Abstract
Nonylphenol (NP) is an environmental contaminant, which adversely affects the male fertility due to endocrine disruption and generation of oxidative stress. The current research was planned to assess the effects of nobiletin (NOB), a polymethoxyflavone, on NP-induced testicular damages. Twenty-four male rats were divided into 4 groups: control (0.1% DMSO), NP group (50 mg/kg), NP+NOB group (50 mg/kg + 25 mg/kg), and NOB group (25 mg/kg). Our results revealed that NP brought down the activities of catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GPx) and glutathione reductase (GSR), while elevated the level of thiobarbituric acid reactive substances (TBARS). Additionally, NP decreased the level of follicle-stimulating hormone (FSH), luteinizing hormone (LH), plasma testosterone, daily sperm production (DSP), epididymal sperm count, viability, motility, gene expression of testicular steroidogenic enzymes (StAR, 3β-HSD and 17β-HSD) and anti-apoptotic protein (Bcl-2), as well as number of spermatogenic cells belonging to various stages. Whereas, sperm (head, mid-piece/neck and tail) abnormalities, expression of apoptotic proteins (Bax and caspase-3), and histopathological damages were increased. However, NOB remarkably reversed all the damages caused by NP. Therefore, it is deduced that NOB could be used as a potential therapeutic to counter the NP-prompted oxidative stress and apoptotic damages in testes.
Introduction
About 90% of infertility issues in males are instigated due to environmental pollutants, genetic abnormalities, and hormonal imbalances. These exogenous and endogenous issues result in defected sperm profile and male reproductive dysfunction. 1 Endocrine-disrupting chemicals are harmful substances which interfere with biosynthesis, secretion, transportation, metabolism, binding ability, action, and removal of natural hormones in the body. 2 Nonylphenol (NP) is an environmental pollutant and endocrine disrupting compound, which causes impairment of endocrine system 3 and induces oxidative stress. 4 NP is a combination of isomers, mainly 4-NP, which originates from degradation of nonylphenol ethoxylates (NPEOs). NPEOs are extensively used in formulation of synthetic products such as detergents, resins, plastics, lubricating oily additives, paper, dyes, pesticides, cosmetics and other industrial products. The big-scale use of pesticides on crops leads to incorporation of NP in environment. 5 –8 The traces of NP can be found in vegetables, fruits, cereals, milk and fish. 9,10 NP causes a number of reproductive and developmental damages in fishes and mammals. 11 This toxicant along with its metabolites stays in blood and body cells of humans and aquatic species, as it holds strong lipophilic potential and long half-life. 12,13
Generation of oxidative stress leads to partial or complete dysfunction of male reproductive system. 14 This oxidative stress is generated by excessive production of ROS, for instance, superoxide anion and hydrogen peroxide. This ROS generation initiates a cascade of damaging processes in cells and distorts the biomolecules, such as protein, DNA and lipid. 15,16 NP has been reported to promote the generation of ROS and induce oxidative stress. 17 In the previous studies, NP exposure led to a reduction in size of testes, decrease in level of testosterone, decline in epididymal sperm count, disturbance in antioxidant enzymes profile, as well as suppression of spermatogenesis. Additionally, it caused hypertrophy of Sertoli cells, escalation of apoptotic degeneration, damage in structure of testes, and ultimately male infertility. 18 –21
Flavonoids, a group of hydroxylated phenolics known as effective ROS scavengers, have evoked a sensational interest as possible therapeutics against free radical mediated diseases. 22 Nobiletin (NOB), 3′,4′,5,6,7,8-hexamethoxyflavone, is a flavonoid, which is present in peels of citrus fruits. 23 NOB holds diverse pharmacological features and displays anti-inflammatory, 24 anti-atherogenic, 25 anti-diabetic, 26 immunomodulatory, 27 nephroprotective, 28 neuroprotective and antioxidant properties. 29
Therefore, present research was planned to assess the protective effect of NOB against NP-prompted male reproductive toxicity.
Materials and methods
Chemicals
Both NP and NOB were bought from the Sigma-Aldrich (Germany).
Animals
Experiment was conducted on 24 male Sprague-Dawley rats weighing 200–250 g. Rats were housed in Animal house of University of Agriculture, Faisalabad and kept in 12/12-h light and dark cycle. The temperature was maintained at (24 ± 2°C) and access to water and food pellets was given. Animals were treated in compliance with the European union of animal care and experimentation (CEE Council 86/609) protocol.
Experimental design
24 rats were divided into four equal groups (n = 6). They were given following treatments for 8 weeks: control group (0.1% DMSO), NP-treated group (50 mg/kg of NP was dissolved in 0.1% DMSO and provided orally), NP + NOB treated rats (Both 50 mg/kg. of NP and 25 mg/kg of NOB were dissolved in 0.1% DMSO and administered by oral gavage), and NOB-treated group (25 mg/kg of NOB was dissolved in 0.1% DMSO and given orally). The 50 mg/kg dose of NP was used in accordance with a previous investigation, 30 while a 25 mg/kg dose of NOB was used in accordance to Bi et al. 31 Rats were given anesthesia by diethyl ether and then decapitated. To separate plasma, trunk blood was drawn into heparinized syringes. Blood centrifugation was carried out for 10 minutes at 3000 revolutions per minute (rpm). Plasma was stored at −20°C after isolation for further analysis. After dissection, left testis was fixed in 10% formalin buffer for histopathological evaluation and right testicle was kept at −80°C to assess the activities of biochemical enzymes. Testicular tissues were homogenized in Na3PO4 buffer at 12,000 rpm for 15 minutes and temperature was maintained at 4°C. This supernatant was ultimately used to assess multiple parameters.
Biochemical evaluation
Catalase (CAT)
The CAT activity was computed by a procedure described by Chance and Maehly, 32 with few modifications. Various compounds, including 2.5 mL of 50 mM phosphate buffer (pH 5.0), 0.4 mL of 5.9 mM H2O2, and 0.1 mL enzyme extract were mixed to make reaction mixture. Absorbance changes in the mixture were checked at 240 nm. One unit of CAT activity was considered as an absorbance change of 0.01 as units/min.
Superoxide dismutase (SOD)
The SOD activity was evaluated by a procedure described by Kakkar et al. 33 Reaction mixture consisted of 1.2 mL of sodium pyrophosphate buffer (0.052 mM; pH 7.0) and 0.1 mL of phenazine methosulphate (186 μM). 0.3 mL of supernatant after centrifugation (1500 × g for 10 min followed by 10000 × g for 15 min) of homogenate was added to the reaction solution. Then, 0.2 mL of NADH (780 μM) was added to initiate enzyme reaction, which was later on terminated by adding 1 mL of glacial acetic acid. Finally, chromogen’ amount was assessed by noticing the change in color intensity (at 560 nm). The values of SOD activity were presented as unit/mg protein.
Glutathione peroxidase (GPx)
The GPx activity was assessed by the methodology of Rotruck et al. 34 Reaction mixture of GPx activity consisted of 0.01 mL of 10 mM sodium azide, 2.0 mL of 0.4 M Tris-HCl buffer (pH 7.0), 0.5 mL of 0.2 mM. H2O2 and 0.2 mL of 10 mM glutathione. Incubation was carried out at 37°C for about 10 minutes and then 0.4 mL 10% (v/v) TCA was added for completion of reaction. The mixture was centrifuged at 5000 rpm for about 5 minutes and absorbance was noticed at 430 nm. Its final values were exhibited as unit/mg protein.
Glutathione reductase (GSR)
The GSR activity was determined by the procedure of Carlberg and Mannervik. 35 Reaction mixture consisted of 0.1 mL EDTA (0.5 mM), 0.05 mL oxidized glutathione (1 mM), 0.1 mL NADPH (0.1 mM), 1.65 mL phosphate buffer (0.1 M, pH 7.6) and 0.1 mL of 10% homogenate in a volume of 2 mL. Enzymatic activity was measured at 25°C by noticing NADPH disappearance at about 340 nm. The values obtained were displayed as nM NADPH oxidized/min/mg tissue.
Thiobarbituric acid reactive substances (TBARS)
The level of TBARS was evaluated by following the protocol stated by Wright et al., 36 but with few modifications. TBARS is produced as a result of lipid peroxidation (LP), therefore, degree of LP was indicated by TBARS level. 1.0 mL total volume of reaction mixture consisted of 0.2 mL homogenate sample, 0.02 mL ferric chloride (100 mM), 0.58 mL phosphate buffer (0.1 M pH 7.4) and 0.2 mL ascorbic acid (100 mM). Incubation was carried out at 37°C for about 1 hour in a shaking water bath. Addition of 1.0 mL 10% trichloroacetic acid terminated the reaction. After adding 1.0 mL 0.67% thiobarbituric acid, tubes were boiled in water bath for about 20 minutes. Then, mixture was transferred to crushed ice-bath prior centrifuging at 2500 × g for about 10 minutes. The TBARS level was determined by measurement of optical density of supernatant at 535 nm with spectrophotometer against reagent blank. Its final values were shown as nM TBARS/min/mg tissue at 37°C using molar extinction coefficient of 1.56 × 105 M−1cm−1.
Evaluation of spermatogenic parameters
Epididymal sperm count
The epididymal sperm count was assessed using hemocytometer following the modified protocol stated by Yokoi et al. 37 The caudal region of the epididymis was minced using anatomical scissors in 5 mL of physiological saline solution, kept in a rocker for 10 min, and incubated at room temperature for about 3 min. The supernatant was diluted 1:100 using a solution containing 1 mL formalin (35%), 5 g sodium bicarbonate, and 25 mg eosin per 100 mL of distilled water. A 10 µL drop of this mixture was placed in a sperm-counting chamber. Finally, at least 10 fields were observed under light microscope at 400x.
Sperm motility
The sperm motility was assessed by following the modified protocol of Moumeni et al. 38 The cauda epididymis was sliced and placed in 0.5 mL of pre-warmed phosphate-buffered solution (pH 7.3) containing a drop of nigrosin stain. 50 µL of homogenate was kept in pre-heated (35°C) slide for observation under light microscope at 200x. Abouts 10 fields and 100 sperms per sample were observed. Each sperm was categorized either as progressive motile sperm (PMS), non-progressive motile sperm (NPMS), or non-motile sperm (NMS). Semen samples were assessed in triplicate and then mean of these 3 estimated values was considered as final sperm motility.
Sperm viability
The 25 µL of eosin-nigrosin stain was mixed with the semen sample. 15 µL of aliquot from this sample was placed on slide, and a smear was prepared and dried at room temperature. Finally, the slides were observed under the microscope at 40x. Unstained or white sperms were classified as alive, while (red) stained sperms were considered dead. About 300 spermatozoa were observed, and percentage of dead sperms was shown as percentage. 39
Sperm morphological abnormality
Sperm morphology was assessed using Kwik-Diff™ staining kit (Thermo Scientific, Pittsburgh, PA, USA). Smears were prepared using 5 µL of semen sample, then air dried and stained with eosin-nigrosin. Finally, slides were rinsed with water to remove excess of stain and observed under light microscope at 400x. 300 random cells were carefully examined on all slides and partial (head, mid-piece/neck, and tail) as well as complete morphological abnormalities of sperms were assessed. 40
Evaluation of hormonal levels
The levels of FSH (serial number-H101), LH (serial number-H206) and plasma testosterone (serial number-H090) were assessed by enzyme linked immune-sorbent assay (ELISA) kits (Los Angeles, CA USA) according to manufacturer’s guidelines. 50 mL of assay diluent and 10 mL of plasma were added to 96-well ELISA plate and incubation was performed for about 2 hours at room temperature. Then, plates were rinsed with the deionized water and before adding 100 mL of peroxidase-conjugated immunoglobulin G (IgG) anti-FSH solution, anti-LH or anti-testosterone in each well, incubation was carried out for maximum 2 hours. Plates were again rinsed with the deionized water, substrate solution was added in wells and incubated for about 25 minutes at room temperature. 50 mL of stop solution was added into each well to terminate the reaction. Finally, the absorbances of FSH, LH and plasma testosterone were recorded at 450 nm. All samples were run in triplicates and conducted at same time under same conditions to avoid inter-assay variation.
Evaluation of DSP
Count of homogenization-resistant spermatids was recorded by procedure described by Robb et al. 41 Primarily, frozen testicular tissues were defrosted, and parenchyma was weighed after removing tunica albuginea. Then its homogenization was carried out in 5 mL of solution (0.9% NaCl + 0.5% Triton X-100) for 30 seconds. Following 5-fold dilution, 20 µL sample was kept in Neubauer chamber and spermatids count was assessed under microscope at 400x. DSP was calculated by dividing spermatid count at 19th stage by 6.1, which represent total days of seminiferous cycle in which spermatids are existing in seminiferous epithelium.
Real-time polymerase chain reaction (qRT-PCR)
To detect changes in gene expression of StAR, 3β-HSD (Hydroxysteroid dehydrogenase), 17β-HSD, Bax, Bcl-2, and caspase-3, 95 mg of tissues from testicles were used. TRIzol (Invitrogen) reagent (Life Technologies, New York, USA) was used to isolate total RNA, while RNA concentrations were evaluated by Nano-Drop 2000c spectrophotometer. Total RNA with A260/A280 ratio between 1.8 and 2.0 was used in reverse transcriptase PCR. Reverse transcription of RNA transformed it into complementary DNA (cDNA) by using total RNA by Fast Quant RT kit (Takara, China). The qRT-PCR was performed in 25 µL of reaction volume using the SYBR Green. The assay was conducted in triplicate. β-actin was used as a standard. Relative expression levels were analysed by 2−ΔΔCT. 42 Primer sequences of target genes as well as β-actin are displayed in Table 1.
Primers sequences for RT-qPCR.

qRT-PCR: real time–polymerase chain reaction; 3β-HSD: 3β-hydroxysteroid dehydrogenase; 17β-HSD: 17β-hydroxysteroid dehydrogenase; StAR: steroidogenic acute regulatory protein.
Histopathological examination of testes
The fixing of testicles and epididymal tissues was carried out in 10% formalin and paraffin wax was used for embedding. 5 µm thick sections were sliced by Richert microtome and then mounted on albumenized slides. These slides were kept at 60°C overnight for deparaffinization and haematoxylin-eosin staining of tissues was performed. Finally, these slides were observed under light microscope (Nikon, Japan), which was furnished with automatic microphotographic system. ImageJ software was used to analyse the photographs.
Statistical analysis
Final values were exhibited as Mean ± SEM. After subjecting values to one-way analysis of variance (ANOVA) for statistical analysis, Tukey’s test was applied for comparing the varying groups by usage of GraphPad prism 5 software. Significance level was adjusted at p < 0.05.
Results
Effect of NP and NOB on biochemical profile
The results of biochemical analysis are exhibited in Table 2. NP administration remarkably (p < 0.05) reduced the activities of CAT, SOD, GPx and GSR, but substantial (p < 0.05) increment was seen in level of TBARS in NP group compared with control group. However, NOB provision alone and together with NP resulted in significant (p < 0.05) rise in activities of CAT, SOD, GPx as well as GSR, whereas TBARS level was decreased compared to NP-induced group.
Effect of NP and NOB on biochemical parameters in rat testes.

Values having dissimilar superscripts in columns are substantially different from other values (p < 0.05)
Effect of NP and NOB on spermatogenic parameters of rat testes.
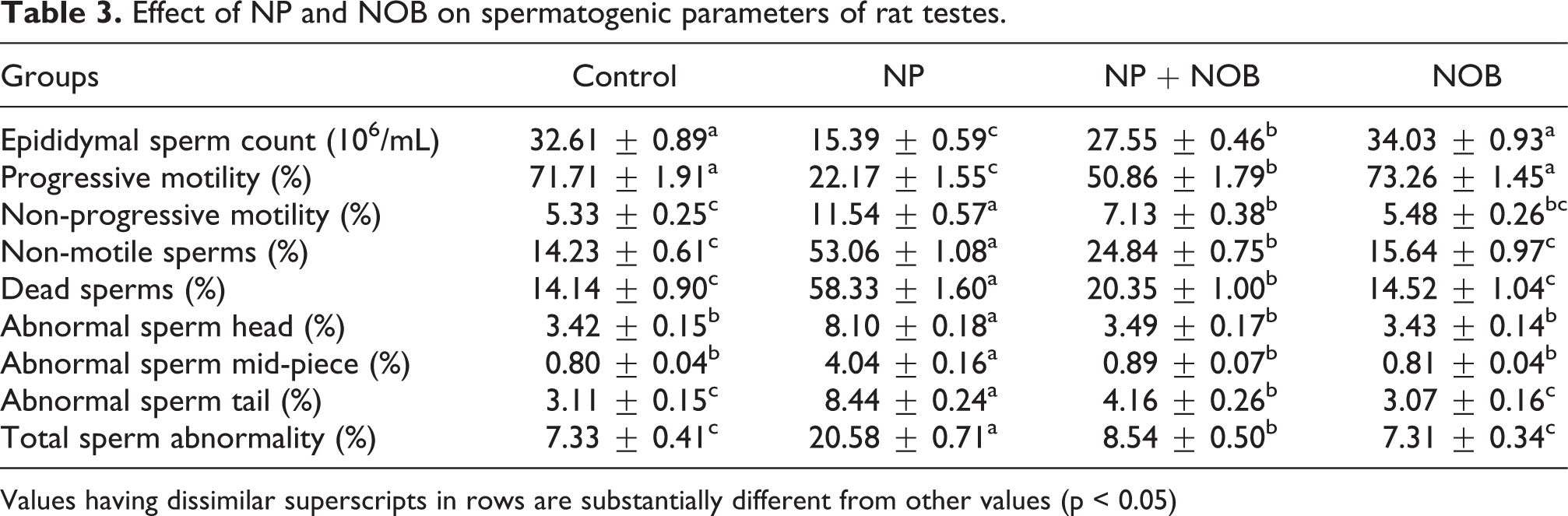
Values having dissimilar superscripts in rows are substantially different from other values (p < 0.05)
Effect of NP and NOB on spermatogenic parameters
The effect of NP and NOB treatments on epididymal sperm count, sperm motility (PMS, NPMS, NMS), viability, as well as partial (head, mid-piece/neck, and tail) and complete morphological abnormalities of sperms are presented in Table 3. The NP exposure led to a significant (p < 0.05) decline in epididymal sperm count, and percentage of PMS and dead sperms. Whereas NPMS, NMS, partial (head, mid-piece/neck, and tail) as well as complete morphological abnormalities of sperms were elevated as compared with control group. Nevertheless, supplementation of NOB in both groups (NOB and NP + NOB) significantly (p < 0.05) augmented the epididymal sperm count, as well as percentage of PMS and dead sperms, meanwhile NPMS, NMS, partial (head, mid-piece/neck, and tail) as well as complete morphological abnormalities of sperms were reduced in both NOB-treated groups in comparison to NP-administered rats.
Effect of NP and NOB on hormonal levels
Variations in hormonal levels are displayed in Table 4. Level of FSH, LH, and plasma testosterone was significantly (p < 0.05) lowered after administration of NP into bodies of rats in NP group compared to control group. Conversely, the NOB exposure upturned this change in hormonal concentrations and significantly (p < 0.05) scaled up the level of FSH, LH, and plasma testosterone in co-treated group (NP + NOB) and NOB-treated group compared with NP-administered rats.
Effect of NP and NOB on hormonal trends of rat testes.

Values having dissimilar superscripts in columns are substantially different from other values (p < 0.05)
Effect of NP and NOB on DSP
The effect of NP and NOB on DSP is exhibited in Table 4. NP-treatment substantially (p < 0.05) lowered the DSP in NP-exposed rats compared to control group. Nonetheless, NOB administration remarkably (p < 0.05) elevated the DSP in NP + NOB and NOB group compared with the NP-exposed rats.
Effect of NP and NOB on gene expression of testicular steroidogenic key enzymes
The changes in expression of steroidogenic enzymes in testes due to NP and NOB are displayed in Figure 1. Administering NP significantly (p < 0.05) minimized the expression of StAR (3.8-fold), 3β-HSD (5.5-fold) and 17β-HSD (6.5-fold) in testicles in NP-induced group compared to control group. However, NOB administration significantly (p < 0.05) regained the level of the expression patterns of StAR (1.50-fold), 3β-HSD (3.5-fold) and 17β-HSD (4-fold) in testicles of co-treated group (NP + NOB) compared with NP group.

Effect of NP and NOB on expression of a) 3β-HSD, b) 17β-HSD, and c) StAR. Bars are displayed on basis of Mean ± SEM values (n = 6). Various subscripts on bars showing significant difference at p < 0.05. NP: nonylphenol; NOB: nobiletin; 3β-HSD: 3β-hydroxysteroid dehydrogenase; 17β-HSD: 17β-hydroxysteroid dehydrogenase; SEM: standard error of mean.
Effect of NP and NOB on gene expression of testicular apoptotic marker
The effect of NP and NOB on apoptotic and anti-apoptotic proteins in testicles is displayed in Figure 2. Expression of apoptotic markers, Bax (1.30-fold) and caspase-3 (0.44-fold), was significantly (p < 0.05) elevated, while expression of anti-apoptotic protein, Bcl-2, was significantly (p < 0.05) reduced (7-fold) in NP-induced group compared to control group. Nevertheless, the expression pattern of all three proteins was overturned following the treatment of NOB. The introduction of NOB in combination with NP significantly (p < 0.05) reduced the expression of apoptotic markers, Bax (0.87-fold) along with the caspase-3 (1.31-fold), while significant (p < 0.05) elevation was seen in expression of anti-apoptotic marker, Bcl-2 (4-fold), when compared to NP group.
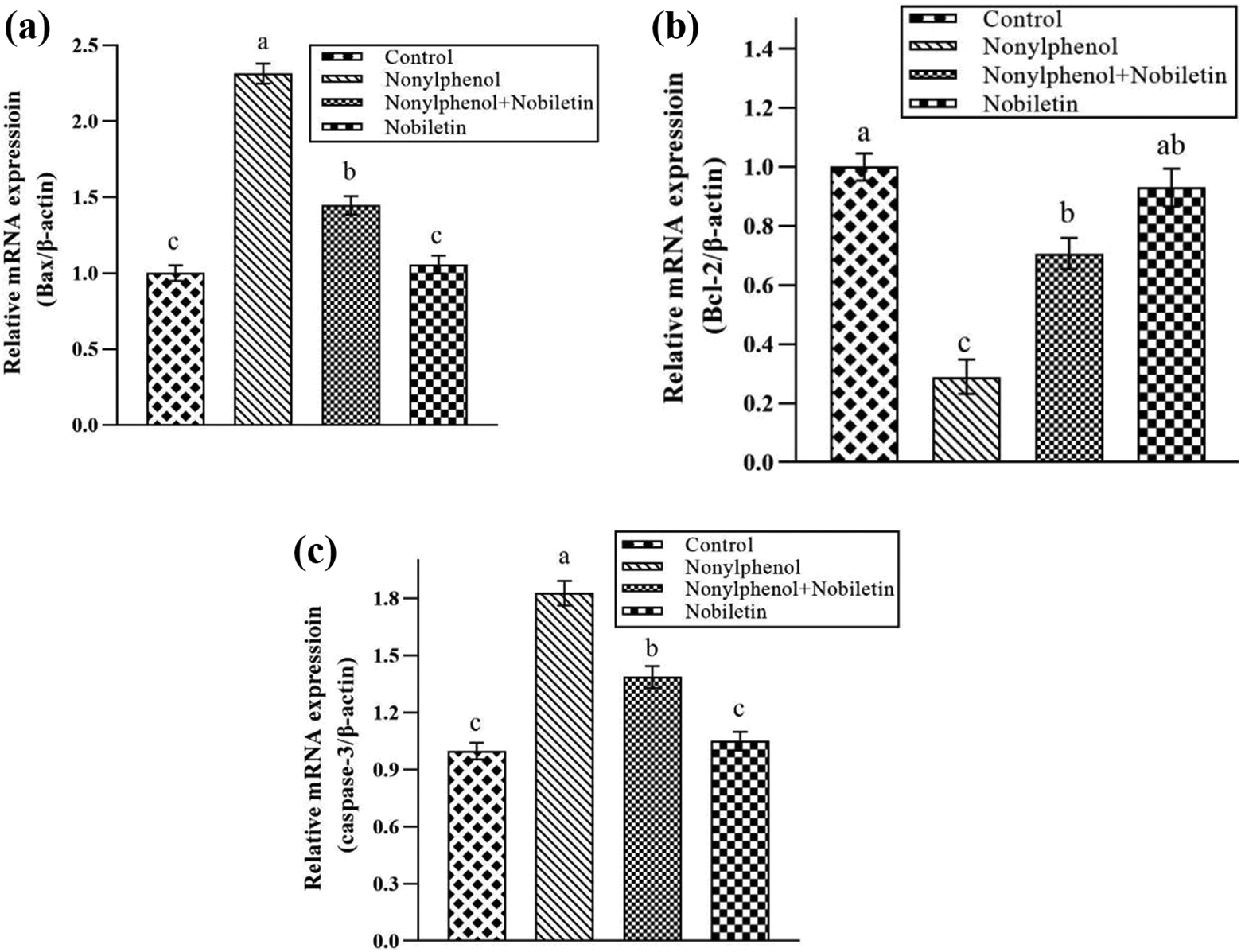
Effect of NP and NOB on expression of a) Bax, b) Bcl-2 and c) Caspase-3. Bars are displayed on basis of Mean ± SEM values (n = 6). Various subscripts on bars showing significant difference at p < 0.05. NP: nonylphenol; NOB: nobiletin; SEM: standard error of mean.
Effect of NP and NOB on histomorphometry of testicular tissues
Changes in histomorphometry of testicles are demonstrated in Figure 3. NP caused a significant (p < 0.05) reduction in epithelial height and diameter of seminiferous tubules, together with tunica albuginea height. Moreover, a significant elevation was observed in tubular lumen plus interstitial spaces in NP group in comparison to control group. However, supplementation of phytomolecule, NOB, significantly (p < 0.05) rectified these adverse morphological alterations and increased the epithelial height and diameter of seminiferous tubules in addition to tunica albuginea height, while tubular lumen and interstitial spaces were reduced in both NP + NOB and NOB treated groups compared with NP-intoxicated rats (Table 5).

Histoarchitectural variations by NP and NOB in rat testicles stained with H&E (200x). (A) Control group displaying thick epithelial layer and normal spermatogenesis; (B) NP group displaying sloughing of epithelium, degenerated area of interstitial spaces, and luminal space with few spermatogenic cells; (C) NP + NOB group exhibiting restoration in epithelial part, improvement in degenerated interstitial cells, and luminal space consisting of more germ cells as compared to NP group; and (D) NOB group revealing resettlement in epithelium, compactness in interstitial area, and luminal part stuffed with all stages of spermatogenic cells. NP: nonylphenol; NOB: nobiletin; H&E: haematoxylin and eosin; TA: tunica albuginea; EH: epithelial height; TL: tubular lumen; IS: interstitial spaces; SG: spermatogonia; ST: spermatids; SS: secondary spermatocyte; and PS: primary spermatocyte.
Effect of NP and NOB on morphometry of rat testes.

Values having dissimilar superscripts in columns are substantially different from other values (p < 0.05)
Apart from structural damages, NP-toxicity also significantly (p < 0.05) minimized the count of various stages of spermatogenic cell i.e. spermatogonia, primary as well as secondary spermatocytes, and spermatids, in NP-treated group compared with control group. But NOB treatment significantly (p < 0.05) escalated number of all stages of germ cells in NP + NOB and NOB treated rats in comparison to NP-intoxicated group (Table 6).
Effect of NP and NOB on number of different stages of germ cells in seminiferous tubules of rat testes.

Values having dissimilar superscripts in columns are substantially different from other values (p < 0.05)
Discussion
In current study, attenuative effects of NOB were investigated against NP-induced toxicity in male reproductive system. NP administration in rats prompted the toxicity in status of antioxidant enzymes, hormonal and sperm parameters, apoptotic and anti-apoptotic proteins, steroidogenic testicular enzymes, morphological integrity of testicles as well as spermatogenic cells of male reproductive system. Multiple in vivo and in vitro investigations have revealed that NP displays toxic effects in reproductive system due to oxidative stress. 43,44 During production of several industrial items, NP is generated as a by-product during alkylphenol polyethoxylates’ degradation. NP exposure culminates in tissue damage due to excessive generation of ROS. 45 It is reported that human body is exposed to NP from the environment. 46 NP is capable of binding with multiple cellular targets and disturb important pathways, resulting in disruption of male reproductive potential. However, NOB treatment shielded the testicular tissues against adverse effects of NP and lessened the oxidative stress. According to Bi et al., 31 NOB has been proved to reduce isoflurane-induced oxidative stress in aging rats due to its antioxidative and anti-apoptotic properties. Therefore, it is assumed that NOB might play role as a pharmacologic tool for averting the toxic effects of NP.
NP administration reduced the activities of antioxidant enzymes such as CAT, SOD, GPx and GSR, while TBARS level was elevated. Imbalance between pro-oxidant and antioxidant leads to generation of oxidative stress. ROS such as O2−, H2O2, and ·OH are generated by mitochondria, which can damage cells in body 47 by damaging macromolecules for instance, polyunsaturated fatty acids (PUFAs) in lipids and proteins in cell membrane. 48 Endogenous antioxidant enzymes, for instance, CAT, SOD and GPx are mandatory to reduce ROS and oxidative stress to prevent further harm. 49 CAT is one of most crucial antioxidant enzymes, which transforms H2O2 to H2O. It also segregates superoxide anion generated by NADPH oxidase from neutrophils as well as protect sperms against oxidative damage. 50 SOD assists in catalysis of superoxide radical to H2O2 and oxygen. 51
Glutathione (GSH) is a cofactor for GPx, which also shields mammalian cells against oxidative stress by minimizing levels of H2O2 as well as other peroxides. A previous study indicated that 5 mM of GSH protected the spermatozoa against freezing and enhanced motility after defrosting. 52 The frequency of LP is proportional to production of superoxide radicals, which is indicated by level of TBARS (final product of LP). 53 Apart from endogenous antioxidant system, these antioxidants can also be supplemented from plant sources to suppress oxidative stress. 54 Therefore, co-treatment with NOB attenuated detrimental effects of NP by reducing oxidative stress in the testicular tissues. Current study revealed that NOB escalated the activities of CAT, SOD, GPx and GSR whereas TBARS level was significantly lowered. The mitigative effects of NOB on biochemical enzymes might be attributed to its antioxidant and free radical scavenging properties. Malik et al. 55 also reported that NOB holds real potential in mitigation of cisplatin-induced oxidative stress and reduction of ROS in kidney due to its antioxidant ability.
Administration of NP clearly scaled down the epididymal sperm count, and percentage of PMS and dead sperms. While an escalation was seen in NPMS, NMS, partial (head, mid-piece/neck, and tail) as well as complete morphological abnormalities of sperms. According to Uguz et al., 56 NP-generated spermatotoxicity was indicated by morphological damage to sperms, aggravated number of dead sperms, and decreased sperm motility. Sperm dysfunction is the major reason behind male infertility. Male gametes are highly prone to ROS due to their increased mitotic events. Several studies have indicated that oxidative burden damages the sperm vitality, motility and structure. 57 Lipids in testes are specifically vulnerable to oxidative insult as they are high in PUFAs. 58 PUFAs are vital to maintain sperm membrane integrity and function. 59 ROS adversely affects the viscosity and permeability of membrane of male gametes, resulting in PUFAs peroxidation. Oxidative burden-induced toxicity reduces ATP generation by causing a direct effect on sperm cell mitochondria. The decline in ATPs in male gametes damages function of flagella, leading to sperm immobility and apoptosis. 60 These NP-prompted spermatogenic impairments including reduction in epididymal sperm count, percentage of motility, viability and morphological abnormalities of sperms could be attributed to elevated ROS levels and degenerated testicles. However, these damaging alterations were effectively mitigated by administration of NOB, probably due to its ROS scavenging nature.
NP exposure led to a reduction in expression of steroidogenic enzymes, 3β-HSD, 17β-HSD and StAR. 3β-HSD and 17β-HSD are key enzymes to maintain androgenesis in testes. The under expression of androgenic enzymes, 3β-HSD and 17β-HSD, leads to reproductive dysfunction in males due to decreased generation of testosterone. 61 StAR is a rate-limiting steroidogenic enzyme in testosterone synthesis, 62 which is used for shifting cholesterol (a testosterone precursor) inside mitochondria. 63 Thus, reduction in expression of these steroidogenic enzymes was reflected by the decreased level of testosterone. On the other hand, NOB brought a profound rise in expression of steroidogenic enzymes. It was deduced that NOB has ability to increase the level of cyclic adenosine monophosphate (cAMP), 64 and this cAMP potentially stimulates expression of genes encoding steroidogenic enzymes and upregulate their activity. 65 This might be underlying reason behind the elevated expression of steroidogenic enzymes by NOB.
Our research findings indicated that NP administration brought down the level of FSH, LH, plasma testosterone, and DSP. FSH stimulates the maturation of sperms and indirectly mediates function of testes. 66,67 LH instigates Leydig cells to produce testosterone, 68 which is essential for production of sperms. 69 Therefore, spermatogenesis depends on accurate proportion of FSH, LH and testosterone in body. 70 NP-exposure disturbed the hypothalamus–pituitary–testicular axis that mediates the function of Sertoli and Leydig cells via hormones production, thereby adversely affecting spermatogenesis. 71,72 Thus, normal level of these aforementioned hormones is necessary to maintain DSP. The findings of our investigation indicated that administering NOB restored the levels of these NP-induced hormonal changes and ultimately recovered normal range of DSP. NOB could alleviate these harmful alterations in hormonal concentrations most likely due to retraction in suppression of hypothalamus–pituitary–testicular axis.
NP-generated testicular toxicity along with attenuative effect of NOB was specifically detected by noticing the gene expression of apoptotic markers by qRT-PCR in testicles. Administration of NP led to over-expression of apoptotic proteins, Bax and caspase-3, along with under-expression of anti-apoptotic protein, Bcl-2. Apoptotic events occurring in body tissues are considered as biological markers of oxidative stress and its associated damages. 73 Caspase-3 plays integral role in apoptosis. 74 The findings of current research study indicated that NP exposure resulted in oxidative stress, which triggered caspase-3, ultimately leading to apoptosis. The intrinsic events which mediate caspase-induced apoptosis are generally called as mitochondrial and death receptor pathways. 75 Genetic expression of two additional proteins, i.e. Bax and Bcl-2, is used as apoptotic marker. Bax and Bcl-2 are vital component to regulate (mitochondria-dependent) apoptosis during spermatogenesis in testes. 76,77 Bax works as pro-apoptotic protein. While Bcl-2 plays its role as anti-apoptotic protein. 78 NOB administration helped to upregulate expression of anti-apoptotic protein, Bcl-2, and downregulate the expression of apoptotic markers, Bax and caspase-3. This finding indicated that NOB exhibited the anti-apoptotic effect against NP-intoxication plausibly due to modulation of these apoptotic markers. Cho et al. 79 also reported that NOB treatment reduced the expression of Bax and caspase-3, as well as augmented the Bcl-2 expression in HT22 hippocampal neuronal cells.
The histological examination elicited that NP administration led to architectural alterations in testicular tissues and spermatogenesis. 80 NP treatment caused significant decline in seminiferous tubules epithelial height and diameter, together with height of tunica albuginea. Whereas, interstitial spaces and tubular lumen diameter were elevated. The degenerated testicular structure and instability in levels of pro-oxidants and antioxidants result in oxidative stress-induced apoptosis and reduction in germ cells. 81 Additionally, number of germ cells, i.e. spermatogonia, primary plus secondary spermatocytes, as well as spermatids was lowered due to this toxic compound. According to Takahashi and Oishi, 82 the inhibition in activities of steroidogenic enzymes and subsequent reduction in testosterone level is the reason behind disrupted spermatogenesis. However, NOB effectively elevated the number of germ cells belonging to all stages and amended various testicular damages.
Conclusion
Taken together, NP exposure damaged the biochemical profile, elevated apoptotic cell death, reduced germ cell count, and resulted in structural degeneration of testes. It predominantly elicited detrimental impact on the hypothalamic-pituitary-testicular axis in rats. Furthermore, gene expression of steroidogenic enzymes and levels of FSH, LH and plasma testosterone were down-regulated, which ultimately affected sperm quality and quantity. However, NOB offered protection against all these testicular damages via its androgenic, antioxidant and anti-apoptotic properties.
Footnotes
Author contributions
MUI and AS designed the experiment. MUI and AS performed the experiments. HA helped in data interpretation. AT and MUI wrote the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
