Abstract
Malathion is an organophosphate pesticide widely used for agricultural crops and for vector control of Aedes aegypti. Humans are exposed to this environmental contaminant by ingesting contaminated food. The juvenile and peripubertal periods are critical for the postnatal development of the epididymis and are when animals are most vulnerable to toxic agents. Since juveniles and adolescents are developing under exposure to the insecticide malathion, the aim of the present study was to evaluate the effects of exposure to low doses of malathion on postnatal epididymal development in rats. Male Wistar rats were exposed to malathion daily via gavage at doses of 10 mg kg−1 (M10 group) or 50 mg kg−1 (M50 group) for 40 days (postnatal days (PNDs) 25–65). The control group received the vehicle (0.9% saline) under the same conditions. On PND 40, the epididymides were removed, weighed and used for histological analysis and determination of the inflammatory profile and sperm count. Sperm from the vas deferens were subjected to sperm motility analysis. The M50 group showed tissue remodelling in the caput and cauda epididymides and increased neutrophil and macrophage migration in the caput epididymis. The M10 group showed decreased motile spermatozoa and IL-6 levels in the caput epididymis. Both doses decreased the IL-1β level and altered the morphology of the same region. These results show that malathion exposure may impair postnatal epididymal development. Furthermore, alterations of the immune system in the epididymal environment are presented as new findings regarding the action of malathion on the epididymis.
Introduction
External factors, especially environmental pollutants, may impair epididymal function. 1 –3 Environmental pollution from pesticides is often reported, and humans are exposed to pesticides by ingesting contaminated food, inhaling contaminated air or contacting the substance directly via the skin. 4 Malathion is an organophosphate pesticide widely used on agricultural food and feed crops, 5 is used to treat head lice in humans 6 and is used for vector control to prevent disease outbreaks, especially those caused by Aedes spp. 7
In 2003, the Agency for Toxic Substances and Disease Registry (ATSDR) suggested that malathion was not toxic to the male reproductive system. 6 However, Choudhary et al. 8 showed that malathion exposure (150 and 250 mg mL−1 body weight (b.w.)) for 60 days decreased the epididymis weight and sperm motility in adult rats. Another study with adult rats exposed to malathion (170 mg mL−1 b.w.; 13 days) also reported a reduction in epididymal weight and in sperm count in the same tissue. 9 Even then, it has been reported that the oral lethal dose 50 (LD50) of malathion for adult male rats is approximately 5400 mg kg−1, 10 and the no observed adverse effect level (NOAEL) for the reproductive system in rats in the developmental period is 130 mg kg−1. 11
An acute exposure model showed that malathion (500 mg mL−1 b.w.; 3 days) led to a decrease in epididymis weight and sperm motility in mice (postnatal day (PND) 45). 12 The same authors showed a reduction in sperm count in the epididymis and sperm motility after mice were exposed to malathion (200 mg mL−1 b.w.; PND 21) for 30 days. 13
During the juvenile and peripubertal period (PNDs 22–65 in rats), maturation of the hypothalamic–pituitary–testicular axis and the beginning of the reproductive cycle occur. 14 Furthermore, between PNDs 16 and 44, narrow and columnar epididymal cells differentiate, followed by an expansion period. 15 Between PNDs 49 and 63, the first increase in epididymal weight occurs. 16 Thus, this period is a window of major epididymal susceptibility to toxic agents.
It has been estimated that approximately 7% of cases of male infertility have inflammatory involvement, including epididymitis. 17,18 However, the relationship between the male reproductive system and immune system is poorly understood, 19 including after malathion exposure. At the systemic level, Lasram et al. 20 showed that malathion exposure (200 mg mL−1 b.w.; 28 days) in adult male mice increased the number of leucocytes, neutrophils and pro-inflammatory cytokine levels (IL-1β, IL-6 and INF-γ) in the liver.
Although there are some studies assessing malathion exposure in rats related to damage due to oxidative stress, no study has shown the association between the immune system and exposure to low doses of malathion during postnatal epididymal development. Furthermore, the present study used lower doses of malathion exposure than studies previously published in the scientific literature. Thus, the aim of the study was to evaluate whether exposure to low doses of malathion during the juvenile and peripubertal periods could impair epididymal development in male rats.
Materials and methods
Animals and experimental conditions
Forty-five juvenile male Wistar rats at PND 21 and a body weight of approximately 52 g were provided by the Central Animal House, Londrina State University (CCB – UEL), Paraná, Brazil. The animals were acclimated to their new environment (at the Animal House of Laboratory of Toxicology and Metabolic Dysfunction of the Reproduction) for 4 days before the experiments commenced. During the experimental period, rats were placed into polypropylene cages (43 × 30 × 15 cm3) with laboratory-grade pine shavings as bedding and maintained under controlled light (12-h light–dark cycle; lights off at 07:00) and temperature (±23°C) conditions. Rats had free access to standard commercial laboratory chow and drinking water. Animal care and handling procedures were in accordance with the EPA Guidelines for Reproductive Toxicity Risk, and the experiments were approved by the Ethics Committee on Animal Use of State University of Londrina (CEUA/UEL protocol numbers 12305.2016.65 and OF.CIRC. CEUA n 137/2016).
Experimental design
The animals were randomly assigned to three experimental groups (n = 15 animals per group): a control group (C), one group treated with malathion 10 mg kg−1 b.w. (M10) and another group treated with malathion 50 mg kg−1 b.w.(M50). The malathion doses were adapted from Geng et al. 21 and administered from PNDs 26 to 65. The M10 and M50 doses represent 0.5% and 2.5% of oral LD50 for adult male rats, respectively. 10 Therefore, the doses used in the present study are considered to be low doses for male rats. Furthermore, the doses were lower than the NOAEL dose (130 mg kg−1 b.w.) for reproductive toxicology in rats. 11 The peripubertal period was selected according to Ojeda et al. 22 Thus, the animals were treated orally (gavage) with either 10 or 50 mg kg−1 malathion (dietil-dimetoxitiofosforiltio; CAS no. 121-75-5; Cheminova, Lemvig, Denmark; diluted in 0.9% saline) or vehicle solution (control group). The groups were treated daily for 40 consecutive days.
Body weight and weight of some reproductive organs
At the end of treatment, rats were anaesthetized with a combination of ketamine 75 mg kg−1 (Sedomin® 10%, Avellaneda, Argentina) and xylazine 10 mg kg−1 (Anasedan®, Paulínia, Brazil), weighed and euthanized by cardiac puncture. The epididymis was removed, and the weights (absolute and relative to body weights) were determined (n = 10 rats per group). The right epididymis was used for determining sperm counts (n = 10 per group) and histopathological and stereological analysis (n = 5 per group). The left epididymis was used to determine the inflammatory cell profile (n = 6 per group). Spermatozoa from the left vas deferens were subjected to sperm motility analysis (n = 10 per group).
Sperm number and transit time in the epididymis
To evaluate the transit time in the epididymis, the right epididymis was decapsulated, weighed and homogenized in 5 mL of NaCl 0.9% containing Triton X-100 0.5% (Sigma-Aldrich Co.®, St. Louis, Missouri, USA) according to the method described by Robb et al., 23 with adaptations as described by Siervo et al. 24 After 10-fold dilution of the homogenate, a small sample volume was transferred to a Neubauer chamber, and late spermatozoa were counted (four fields of view per animal). To calculate sperm transit time through the epididymis, the number of sperm in each portion was divided by the daily sperm production.
Sperm motility
Sperm motility was evaluated according to methods described by Siervo et al. 24 Briefly, the left vas deferens was rinsed with 1.0 mL modified human tubal fluid medium with gentamicin (Irvine Scientific®, Santa Ana, California, USA) at 34–36°C to obtain spermatozoa. A warmed Makler counting chamber (Sefi-Medical, Haifa, Israel) was loaded with 10 µL of the aliquot of sperm solution. Sperm motility was assessed by visual estimation (100 spermatozoa per animal) under a light microscope (Motic®, Carlsbad, California, USA) at 100× magnification and was performed by the same person throughout the study. Spermatozoa were classified as motile or immotile.
Morphometric and histopathological analyses
The left epididymis (five per group) was removed and fixed in Metacarn solution (10% acetic acid, 60% methanol and 30% chloroform; Sigma-Aldrich Co.®, St. Louis, Missouri, USA) for 3 h at 30°C. The epididymides were embedded in Paraplast® (Sigma-Aldrich Co.®, St. Louis, Missouri, USA) and sectioned at 5 μm. The sections were stained with haematoxylin and eosin and examined by light microscopy for histopathological and stereological analysis as described by Favareto et al. 25
Histopathological inspection was conducted using cross-sections of the caput and cauda epididymides of each animal which were evaluated qualitatively using an Opton microscope (100× and 400× magnification). In the stereological analysis, 10 random cross-sections per animal of the caput (Region 2A) and cauda (Region 5A/B) epididymides 26 were captured using a photomicroscope (Opton, Cotia, Sao Paulo, Brail) and BELView version 6.2.3.0 (BEL Engineering, Monza, Monza and Brianza, Italy) for Windows at a magnification of 400× and analysed. This analysis was performed by means using Weibel’s multipurpose graticule with 169 points 27 to compare relative proportions among the epididymal components (epithelium, stroma and lumen) in the experimental groups (50 sections per group for each 4 epididymal region).
Inflammatory profile
Myeloperoxidase activity
Neutrophil recruitment to the epididymis was determined by the myeloperoxidase (MPO) kinetic-colorimetric assay. 28,29 Thus, epididymis was separated at the caput/corpus and cauda and the frozen samples were homogenized using a Tissue Tearor (BioSpec, Bartlesville, Oklahoma, USA) in ice-cold K2HPO4 buffer (400 µL, 50 mM, pH 6.0) containing HTAB (0.5% weight/volume). The homogenates were centrifuged (16,100 × g, 2 min, 4°C) and the supernatants (30 µL) were mixed with K2PO4 buffer (200 µL, 50 mM, pH 6.0) and hydrogen peroxide (0.05%, v/v) and placed in a 96-well plate. The absorbance was determined after 5 min at 450 nm (Multiskan GO Microplate Spectrophotometer, Thermo Scientific, Vantaa, Finland). MPO activity is expressed as the number of neutrophils per mg tissue, read off a standard curve constructed over the range 196–400,000 neutrophils.
N-acetyl-β-d -glucosaminidase activity
The N-acetyl-β-
Cytokine measurement
The frozen samples were homogenized in saline and centrifuged (3600 r min−1, 4°C, 15 min) according to Staurengo-Ferrari et al. 31 The supernatants were used to determine the levels of IL-6, IL-1β, TNF-α and IL-10. All measurements were performed using enzyme-linked immunosorbent assay kits according to the manufacturer’s instructions (R&D Systems, Minneapolis, Minnesota, USA). The results were expressed as pg per 100 mg of tissue.
Statistical analysis
The parameters were submitted to the Shapiro–Wilk test for normality and classified into parametric and non-parametric data. For parametric results, the results from malathion-treated groups were compared to the control group by one-way analysis of variance (ANOVA) with post hoc Dunnett’s test. Non-parametric results were statistically analysed by Kruskal–Wallis test followed by Dunn’s post hoc method. The variance among the experimental groups was compared by Bartlett’s test. Differences were considered significant when p < 0.05. The statistical analyses and graph design for the results were performed by GraphPad Prism for Windows (version 7.01 – GraphPad Software, La Jolla, California, USA). Data are presented in the text as the mean ± SEM.
Results
Epididymis weight, sperm motility, sperm number and sperm transit time in the epididymis
Table 1 shows the epididymal weight, sperm motility, sperm concentration and sperm transit of the caput/corpus and cauda epididymal portions. Compared to the control group, the group exposed to malathion did not have a significantly altered epididymal weight. The treatments did not show any alteration in the sperm count or transit time in the caput and corpus or cauda epididymis. On the other hand, although there were no alterations in sperm motility after 50 mg kg−1 malathion exposure, exposure to the minor dose (10 mg kg−1) increased the percentage of immobile spermatozoa.
Epididymal weight, sperm motility, sperm count and transit time in the epididymis.a

ANOVA: analysis of variance; M10: rats treated with 10 mg kg−1 malathion; M50: rats treated with 50 mg kg−1 malathion.
a Values are expressed as the mean ± SEM. One-way ANOVA test with a posteriori Dunnett’s test.
b p < 0.05, groups that differ statistically from the control group.
Histopathological and stereological analyses of the epididymis
Histopathological analysis (Figure 1) showed the presence of multinucleated cells (Figure 1(b) and (d)) in the lumen of the caput epididymis and vacuoles (Figure 1(b) and (c)) on the epithelium in the same epididymis region after both malathion treatments. On the other hand, the cauda epididymis was not susceptible to malathion (Figure 1(e) to (g)).

Histopathological analysis of caput and cauda epididymis. Photomicrograph of (a to d) caput and (e to g) cauda epididymis sections from (a and e) the control, (b and f) M10 and (c, d and f) M50 groups. (a, e, f and g) Epithelium, lumen and stroma with normal aspects. (b to d) The presence of vacuoles in the epithelium (asterisk), multinucleated (arrow) and round cells (arrowhead) in the lumen. Haematoxylin and eosin staining. Magnification (a and e) ×100 and (b, c, d, f and g) ×400. L: lumen; Ep: epithelium; S: stroma.
Stereological analysis (Table 2) showed that compared to control group animals, animals exposed to 50 mg kg−1 malathion had an increase in the percentage of the caput stromal compartment and in the caudal lumen compartment. The toxicant at the lower dose did not alter the compartments of the caput or cauda epididymis.
Epididymal stereological analysis.a

ANOVA: analysis of variance; M10: rats treated with 10 mg kg−1 malathion; M50: rats treated with 50 mg kg−1 malathion.
a Values are expressed as the median (Q1–Q3). One-way ANOVA test with a posteriori Dunnett’s test.
b p < 0.05, groups that differ statistically from the control group.
Inflammatory profile: MPO and NAG activity and cytokine levels
The evaluation of the MPO and NAG activities (Figure 2) showed that malathion at a dose of 50 mg kg−1 increased the number of neutrophils (Figure 2(a)) and macrophages (Figure 2(b)) in the caput epididymis, whereas the minor dose did not have the same effects. However, compared to the control group, the malathion-exposed groups did not have altered MPO (Figure 2(c)) and NAG (Figure 2(d)) recruitment in the cauda epididymis at either dose.

Inflammatory profile of the epididymis. Number of (a and b) neutrophils and (b and d) macrophages in the (a and b) caput or (c and d) cauda epididymides. Data are the mean ± SEM. *p < 0.05 and **p < 0.01 in relation to the control group. One-way ANOVA was performed with Dunnett’s post hoc test. M10: rats treated with 10 mg kg−1 malathion; M50: rats treated with 50 mg kg−1 malathion; MPO: myeloperoxidase; NAG: N-acetyl-β-
Figure 3 shows the levels of IL-1, IL-6, IL-10 and TNF-α. The levels of IL-1 (Figure 3(a)) in the caput epididymis were reduced in animals exposed to both malathion doses, while the IL-6 levels (Figure 3(e)) decreased after exposure to only 10 mg kg−1 malathion. These alterations were not observed in the cauda epididymis. The IL-10 and TNF-α levels (Figure 3(c), (d), (g) and (h)) were not altered by either of the doses in any epididymal region.
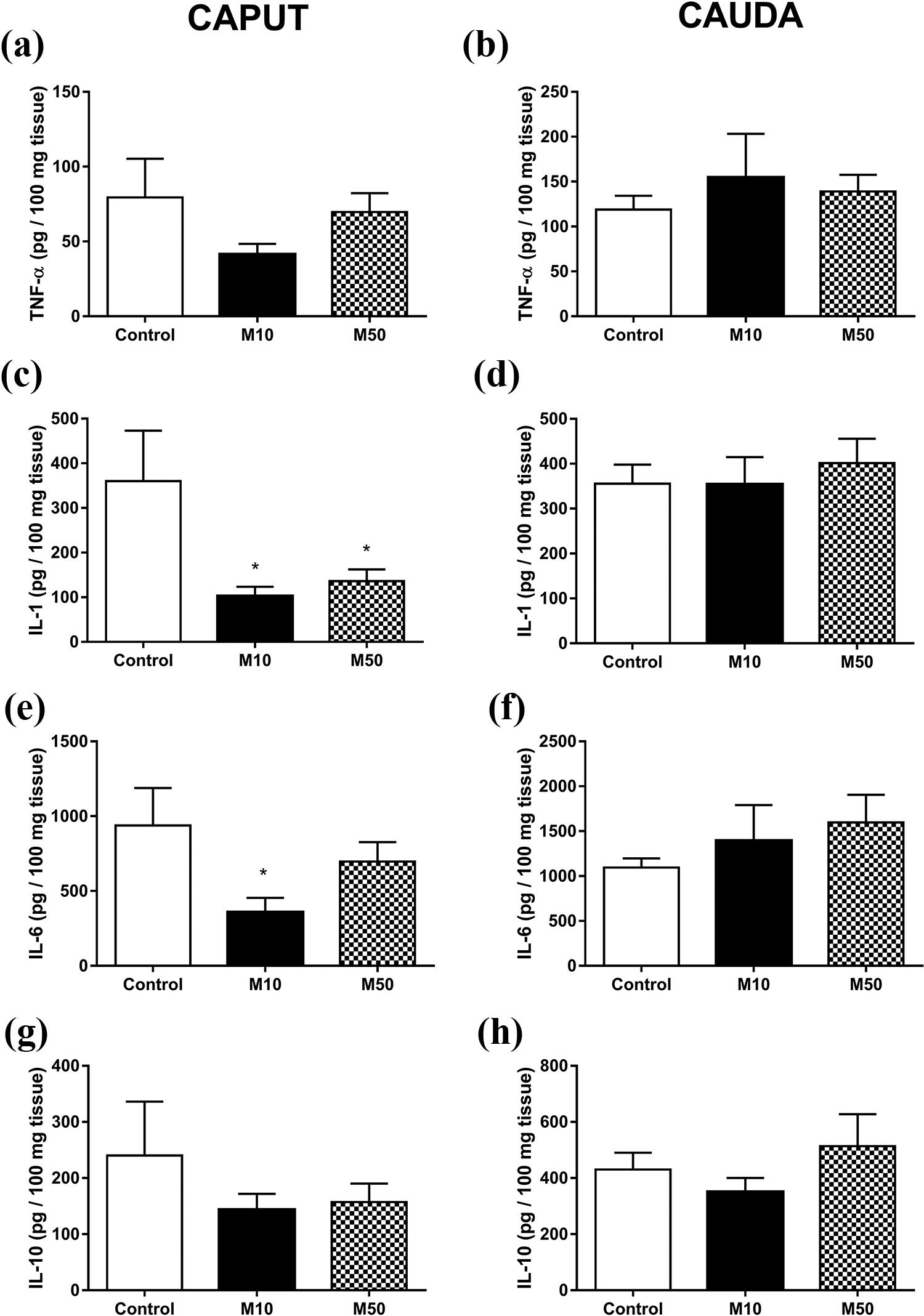
Cytokine levels in the epididymis. (a, c, e and g) Caput and (b, d, f and h) cauda epididymis from control and malathion-treated rats. (a and b) IL-1, (c and d) IL-10, (e and f) IL-6 and (g and h) TNF-α. Data are the mean ± SEM. *p < 0.05 in relation to the control group. One-way ANOVA was performed with Dunnett’s post hoc test. M10: rats treated with 10 mg kg−1 malathion; M50: rats treated with 50 mg kg−1 malathion; ANOVA: analysis of variance.
Discussion
The present study shows that malathion exposure during the juvenile and peripubertal periods impairs epididymal development through an increased inflammatory response.
In general, the epididymis shows an increased susceptibility to inflammatory and autoimmune responses in relation to the testis. 19 Studies have observed a greater presence of macrophages and T lymphocytes in proximal epididymal regions than in distal regions of the tissue. 32,33 In our study, the increase in neutrophil and macrophage migration to the caput epididymis after 50 mg kg−1 malathion exposure demonstrated that there was some tissue injury, and these cells were involved in an inflammatory process. Macrophages in the epididymal lumen have been shown to phagocytose abnormal sperm. 19,34 Furthermore, Serre and Robaire 35 reported that the increase in lymphocyte number, especially in the proximal epididymis, was related to disturbance of spermatocytes in Brown Norway rats. The literature is clear in showing that malathion at higher doses than those used in the present study may lead to alterations in normal sperm morphology. 9,12,13 From this, the increase in immune cells in the caput epididymis is probably a mechanism to phagocytose abnormal sperm resulting from altered spermatogenesis and tissue injury.
A previous study by our group showed that the increase in neutrophils and macrophages after bisphenol A (BPA) exposure, a toxicant and environmental contaminant, was associated with tissue changes during epididymal development in peripubertal rats. 1 Similarly, our study showed alterations in the epididymal epithelium after both malathion treatments, and the abnormalities found may be related to the inflammatory response, which leads to tissue damage. Although the lower dose of malathion did not show an increase in inflammatory cells, we observed abnormalities in the tissue of 10 mg kg−1 malathion-treated animals. This implies that this lower dose was less antigenic, but that there were some possible inflammatory responses before this time point of analysis leading to tissue damage.
Corroborating Ogo et al., 1 who concentrated on an experimental model of exposure to BPA in rats, we also correlated inflammatory cell migration with tissue remodelling in both epididymal regions after malathion treatment (50 mg kg−1 b.w.). Thus, malathion can lead to epididymitis, altering the tissue morphology and compromising the sperm maturation process.
Although IL-1 and IL-6 are considered pro-inflammatory cytokines at a systemic level, the present study showed a decrease in IL-1 levels in caput epididymis after both malathion treatments and in IL-6 levels after M10 treatment. In this context, the indoleamine 2,3-dioxygenase (IDO) enzyme is highly expressed in the caput epididymis by principal and apical cells, 36 and the absence of IDO leads to higher expression of pro-inflammatory cytokines, while Ido1−/−-deficient mice have decreased white blood cells in their epididymal fluid, impairing sperm quality. 37 From this, IDO expression was probably altered in the present study, as noted by the lower expression of IL-1 and IL-6 at the lower dose and the increase in white blood cells in the caput epididymis.
The decrease in IL-6 levels in the caput epididymis after M10 exposure may be associated with the increase in immotile sperm after the same treatment. Lampiao and du Plessis 38 showed that IL-6 or TNF-α did not reduce sperm total motility but reduced progressive motility. They suggested that this sperm parameter reacts in a dose- and time-dependent manner to cytokines. In contrast, another study determined the IL-6 levels, which enhanced the fertilizing capacity of human sperm by acting on sperm motility parameters, showing its role in sperm capacitation in the epididymis. 39 The differences in exposure time and action pathway of malathion may characterize a different immune response in the epididymis, which may explain the different correlation between the IL-6 levels and sperm motility from other studies.
Furthermore, it was reported that IL-6 and IL-1 exert fundamental roles in spermatogenesis, but the role of these cytokines in the epididymis remains unclear. 19 In this sense, there are some epididymal proteins belonging to the defensin-like family, as bin1b has antimicrobial properties. 40 Fei et al. 41 suggested that mBin1b has a role in the regulation of the inflammatory response in the epididymis, showing that the expression of IL-1β was decreased in animals overexpressing mBin1b. Thus, a possible mechanism to decrease IL-1β may be the overexpression of Bin1b in defence against malathion damage.
In the present study, unchanged epididymal weight, sperm count and sperm transit in the epididymis were associated. However, studies exposing animals to higher doses of malathion than those used in the present study showed a reduction in sperm count in the epididymis. Selmi et al. 13 showed a reduction in sperm count per gram of epididymis after malathion exposure (200 mg kg−1 b.w., PND 21) for 30 days in rats. Espinoza-Navarro and Bustos-Obregón 9 also showed a decrease in sperm count in the epididymis in adult rats treated with 170 mg kg−1 b.w. for 13 days. These different results between our data and those of previous studies are because of the lower doses used in the current study, which best reflect the reality of malathion exposure.
Conclusion
The results of the present study show that malathion exposure during the postnatal period is critical to epididymal development. At higher doses, these alterations occur through increased neutrophil and macrophage migration and remodelling of epididymal tissue. Furthermore, alterations of the immune system in the epididymal environment are presented as new findings regarding the action of malathion on the epididymis.
Footnotes
Acknowledgement
The authors are grateful to State to the Coordinating Body for the Improvement of Postgraduate Studies in Higher Educations (CAPES) for providing a Master’s scholarship to RPE.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
