Abstract
Talinum paniculatum (Jacq.) Gaertn. (Talinaceae), popularly known as “major gomes,” is a Brazilian Cerrado plant used in traditional medicine and as a food source. Recent studies have demonstrated its diuretic effects. However, no studies have been performed on its effects on the reproductive system. Therefore, we aimed to investigate the effects of the ethanol-soluble fraction of T. paniculatum leaves (ESTP) on general toxicity and on the pubertal development of male and female Wistar rats. For this purpose, the uterotrophic and the pubertal assays were performed. In the uterotrophic test, female immature rats were treated for three consecutive days with 30, 100, or 300 mg/kg of ESTP. Uterus without luminal fluid was weighed and the relative weight calculated. For the pubertal assay, male and female immature rats were submitted to 30-day treatment with 30 or 300 mg/kg of ESTP. Clinical signs of toxicity, biochemical, and histopathological parameters were evaluated. ESTP treatment did not promote estrogenic effects in female rats. In the pubertal test, no daily signs of toxicity or weight loss were observed. Moreover, ESTP did not affect the onset of vaginal opening and preputial separation and did not cause significant changes in biochemical parameters as well as in organ weight and histopathological analyses of animals.
Introduction
Talinum paniculatum (Jacq.) Gaertn. (Talinaceae) is a weedy species from the Brazilian Cerrado popularly known as “major gomes” and “erva gorda.” Its leaves are widely used in folk medicine and as a food source. 1 In Mato Grosso do Sul State, Brazil, local healers usually prescribe the infusion of T. paniculatum leaves to treat several cardiovascular and urinary tract diseases. 2 Bioactive compounds of T. paniculatum leaves include tannins, triterpenes, steroids, saponins, and phytosterols. 1 In addition, a recent study performed by our research group with the ethanol-soluble fraction from T. paniculatum (ESTP) showed the presence of chlorogenic acids, amino acids, nucleosides, O-glycosylated flavones, and organic acids in its composition. 3
Previous pharmacological studies have proved its potential as antidiabetic, antinociceptive, antimicrobial, and to treat gastrointestinal disorders. 1,4 Moreover, T. paniculatum has also demonstrated renal effects as it promoted an important diuretic effect after prolonged exposure and no signs of toxicity after a single exposure in Wistar rats. 3 These findings demonstrate an important pharmacological action of the species, showing its great potential in becoming raw material for the development of new diuretic agents. However, no studies were performed to detect (potential) endocrine and toxicological effects of T. paniculatum leaves in rodents.
Medicinal plants and plant-derived compounds can display a wide range of toxicological effects, including hepatic, renal, cardiac, pulmonary, reproductive, and endocrine toxicity. Some of these effects may not be manifested following acute exposures but may be evident after long-term use. In addition, sensitive populations, such as infants and children, may be more susceptible to adverse effects induced by drugs, including medicinal herbs. 5 Effects on the endocrine system are of particular concern, because hormones have essential roles in the homeostatic control of all organ systems, in tissue differentiation, and growth during development. As known, endocrine-disrupting chemicals (EDCs) can cause a wide range of negative effects, including developmental, reproductive, neurological, and immunological changes in animals and humans. These substances may disrupt the hypothalamic–pituitary–adrenal and the hypothalamic–pituitary–gonadal axis as they may act via hormone receptors as well as in the production, storage, release, transport, metabolism, and also in the elimination of hormones, promoting hormonal imbalance. 6,7
EDCs are considered ubiquitous as they can be found in man-made products such as plasticizers, toiletries, toys, cosmetics, drugs, and pesticides as well as in natural sources such as hormones and plant bioactive compounds. 8 For instance, many plants and their extracts contain phytoestrogens that can interact with estrogen receptors and change the development and function of the endocrine and reproductive systems, which are particularly sensitive to hormonal imbalances. Industrial and natural occurring endocrine disruptors have become a major concern worldwide as they can affect the reproductive development, leading to reproductive disorders and infertility. 9 In vivo male and female pubertal and uterotrophic bioassays are among the recommended screening tests to identify substances with potential to interact with the endocrine system. Based on that, we aimed to investigate the possible effects of T. paniculatum leaf extract on general toxicity and on the development and maturation of male and female rat reproductive system following repeated-dose toxicity protocols as well as its estrogenic potential in the uterotrophic assay using immature female rats.
Materials and methods
Plant material and extract preparation
Talinum paniculatum aerial parts were collected in February 2017 in a Cerrado area in Dourados, Mato Grosso do Sul State, Brazil, at 458 m above sea level (22°12′22.6″S 54°47′43.1″W). A voucher specimen was then prepared, authenticated by Dr Maria do Carmo Vieira, and deposited in the Herbarium of the Federal University of Grande Dourados (UFGD) under the number 5539.
For the extract preparation, T. paniculatum leaves were carefully separated from the stems, washed in running water, and then air-dried in an oven at 40°C for 7 days. Afterwards, dried leaves were ground into fine powder and the infusion was made by pouring 1 L of boiling water (97°C) on each 100 g of ground leaves. The extraction occurred until room temperature was reached (∼5 h) and then the infusion was treated with three volumes of ethanol, originating a precipitate and an ESTP (3.42% yield). ESTP samples were freeze-dried and stored in a freezer at −18°C for further experiments. The chemical characterization of the ESTP used in this study was recently described by Tolouei and collaborators. 3
Chemical analysis of the extract
ESTP was previously analyzed by our research group and the results are already published. Its chemical profile was determined by liquid chromatography using diode-array detection and mass spectrometry. 3
Animals
Forty male and eighty female Wistar rats—postnatal day (PND) 21—were obtained from the animal facility of the Federal University of Paraná (UFPR). Animals were housed under standard conditions of temperature (22 ± 3°C), light (12-h light/dark cycle), humidity (50–60%), and had ad libitum access to filtered water and food pellets. All procedures involving animals were previously approved by the Ethics Committee in Animal Experimentation from UFPR (protocol: 1076).
Uterotrophic bioassay
This test was performed in accordance with the protocol 440 described by the Organization for Economic Co-operation and Development (OECD) in 2007. 10 Forty immature female rats (PND 21) were randomized and divided into five experimental groups as follows: negative control (vehicle, 2 ml/kg), ESTP 30 mg/kg, ESTP 100 mg/kg, ESTP 300 mg/kg, and 17α-ethinylestradiol (3 μg/kg, positive control). Doses were chosen based on the work performed by Tolouei et al., 3 where 30, 100, and 300 mg/kg of ESTP were used for the investigation of the renal and cardiovascular effects in Wistar rats. For the acclimation period, we consider that weaning of animals from the UFPR Bioterium is always done on PND 21. Moreover, the OECD protocol 440 states that the treatment should be completed prior to PND 25. Therefore, treatment started after a single day of acclimation. Rats were treated for three consecutive days by oral gavage (PND 22–24) and doses were daily calculated according to each animal body weight. Twenty-four hours after the last treatment (PND 25), rats were individually weighed and humanely euthanized by isoflurane anesthesia (inhalation) followed by decapitation. Necropsy was performed and uterine horns were carefully detached from the body wall and transferred to a petri dish. Uterus without luminal fluid was weighed and the relative weight calculated.
Pubertal bioassay
Both male and female pubertal assays were conducted simultaneously and were performed in accordance with protocols established by Marty, Crissman, and Carncy, with some modifications. 11,12
Forty male and forty female rats at 21 days of age were randomized and divided into eight experimental groups (n = 10). Animals were treated by oral gavage from PND 23 to PND 53 with two doses of ESTP (30 and 300 mg/kg), vehicle (filtered water, 5 ml/kg), di (2-ethylhexyl) phthalate (DEHP 750 mg/kg, male positive control), or 17α-ethinylestradiol (3 μg/kg, female positive control). The ESTP doses were based on a previous study performed by Tolouei et al. 3 The DEHP dose was based on previous studies performed by Kita et al. 13 and Venturelli et al., 14 while the 17α-ethinylestradiol dose was chosen on the basis of recent uterotrophic assays conducted in our laboratory (unpublished).
Throughout the experimental period, all animals were carefully observed once daily for any clinical signs of toxicity according to the Hippocratic screening as follows: general activity, reflexes, response to tail touch and grip, straightening, strength to grab, tremors, Straub tail reaction, sedation, lacrimation, cyanosis, salivation, and piloerection. 15 Body weights were measured daily for dose calculation and statistical analysis at 23, 29, 36, 43, and 50 days of age. In addition, food and water consumption were evaluated on days 23, 29, 36, 43, and 50 after birth.
Vaginal opening (VO) and preputial separation (PPS), which are external markers of puberty onset, were examined from PND 30 and PND 35 onwards in female and male rats, respectively. On the day VO and PPS were achieved, weight and age of animals were recorded.
After the last treatment (PND 54), overnight fasted animals were first weighed and then euthanized by isoflurane anesthesia (inhalation) followed by decapitation. Blood samples were collected for further analysis. On necropsy, vital (heart, spleen, liver, and kidneys) and reproductive organs (uterus, ovaries, testes, epididymis, prostate, glans penis, levator ani muscle, bulbourethral glands, and seminal vesicles) were removed, carefully cleaned, weighed, and macroscopically analyzed for any gross changes. Absolute (g) and relative (%) organ weights were calculated based on animal body weights determined right before euthanasia. Thyroid was also removed but as it was still attached to the trachea, absolute, and relative weights were not calculated. Thyroid separation was only performed before histopathological evaluation.
After necropsy, thyroid, heart, spleen, liver, kidneys, uterus, ovaries, testes, and epididymis samples were sent to histopathological analysis that was performed by two veterinary pathologists from the UFGD.
Biochemical analysis
Analysis was performed using commercial kits from Bioclin brand (Belo Horizonte, Minas Gerais, Brazil) in a Mindray BS-200® automated biochemical analyzer and the following parameters were evaluated: aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase, gamma-glutamyl transpeptidase (GGT), total protein, albumin, blood urea nitrogen (BUN), creatinine, calcium, total cholesterol, triglycerides, glucose, direct bilirubin, total bilirubin, indirect bilirubin, and uric acid.
Histopathology
Vital (heart, liver, spleen, and kidney) and reproductive organs (ovary, uterus, testis, and epididymis) and the thyroid gland attached to the trachea were removed and cut into 5 mm fragments. Fragments of thyroid gland and all vital and female reproductive organs were fixed in 10% buffered formalin. Male reproductive organs were fixed in Bouin’s solution for 6 h. Thereafter, fragments were washed in tap water and immersed in 70% ethanol, which was changed every 24 h for 7 days. Then, fragments were cleaved, dehydrated with increasing absolute ethanol concentrations, diaphanized in xylol, and embedded in paraffin. All sections were cut at a thickness of 4 µm and stained with hematoxylin and eosin (HE) for evaluation under light microscopy (40×). The parameters analyzed were based on the presence or absence of reversible and/or irreversible cellular lesions.
Statistical analysis
Data were analyzed by one-way analysis of variance, followed by Dunnett’s test or by Student’s t-test when applicable. Results are expressed as mean ± standard error of the mean and the level of statistical significance was 5% (p < 0.05). Graphs were drawn and statistical analyses were performed using GraphPad Prism software version 6.0.
Results
ESTP does not induce estrogenic effects in immature female rats
In the uterotrophic assay, female rats exposed to ESTP 30, 100, and 300 mg/kg did not present any significant differences regarding the relative empty uterus weight when compared to the control. As expected, animals from the positive control (17α-ethinylestradiol 3 μg/kg) presented a significant increase in relative uterus weight when compared to the control (Figure 1).

Relative empty uterus weight (%) of female rats treated orally from PND 22 to PND 24 with ESTP 30, 100, and 300 mg/kg. The negative control group received the vehicle (filtered water) alone and the positive control group received 17α-ethinylestradiol (3 μg/kg). Bars represent the mean ± SEM (n = 8). *Represents significantly different from control (p < 0.05) by ANOVA followed by Dunnett’s test. PND: postnatal day; ESTP: ethanol-soluble fraction of Talinum paniculatum leaves; SEM: standard error of the mean; ANOVA: analysis of variance.
ESTP does not cause death or visible signs of toxicity in rats
No deaths nor clinical signs of toxicity, according to the Hippocratic screening, were observed in male and female rats treated orally from PND 33 to PND 53 with ESTP 30 and 300 mg/kg as well as in animals treated with the vehicle, DEHP (750 mg/kg, male negative control), or 17α-ethinylestradiol (3 μg/kg, female positive control). The behavior of animals was recorded daily and no changes were observed (data not shown).
ESTP does not cause animal weight loss or changes in food and water consumption
Our data show that all animals submitted to this research gained weight along the experimental period (Table 1). When statistical analyses were performed, it is possible to observe that animals treated with ESTP 30 and 300 mg/kg gained weight along the 30-day treatment (Figure 2). Thus, no significant differences were observed in relation to final body weight of animals treated with both doses of ESTP when compared to the control (Table 1). On the other hand, animals treated with 17α-ethinylestradiol (female positive control) and DEHP (male positive control) presented a significant decrease in body weight gain when compared to the control (Table 1; Figure 2). Regarding food and water consumption, no significant differences were observed in groups treated with ESTP 30 and 300 mg/kg when compared to the control (Table 1).
Effects of a 30-day administration of ESTP from Talinum paniculatum on body weight gain (g), food (g/day), and water (ml/day) consumption of female and male rats in the pubertal assay.a

ESTP: ethanol-soluble fraction of Talinum paniculatum leaves; SEM: standard error of the mean; ANOVA: analysis of variance; DEHP: di (2-ethylhexyl) phthalate.
a The negative control group received the vehicle (filtered water). The positive control was treated with 17α-ethinylestradiol (3 μg/kg) for female rats and with DEHP (750 mg/kg) for male rats. Data are shown as mean ± SEM (n = 8).
* Represents significantly different from negative control (p < 0.05) by ANOVA followed by Dunnett’s test.

Mean body weight gains of female (a) and male (b) rats during the pubertal assay. Body weights were daily monitored during the 30-day treatment period and body weight gains were analyzed statistically for the intervals 23–29, 29–36, 36–43, and 43–50 days of age. Animals were treated orally from PND 23 to PND 53 with ESTP 30 and 300 mg/kg. The negative control group received the vehicle (filtered water) alone and the positive control group received 17α-ethinylestradiol (3 μg/kg) for females and DEHP (750 mg/kg) for males. Bars represent the mean ± SEM (n = 10). *Represents significantly different from control (p < 0.05) by ANOVA followed by Dunnett’s test. PND: postnatal day; ESTP: ethanol-soluble fraction of Talinum paniculatum leaves; SEM: standard error of the mean; ANOVA: analysis of variance; DEHP: di (2-ethylhexyl) phthalate.
ESTP does not affect the onset of VO and PPS
ESTP (30 and 300 mg/kg) did not cause any significant differences in the onset of VO or in PPS when compared to the control (Figure 3). As expected, the positive control for males (DEHP, 750 mg/kg) delayed the age of PPS as it was significantly different from the negative control group (Figure 3). In females, VO occurred earlier in animals treated with the positive control (17α-ethinylestradiol, 3 μg/kg), but this change was not statistically different from the control.
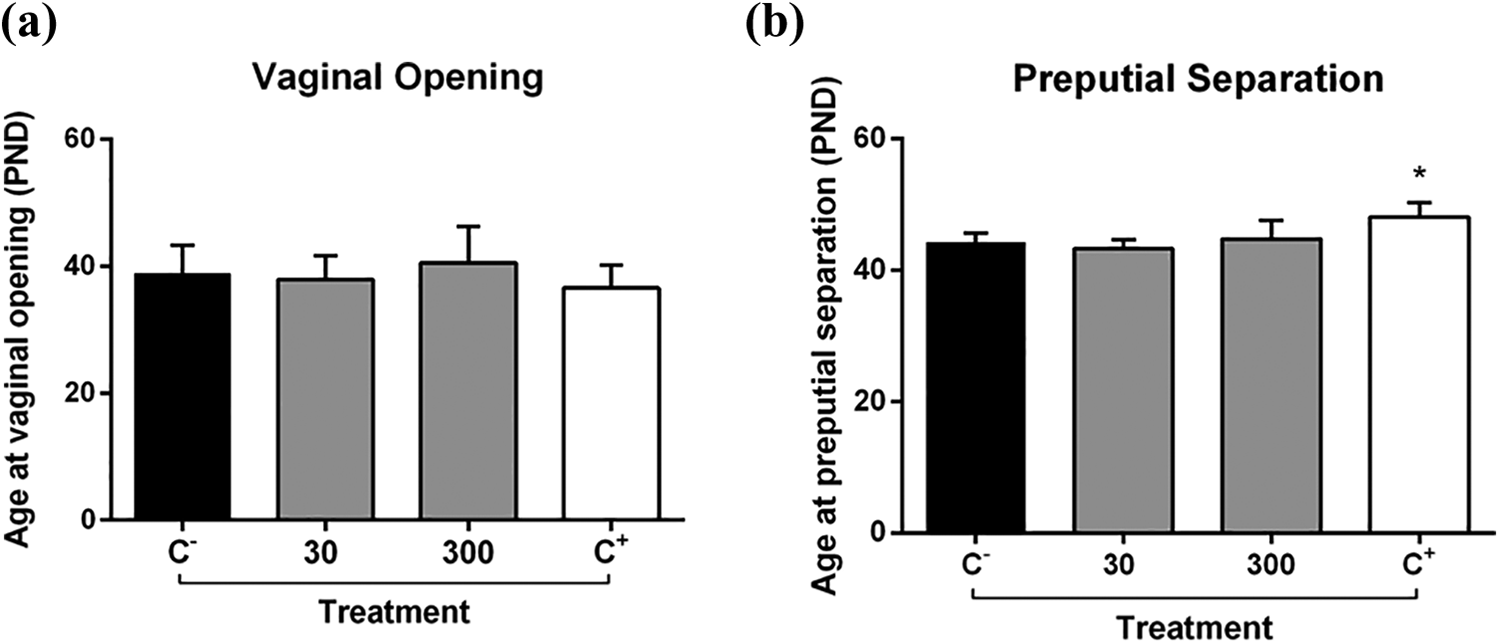
VO (a) and PPS (b) of female and male rats treated orally from PND 23 to PND 53 with ESTP 30 and 300 mg/kg. The negative control group received the vehicle (filtered water) alone and the positive control group received 17α-ethinylestradiol (3 μg/kg) for females and DEHP (750 mg/kg) for males. Bars represent the mean ± SEM (n = 10). *Represents significantly different from control (p < 0.05) by ANOVA followed by Dunnett’s test. VO: vaginal opening; PPS: preputial separation; PND: postnatal day; ESTP: ethanol-soluble fraction of Talinum paniculatum leaves; SEM: standard error of the mean; ANOVA: analysis of variance; DEHP: di (2-ethylhexyl) phthalate.
ESTP does not induce significant changes in relative organ weight
No significant changes were observed in relative organ weights from female and male rats treated orally with ESTP 30 and 300 mg/kg when compared to the control (Table 2). However, animals treated with DEHP 750 mg/kg (positive control for male rats) showed significant differences in heart, liver, spleen, testicles, epididymis, empty seminal vesicle, and levator ani muscle relative weights when compared to the control (Table 2).
Effects of a 30-day administration of ESTP from Talinum paniculatum on relative organ weight (g/100 g body weight) of female and male rats in the pubertal assay.a

ESTP: ethanol-soluble fraction of Talinum paniculatum leaves; SEM: standard error of the mean; ANOVA: analysis of variance; DEHP: di (2-ethylhexyl) phthalate.
a The negative control group received the vehicle (filtered water). The positive control was treated with 17α-ethinylestradiol (3 μg/kg) for female rats and with DEHP (750 mg/kg) for male rats. Data are shown as mean ± SEM (n = 8).
* Represents significantly different from negative control (p < 0.05) by ANOVA followed by Dunnett’s test.
Biochemical analysis
The effects of a 30-day administration of ESTP from T. paniculatum on biochemical parameters of female and male rats in the pubertal assay are presented in Table 3. No significant differences were observed in levels of GGT, total protein, albumin, total cholesterol, triglycerides, glucose, direct bilirubin, indirect bilirubin, and uric acid from females treated with two doses of ESTP (30 and 300 mg/kg) when compared to the control. However, it is possible to observe a significant decrease in levels of ALT in females treated with both doses of ESTP (30 and 300 mg/kg) when compared to the control. Thus, a subtle but significant increase in levels of calcium was observed in females treated with ESTP 30 mg/kg when compared to the control. Significant differences were observed in levels of AST, ALT, alkaline phosphatase, BUN, creatinine, and total bilirubin from female rats from the positive control (17α-ethinylestradiol) when compared to the negative control group.
Effects of a 30-day administration of ESTP from Talinum paniculatum on biochemical parameters of female and male rats in the pubertal assay.a

ESTP: ethanol-soluble fraction of Talinum paniculatum leaves; SEM: standard error of the mean; ANOVA: analysis of variance; DEHP: di (2-ethylhexyl) phthalate; AST: aspartate aminotransferase, ALT: alanine aminotransferase, A. phosphatase: alkaline phosphatase, GGT: gamma-glutamyl transpeptidase, T. protein: total protein, BUN: blood urea nitrogen, T. cholesterol: total cholesterol, D. bilirubin: direct bilirubin, T. bilirubin: total bilirubin, I. bilirubin: indirect bilirubin..
a The negative control group received the vehicle (filtered water). The positive control was treated with 17α-ethinylestradiol (3 μg/kg) for female rats and with DEHP (750 mg/kg) for male rats. Data are shown as mean ± SEM (n = 8).
* Represents significantly different from negative control (p < 0.05) by ANOVA followed by Dunnett’s test.
Regarding the biochemical parameters evaluated in male rats, only creatinine was significantly different in groups treated with ESTP (30 and 300 mg/kg) when compared to the control. Besides, alkaline phosphatase and creatinine were significantly different in animals from the positive control group (DEHP) when compared to the negative control (Table 3).
Histopathology
No signs of toxicity were observed in the thyroid, liver, ovary, uterus, testis, and epididymis of all ESTP-treated rats submitted to the pubertal assay (Figures 4 and 5). In addition, no toxic effects were observed in the heart, spleen, and kidney of treated animals (Supplementary Material S1). However, all male rats from the positive control group presented a pronounced diffuse degeneration of the germinal epithelium of the seminiferous tubules (indicated by arrowheads) where only remaining Sertoli cells (arrow) are observed (Figure 5). In the epididymis of these animals, no degenerative changes were found, only eosinophilic amorphous material and cellular debris in the epididymal light-duct (arrow) (Figure 5).

Histopathological assessment of thyroid, ovary, uterus, and liver from female rats treated orally with the vehicle (2 ml/kg, negative control), ESTP 30 mg/kg, ESTP 300 mg/kg, and 17α-ethinylestradiol (3 μg/kg, positive control) in the pubertal assay. HE (40×). ESTP: ethanol-soluble fraction of Talinum paniculatum leaves; HE: hematoxylin and eosin.

Histopathological assessment of thyroid, testis, epididymis, and liver from male rats treated orally with the vehicle (2 ml/kg, negative control), ESTP 30 mg/kg, ESTP 300 mg/kg, and DEHP (750 mg/kg, positive control) in the pubertal assay. Arrows and arrowheads indicate significant changes. HE (40×). ESTP: ethanol-soluble fraction of Talinum paniculatum leaves; DEHP: di (2-ethylhexyl) phthalate; HE: hematoxylin and eosin.
Discussion
The use of medicinal plants for the prevention and treatment of various diseases is a secular tradition performed worldwide. This practice has gained more emphasis nowadays due to the belief that natural compounds are free from toxic effects. However, although natural, plant species used medicinally can cause toxic effects on the renal, hepatic, cardiac, pulmonary, endocrinem and reproductive systems. These effects seem to be more prevalent in sensitive populations, such as infants and pubescent adolescents, as they are particularly vulnerable to adverse effects induced by synthetic and natural drugs. 5
Previous reports have demonstrated several scenarios in which medicinal plants proved to be toxic. In the early 1990s, the widespread use of Teucrium chamaedrys L. was responsible for the occurrence of a hepatitis epidemic in France. High incidence of this serious toxic effect was attributed to the presence of neo-clerodane-type diterpenoids in the species, which are transformed by cytochrome P450 enzymes into hepatotoxic metabolites. 16 In South Africa, the decoction of the roots of Polygala fruticose, popularly used to treat tuberculosis, gastrointestinal disorders, gonorrhea, and to improve general circulation 17 demonstrated significant toxicity as it caused anti-hemopoietic, hepatotoxic, and nephrotoxic effects in female rats and mice. 18
Other medicinal plants have demonstrated to be potentially toxic, such as Ruta graveolens, Zengiber officinalis, and Symphitum officinalis, which had an abortive effect by stimulating uterine motility, Lantana camara L., known for its hepatotoxicity, and Datura suaveolens Humb. & Bopl ex Willd., which has the potential to damage the central nervous system. 19
Concern over the possible harmful effects of substances used for medicinal purposes gained momentum in the mid-twentieth century after the thalidomide tragedy. Although disastrous, this episode served as a starting point for the insertion of more effective safety assessment methods, which have been widely applied so far. 20 However, the number of preclinical studies on medicinal plants regarding possible systemic toxicity and negative effects during pubertal development is still scarce. 21 Based on that, we aimed to investigate the possible effects of T. paniculatum leaf extract on general toxicity and on the pubertal development of male and female Wistar rats.
For this reason, the first step of this work consisted in the evaluation of possible estrogenic effects of the ESTP in female rats through the uterotrophic assay. In this test, different doses of ESTP did not cause estrogenic effects in immature female rats treated for three consecutive days. This is in contrast to a study performed by Thanamool and collaborators 22 in which T. paniculatum leaf extract promoted estrogenic effects in ovariectomized adult rats. Although both studies were performed with the same part of the same plant species, animals from this study were treated for 42 consecutive days whereas ours were treated for only 3 days as previously described by the OECD protocol 440. 10 Another reason why contradictory results were observed must be related to a variation of the major secondary metabolites produced by each sample. In the chemical characterization of T. paniculatum extract conducted by Thanamool et al., 22 nonsteroidal phytoestrogens (campesterol, B-sitosterol, stigmasterol, stigmastan-3-ol, stigmast-22-en-3-ol, and stigmastanol) were identified as the major metabolites. As known, phytosterols have been claimed to possess estrogenic activity due to their affinity to estrogen receptors. 23 –26 Therefore, differences in the phytochemical composition in both extracts may be the reason why an estrogenic effect was observed in their study and not in ours. Such variation in the amount and the type of secondary metabolites produced by plants usually occurs due to differences in climate, temperature, water availability, altitude, UV radiation, and nutrients of the collection site. 27,28 Furthermore, as the dose that presented estrogenic effect in Thanamool’s study was three times higher (1000 mg/kg) than the highest dose used in our study (300 mg/kg), it is also believed that the estrogenic effect may have occurred in a dose-dependent manner.
In the second stage of this work, we investigated the effects of ESTP on general toxicity and on the development and maturation of the reproductive system through the pubertal assay. Our data showed that male and female rats treated orally with two doses of ESTP gained weight along the 30-day treatment. Thus, weekly evaluation of feed and water consumption have shown that animals treated with ESTP had similar intakes when compared to the control. Such parameters are of great importance as reduced body weight and food and water intake are clear signs of systemic general toxicity. 29,30 Besides, pubertal exposure to endocrine-disrupting substances might lead to metabolic disorders such as exacerbated weight gain and even obesity. 31,32
When it comes to pubertal development of male and female rats, it is well known that PPS and VO are external signs of such development. According to Stoker et al., 33 substances with toxic and endocrine-disrupting potentials can affect the onsets of such signs. In this study, both doses of ESTP did not advance nor delay the onset of VO nor PPS in animals tested. However, treatment of male rats with a high-DEHP dose (750 mg/kg/day) significantly delayed the age at PPS, which is in accordance with the antiandrogenic mode of action of this compound. On the other hand, although 17α-ethinylestradiol (3 μg/kg) was able to increase the uterine weight in the uterotrophic assay, it was unable to change the age at VO or other estrogen-sensitive parameters in female rats treated throughout puberty. These results may indicate that higher estrogen oral doses are needed to change reproductive parameters in older animals, perhaps because of increased biotransformation. 34,35
Regarding the relative organ weight of animals exposed to different ESTP doses, no significant differences were observed in vital or in the reproductive organs of males and females when compared to the control group. However, some significant differences were observed in the relative organ weights of the male positive control group when compared to the control group. As known, organ weight is an essential endpoint for the identification of potentially toxic chemicals. 36 Besides, relative reproductive organ weights are sensitive measures of detrimental effects of substances on the reproductive and endocrine systems and serve as crucial bases in toxicological investigations. 37
We also evaluated the biochemical parameters of male and female rats submitted to the pubertal assay aiming to investigate target organs and a possible systemic toxicity. These parameters are essential as they provide information on major harmful effects in tissues, mainly in kidney and liver. 38 In this study, female rats treated with ESTP in all tested doses presented a significant decrease in levels of ALT when compared to the control. As known, ALT ratio is one of the most reliable indicators of liver damage. Therefore, the observed decrease of ALT activity in ESTP-treated animals is possibly not related to any liver injury, especially considering that the measured values are within the normal range for the species. 39 In relation to the male rats, only creatinine was significantly increased in groups treated with both doses of ESTP when compared to the control. Blood measurement of creatinine is an important indicator of renal health. 40 However, as creatinine values obtained in male rats treated with ESTP-highest dose (300 mg/kg) were within the normal range for the species, 39 we did not identify any relevant clinical impairment derived from this change. In addition, further histopathological assessments revealed the absence of tissue damage in kidneys from both groups.
Finally, a detailed histopathological analysis was performed in vital and reproductive organs to investigate possible pathological changes or signs of toxicity in animals exposed to ESTP. Corroborating the data already presented; no treatment-related effects were observed. Thus, the data obtained extend previous ethnopharmacological knowledge and provide important safety data for the prolonged use of T. paniculatum.
Our data demonstrate that ESTP did not cause any signs of toxicity after prolonged exposure and does not play a direct effect on estrogen- and androgen-dependent organs as it did not delay or accelerate the onset of puberty of male and female rats. Therefore, T. paniculatum can be considered safe in Wistar rats at all doses tested. Further studies on the genotoxicity and reproductive toxicology of this plant are required to proceed to clinical trials.
Supplemental material
Supplementary_materials_HET - Effects of Talinum paniculatum (Jacq.) Gaertn. leaf extract on general toxicity and pubertal development of rats
Supplementary_materials_HET for Effects of Talinum paniculatum (Jacq.) Gaertn. leaf extract on general toxicity and pubertal development of rats by SEL Tolouei, GN da Silva, TZ Curi, MT Passoni, DCK Ribeiro, HG Meldola, N Grechi, GS Hey, RIC Souza, AC dos Santos, OC Beltrame, PR Dalsenter, AJ Martino-Andrade and A Gasparotto Junior in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brazil, 449464/2014-8) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, Brazil).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
