Abstract
Introduction:
The aim of this study was to assess the relationship between polymorphisms of genes encoding enzymes involved in arsenic metabolism and urinary arsenic concentration in people occupationally exposed to arsenic.
Materials and Methods:
The data from 113 employers directly exposed to lead, cadmium, and arsenic in copper smelter in Legnica and Glogow were collected. Urinary arsenic concentration was measured. In addition, blood level of cadmium, lead, and zinc protoporphyrins was assayed. Genetic analyses included polymorphism of PNP (rs 1130650), GSTO-1 (rs 4925), AS3MT (rs 11191439), and ADRB3 (rs4994) genes.
Results:
Individuals occupationally exposed to arsenic compounds, who have allele T in homozygous constellation in locus rs 1130650 of PNP gene, are predisposed to lower urinary arsenic concentration, while AA homozygosity in locus rs 4925 of GSTO-1 gene may result in statistically significant higher urinary arsenic concentration. Polymorphisms of AS3MT and ADRB3 genes showed no statistically significant correlation with urinary arsenic, however, there was a tendency to higher arsenic concentration in allele A carriers in locus rs4994 of ADRB3 gene and in allele T carriers in rs 11191439 of AS3MT gene.
Conclusion:
This study indicates that arsenic absorption and metabolism depend on polymorphisms of genes encoding PNP and GSTO-1. Individuals with disadvantageous constellation of polymorphisms are more susceptible to harmful effects of arsenic exposure.
Keywords
Introduction
Arsenic is one of the most common elements incorporated in the Earth’s crust. It is emitted to the environment by volcanic and industrial activity. Inorganic arsenic is classified as a group I human carcinogen. 1 The primary route of arsenic exposure is via the ingestion of contaminated food or water. In general, the daily intake of arsenic from food and beverages ranges between 20 μg/day and 300 μg/day. 1 –3 The amount of arsenic inhaled from ambient air constitutes a minor exposure for the general population, however, it is the main route of occupational exposure. It is worth noting that the amounts of arsenic consumed per day vary, for instance, the daily arsenic intake in rural areas ranges between 20 ng and 200 ng, it increases to 400–600 ng in cities without substantial industrial emission of arsenic and is even higher in the vicinity of industrial sources. Moreover, it is influenced by tobacco consumption and can vary from 1 μg/day in a nonsmoker to approximately 10 μg/day in a smoker. 1,3,4 . Epidemiological studies indicate that long-lasting exposure to arsenic is associated with skin, lung, liver, kidney, prostate, and bladder cancer as well as vascular diseases and diabetes. Arsenic is also hepato- and neurotoxic and is responsible for impaired fetal and child development. 5
The absorbed dose of arsenic can be measured in hair, nail, blood, or urine samples. Due to the fact that arsenic accumulates in keratin-rich tissue, total arsenic level in hair, fingernails, or toenails is used as the indicator of long-lasting exposure. In contrast, levels of arsenic in blood and urine and urinary arsenic metabolites (monomethylarsonic acid (MMA) and dimethylarsinic acid (DMA)) are typically used as indicators of recent exposure. In addition, it has been reported that average occupational exposure to airborne arsenic trioxide is significantly correlated with the inorganic arsenic metabolites in urine samples collected immediately after shift. 6
After absorption, arsenic is metabolized by stepwise methylation, mainly in the liver. In general, two pathways of arsenic methylation have been suggested—reductive proposed by Hayakawa et al. 7 and oxidative proposed by Challenger et al. 8 Both models result in DMA or MMA induction. 9 To date, arsenic (+3 oxidation state)-methyltransferase (AS3MT), purine nucleoside phosphorylase (PNP), glutathione S-transferase omega 1 (GSTO1), and glutathione S-transferase omega 2 (GSTO2) have been proposed to be involved in arsenic metabolism.
AS3MT is thought to catalyze the conversion of arsenite (iAs3+) methylate to MMA5+ and trivalent monomehtylarsonic acid (MMA3+) methylate to DMA5+ using S-adenosyl-methionine (SAM) as the methyl donor. However, PNP, GSTO1, and GSTO2 are suggested to be involved in reducing the pentavalent arsenic species, including arsenate (iAs5+), MMA5+, and DMA5+ to trivalent arsenicals. 10 –13
Arsenic methylation produces both highly reactive metabolites containing trivalent arsenic (As3+) and less reactive metabolites containing pentavalent arsenic. 14 Studies on arsenic toxicity yield inconsistent results. Numerous studies indicate that inorganic arsenic, such as iAs5+ and iAs3+, is more toxic than organic arsenic, such as MMA, DMA, arsenobetaine, arsenocholine, and arsenosugars. 15 However, other studies indicate that certain types of organic arsenic, such as the MMA3+, are more toxic than As3+. 13,16
Arsenic-induced health effects differ greatly between individuals, partly due to the individual ability to metabolize arsenic which affects retention and distribution of toxic metabolites. 17 Arsenic absorption and toxicity are thought to depend on specific configurations of PNP, glutathione-S-transferase omega 1, and AS3MT genotypes. 1,4,6,17 –22
According to the literature, there are six polymorphisms in the exonic region of PNP gene which in general contains six exons and is located on chromosome 14q11. Among these, one is located in the 5’UTR (rs17881206), three (His20His, Gly51Ser, Pro57Pro) in exon 2, one in exon 5 (Ala174Ala), and one in the 3’UTR (rs7785). 23 Hsieh et al. found a significantly higher risk of carotid atherosclerosis in people who carried the PNP A–T haplotype. 24
For AS3MT, several exonic single nucleotide polymorphisms were found on chromosome 10q24 (Arg173Trp, Met287Thr, Thr306Ile, Ile132Phe, Tyr135Asn, Gly140Ala). 9,25 Meza et al. pointed out that in Mexican subjects, three AS3MT gene polymorphisms—in locus rs12767543, rs3740393, and rs11191453 of chromosome 10—were associated with high uDMA/uMMA ratio. 9,26 In studies carried out by Gongand O’Bryant, GG polymorphism in locus rs10748835 was associated with higher incidence of cardiovascular diseases in people exposed to low doses of arsenic in drinking water. 22
Free oxygen radicals produced in arsenic biotransformation contribute to arsenic toxicity and carcinogenicity. Glutathione S-transferase (GST) appears to have a key function in the defense system against oxidative damage and, in consequence, in maintenance of cell homeostasis. GST is a phase II enzyme that can detoxify xenobiotics by catalyzing their conjugation with reduced glutathione. GST is involved in the reduction of As(5+), MMA(5+), and DMA(5+). 14,15
Two genes from the GST family, GSTO1 and GSTO2, are located on chromosome 10q and contain six exons each. In GSTO1, six exonic polymorphisms have been identified in various world populations (Cys32Tyr, Ala140Asp, –/AGG, Glu208Lys, Thr217Asn, Ala236Val) 27 and there has been a single case of a Thr217Asn variant in the dbSNP, but not yet reported in any population. 12 Four nonsynonymous exonic SNPs (Val114Ile, Cys130Tyr, Asn142Asp, Leu158Ile) were also reported in GSTO2. In a study by Paiva et al., Asn142As polymorphism appears to be correlated with an increase in DMA excretion and seems to modulate arsenic biotransformation and thereby arsenic toxicity. 28
Objectives
The aim of this study was to analyze the link between single nucleotide polymorphisms in locus rs 1130650 in PNP gene, locus rs 4925 in GSTO-1 gene (Ala140Asp), and locus rs 11191439 in AS3MT gene (Met287Thr) and the total urinary arsenic concentration in people occupationally exposed to arsenic and other heavy metals, particularly lead and cadmium. Moreover, we attempted to find the possible connection between occupational arsenic exposure and polymorphism of Trp64Arg in ADRB3 gene, which is associated with atherogenic risk factors, namely weight gain, insulin resistance, and diabetes. 29 –31
Materials and methods
Study population
The study was carried out in copper smelter and refinery in Legnica and Glogow. Ethical approval from the Bioethics Committee of Wroclaw Medical University (No. KB-156/2010) was obtained. All the study participants provided written informed consent.
The data of 113 workers directly exposed to lead, cadmium, and arsenic were collected. Precisely, the group consisted of refinery workers, smelters, crane operators, converter plant operators, dust removal device operators, electrode molders, electrolyzers, outfall operators, feedstock operator, and electrolyte cleaner operator. A total of 109 males and 4 females were enrolled in the study with the age range between 26 years and 63 years. The mean occupational exposure was 21 years. Fifty-five employees smoked cigarettes. All the subjects participating in the study were asked to complete the special questionnaire to obtain the relevant information on occupational history (work department, type of work, and exposure duration), modifying lifestyle factors (type of diet, physical activity, smoking, and alcohol consumption habits), health conditions, and medical history. Physical examination included general medical checks, blood pressure measurement, and body mass index evaluation. The characteristics of the study population are given in Table 1.
Clinical characteristics of the study population.
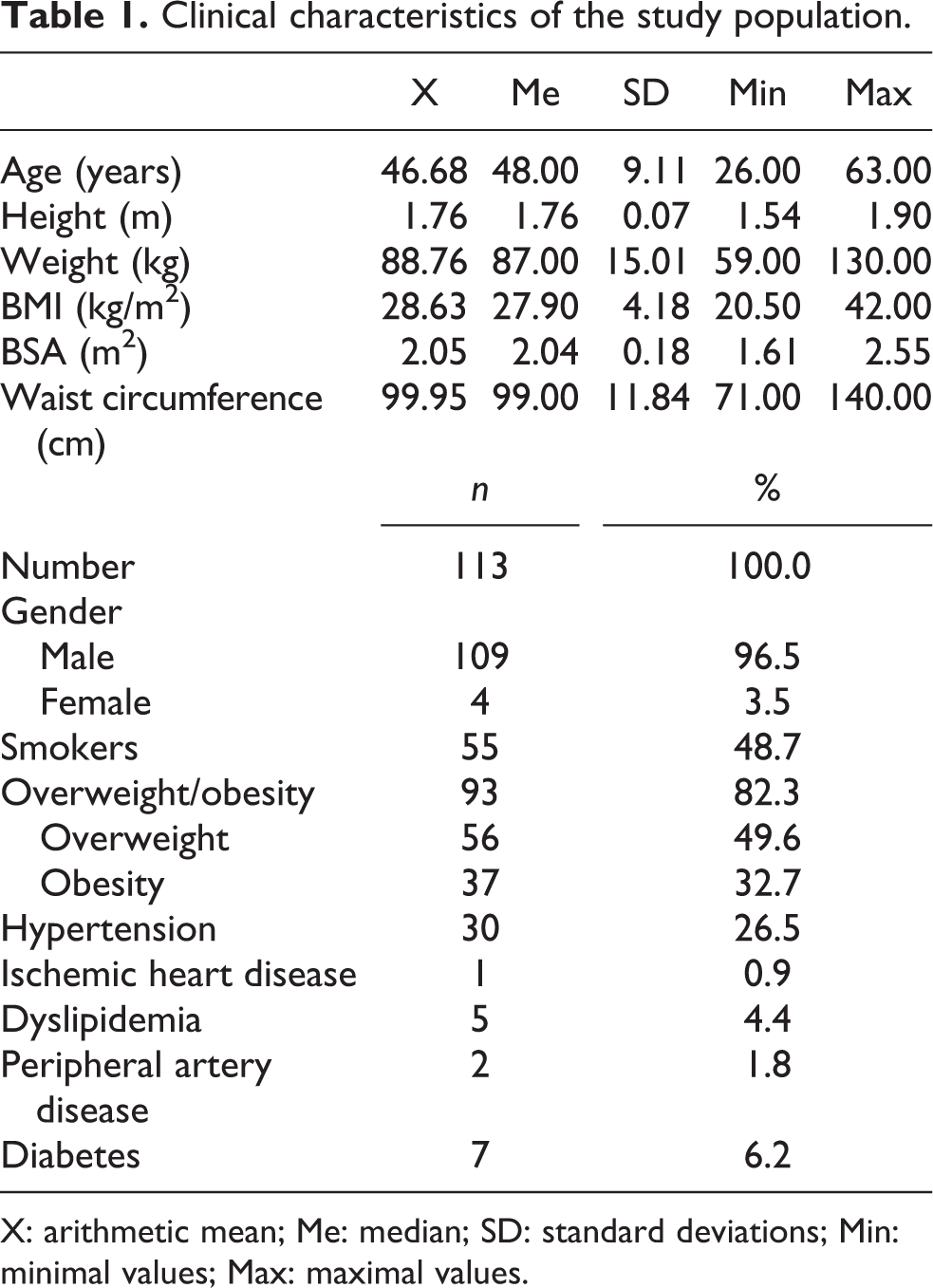
X: arithmetic mean; Me: median; SD: standard deviations; Min: minimal values; Max: maximal values.
We collected 25 ml of venous blood and standard urine sample from each worker right after the end of a shift. The lab tests included:
– blood lead (Pb-B) and cadmium (Cd-B) concentration,
– urinary total arsenic (As-U) concentration,
– blood zinc protoporphyrins (ZnPP) concentration,
– serum zinc (Zn-S) and copper (Cu-S) concentration,
– conventional biochemical tests detecting serum creatinine, urea, aminotransferases, bilirubin, glucose, total cholesterol with low-density lipoprotein (LDL) / high-density lipoprotein (HDL) fractions and triglycerides, and
– complete blood count.
Laboratory and toxicological characteristics of the study population are presented in Tables 2 and 3.
Conventional lab tests in the study population.

X: arithmetic mean; Me: median; SD: standard deviations; Min: minimal values; Max: maximal values; EGFR: estimated glomerular filtration rate.
Basic toxicological parameters in the study population.

X: arithmetic mean; Me: median; SD: standard deviations; Min: minimal values; Max: maximal values; Pb-B: blood lead concentration; Cd-B: blood cadmium concentration; ZnPP: blood zinc protoporphyrins concentration; Zn-S: serum zinc concentration; Cu-S: serum copper concentration; As-U: urinary total arsenic concentration.
Determination of Pb-B and Cd-B concentrations
Pb-B and Cd-B concentrations were measured by graphite furnace atomic absorption spectrometry (Solaar M6 of Thermo Elemental, UK). Analytical procedures were based on the methods described in the work by Trzcinka-Ochocka et al. 32 Calibration curves of lead and cadmium were prepared with blood standards: certified reference material (BCR® IRMM). Both methods were routinely monitored by determination of reference material (Recipe) and participation in intercomparison programme for toxicological analyses in biological materials G-EQUAS.
All the measurements were presented as micrograms per liter (μg/L) and in accordance to the recommendations of Nofer Institute of Occupational Medicine in Łódź, the biological exposure limit to lead is 500 μg/L and 5 μg/L to cadmium.
Determination of urinary arsenic concentration
Total urinary As concentration was measured by hydride generation atomic absorption spectrometry (HGAAS) using the VP100 Continuous Flow Vapour System (Thermo Fisher Scientific Inc., Waltham, Massachusetts, USA). The method used was described in the work by Jakubowski et al. 32 To determine the calibration curve, the reference material—ClinCal® Urine Calibrator; Recipe—was used. We monitored the accuracy of the method by analyzing samples of a reference material: Seronorm Trace Elements Urine (SERO AS, Oslo, Norway) and by participating in intercomparison programme for toxicological analyses in biological materials G-EQUAS.
Biological exposure limit to arsenic, measured in urine, proposed by Nofer Institute of Occupational Medicine in Łódź, is 35 μg/L (35 μg/g creatinine).
Determination of zinc and copper serum concentration
Cu and Zn serum concentrations were assessed by flame atomic absorption spectrometry (ThermoElemental, Solaar M6, UK) by measuring the absorbance at wavelength of 324.8 (213.9) nm and using air-acetylene flame with the deuterium background correction. Single-element copper (Zinc) standard 1000 μg/mL certified by the CPI International; Peak Performance USA was used as a reference material for calibration.
These methods were routinely monitored by analyzing the samples of reference material: Seronorm™ Trace Elements Serum and by participating in intercomparison program for toxicological analyses in biological materials G-EQUAS.
Determination of blood zinc protoporphyrins concentration
ZnPP erythrocyte concentration was measured by means of ProtoFluor Z hematofluorometer (Helena Laboratories Corporation, Beaumont, Texas, USA) in accordance with the manufacturer’s instructions. The measurement was performed in one drop of patient’s whole blood previously collected into test tubes with anticoagulant (ethylenediaminetetraacetic acid (EDTA)). To obtain total oxygen saturation of hemoglobin, ProtoFluor reagent with cyanide salt and stabilizers in aqueous solution were added. Hemoglobin converts into oxyhemoglobin. The wavelengths for fluorescence were as follows: excitation 415 nm, emission 595 nm, range 0–270 µg/dL. Equipment calibration was performed with two ProtoFluor calibrators: ProtoFluor Low and ProtoFluor High. Both solutions contain ZnPP, pyridine, stabilizers, and preservatives. The results were adjusted for patient’s hematocrit.
DNA isolation
Blood samples were collected by venipuncture into standard 2.7 mL tubes containing EDTA and then frozen at −80°C.
Genomic DNA was manually extracted from leukocytes by a magnetic-bead technology incorporated in a NucleoMag® Blood of MACHEREY-NAGEL workstation.
Determination of the PNP (rs 1130650), GSTO-1 (rs 4925), and AS3MT (rs 11191439) gene SNP
A multiplex assay based on the polymerase chain reaction (PCR) was used for amplification of PNP, GSTO-1, and AS3MT gene fragments with the following primers:
PNP gene: F/forward/5′-CTC AGT ATA CCT GCC AGC CTT T-3′
R/reverse/5′-CTC TGA CTC ATG GTG GGC TT-3′
GSTO-1 gene: F/forward/5′-GAC CAA GCC AGC ATT TTA GG-3′
R/reverse/5′-AGC AAG CCC ATG ACA AAG TC-3′
AS3MT gene: F/forward/5′-TCG TTT TGT TTC TGC AAC AT-3′
R/reverse/5′-AGG CAA TGC AAA GTC AAG AA-3′.
All the amplified products were digested in the multistage process by the PCR clean-up restriction enzyme, labeled with probes, and finally digested with the FastAP enzyme. PCR products were separated by the ABI 3100 analyzer (Applied Biosystems, Foster City, California, USA). Collected data concerning PCR products were analyzed with Gene Mapper ver. 3.5 (Applied Biosystems).
Determination of the ADRB3 (rs4994) gene SNP
Genotyping was performed by means of Fluidigm 192.24 Dynamic Array with BioMark HD Systems and EP1 (Fluidigm Corp., San Francisco, California, USA). We applied SNP-type assay (Fluidigm Corp.) which includes allele-specifically designed fluorescence primers (fluorescein amidites with emission of wavelength 518 λ max/nm) and a common reverse primer. The data were analyzed by the BioMark SNP Genotyping Analysis software to obtain genotype calls.
Statistical analysis
Statistical analysis was performed using the Statistica 12.0 software. Quantitative variables established for the study group included arithmetic means (X), medians (Me), standard deviations (SD), minimal values (Min), and maximal values (Max). Distribution of variables was tested with the W-Shapiro–Wilk test. For independent, quantitative variables manifesting normal distribution, analysis of variance (ANOVA, unifactorial parametric) was performed. In case of variables manifesting distribution distinct from normal, for independent qualitative variables, a non-parametric equivalent of Kruskal–Wallis analysis of variance was used. Significant differences between the arithmetic means were estimated using post hoc tests. Results for the qualitative variables were expressed in percentages. For the independent qualitative variables, in further statistical analysis, the highest credibility χ 2 test was used. The values of p < 0.05 were regarded as statistically significant.
Results
Mean urinary arsenic concentration in the occupationally exposed group was within the range of maximum allowable concentration and amounted to 31.34 ± 27.52 μg/g creatinine (CREA). The job seniority of people exposed to arsenic was 21.01 ± 11.73 years. Concentrations of other toxicological parameters were also within the range of maximum allowable concentration and were as follows: mean Pb-B concentration 292.11μg/L, mean ZNPP concentration in erythrocytes 44.32ug/dL, mean Cd-B concentration 1.78μg/L. Basic toxicology parameters of the exposed group are presented in Table 3.
The results of genotypes distribution for PNP, GSTO-1, AS3MT, and ADRB3 gene polymorphisms in exposed individuals are presented in Table 4.
Selected polymorphisms of PNP, GSTO-1, AS3MT, and ADRB3 gene in the study population.
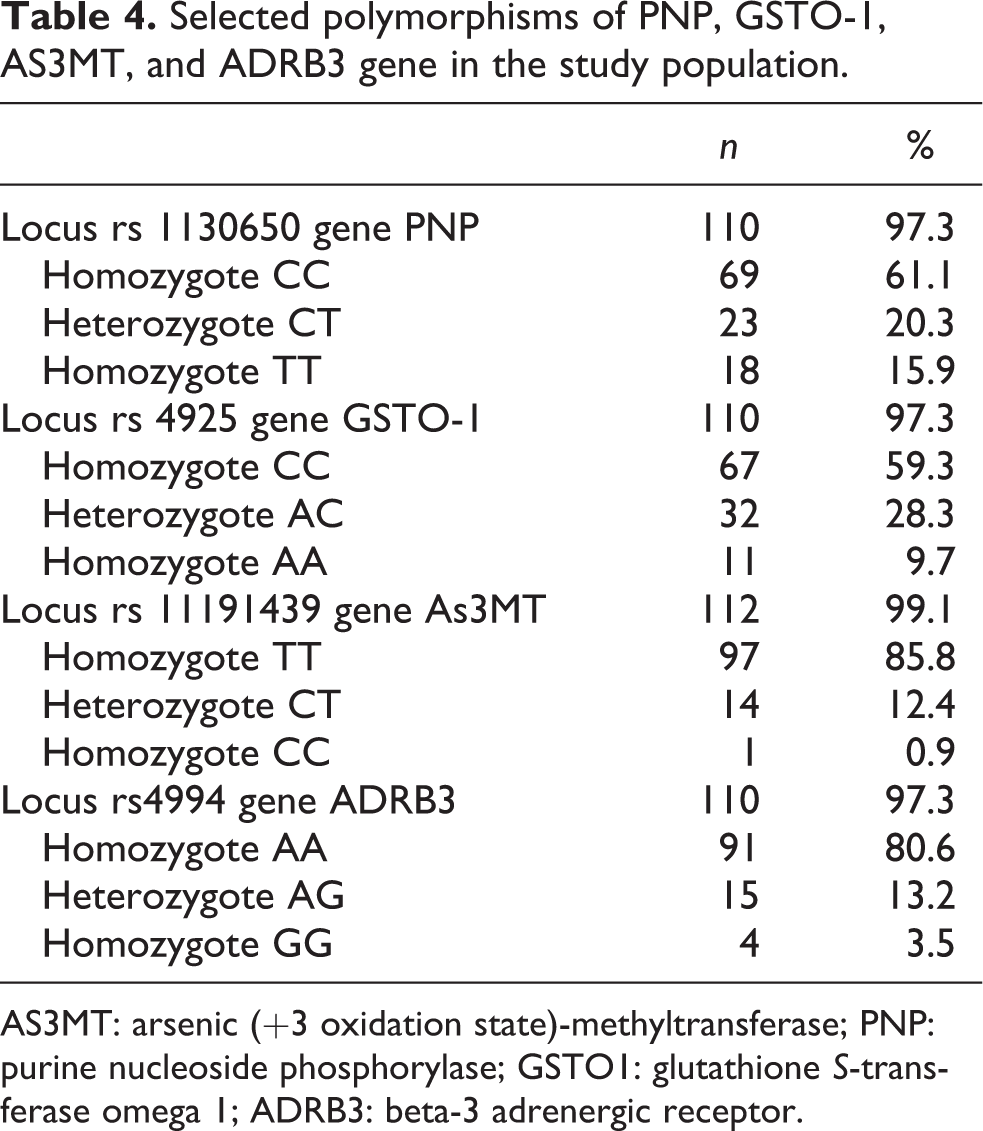
AS3MT: arsenic (+3 oxidation state)-methyltransferase; PNP: purine nucleoside phosphorylase; GSTO1: glutathione S-transferase omega 1; ADRB3: beta-3 adrenergic receptor.
Comparative analysis of subgroups divided on the basis of rs 1130650 PNP gene polymorphism showed that arsenic levels equal or higher than median for the whole group were statistically more common in homozygotes CC and heterozygotes CT when compared to the subgroup of homozygotes TT. As-U values for described subgroups are presented in Table 5.
As-U concentration in subgroups divided according to selected polymorphisms of genes.

AS3MT: arsenic (+3 oxidation state)-methyltransferase; PNP: purine nucleoside phosphorylase; GSTO1: glutathione S-transferase omega 1; As-U: urinary total arsenic concentration; ADRB3: beta-3 adrenergic receptor.
a Values are represented as mean ± standard deviation.
b Values are represented as number of persons/percentage.
The comparative analysis of subgroups based on the type of 4925 GSTO-1 gene polymorphism revealed that homozygotes AA were characterized by statistically significant higher mean values of As-U than heterozygotes AC and homozygotes CC. Moreover, the subgroup of homozygotes AA, when compared to heterozygotes CT and homozygotes TT, had statistically significant higher frequency of As-U levels equal or higher than median or the third quartile for the whole group and higher MAC value. As-U values for subgroups with described type of the GSTO-1 gene polymorphism are presented in Table 5.
The comparative analysis of subgroups divided on the basis of rs 11191439 AS3MT gene polymorphism and rs4994 ADRB3 gene polymorphism revealed no statistically significant differences. As-U values in subgroups divided on the basis of rs 11191439 AS3MT and rs4994 ADRB3 gene polymorphisms are presented in Table 5.
The comparative analysis showed no effect of the studied polymorphisms of the PNP, GSTO-1, AS3MT, and ADRB3 genes on the concentrations of Cu, Zn, Cd, Pb, and ZnPP. Cu-S, Zn-S, Cd-B, Pb-B, and ZnPP values in subgroups divided on the basis of PNP, GSTO-1, AS3MT, and ADRB3 gene polymorphisms are presented in Table 6.
Basic toxicological parameters other than urine arsenic concentration in subgroups divided according to selected polymorphisms of genes.

PNP: purine nucleoside phosphorylase; GSTO1: glutathione S-transferase omega 1; Pb-B: blood lead concentration; Cd-B: blood cadmium concentration; ZnPP: blood zinc protoporphyrins concentration; Zn-S: serum zinc concentration; Cu-S: serum copper concentration; AS3MT: arsenic (+3 oxidation state)-methyltransferase; ADRB3: beta-3 adrenergic receptor.
a Values are represented as mean ± standard deviation.
Discussion
Humans typically excrete absorbed arsenic in urine; 10–30% as inorganic, 10–20% as MMA, and 60–80% as DMA, although excretion patterns vary among populations. 10,17,25,28 Furthermore, there are significant differences in arsenic excretion among populations exposed to similar arsenic concentration. For instance, Indians from South America tend to excrete lower (about 5%) amounts of MMA when compared to other populations. 33 A number of factors have been hypothesized to impact As metabolism, including exposed dose, age, gender, race, exposure route, nutritional status, and some environmental circumstances of exposure. 13,34 Taking into account the role of AS3MT, GSTO, and PNP in arsenic biotransformation and metabolism, polymorphisms of genes encoding these enzymes are likely to produce interindividual variations in arsenic metabolism and thus influence susceptibility toward arsenic toxicity, in both in vivo and in vitro studies. 9,12
Glutathione is the most widespread particle with thiol group that can reduce arsenic (5+) to arsenic (3+) and create a stable complex. Both GSTO and PNP are enzymes catalyzing reduction of arsenic (5+). AS3MT catalyzes the methylation of iAs3+ using SAM as the methyldonor.
A comprehensive retrospective meta-analysis concerning influence of selected polymorphisms of AS3MT, PNP, and GSTO on arsenic methylation and its possible toxicity were published in 2014 by Antonelli et al. 10 He concluded that polymorphism of GSTO gene has no influence on arsenic concentration in urine. However, this influence was noticeable in animal studies and in vitro studies. The authors suggested that humans have arsenic reduction pathways other than just GSTO which makes it even more difficult to determine the role of GSTO in arsenic metabolism. On the other hand, Fu et al. discovered a negative influence of Ala/Asp polymorphism of GSTO1 gene on the level of DMA excreted in urine in a study on 155 Chinese people exposed to high-dose inorganic arsenic in drinking water. 19
Our study suggests that AA homozygosity in locus rs 4925 of GSTO1 gene is related to higher, exceeding maximum allowable concentration, urinary arsenic, hence individuals with such constellation should be thoroughly monitored for distant negative impact of arsenic exposure.
Genes encoding AS3MT and GSTO1 are located on the same region of chromosome 10 (10q25). Sampayo-Reyes et al. and Hernandez et al. studied polymorphisms of these genes in Mexicans exposed to arsenic in drinking water. They discovered a straight relationship between arsenic concentration in urine and level of DNA damage. This relation was modified by Met287Thr polymorphism in AS3MT gene. 15,28
Schlawicke Egstrom et al. also confirmed a strong influence of polymorphism of AS3MT in locus rs 3740400 on level of arsenic methylation and its detoxification. 17 Hwang et al. proved that polymorphism G > C in intron 6 and T > C in intron 10 of AS3MT gene is related to significant difference in excretion of methylated forms of arsenic, especially of DMA. 1
Agusa et al. studied the frequency of particular AS3MT gene polymorphisms in populations of various countries (Mexico, India, Chile, Argentina, Taiwan, and others). 9 To describe this gene’s polymorphic model in East-Central Europe Lindberg research studies were used. Lindberg et al. showed that allele C in locus rs 11191439 of AS3MT gene is related to higher MMA (V) and lower DMA (V) concentration in urine, especially in men. 34
Our study yielded no statistically significant correlation between AS3MT polymorphism and urinary arsenic, nevertheless subjects with allele T in locus rs 11191439 had their total arsenic concentration more than twice as high as homozygous CC. No direct relationship does not exclude the relevance of this result. Sampayo-Reyes et al. showed that arsenic affects DNA damage and that AS3MT polymorphism has an important impact on this damage, however, mostly in children. 15 In a research from 2017, Hsieh et al. discovered a link between AS3MT polymorphism, decrease in arsenic methylation, and child development retardation. 35
The role of PNP polymorphism is even harder to determine and scientific data on the subject is limited. In the aforementioned Antonelli’s meta-analysis, this polymorphism was not included in the study due to the lack of bibliographic data. 10 However, research from 2015 by Gaxiola-Robles et al. showed that increased PNP activity induces increase in glutathione peroxidase and reductase activity but not in aforementioned transferase. 20 Nevertheless, due to limited data, it is hard to univocally assess connections between processes of arsenic detoxification, GSTO activity, and polymorphic constellation of GSTO1 gene. Chaudhuri et al. provided unambiguous proof of dependence between arsenic toxicity and PNP gene polymorphism, namely polymorphism His20His-, Gly51Ser-, and Pro57Pro-induced skin cancer. 12
Our study showed a correlation between homozygous T allele in locus rs 1130650 of PNP gene and lower urinary arsenic concentration which could be the result of lower absorption of arsenic among the carriers and as a consequence may contribute to lower arsenic toxicity.
The adrenergic beta-3 receptor is the protein that works as a regulator of lipolysis as well as thermogenesis. From all adrenergic receptor, ADRB3 is the most interesting to investigate in terms of sky-rocketing frequency of metabolic syndrome. Until now, connection between Trp64Arg polymorphism of gene ADRB-3 (rs4994) and development of abdominal obesity, diabetes, insulin resistance, and atherogenic dyslipidemia was found. 29 –31 This polymorphism was also reported to influence the effectiveness of pharmacological treatment of obesity. 36 In addition, relationship of Gln27Glu polymorphism of gene ADRB-2 with lower body mass was observed. 37 On the other hand, the role of xenobiotics, including biotoxic metals, is getting more and more significant when discussing pathogenesis of metabolic diseases and atherosclerosis. 22,25 Therefore, investigating the relationship between arsenic concentration and aforementioned ADRB-3 (rs 4994) polymorphism seems both reasonable and interesting. Our study showed no statistically important relationship, however, we observed a marked tendency to higher urinary arsenic concentration in allele A carriers in locus rs 4994 of ADRB-3 gene. Considering the fact that most (82.3%) of studied copper smelter workers were obese, 26.5% had hypertension, 6.2% had diabetes, and 4.4% had atherogenic dyslipidemia, it might be concluded that metabolic syndrome is caused not only by behavioral factors (their impact is without doubt the most crucial) but also by genetic factors and environmental pollution.
This study has significant limitations that may prompt other researchers to continue our work. In the scope of the studied group, the restrictions are no control group (people who are not professionally exposed to arsenic) and overrepresentation of men. In case of testing health consequences of arsenic exposure, a control group of unauthorized subjects would be necessary. In studies on genetic predisposition, lack of such group is a significant limitation, however, it does not exclude the possibility of valuable inference concerning the examined group of those professionally exposed to arsenic. A large overrepresentation of men among the subjects was a reflection of the gender structure of employees within the examined factory. Due to the substantially lower number of women in the examined group, the analysis of the impact of gender on observed relationships was not performed. In terms of methodology, the limitations include no determination of inorganic arsenic (AsIII and AsV) and metabolites of arsenic (MMA and DMA), as well as glutathione (GSH and GSSG). Additional laboratory analyzes were not performed due to the limited amount of biological material.
In conclusion, our study has proved that individual tendency to absorb toxic metals (especially arsenic) in workplaces with the same standard of occupational hygiene is determined by genetic factors. Thus, based on frequency of particular polymorphisms, a group of workers mostly predisposed to extensive arsenic absorption can be extracted and their work can be planned in more rational way (for instance by avoiding high-exposure worksites). Another step would be to asses direct health influence of chronic exposure to arsenic and other metals, especially in terms of cancer morbidity, diabetes, and cardiovascular disease. Notwithstanding, it must be remembered that exposure to toxic metals is only one of the many factors involved in etiopathogenesis of these diseases, next to a number of detrimental behavioral factors such as smoking cigarettes, minimal physical activity, and improper diet. Therefore, the right prophylaxis should focus on both wide educational actions and systematic toxicological monitoring as well as, when possible, directing workers to less exposed worksites.
Conclusions
Correlation between polymorphisms of PNP and GSTO-1 genes and urinary arsenic concentration was found in subjects occupationally exposed to arsenic which confirms the hypothesis that tendency to absorb toxic metals is genetically determined.
People occupationally exposed to arsenic with homozygous allele T in locus rs 1130650 of PNP gene are predisposed to lower urinary arsenic concentration.
AA homozygosity in locus rs 4925 of GSTO-1 gene may result in higher urinary arsenic concentration.
In people occupationally exposed to arsenic, polymorphism in locus rs 11191439 of AS3MT gene and in locus rs4994 of ADRB3 gene has no statistically significant influence on urinary arsenic concentration, however, the presence of some alleles in these loci indicates the tendency toward higher absorption of arsenic.
In people particularly predisposed to higher arsenic absorption, that is, having certain polymorphisms in genes PNP, AS3MT, GSTO-1, and ADRB-3, a comprehensive assessment of distant health effects of arsenic exposure should be performed, including oncological screening, as well as blood pressure, lipid, and glucose control. In this group, early non-pharmacological prevention of arterial hypertension and other metabolic syndrome components, such as diabetes, dyslipidemia, and obesity should be introduced.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Wroclaw Medical University.
