Abstract
The declining rate of male fertility is a growing concern. Tributyltin (TBT) is a well-known endocrine disruptor (ED), that induces imposex in female gastropods and is widely used in various industrial applications. The aim of this study was to evaluate the toxic effects of TBT on the testes of adult albino rats and the possible role of omega-3. Forty two adult male albino rats were divided into five groups; control group (Group I) and four experimental groups: omega-3 treated group, TBT treated group, TBT & omega-3 treated group and follow up group. At the end of the study, the rats were subjected to biochemical, histological, immunohistochemical staining for Ki-67 and seminal examinations. Our results clarfied that TBT induced a significant decrease in testosterone, FSH, LH and serum glutathione peroxidase levels and a significant increase in the serum Malondialdehyde as compared to the control group. Tributyltin induced disorganization and shrinkage of seminiferous tubules, apoptosis, cellular damage and marked reduction in the germinal epithelium. A significant decrease in the cell proliferation and arrested spermatogenesis were also detected. Seminal analysis of TBT group showed a significant affection of all parameters as compared to other groups. Omega-3 ameliorated all of these hazardous effects. Follow up group still showed toxic effects. In conclusion, TBT has a toxic effect on the testis. Increased testicular oxidative stress, cellular damage and arrest of spermatogenesis with attenuation in antioxidant defenses are all contributing factors. Omega-3 can protect against TBT induced reproductive toxicity.
Introduction
The rapid increase in the prevalence of male infertility suggests the possible involvement of environmental factors such as endocrine disruptors. 1 Exposure to ED is ubiquitous and unavoidable, so concerns are growing that living in an ED contaminated world may be contributing to adverse health trends such as declining male fertility. 2 Tributyltin is recognized as one of the most toxic anthropogenic chemicals 3 that has contaminated the aquatic environment globally. 4 Being a well-known ED, TBT pollution is rampant worldwide, causing growing concerns in evaluating increasing trend of reproductive failure. 3 Tributyltin is a member of organotin family. It is widely used as an antifouling agent in boat paints, agricultural fungicide, wood preservative and many other industrial applications. 5,6
Reports on imposex of female gastropods, a phenomenon that is considered the best example of endocrine disruption in wildlife and has received tremendous attention over the past several years, 7 led to the ban of TBT usage since 2008 but its environmental persistence and long half-life will ensure human exposure for the next few decades. 3,4 Humans are getting exposed from various sources, ingestion of contaminated seafood, as TBT is lipophilic and tends to accumulate in marine species, drinking contaminated water and beverages, dermal contact and inhalation among occupationally vulnerable population, especially in countries where TBT usage is not regulated at all. 3,4,6 Considerable amounts of TBT residues have been detected in human tissues all over the world. 3
Omega-3 polyunsaturated essential fatty acids PUFAs are found in fish oil in large amounts. It contains eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) which are essential fatty acids vital for many functions including growth, reproduction, vision, and brain development. 8 Previous studies documented the antioxidant, anti-inflammatory and anti- apoptotic effects of omega-3 fatty acids in various tissues exposed to stress and damage. Omega-3 PUFAs have been shown to exert beneficial activity for cardiovascular disease, mental disorders, obesity, inflammatory diseases, and various diseases as cancer. 9,10
Materials and methods
A—Chemicals
▪ ▪ ▪ ▪ ▪ ▪
B—Animals and experimental design
The study was carried out on 42 adult male albino rats, 6 rats in each group, each weighing 150–200 gm with an average age of 50–60 days. They were obtained from the animal house, Faculty of Medicine, Zagazig University.
The rats received balanced food rich in all stuffs necessary to maintain their health before and during drug administration. It consisted of bread, barley and milk. Water was offered in separate clean containers.
All animals received humane care in compliance with the animal care guidelines of the National Institutes of Health (NIH), and the local committee approved the design of the experiment.
C—Grouping of animals
The rats were classified into 5 groups:
Group I (control group) (18 rats): was further subdivided into 3 equal subgroups
Group II (Omega-3 group) (6 rats)
Rats of this group were given omega-3 fatty acid dissolved in sesame oil 11 in a dose of 250 mg/kg/day, 12 by oral gavage for 8 weeks.
Group III (TBT-treated group) (6 rats)
Each rat in this group received Tributyltin chloride in a dose of 5 mg/kg/day 5 dissolved in corn oil orally by gavage for 8 weeks.
Group IV (TBT & Omega-3 group) (6 rats)
Rats of this group received omega-3 (250 mg/kg body weight) 12 dissolved in sesame oil then 5 mg/kg tributyltin chloride 5 dissolved in corn oil orally by gavage for 8 weeks.
Group V (Follow up group) (6 rats)
Rats in this group received tributyltin chloride dissolved in corn oil in a dose of 5 mg/kg 5 for 8 weeks then TBT was stopped. Rats were examined 4 weeks after its discontinuation.
At the end of the study, rats from each group were subjected to measurement of body and testicular weights, blood samples were collected to estimate the hormonal levels of testosterone, FSH and LH. Malondialdehyde and glutathione peroxidase were also assessed. Seminal analysis was done to estimate sperm count, motility, viability and abnormal forms. The testicular tissue was processed for histological study and Immunohistochemical examination for detection of Ki-67 as a marker of proliferation.
D—Methods
Assessment of serum testosterone, FSH and LH levels
Blood was collected from the retro-orbital plexus, then was centrifuged for 15 minutes, after that serum was kept at −20°C for analysis of hormones by ELISA, testosterone 13 and FSH & LH by. 14
Antioxidant enzymes and lipid peroxidation
For assessment of markers of oxidative stress, blood samples were collected into a clean centrifuge tube and incubated at 37°C until blood clotted and then centrifuged for 10 minutes to separate the serum, and a pipette was used to separate the top layer.
Assessment of serum malondialdehyde (MDA) level (nmol/ml)
Serum MDA was assayed using MDA biodiagnostic kit according to the method described by Draper and Hadley, 15 where MDA can react with Thiobarbituric Acid (TBA) and give pink colored trimethine complex. Malondialdehyde content in the serum was determined from the absorbance on spectrophotometer at 530–532 nm.
Assessment of serum Glutathione peroxidase (GPX) (ng/ml)
The GPX activity was measured using GPX biodiagnostic kit according to the method of Paglia and Valentine. 16 The principle is based on the enzymatic reaction involving B-nicotinamide adenine dinucleotide phosphate (B-NADPH, reduced form), Glutathione Peroxidase and a sample or a standard which was initiated by addition of hydrogen peroxide. The change in the absorbance was monitored spectrophotometrically at 430 nm. A standard curve was plotted for each assay.
Epididymal (spermatozoa) analysis
Spermatozoa collection was done as described by Kuriyama et al.
17
The epididymal content of each rat was obtained immediately by cutting the tail of the epididymis and squeezing it gently to obtain the fresh undiluted semen in a clean Petri dish and incubated at 37ºC for half an hour for liquefaction to proceed to the following examinations: Sperm count:
Sperm cell concentration was estimated by multiplying the counted number of sperms by 100 (depth) and 1000 (dilution). 18,19
Progressive motility:
The progressive motility of sperms was estimated according to the method reported by Bearden and Flyquary. 20
Sperm viability:
Two hundred sperms were counted per rat under high power lens of light microscope and the number of live (unstained) and dead (stained) sperms were estimated among the 200 sperms, then the percentage of viability was calculated according to the method reported by Björndahl et al. 21
Sperm abnormalities:
Sperm morphology was estimated according to the method reported by Brown et al. 22 The description of the sperm abnormal forms observed in this study was done according to Mori’s classification 23 who classified sperm abnormal forms into tailless sperm, sperms with abnormal heads and sperms with deformed tails.
Histopathological study
At the time of sacrifice, all animals were weighed then anaesthetized by ether inhalation, then were sacrificed. The testes and epididymis were immediately dissected out, grossly inspected to assess any gross abnormalities and testicular weight was recorded. Both testes were fixed in Bouin’s solution, After fixation, testes were embedded in paraffin blocks and processed for the preparation of 5 μ thickness sections. These sections were stained with Hematoxylin and Eosin and examined by light microscope. 24
Immunohistochemical study
Detection of Ki-67 antibodies was carried out using streptavidin–biotin complex immunoperoxidase system. Paraffin-embedded sections of specimens were dewaxed and incubated in 0.1% hydrogen peroxide for 30 min to block the endogenous peroxidase followed by rinsing three times in phosphate-buffered saline (PBS). Antigen-retrieval was achieved by microwave treatment (20 min, 0.01 Mol/L citrate buffer, pH 6). After demasking, the slides were incubated overnight with the primary antibody. Sections were incubated with rabbit monoclonal Ki-67 Antibody (SP6) (Cat. No. MA5- 14520, Thermo Fisher Scientific, Rockford, USA), diluted 1:200 and applied to sections for 12 h at 4 C. After several washes with PBS, primary antibodies were detected by incubation with biotinylated anti-rabbit antibodies (versal kits, Zymed laboratories) for 30 min at room temperature. 25
Histo-morphometric study
The image analyzer computer system Leica Qwin 500 (Leica Ltd, Cambridge, UK) at the Image analyzing unit of Pathology Department, Faculty of Dentistry, Cairo University, was used to evaluate the area percent of Ki 67 immune-reaction, the perimeter of the seminiferous tubules, and the epithelial height of the germinal epithelium. They were measured using the interactive measure menu. Ten readings from five non-overlapping sections from each rat of all groups were also examined.
Statistical analysis
Analysis of data was done using SPSS program (Statistical Package for Social Science) version 25.0. For normally distributed data, comparison between the five studied groups was analyzed using one way analysis of variance (ANOVA or F-test) and least significant Difference (LSD) test. All data were expressed as mean ± SD (Standard deviation). P value less than 0.05 was considered statistically significant. 26
Results
Statistical comparison among (–ve) control, (+ve) control groups and omega 3 group in mean of body & testicular weight and relative testicular weight using ANOVA test.
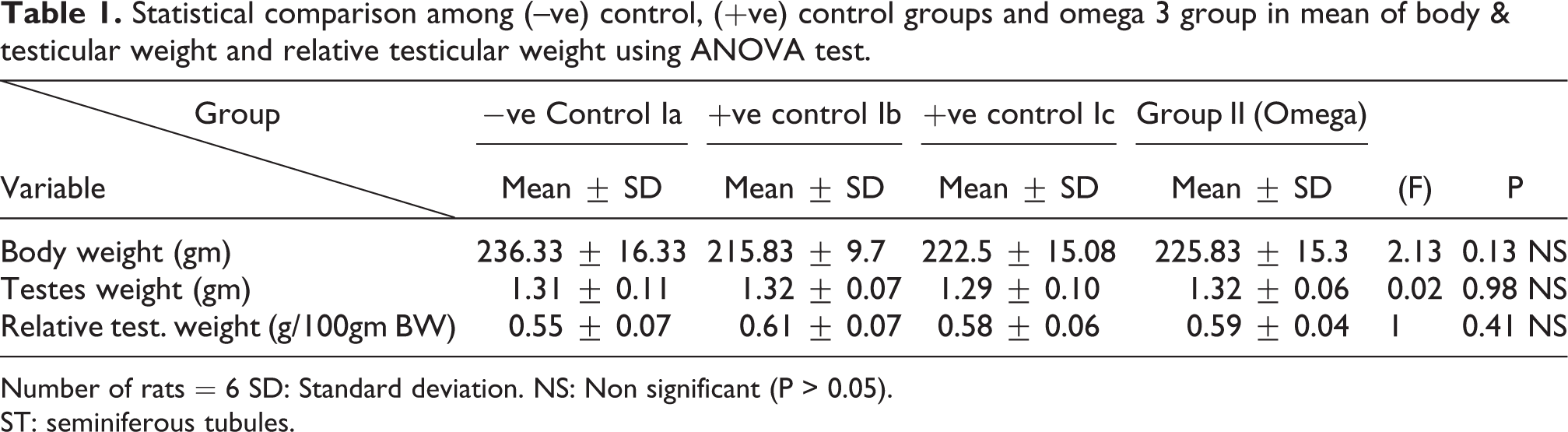
Number of rats = 6 SD: Standard deviation. NS: Non significant (P > 0.05).
ST: seminiferous tubules.
Statistical comparison among (–ve) control, (+ve) control groups and omega 3 group in mean of hormones level (testosterone, FSH and LH) and oxidative stress markers (MDA& GPX) using ANOVA test.
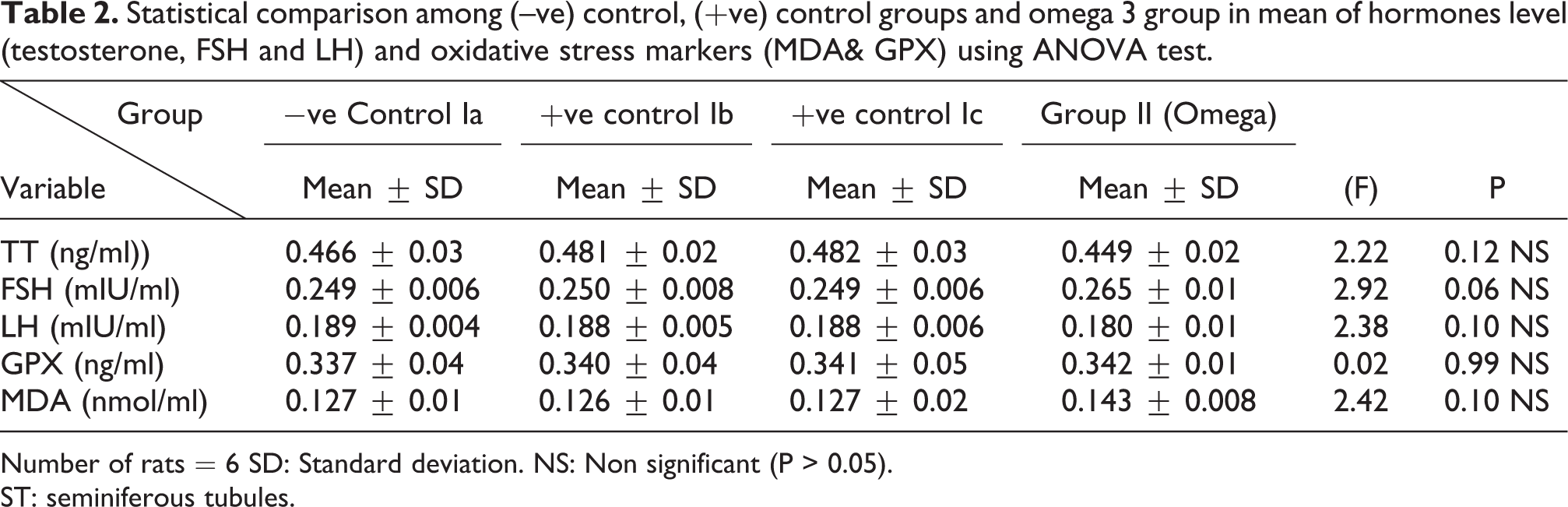
Number of rats = 6 SD: Standard deviation. NS: Non significant (P > 0.05).
ST: seminiferous tubules.
Statistical comparison among (–ve) control, (+ve) control groups and omega 3 group in mean of semen analysis parameters using ANOVA test.

Number of rats = 6 SD: Standard deviation. NS: Non significant (P > 0.05).
ST: seminiferous tubules.
Statistical comparison among (–ve) control, (+ve) control groups and omega 3 group in mean of perimeter, height of seminiferous tubules (ST) and area% of Ki 67 using ANOVA test.
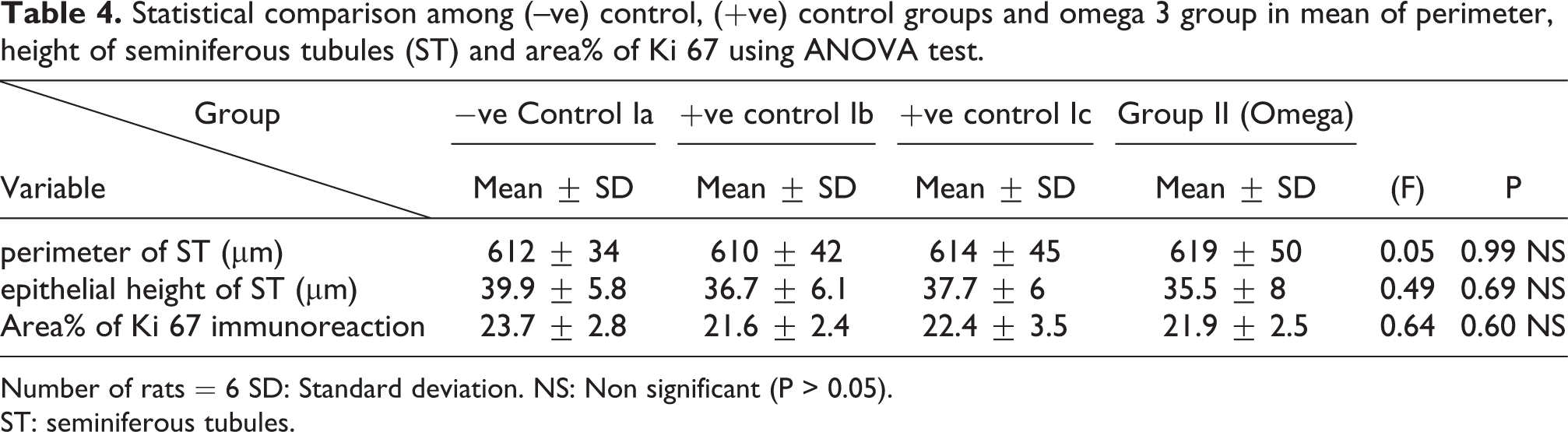
Number of rats = 6 SD: Standard deviation. NS: Non significant (P > 0.05).
ST: seminiferous tubules.
Regarding the body weight, there was an increase in the body weight in TBT group & Follow up group as compared to other groups but with no statistically significant difference (P > 0.05). No statistically significant difference between the control group and TBT& omega-3 group. Regarding both testicular and relative testicular weight, there was a highly statistically significant decrease in TBT group as compared to the control group (P < 0.001). A highly statistically significant increase of both testicular and relative testicular weight was observed in TBT& omega-3 group as compared to TBT group (P < 0.001). There was no significant difference between TBT& omega-3 group and the control group (P > 0.05). There was a significant decrease in the follow up group when compared to TBT& omega-3 and the control groups (P < 0.05) (Table 5).
Statistical comparison among negative control group, TBT group and TBT& Omega 3 group and Follow up group in mean of body & testicular weight and relative testicular weight using ANOVA test.

Number of rats = 6 SD: Standard deviation.
NS: Non significant (P > 0.05) **: Highly significant (P < 0.001).
Groups with different letters are statistically different (p < 0.05).
Regarding testosterone, FSH and LH hormonal levels among the studied groups, there was a highly statistically significant decrease in the mean hormonal levels in TBT group as compared to the control (P < 0.001) while there was a highly significant increase in TBT& omega-3 group when compared to TBT group (P < 0.001). There was non-significant difference in TBT& omega-3 group when compared to the control group (P > 0.05). A significant decrease in the follow up group when compared to other groups was noted (P < 0.05) (Table 6).
Statistical comparison among negative control group, TBT group, TBT& Omega 3 group and Follow up group in mean of hormones levels (testosterone, FSH and LH) and oxidative stress markers (MDA& GPX) using ANOVA test.

Number of rats = 6 SD: Standard deviation.
NS: Non significant (P > 0.05) **: Highly significant (P < 0.001).
Groups with different letters are statistically different (p < 0.05).
As regard GPX activity, there was a highly statistically significant decrease in TBT group as compared to the control and TBT& omega-3 groups (P < 0.001). There was non-significant difference in TBT& omega-3 groups when compared to the control group (P > 0.05). A significant decrease in the follow up group when compared to other groups (P < 0.05) (Table 6).
Regarding MDA levels among the treated groups, there was a highly significant increase in TBT group when compared to control group (P < 0.001). There was a highly significant decrease in TBT& omega-3 groups when compared to TBT group (P < 0.001). There was non-significant difference in TBT& omega-3 groups as compared to the control group (P > 0.05). There was a significant increase in follow up group when compared to other groups (P < 0.05) (Table 6).
Tributyltin treated group showed a highly statistically significant decrease in the mean values of sperm count, sperm motility and viability percent and a highly significant increase in the sperm abnormal forms percent in comparison to negative control group (P < 0.001). In TBT& omega-3 group, a highly statistically significant increase in the mean values of sperm count, sperm motility and viability percent and a highly statistically significant decrease in the mean values of sperm abnormal forms percent were noted when compared with TBT group (P < 0.001). While, there was non-significant difference in TBT& omega-3 group when compared to control group (P > 0.05). There was a significant decrease in the follow up group when compared to other groups (P < 0.05) (Table 7). Figures (3a,3b,3c,3d & 3e) showed examples of the sperm abnormal forms detected in TBT group as compared to the normal sperm forms.
Statistical comparison in negative control group, TBT group, TBT& Omega 3 group and follow up group in mean of semen analysis parameters using ANOVA test.

Number of rats = 6 SD: Standard deviation.
NS: Non significant (P > 0.05) **: Highly significant (P < 0.001).
Groups with different letters are statistically different (p < 0.05).
Histological examination of the control subgroups showed the same histological results, so -ve control group was considered as the control group.
H&E stained sections of the control testes showed numerous packed seminiferous tubules that were covered by connective tissue capsule. These tubules were lined by stratified germinal epithelium. Spermatozoa were present in the lumen of seminiferous tubules. In-between the seminiferous tubules, narrow interstitium containing interstitial cells was detected (Figures 1 a & b).
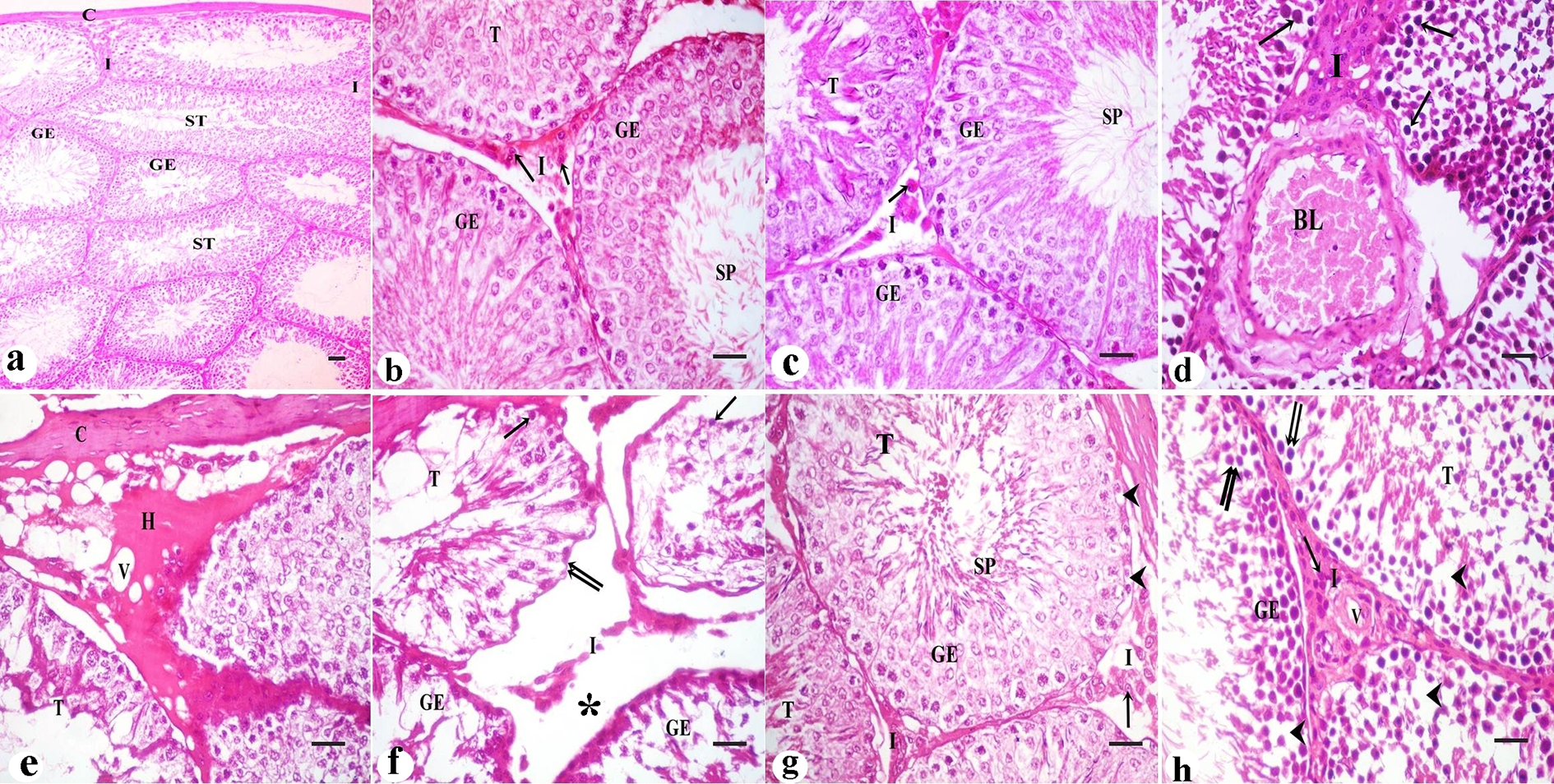
H&E stained sections of omega-3 treated group revealed also normal histological structure. Packed seminiferous tubules were lined by germinal epithelium. Spermatozoa were present in the lumen of the tubules. Interstitium containing Leydig cells was also detected (Figure 1c).
Sections in TBT treated group revealed marked disorganized seminiferous tubules. Some sections showed germinal epithelial cells with dark pyknotic nuclei. Blood vessels in the interstitium were dilated and congested (Figure 1d). Other sections showed thickened connective tissue capsule and decreased height of the lining germinal epithelium, other tubules had marked vacuolations. Acidophilic hyaline material was detected in the interstitium in between the seminiferous tubules (Figure 1e). Shrinkage of the tubules with irregular basement membrane, wide separation between the tubules, marked decrease in the height of the lining germinal epithelium (Figure 1f). Examination of sections in TBT & omega-3 treated group showed improvement in the histological picture of the germinal epithelium of seminiferous tubules. Tubules were lined by germinal epithelium. Spermatozoa were present in the lumen of the tubules. Interstitium containing Leydig cells and blood vessels was also detected. Few vacuolated cells were also seen (Figure 1g). Sections in the follow up group still showed tubules lined by germinal epithelial cells; most of them had dark pyknotic nuclei and separation between the cells. Interstitium revealed Leydig cells and blood vessels (Figure 1h).
Immunohistochemical stained sections for ki67 protein in the testicular tissue of the control and omega-3 treated group showed strong positive reaction in the nuclei of germinal epithelial cells (Figures 2a & 2b). In TBT treated group, positive reaction for ki67 in the nuclei of few germinal epithelial cells was seen (Figure 2c). In TBT & omega-3 treated group, there was strong positive reaction in the nuclei of most of the germinal epithelial cells (Figure 2d), however, in the follow up group, positive reaction was seen in the nuclei of some germinal epithelial cells (Figure 2e).

In the control and omega-3 treated groups, normal sperm morphology was detected. Sperms had smooth heads with hooked tapering apex and normal long tails (Figures 3a & 3b). In TBT treated group, sperm abnormalities were detected in the heads and tails; a hummer shaped heads, banana shaped heads and headless tails were seen (Figure 3c). In TBT & omega-3 treated group, marked improvement in sperm morphology as the sperms were with normal heads and tails (Figure 3d). In the follow up group, some sperms still had sperm abnormalities such as headless tails and detached heads (Figure 3e).

There was a highly statistically significant decrease in the perimeter and epithelial height of seminiferous tubules and area% of Ki 67 immunoreaction in TBT treated group as compared to the control group while there was highly significant increase in TBT& omega-3 groups when compared to TBT group (P < 0.001). There was non-significant difference in TBT& omega-3 groups when compared to control group (P > 0.05). There was a significant decrease in the follow up group when compared to other groups (Table 8).
Statistical comparison among negative control group, TBT group, TBT& Omega 3 group and follow up group in mean of perimeter and height of seminiferous tubules (ST) and area% of Ki 67 immunoreaction using ANOVA test.
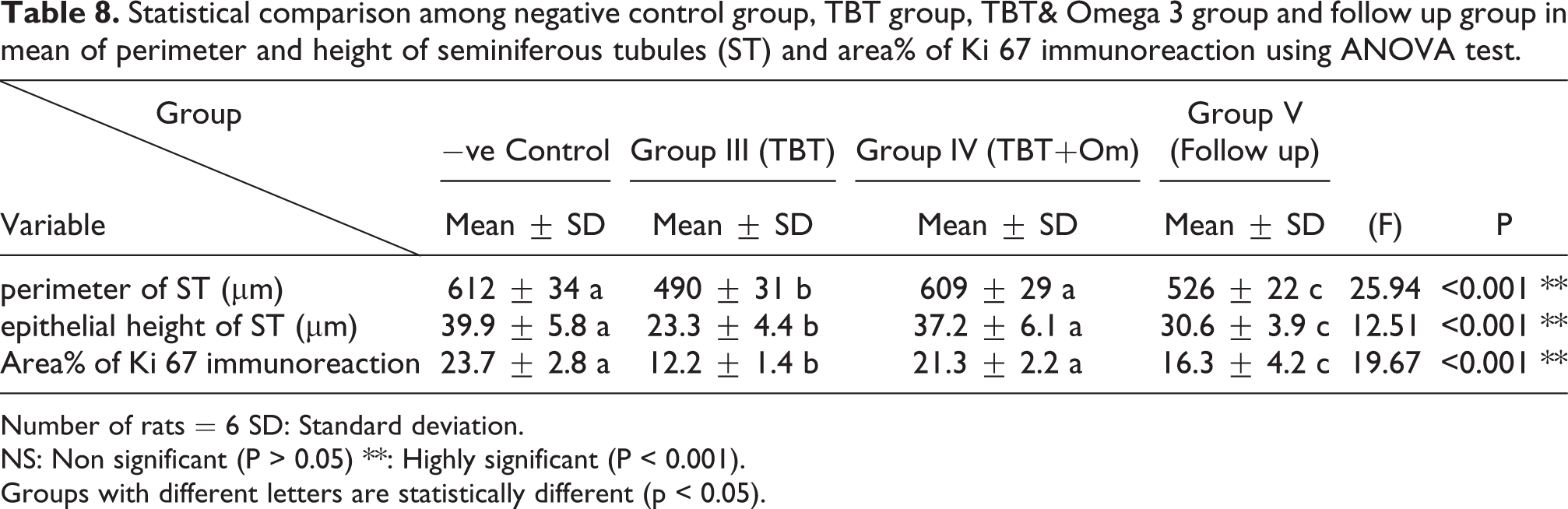
Number of rats = 6 SD: Standard deviation.
NS: Non significant (P > 0.05) **: Highly significant (P < 0.001).
Groups with different letters are statistically different (p < 0.05).
Discussion
The present study showed that, in TBT treated rats, there was an increase in body weight, compared to control group but with no statistically significant difference. This can be explained by the fact that tributyltin chloride has been identified recently as an obesogen. 27 –29 Pivonello et al. 29 mentioned that TBT activates peroxisome proliferator-activated-receptor gamma, a master regulator of adipogenesis. Guo et al. 30 reported that TBT exposure (once every 3 days from postnatal days (PNDs) 24 to 54) resulted in increased weight gain in mice and dyslipidemia. Sena et al. 28 investigated TBT effects on the hypothalamus-pituitary-gonadal (HPG) axis function in female rats and reported an increase in the body weight gain.
A highly statistically significant decrease in both testicular and relative testicular weight was recorded in tributyltin -treated group compared with control group. Kanimozhi et al. 31 reported that significant reductions in testicular weights were observed among TBT-treated syrian hamsters compared with control. These findings are in agreement with those of Omura et al. 32 who reported that in two generations study, male rats treated with oral tributyltin chloride had decreased weights of testes and epididymis. According to Hajizadeh et al., 33 testicular weight largely depends on the population of differentiated spermatogenic cells. Therefore, significant reduction in testicular weight could be attributed to testicular hypocellularity and spermatogenesis suppression induced by tributyltin.
Co administration of omega-3 normalized the body weight, however with no statistically significant difference, and caused a highly significant increase in both testicular and relative testicular weight when compared with TBT group. Dogan et al. 12 reported that the weights of rats in TBT and omega-3 group slightly increased however not being statistically significant. Li et al. 34 reported that after 4 weeks of omega-3 PUFA treatment, the weights of the testes and epididymis in rats were significantly increased compared with non-treated rats.
In the current work, a highly significant decrease in the serum testosterone, FSH, LH was recorded in tributyltin -treated group when compared with control group. This significant decline in these reproductive hormones indicates that TBT has an inhibitory effect on reproduction. There are various hypotheses to explain TBT induced reproductive toxicity.
Under the effect of the hypothalamic gonadotropin releasing hormone
So, the low serum testosterone concentration may be linked to the inhibitory effect of TBT on the secretion of pituitary gonadotropins (FSH and LH) which are involved in testosterone biosynthesis as reported by 4,28,36 that TBT was found to exert toxic effect on hypothalamic pituitary axis disrupting its function at different levels directly and or indirectly by abnormal metabolic regulation of the axis.
Another theory is by direct inhibition of leydig cells as stated by Wu et al. 37 that a short-term exposure to TBT blocked Leydig cell proliferation via down-regulating steroidogenesis-related proteins in adult rat testes resulting in significant reduction in serum testosterone levels, or by inhibition of steroidogenesis in the testis as TBT has a detrimental effect on steroidogenesis resulting in decreased testosterone production, as Diemer et al., 38 Mitra et al. 5 and Mitra et al. 3 stated that it severely decreased the expression of steroidogenic key proteins which are 3β-hydroxysteroid dehydrogenase (3β-HSD), StAR and translocator protein (TSPO).
Furthermore, the increased oxidative stress associated with an impaired antioxidant defense status initiates a cascade of reactions might be responsible for TBT induced reproductive toxicity as ROS can have detrimental effects on critical components of steroidogenic pathway. 3,5,7 Additionally, TBT significantly depletes GSH and since Leydig cells possess high levels of GSH as potent intracellular antioxidant, on depletion, oxidative stress occurs in the cells, resulting in decreased testosterone production. 5
Kanimozhi et al. 31 reported that testosterone levels were significantly decreased after TBT administration in hamsters through upregulation of ApoE (a lipoprotein central to sex hormone synthesis) which in turn negatively affects the testis function. Rantakokko et al. 39 evaluated the impact of antenatal exposure to organotins (OT) on testicular descent at birth and reproductive hormone levels in newborn boys from Denmark and Finland. They reported that higher TBT and OT concentrations were consistently associated with lower serum LH and testosterone levels and that OTs appeared to be a risk factor for cryptorchidism and lower androgen levels.
On the other hand, it was suggested that tributyltin is supposed to be an inhibitor of aromatase (enzyme which metabolize testosterone to17b-estradiol), which increases testosterone levels and then cause imposex in female. 40,41 This disparity in the change of serum T levels induced by TBT is probably due to differences in the route and the time of exposure.
Upon administration of omega-3 fatty acids to TBT treated rats; there was a highly significant increase in serum testosterone, FSH, LH in tributyltin and omega-3 group when compared with TBT group. The results of the present study coincide with Dogan et al. 12 who reported omega-3 as a protective or alleviating substance against the hazardous effects of TBT duo to its profound antioxidant properties.
Mahmoud et al. 34 reported that omega-3 administration resulted in a significant increase in the levels of testosterone and LH hormones to normal levels.
Moreover, omega-3 seems to have a direct effect on leydig cells stimulating the production of testosterone and modulating testicular steroidogenesis. 34,42
Omega-3 was found to increase nitric oxide release as reported by Ross et al. 43 and Mahmoud. 8 Nitric oxide activates Guanylate cyclase enzyme that induces the release of cyclic guanosine monophosphate leading to eventually raising GnRH, LH and FSH, enhancing sperm motility. 44 Moreover, omega-3 was reported to prevent the reduction of plasma testosterone level by inhibiting 5 alpha-reductase with subsequent prevention of conversion of testosterone to dihydrotestosterone. 45 Hu et al. 46 demonstrated that omega-3 potentiates the cellular development and steroid biosynthesis via CYP51 up-regulation.
In the current study, we evaluated the protective role of omega-3 against the oxidative stress changes resulting from the administration of TBT in rats, through measuring the levels of MDA (lipid peroxidation marker) and GPX (marker of antioxidant activity) in the serum. A highly significant increase in serum MDA and a highly significant decrease in serum GPX were noted in TBT -treated group when compared with control group. These results are consistent with previous studies done by Mitra et al., 3 Mitra et al., 47 Mitra et al., 5 Zhang et al., 48 Li et al., 49 Kanimozhi et al., 6 Dogan et al. 12 and El Dine et al. 1 who suggested that the generation of ROS is one of the major mechanisms involved in TBT toxicity causing damage to mitochondrial and other cytoplasmic organelle membrane structures through peroxidation of phospholipids, proteins and nucleotides. The results of the present study are supported by Dogan et al. 12 who reported that TBT caused an increase in the lipid peroxidation, a decrease in the GSH, and a loss of antioxidant defense enzyme activity. Kanimozhi et al. 6 reported that TBT increased production of ROS and decreased the enzymatic and non-enzymatic antioxidant defense systems, leading to lipid peroxidation and cell death in hamsters while Li et al. 43 detected a significant elevation of the oxidative stress markers was observed following TBT exposure. Mitra et al. 47 stated that TBT depleted cellular GSH, increased ROS generation. They concluded that immediate rise in calcium and affection of mitochondrial stability are the causal factor for ROS generation.
Co administration of omega-3 fatty acids with TBT remarkably attenuated the effect of TBT on biochemical markers of oxidative stress and lipid peroxidation, indicating a profound antioxidant role of omega-3 as there was a highly significant decrease in serum MDA and a highly significant increase in serum GPX in TBT& omega-3 group when compared with TBT group. The antioxidant effect of omega-3 fatty acids has been reported previously by multitude of studies. The results of the present study were supported by Dogan et al. 12 who reported that omega-3 ameliorated the increase in the lipid peroxidation and loss of antioxidant defense enzyme activity induced by TBT.
Moreover, Hu et al. 50 reported that omega-3 PUFAs alleviated ROS generation and DNA damage in sertoli cells.
Since spermatozoa are very rich in polyunsaturated fatty acids, they are extremely sensitive to oxidative stress and lipid peroxidation 42 hence reproductive toxicity can be attributed to free radicals initiating lipid peroxidation of unsaturated fatty acids in cell membrane. Even though the human semen is rich in vitamin n C, omega-3 fatty acids exert cellular antioxidant rather than pro oxidant actions. 51
Another theory is that since sperms contain Nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 52 and DHA is an inhibitor of NADPH oxidase in endothelial cells, 51 so omega-3 might prove beneficial effects both because it provides essential fatty acids necessary for sperm survival and because it would lower the production of ROS by sperm itself.
Tributyltin treated rats showed a highly significant decrease in sperm count, viability and motility compared to control group. Moreover, sperms abnormalities were significantly increased. A hummer shaped heads, banana shaped heads and headless tails were detected. The attributed causes for the previous changes are multifactorial. Mitra et al. 5 reported that TBT has the potential to alter spermatogenesis by evoking disturbance of hormonal axis or damage to important testicular cells such as sertoli cells.
Decreased levels of testosterone, FSH, and LH also play a role since these hormones are critical for spermatogenesis. This explanation agree with Kanimozhi et al. 6 who reported that decreased FSH might be one of the reasons causing dysfunction of sertoli cells which play a critical role during spermatogenesis.
Mitra et al. 3 reported that TBT exposure decreased the expression of Tesmin, a marker of spermatogenesis, and induced germ cell death and that may be a major reason behind its spermicidal effect. Moreover, TBT was found to disrupt the blood testicular barrier (BTB), which playa a very pivotal role in spermatogenesis, by 3 folds as stated by Mitra et al. 47
Uygur et al. 42 reported that sperm membranes harbor a higher concentration of polyunsaturated fatty acids which are extremely sensitive to oxidative stress causing ultimate damage that might cause loss of sperm motility, abnormal sperm morphology.
In the work done by Si et al. 53 they reported that TBTC perinatal exposure produced significant and dose dependent decrease in all sperm parameters compared to control.
Upon supplementation with omega-3 fatty acids, there was a highly significant increase in the mean values of sperm count, viability, motility and a highly significant decrease in the percent of sperm abnormal forms as the sperms were with normal heads and tails when compared with tributyltin group. This improvement in semen analysis may be related to their antioxidant effect in reducing oxidative damage and improvement of testicular tissue integrity, which were noted in the current study.
Putri et al. 54 reported that the administration of omega-3 increased the motility and sperms count in obese rats. Khavarimehr et al. 55 reported that omega-3 effectively improved the sperm quality in rats by up-regulating the testicular endocrine and antioxidant statuses and preventing mitochondria-dependent apoptosis pathway. Soliman et al. 56 mentioned that Omega-3 also stimulates spermatogenesis and increases the activity of sertoli cells.
Mendeluck et al. 57 reported that the lipid composition of the sperm cell membrane has a significant effect upon the functional quality of spermatozoa so; higher intake of omega-3 fats was positively associated with better sperm morphology and function. Moreover, oxidative stress at the level of the testicular microenvironment may result in decreased spermatogenesis and sperm damage.
In the work done by Hu et al., 50 they suggested a candidate therapeutic approach for male infertility by the use of ω-3 PUFAs as they reported their protective effect with regard to sertoli cell viability. Moreover, Esmaeili et al. 44 reported that omega-3 improved semen quality and sperm characteristics and it may be due to effective incorporation of unique fatty acids, especially DHA, to membrane.
Several studies 58 –63 have demonstrated the beneficial influence of PUFAs supplementation on normal sperms, recommending that PUFAs dietary supplementation might provide a suitable choice for treatment of male infertility. Oborna et al. 58 and Safarinejad et al. 60 found that men with defects in sperm quality or counts typically had low levels of omega-3, in their semen and sperm cell composition. It was also reported by Vujkovic et al. 64 that omega-3 rich mediterranean style diet boosts the chances of successful pregnancy in previously infertile couples by 40%. On the other hand, Yeste et al. 65 reported that omega-3 administration to boars did not affect sperm concentration, viability or motility compared with control.
Light microscopic examination of sections of the testes of TBT treated group revealed the following histopathological changes: marked disorganized seminiferous tubules, wide separation between the tubules with dark pyknotic nuclei. Blood vessels in the interstitium were dilated and congested. Other sections showed tubules that had lost their lining germinal epithelium, others had marked vacuolations and disorganized epithelial lining. Acidophilic hyaline material was present in the interstitium in between seminiferous tubules.
Similar results were reported by Mitra et al., 47 Mitra et al. 5 and Kanimozhi et al. 6
Kanimozhi et al. 6 found that TBT treatment resulted in regression of seminiferous tubule diameter, necrosis and edema of Leydig cells and degeneration of seminiferous epithelium. Mitra et al. 47 found that TBT exposure resulted in blood–testis barrier disruption and marked tissue damage.
Kanimozhi et al. 31 reported that TBT treatment resulted in sloughing of epithelial cells, outer membrane shrinkage, presence of vacuoles, dead cell debris and distorted seminiferous epithelium. Atrophy of seminiferous tubules and the decrease in spermatogenic cells were morphological indicators of spermatogenesis failure as recorded by Uygur et al. 42
Cytoplasmic vacuolation and degeneration could be considered an indication of cell necrosis. According to Manivannan et al., 66 vacuolations of germ cells could be duo to metabolic disturbance in these cells with a subsequent change in their morphology. Several germ cells had deeply stained pyknotic nuclei. Such finding was confirmed by Atli et al. 67 who considered pyknosis as a sign of germ cell degeneration.
The presence of wide spaces between the tubules in this study suggests the presence of interstitial edema, as had been previously reported by Kanimozhi et al., 6 The marked deposition of collagen fibers might be attributed to the oxidative stress induced by TBT. This explanation coincided with the finding of El-Din and Abd-El Aty 68 who reported that the formation of highly reactive oxidizing molecules led to lipid peroxidation causing damage to proteins and nucleic acids. The end results of these reactions led to an increase in collagen fibers and ground substance formation.
Upon administration of omega-3 fatty acids with TBT, there was a significant improvement of all histopathological changes compared with tissues from rats treated with TBT alone.
Soliman et al. 56 and Mahmoud et al. 35 reported that administration of omega-3 resulted in marked improvement in testicular architecture as seminiferous tubules were more or less similar to those of control with regular basement membrane and healthy looking spermatogenic lineages.
Yan et al. 69 found that in omega-3 treated group, spermatocytes developed better than control group, chromatin around nuclear membrane had more uniform distribution, nucleoli ere prominent, with better spermatogonial development.
Ki-67 is a marker of proliferation. Previous studies have used the immunodetection o Ki-67 as a proof of enhanced or suppressed proliferation. 70 –74
Immunohistochemical staining of ki67 protein in TBT-treated rats showed decreased expression of ki67 antibody as positive reaction for ki67 was only present in the nuclei of very few germinal epithelial cells compared to control group indicated the decrease in proliferative activity and spermatogenesis.
The results of the present study are supported by Dogan et al. 12 who reported that TBT also leads to generation of ROS, death of proteins and nucleic acids inside the cell and finally inhibition of cell proliferation. Wu et al. 37 mentioned the inhibitory role of TBT on cell proliferation which explains the damaged seminiferous tubules in rats treated with TBT.
Mitra et al. 5 reported that TBT reduced cell survival and induced cell death leading to inhibition of cell proliferation. Cheng et al. 75 reported that TBT chloride induced DNA damage germ cell apoptosis and proliferation arrest.
Coadministration of omega-3 fatty acids with TBT improved immunohistochemical results as testicular sections showed strong positive reaction seen in the nuclei of most of the germinal epithelial cells compared to TBT-treated group. This improvement may be attributed to protection from oxidative damage and restoration of testicular architecture allowing for normal germ cell proliferation.
Previous studies have documented the antioxidant, anti-apoptotic of omega 2 fatty acids and proved that omega-3 produced a highly significant decrease in the percentage of oxidative damage and cell death. 35,42,46,50,56,76
Campelo et al. 77 reported that animals receiving oil mix of ω3 (ALA+DHA) showed increased expression of Ki-67 and increase in cell proliferation. Uygur et al. 42 reported that there was an increase in the proliferative activity and spermatogenesis in testis tissues after administration of omega-3 fatty acids.
On the other hand, Zhang et al. 78 reported that omega-3 fatty acids supplementation for a short duration wasn’t sufficient to produce biologically meaningful changes in Ki-67 levels.
To sum up, the improvement in all studied parameters with omega-3 fatty acids may be related to reduction of oxidative damage and improvement of testicular tissue integrity, which were observed in the present study.
After arrest of TBT treatment, there was persistence of changes in all parameters in follow up group .there was a non-significant increase in body weight, a significant decrease in testicular and relative testicular weight compared with control group. There was also a significant decrease in serum testosterone, FSH, LH, GPX, and a significant increase in serum MDA. Testis still showed pyknotic dark nuclei with separation between the cells and decreased positive Ki67 reaction. Also, there was a significant decrease in the mean values of sperm count, viability; motility with some sperms still had sperm abnormalities when compared with control group.
Si et al. 53 reported that perinatal TBTC exposure is implicated in causing long lasting alterations in male reproductive system and that these changes may persist even after TBTC administration ceased and far into adulthood. Moreover, Si et al. 79 reported the tin compound is eliminated very slowly from the tissues upon cessation of administration and that TBT might remain in tissues for 40–60 days after continuous exposure is ended.
Conclusion
Tributyltin is a global environmental pollutant that should be taken into consideration while evaluating the declining male fertility worldwide. The contributing factors in the occurrence of tributyltin -induced reproductive toxicity are inducing testicular oxidative stress, cellular damage and arrest of spermatogenesis with attenuation in antioxidant defenses. Omega-3 polyunsaturated fatty acids PUFAs can protect against tributyltin -induced reproductive toxicity more than arrest of exposure. It is recommended to use omega-3 fatty acids for protection against tributyltin -induced reproductive toxicity. Also, further studies are needed to evaluate the effectiveness of TBT&omega-3 combination with longer duration, larger sample size and larger protective doses than the used in our study. More importantly, we should increase the awareness to reduce exposure to tributyltin and pay attention to its sources to limit adverse outcomes for future generations.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
