Abstract
Cleft palate (CP), a congenital defect in the oral and maxillofacial regions, is difficult to detect prenatally. This study investigated the correlation between differentially expressed proteins in serum and CP induced by all-trans retinoic acid (atRA) and 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) in mice. We studied 80 mice in the following groups: male mice (male; n = 6), nonpregnant female control mice (NP-CRL; n = 6), healthy pregnant controls (P-CRL; Con; n = 24), pregnant mice with CP induced by atRA (n = 24), or pregnant mice with CP induced by TCDD (TCDD; n = 20). Pregnant mice were given with atRA (100 mg/kg) or TCDD (40 μg/kg), or corn oil by oral gavage at E10.5. The serum samples were collected and eight proteins—including interleukin (IL)-12p40, IL-12p70, receptor for advanced glycation end products (RAGE), interferon (IFN)-γ, IFN-β, IL-10, leukemia inhibitory factor (LIF), and epiregulin—were detected by enzyme-linked immunosorbent assay. Placental tissues were immunostained for IL-12p40 and RAGE from stages E13.5 to E16.5. In P-CRL mice, serum IL-12p40 was significantly increased at E13.5 and declined over E14.5–E16.5. P-CRL had lower IFN-γ levels at E13.5 compared with NP-CRL. The CP groups showed lower concentrations of IL-12p40 at E13.5–E14.5 and clearly higher concentrations of soluble RAGE (sRAGE) at E13.5 when compared with P-CRL. IL-12p40 immunostaining clearly decreased in placental tissue sections obtained from E13.5 to E14.5 in both CP groups. These findings suggest that reduced levels of IL-12p40 and increased levels of sRAGE in serum may be correlated with chemically induced CP in mice, but further studies would be required to establish this.
Introduction
Oral clefts are common congenital defects affecting roughly 1 in 500 live births. 1 Both genetic and environmental factors play key roles in the clefts pathophysiology. 2 Oral clefts are often divided into cleft lip with or without cleft palate (CL/P) and cleft palate (CP) only. Individuals with oral clefts experience problems with feeding, speaking, and social integration.
Close similarities of the embryonic processes in the palate between humans and mice have been noted. 3 The development of the secondary palate starts at embryonic day 12 (E12) in mice and gestation week 6 in humans. Palatal fusion is completed by E16 in mice and around week 12 in humans. Disturbance of the regulated process at any stage may result in CP. 4 Drugs, radiation exposure, infections, smoking, and alcohol consumption during pregnancy enhance the risk of CP. 5 To study the defect, researchers have successfully established the mouse CP model induced by all-trans retinoic acid (atRA) or 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). 6,7
Oral clefts are usually diagnosed through prenatal ultrasonographic examination. 8 However, the accuracy of ultrasonography for prenatal diagnosis of oral clefts is highly variable and dependent on the experience of the sonographer, fetal position, maternal body habitus, the amount of amniotic fluid (AF), and the type of cleft. 8 Cash et al. reported that the detection rate for oral clefts was 67–93% by fetal ultrasonography, while more than 50% of isolated CP were not detected. 9
Biomarker in body fluids is an attractive tool to diagnose disease in biomedical research. 10 –12 A previous study demonstrated that dams with CL/P had significantly lower thiamine concentrations in serum and AF in mice. 13 Another study confirmed that elevated CLP risks were related with abnormal expression levels of pyridoxal phosphate (vitamin B6), choline, and symmetric dimethylarginine in women’s mid-pregnancy serum. 14 Other studies identified the reduced levels of retinol-binding protein 4 and vitamin A in serum from children with nonsyndromic CLP children after birth. 15 Taking these studies into consideration, there may be special components related to CP fetuses in the serum of pregnant women, which can be used as a specific biomarker combined with ultrasound technology in prenatal diagnosis to improve the detection rate of CP and reduce birth defects.
Our previous study had demonstrated that differential expression levels of proteins in AF are correlated to CP embryos induced by atRA, as revealed by label-based mouse antibody array, including interleukin (IL)-12p40, IL-12p70, receptor for advanced glycation end products (RAGE), interferon (IFN-γ), IFN-β, IL-10, leukemia inhibitory factor (LIF), and epiregulin. 16 However, AF detection is an invasive diagnosis and requires certain indications, which greatly limit its clinical application in the field of early diagnosis. Inversely, blood samples are easily accessible in laboratories. Identification of novel biomarkers from blood that can be used for the early diagnosis of CP is urgently needed. In this study, we established the mouse CP model induced by atRA and TCDD. Then, we used the enzyme-linked immunosorbent assay (ELISA) to compare the eight protein levels of serum from pregnant females with CP embryos with those from healthy control, and the immunohistochemical (IHC) staining to explore the source of these proteins in serum correlated to chemically induced CP.
Materials and methods
Chemicals
at RA was purchased from Sigma (St Louis, Missouri, USA). atRA powder was dissolved and stored in dimethyl sulfoxide. All works involving atRA were conducted under dim yellow light to prevent photo-oxidation. TCDD was obtained from AccuStandard (New Haven, Connecticut, USA). TCDD powder was dissolved and stored in toluene. Dose formulations for treatment were prepared on the day of treatment by mixing the atRA solution or TCDD solution in corn oil vehicle.
Animals and exposure
80 females and 30 males of C57BL/6 J mice (age 8–12 weeks; weight 25–30 g) were purchased from Experimental Animal Research Center of Hubei Province (Wuhan, China) and housed under specific-pathogen-free conditions. Female mice were crossed with fertile males overnight, and the presence of a vaginal plug was designated as E0.5. Pregnant females at E10.5 were randomly divided into three groups for treatment: atRA (100 mg/kg) or TCDD (40 μg/kg) dissolved in corn oil by oral gavage or an equal amount of corn oil as a control. Fetuses and serum obtained from 80 mice were harvested in the following groups: male mice (male; n = 6), nonpregnant female control mice (NP-CRL; n = 6), healthy pregnant controls (P-CRL; Con; n = 6 mice per stage from E13.5 to E16.5), pregnant mice with CP induced by atRA (n = 6 mice per stage from E13.5 to E16.5), or pregnant mice with CP induced by TCDD (n = 5 mice per stage from E13.5 to E16.5). The experimental protocols were conducted in accordance with the Guidelines for the Care and Use of Laboratory Animals and approved by the Animal Care and Use Committee of Wuhan University.
Serum samples collection and preparation
Blood was obtained from the orbital vein after anesthesia and collected in centrifuge tubes. The blood samples were allowed to clot for 2 h at room temperature before centrifuging at 2000 ×g for 20 min, after which the supernatant is aspirated and stored in tubes. The serum samples were immediately stored at −80°C until use.
Hematoxylin and eosin staining
Embryo heads obtained from pregnant mice at stages from E13.5 to E16.5 were dissected for hematoxylin and eosin (H&E) staining (n = 5 litters per group). They were fixed in 4% paraformaldehyde, dehydrated through an ethanol series, embedded in paraffin, sectioned by routine procedures (5 μm), and stained with H&E for microscopic observation.
Enzyme-linked immunosorbent assay
The serum levels of the eight proteins were determined with commercially available ELISA kits: IL-12p40, IL-12p70, soluble RAGE (sRAGE) and epiregulin (R&D Systems, Minneapolis, Minnesota, USA); IFN-γ and IL-10 (RayBiotech, Norcross, Georgia, USA); IFN-β and LIF (Enzyme-linked Biotechnology, Co., Ltd, Shanghai, China). Serum samples were collected from the five groups. Each sample was prepared by fivefold dilution and measured in duplicate according to the manufacturer’s protocol.
Immunohistochemistry
Mouse placenta obtained from E13.5 to E16.5 from the three groups were dissected and fixed in 4% paraformaldehyde overnight at 4°C for IHC (n = 5 litters per stage and group). The 5-µm paraffin sections were deparaffinized, rehydrated, and subjected to antigen retrieval using a high-pressure method. A mixture of 30% H2O2 and methanol (1/9, v/v) was used to inhibit endogenous peroxidase activity. The sections were incubated overnight with anti-IL-12p40 antibody (Proteintech Group, Inc., Wuhan, China, 1:40 dilution) and anti-RAGE antibody (Proteintech Group, Inc., 1:50 dilution) at 4°C and then detected using a rabbit SP kit. Staining was performed with diaminobenzidine and counterstained with hematoxylin. Image analysis and comparison were conducted by Image J (NIH, Bethesda, MD) plus IHC Profiler plugin. IHC optical density score (from 1 to 4) for the IHC images was calculated according to the method of Seyed and Hunger. 17
Statistical analysis
Kolmogorov–Smirnov and Shapiro–Wilk tests were used to determine whether data were normally distributed. All quantitative data were presented as mean ± standard deviation, and data were analyzed using one-way or two-way analysis of variance, or Kruskal–Wallis and post hoc Mann–Whitney U-tests at a significance level of p < 0.05.
Results
Effect of atRA or TCDD administration at E10.5 on the development of CP
The morphological outcomes in terms of palatal development of E13.5–E16.5 fetuses treated with atRA and TCDD were examined by H&E staining. Both atRA (100 mg/kg) and TCDD (40 μg/kg) exposure to pregnant mice at E10.5 induced CP in all of the fetuses (Figure 1). H&E staining of embryo heads sectioned at E13.5–E16.5 showed that palatal fusion was completed in the control group (Figure 1). The bilateral palatal shelves grow down vertically along the two sides of the tongue at E13.5 (Figure 1(a)), and then, the shelves elevate above the tongue and grow toward each other at E14.5 (Figure 1(b)). At E15.5, the bilateral shelves adhere to form the midline epithelial seam (MES) (Figure 1(c)). The MES then disappears and palatal fusion is completed by E16.5 (Figure 1(d)). By contrast, the bilateral palatal shelves in the atRA group failed to elevate into a horizontal position above the tongue from E14.5 to E16.5, resulting in the formation of CP (Figure 1(e) to (h)), whereas in the TCDD group, the opposing palatal shelves were delayed in elevating above the tongue until E15.5 and could not successfully adhere to each other due to their abnormally short length, resulting in the defect (Figure 1(i) to (l)).

atRA and TCDD impair palate development and generate cleft palate. (a–d) Palatal fusion was completed in the control group. (e–h) The bilateral palatal shelves in the atRA group failed to elevate into a horizontal position, resulting in the formation of cleft palate. (i–l) In the TCDD group, the opposing palatal shelves could not be adhered onto each other, resulting in the defect (n = 5 litters per group). Scale bar: 200 μm. ps: palatal shelf; p: palate; t: tongue; atRA: all-trans retinoic acid; TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin.
Relationship between gender, pregnancy, gestational age, and protein concentration in maternal serum of untreated mice
The expression levels of proteins in serum from male, NP-CRL and P-CRL mice were detected by ELISA, including IL-12p40, IL-12p70, sRAGE, IFN-γ, IFN-β, IL-10, LIF, and epiregulin (Figure 2). Overall, P-CRL at E13.5 had higher IL-12p40 levels compared with male (Figure 2(a); p = 0.035) and lower IFN-γ levels compared with NP-CRL (Figure 2(c); p = 0.045). Moreover, pregnancy data were analyzed based on gestational age intervals, we found that serum IL-12p40 was significantly increased at E13.5 and declined over E14.5–E16.5 (Figure 2(a); E13.5 vs. E15.5: p = 0.022, E13.5 vs. E16.5: p = 0.003). The serum levels of sRAGE, IFN-γ, IFN-β, IL-10, and LIF in P-CRL mice were relatively stable from E13.5 to E16.5, and no statistical difference was observed (Figure 2(b) to (f)). In addition, we evaluated the expression levels of IL-12p70 and epiregulin in serum, but they were undetectable despite our best efforts (data not shown).
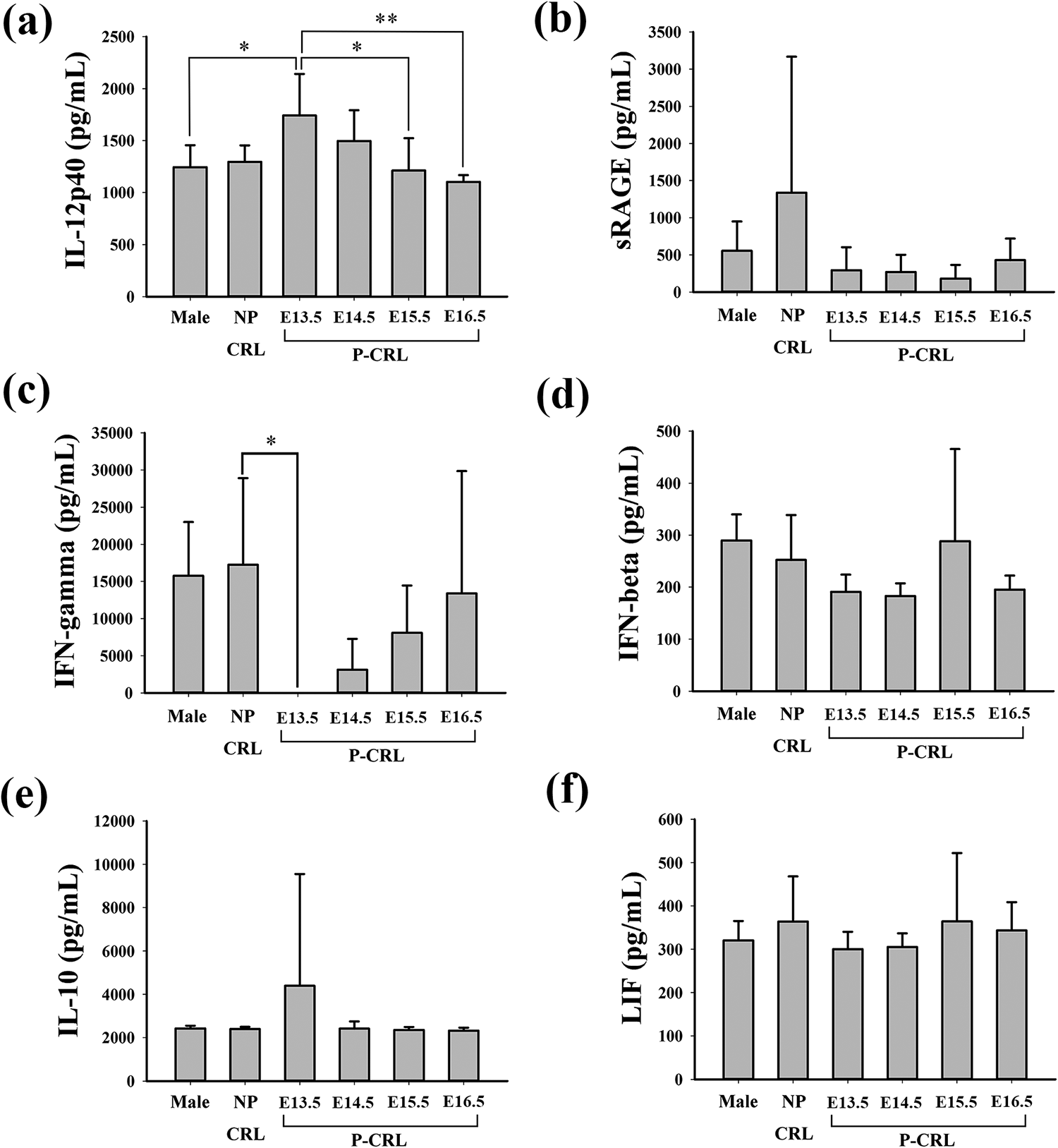
Relationship between gender, pregnancy, gestational age, and protein levels in maternal serum of untreated mice. The expression levels of (a) IL-12p40, (b) sRAGE, (c) IFN-γ, (d) IFN-β, (e) IL-10, and (f) LIF obtained from male, NP-CRL and P-CRL groups were detected by ELISA. Male: male mice (n = 6); NP-CRL: nonpregnant female control mice (n = 6); P-CRL: healthy pregnant controls (n = 6 mice per stage from E13.5 to E16.5). *p < 0.05, **p < 0.01. IL: interleukin; sRAGE: soluble advanced glycation end products; IFN: interferon; LIF: leukemia inhibitory factor; ELISA: enzyme-linked immunosorbent assay.
Maternal serum levels of proteins in CP groups induced by atRA or TCDD and in control groups
Next, the expression levels of the eight proteins in maternal serum were detected by ELISA from E13.5 to E16.5 in healthy pregnant control groups, atRA groups, and TCDD groups.
The expression levels of IL-12p40 in healthy control were gradually decreased from 1742.1 pg/ml to 1103.3 pg/ml during gestation period of E13.5–E16.5 (Figure 3(a)). Specifically, atRA and TCDD groups showed clearly decreased IL-12p40 levels at E13.5 and E14.5 compared with those in the control (E13.5-con vs. atRA: p < 0.001, con vs. TCDD: p < 0.001; E14.5-con vs. atRA: p = 0.005, con vs. TCDD: p = 0.005).

Maternal serum levels of proteins in cleft palate groups induced by atRA or TCDD and controls. The expression levels of (a) IL-12p40, (b) sRAGE, (c) IFN-γ, (d) IFN-β, (e) IL-10, and (f) LIF obtained from Con, atRA, and TCDD groups were detected by ELISA. Con: healthy pregnant controls (n = 6 mice per stage from E13.5 to E16.5); atRA: pregnant mice with cleft palate induced by all-trans retinoic acid (n = 6 mice per stage from E13.5 to E16.5); TCDD: pregnant mice with cleft palate induced by 2,3,7,8-tetrachlorodibenzo-p-dioxin (n = 5 mice per stage from E13.5 to E16.5). *p < 0.05, **p < 0.01, ***p < 0.001. IL: interleukin; sRAGE: soluble advanced glycation end products; IFN: interferon; LIF: leukemia inhibitory factor; ELISA: enzyme-linked immunosorbent assay.
The levels of sRAGE in the control group were relatively stable from E13.5 to E16.5, which were approximately 291.9 pg/ml (Figure 3(b)). Compared with the controls, atRA and TCDD groups had nearly threefold higher sRAGE levels at E13.5 (atRA vs. con: p = 0.046, TCDD vs. con: p = 0.044).
The average serum concentrations of IFN-γ, IFN-β, IL-10, and LIF in the control group from E13.5 to E16.5 were around 6143.7, 214.4, 2871.7, and 328.5 pg/ml, respectively. These relative proportions remained unaffected by CP induced by atRA and TCDD. No statistical difference was observed among the three groups (Figure 3(c) to (f)).
The expression levels of epiregulin and IL-12p70 in serum from the CP groups were below the detectable range as well (data not shown).
Immunostaining of IL-12p40 and RAGE in mouse placenta
The IHC analysis of the mouse placenta at stages E13.5–E16.5 showed that both IL-12p40 and RAGE were expressed in the cytoplasm and membranes of syncytiotrophoblastic and vascular endothelial cells, and macrophages (Figures 4 and 5).

Immunohistochemical localization of IL-12p40 in mouse placenta at E13.5–E16.5 in healthy control (Con), atRA group (atRA), and TCDD group (TCDD). IL-12p40 was expressed in mouse placenta. In healthy pregnant controls, at E13.5 (a) to E14.5 (b), positive staining for IL-12p40 was intensely present in the placental tissue. At E15.5 (c) to E16.5 (d), the staining became faint in placentas. IL-12p40 staining was generally low at E13.5–E14.5 in atRA (e, f) and TCDD (i, j) groups compared with the control. The scoring analysis indicated that both atRA and TCDD groups showed a reduced number of IL-12p40 positive cells in the placental tissue at E13.5–E14.5 (m) (n = 5 litters per stage and group). *p < 0.05, **p < 0.01. Scale bar: 50 μm. atRA: all-trans retinoic acid; TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin.
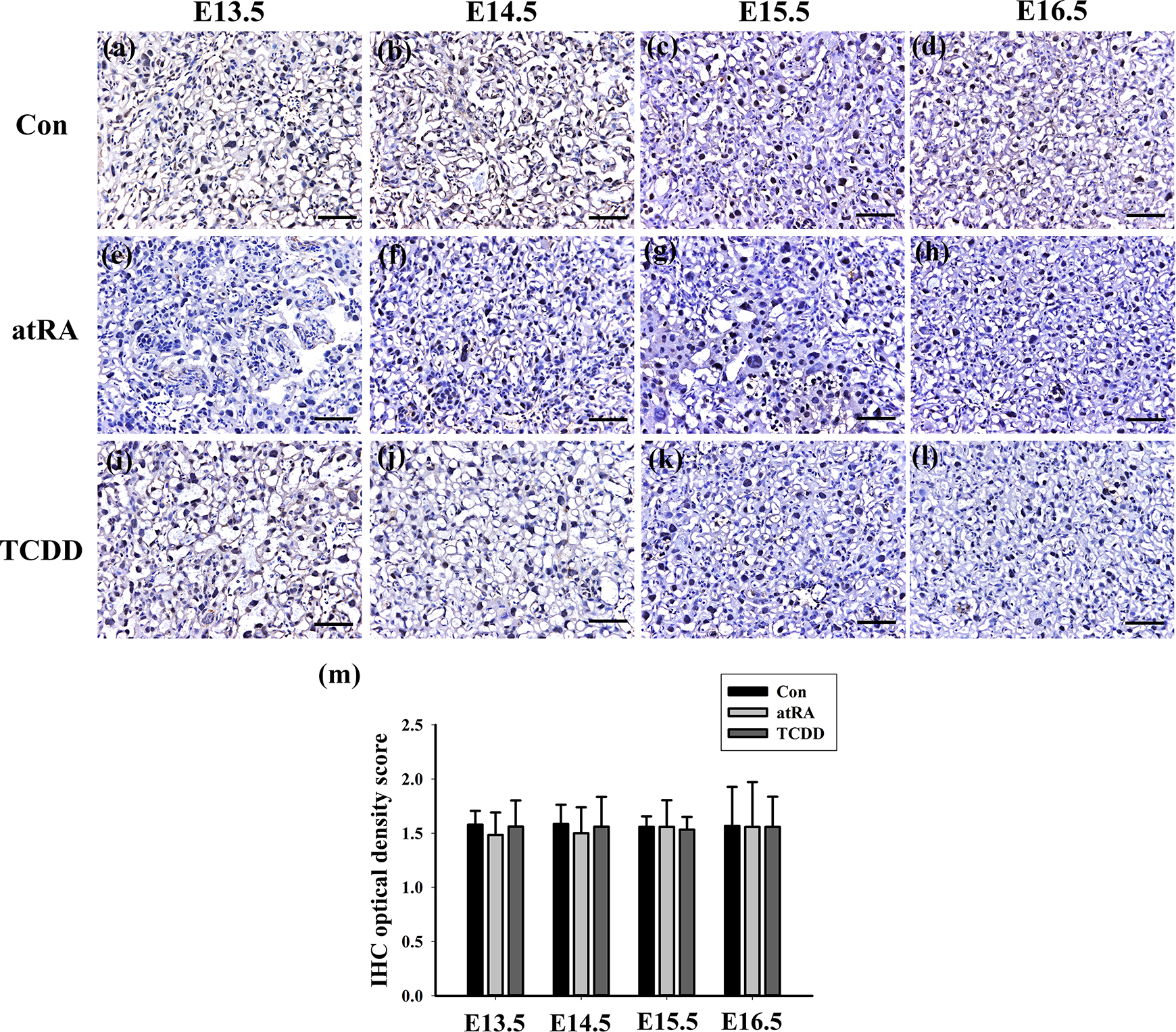
Immunohistochemical localization of RAGE in mouse placenta at E13.5–E16.5 in healthy control (Con), atRA group (atRA), and TCDD group (TCDD). (a–l) Positive staining of RAGE was present in the placentas, and there are no clear differences of RAGE among these three groups. (m) The scoring analysis indicated that there are nonstatistically significant differences among these three groups (n = 5 litters per stage and group). Scale bar: 50 μm. atRA: all-trans retinoic acid; TCDD: 2,3,7,8-tetrachlorodibenzo-p-dioxin.
In the control groups, IL-12p40 was strongly expressed in the placental tissues at E13.5–E14.5 and then the staining weakened toward E15.5–E16.5 (Figure 4(a) and (b)). The intensity of immunoreactivities to IL-12p40 was clearly lower in the placentas from the CP groups compared with those from the control (Figure 4(e), (f), (i), and (j)). The image analysis indicated that both atRA and TCDD groups displayed a reduced score of IL-12p40 positive cells at E13.5 and E14.5 (Figure 4(m); E13.5-con vs. atRA: p = 0.037, con vs. TCDD: p = 0.042; E14.5-con vs. atRA: p = 0.007, con vs. TCDD: p = 0.006).
Positive staining of RAGE was weakly present in the placentas in both CP groups and the controls (Figure 5(a) to (l)). Meanwhile, there are no statistically significant differences when comparing IHC optical density score of RAGE between these three groups (Figure 5(m)).
Discussion
Serum detection is a routine screening method in clinical practice, which has the advantages of convenient sampling, no complications, and low cost. A previous study reported on maternal serum screening of fetal chromosomal abnormalities by α-fetoprotein, unconjugated estriol, human chorionic gonadotrophin (hCG), and free-β hCG. 18 So far, there were no studies about specific protein related to CLP fetuses in maternal serum.
In this study, two proteins, IL-12p40 and sRAGE, were confirmed by ELISA to be differentially expressed in serum from mice carrying embryos with CP. At the same time, decreased immunostaining of IL-12p40 was detected in placental tissue sections obtained from E13.5 to E14.5 in the CP groups.
IL-12 is a cytokine that exerts immune regulatory effects on T and NK cells via inducing IFN-γ production and generating Th1 responses and cytotoxic T lymphocytes. 19,20 It is a 70-kDa heterodimeric protein composed of two disulfide-bonded chains, the p40 and p35 subunits. The IL-12p70 fraction equates with IL-12 biological activity, whereas IL-12p40 has been shown to act as an IL-12 antagonist in vitro. 21 Our study showed that P-CRL at E13.5 had higher serum IL-12p40 levels when compared with male. Moreover, the expression levels of IL-12p40 in P-CRL mice were gradually decreased from E13.5 to E16.5. The IL-12p40 levels in serum are likely to be influenced by gender and hormone in mice. Here, the concentrations of IL-12p70 were too low to be detectable, which is similar to that of Holtan et al. 22 The increased serum IL-12p40 at E13.5 may induce the accumulation of IFN-γ, leading to the increasing IFN-γ over E13.5–E16.5. There is a hypothesis that the time-dependent changes in levels of cytokines as pregnancy normally progresses represent a maternal shift away from immune response and toward an inflammatory response. 22 Mounting evidence suggests that the serum IL-12p40 levels are associated with severity of silicosis, the progression of colorectal cancer, the risk of osteosarcoma and esophageal cancer. 19,23 –25 In our study, IL-12p40 was significantly decreased in the serum from CP groups at E13.5 and E14.5. Considering a number of proteins in the peripheral blood originate from cells that comprise the placenta, we analyzed the IL-12p40 expression in the mouse placenta at stages E13.5–E16.5 by IHC, which showed a similar trend as in the serum. The placenta is thought to be a potential source of circulating inflammatory cytokines. 26 Piccinni demonstrated that the modulation of IL-12 may induce adverse effects on the conceptus either directly or via activating NK cells/macrophages that damage the trophoblast. 27 Although we detected the spatiotemporal expression of IL-12p40 in embryonic palate by IHC, the results showed that IL-12p40 was barely detectable (data not shown). The role of IL-12p40 levels in serum on reproductive outcome in vivo still remains to be clarified.
RAGE is a multiligand transmembrane receptor, a member of the immunoglobulin superfamily. sRAGE is a truncated form of RAGE—the extracellular domain. It plays a role in the pathogenesis of many diseases like diabetes mellitus, coronary artery disease, and inflammatory diseases. 28 –30 Germanova et al. reported that serum sRAGE levels are decreased in pregnancy compared with nonpregnant women. The study also described an increase of sRAGE toward mid-pregnancy and a decrease thereafter. 31 Our study here showed that the serum levels of sRAGE in P-CRL mice were relatively stable from E13.5 to E16.5, and we did not detect any differences between P-CRL mice and NP-CRL mice. This discrepancy maybe due to the sample size, different responding way between humans and mice during pregnancy, different time periods selected, and assay kits used. Meanwhile, sRAGE was significantly increased in the serum from CP groups at E13.5, which showed a different trend from that in AF in our previous study. 16 Chen et al. also reported the changes of sRAGE level in the maternal blood from women with severe preeclampsia compared with the normal pregnancy controls, which showed a reversed trend to that in the umbilical blood. 32 Increasing evidence suggests that RAGE was expressed in placenta. 32,33 Here, we also found that positive staining of RAGE was present in the placentas, and we did not detect any significant differences of RAGE in the placental tissues between the CP groups and the controls. It remains unclear whether the placenta contributed to the increased sRAGE levels in serum from the CP groups. There is a role for RAGE in the development of the palate, as CP groups displayed a clearly lower RAGE expression in embryonic palate in our previous study. 16 The mechanisms for the differentially expressed sRAGE levels in serum remain to be elucidated by further research.
Dysregulated expression of IL-12p40 and sRAGE in maternal serum in dams treated with two chemicals that induce CP, which was seen in our study, has not yet been previously described. It has to be considered whether IL-12p40 and sRAGE were altered in the maternal serum because of some other aspect of toxicity induced in the dams by atRA and TCDD, as atRA produces limb defects and TCDD causes hydronephrosis in addition to inducing CP. 34,35 Using knockout mice as the control might help resolve this issue, such as EGF (−/−) mice, which diminish sensitivity to CP induction by TCDD or atRA but enhance sensitivity to hydronephrosis, missing or short digits and oligodactyly, respectively. 34,35 On the other hand, some studies speculated that atRA and TCDD may have similar mechanisms in causing fetal CP, we cannot rule out the possibility of a similar role of atRA and TCDD on the circulating cytokines. 36,37 However, the morphological outcomes in terms of palatal development of fetuses treated with atRA were different from that with TCDD in our study. It is possible, and indeed likely, that other proteins in serum of pregnant females with CP embryos are differentially expressed, as we only detected eight proteins by ELISA.
Conclusion
In conclusion, reduced concentrations of IL-12p40 and increased concentrations of sRAGE were observed in serum from mice carrying embryos with chemically induced CP, paralleled by reduced IL-12p40 expression in placental tissue. IL-12p40 and sRAGE in maternal serum may be potential biomarkers for CP, but further investigation is needed to assess whether such changes may also apply to women carrying fetuses with CP.
Footnotes
Author contributions
X-HW contributed to the execution of experiments, data analysis, manuscript writing, YP conducted the experiments, L-YM was responsible for experimental design, the funding application, the supervision and management of the project and critically revising the manuscript. All the authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants of the National Natural Scientific Foundation of China [Grant Nos. 81571438 and 81870761] and the Fundamental Research Funds for the Central Universities.
