to cells. Afterward, the
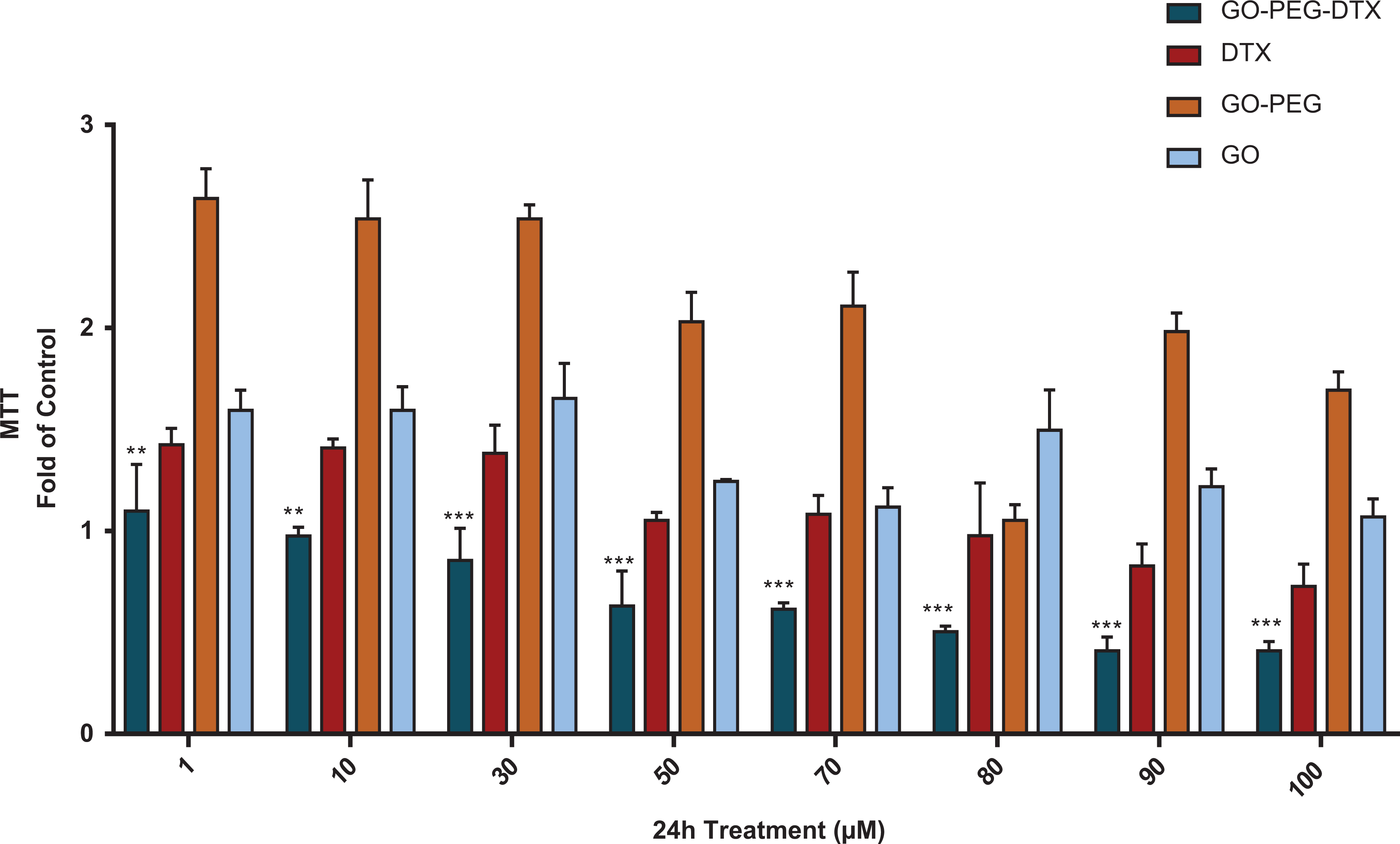
Cytotoxicity study of

Cytotoxicity study of

Cytotoxicity study of GO-PEG-DTX, GO-PEG, GO, and
Compared to the control group (DU-145 cell without the test substance), DU-145 prostate cancer cells that were treated with GO-PEG-DTX, GO, GO-PEG, and DTX significantly reduced the cell survival rate after incubation for 24 hours, 48 hours, and 72 hours. It was determined that while GO-PEG-DTX significantly reduced the viability of DU-145 cells at most in all three time periods (***p < 0.0001, **p < 0.001, and *p < 0.01). Moreover, it was detected to be GO-PEG-DTX after the most active 72-hour incubation in three time periods. Additionally, the IC50 values of the most active molecule, GO-PEG-DTX, for 24 hours, 48 hours, and 72 hours were 4.23 ± 0.26 μM, 2.41 ± 1.37 μM, and 1.54 ± 3.31 μM, respectively (Table 1). No obvious cytotoxicity of plain GO-PEG was found during the experiments, even at high concentrations. These results indicated that loading DTX in GO-PEG did not significantly interfere or reduce cytotoxicity of DTX against cancer cells.
Comparing IC50 values between GO-PEG-DTX, GO-PEG, GO, and DTX on DU-145 following incubating for 24 h, 48 h, and 72 h.

In the present research, when GO-PEG-DTX was compared with DTX, it was determined that GO-PEG-DTX had more effect on DU-145 cancer cells, and it was revealed to be statistically significant (***p < 0.0001, **p < 0.001, and *p < 0.01) (Figures 7 to 9).
The most common way of applying nanoparticle based anticancer agents is intravenous injection. This approach bypasses the stage of absorption from the intestinal epithelium. 31 In the tumor areas, the vascular barrier is disrupted, which causes nano-carriers to accumulate in the tumor tissue. 32 Gaps between tumor vascular endothelial cells may vary depending on tumor type, localization, and environment. Also, due to poor lymphatic drainage, nanoparticles are not rapidly discarded and accumulate in tumor tissue. 33 Thus, this is known as the permeability and retention (EPR) effect, which forms the basis of passive targeting. 34 The accumulation of the drug in the tumor areas is a passive process and the drug must remain in the circulation for a long time for proper drug delivery. The accumulation of nano carriers in tumor tissue is mainly dependent on physicochemical properties such as size, shape, surface charge and surface chemistry. 35 In this study, GO-PEG-DTX synthesized is a preliminary study based on passive targeting. Similar to our study, there are several studies that give information about the successful applications of passive targeting of tumor cells and a successful transformation into clinical therapeutics. 36 –38 Zhu et al. found that GO-PEG-PPT showed very high anticancer activity in both human cervical adenocarcinoma cell line (HeLa) and human hepatoma cell line (SMMC-7721)compared to free podophyllotoxin (PPT). 4 Du et al. (2013) reported that hydroxyethylated graphene oxide (HE-GO) loaded with methotrexate (MTX) had higher anticancer activity in A549 cells than MTX. 39 Liu et al. reported that PEGylated GO was biocompatible without obvious toxicity and has been successfully employed for SN38 and doxorubicin (DOX) delivery. 16 Pei et al.invetigated in vitro the anticancer effect of PEGylated nano-graphene oxide (pGO), pGO-Pt, pGO-DOX, pGO-Pt/DOX, and cisplatin (Pt)/DOX mixture on squamous cell carcinoma cell line (CAL-27), breast cancer cell line (MCF-7) and murine fibroblasts cell line (L929). They found that anticancer activity of the dual-drug delivery system (pGO-Pt/DOX nanoparticles) was obviously higher than that of the single-drug delivery system (pGO-Pt and pGO-DOX). 19 Unlike our study, an other in study, Xu et al. observed that the anticancer activity of GO-PEG-PTX is similar to that of PTX. Their results indicated that loading PTX in GO-PEG did not significantly interfere or reduce cytotoxicity of PTX against cancer cells. 40 But, Arya et al. have reported that carbon nanostructures such as GO and SWNTs can increase the sensitivity of lung cancer cells to PTX when carbon nanostructures and PTX were incubated with cells together. 41 As a result, anticancer activity is quite high water soluble GO-PEG-DTX complex could open up a window to potential use of this drug. Studies on this nano carrier based drug should be developed and supported by in vivo studies.
Conclusions
In summary, we successfully synthesized a drug delivery system using PEG-functionalized GO and anticancer drug (DTX). We demonstrated that GO nanoparticles had enhanced biocompatibility and could be used as a safe nanocarrier. Because, the intelligent design and synthesis of a nanomaterial, the ease of surface functionalizations to increase precise control and specificity on their physicochemical properties, are essential for the success of cancer nanotherapeutics.Moreover, this study showed that GO-PEG-DTX based drug had a higher effect on DU-145 prostate cancer cells than DTX. The water soluble GO-PEG-DTX complex could be an effective way for the potential use of this agents. More in vitro and in vivo studies are needed to make the GO-PEG-DTX nanoparticle-based drug a future candidate for cancer drug. In addition, the GO-PEG-DTX nano-carrier-based drug can be transformed into a targeted drug by marking expression with increased protein in prostate cancer cells and may have the potential to be used as a new therapeutic agent for cancer treatment in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the financial support provided by the Scientific Research Project Fund of Sivas Cumhuriyet University under the project number M-720.
