Abstract
Brimonidine is a first-line topical medication for increased intraocular pressure and glaucoma which may be used alone or in conjunction with other topical therapies. Its structural and pharmacological comparabilities to clonidine give way to the hypothesis that it may cause neuropsychiatric side effects. The majority of case reports citing brimonidine toxicity, either for topical or peripheral exposure, include pediatric age groups but especially infants. Among the latter, a dose-response phenomenon is evident. Dose-response correlates have also been shown among adults. Case series and prospective double-blind treatment studies also give evidence for the occurrence of several central nervous system adverse reactions. Topical ophthalmic brimonidine use should be followed for the occurrence of neuropsychiatric disturbances generally, and enhanced vigilance should be maintained for at-risk populations.
Introduction
Although the spectrum of topical treatments for increased intraocular pressure and glaucoma has become more plenary, brimonidine has remained one of the most common pharmacological agents utilized for single, dual, or add-on therapy. It also remains an important option when other topical pharmacological agents require discontinuation for either adverse events, compliance problems, insufficient efficacy, or contraindications otherwise.
Brimonidine had been initially considered for topical use as early as 1974, but it was largely not available until 1996. 1 –3 It was formerly labeled UK-14304 and AGN 19042-22. Currently, it is available in forms of 0.1–0.2% brimonidine tartrate suspensions alone or in combination when used for glaucoma. 4 An over-the-counter preparation of 0.025% brimonidine has been marketed for relief of eye redness but not for intraocular pressure-lowering capability. 5 The latter preparation has not been cited for toxicity and is not discussed herein. It is classified as an α-adrenoreceptor agonist in ophthalmological pharmacology, and as such, is similar to two other α-agonists, clonidine and apraclonidine. 4,6,7 As such, it activates a G protein-coupled receptor which inhibits adenyl cyclase, and reduces noradrenaline release. Clonidine is a 2-imidazoline derivative, and its use was initially reserved for hypertension. Early studies demonstrated that systemic use from oral ingestion among hypertensive patients was also accompanied by reduced intraocular pressure. Both oral and topical clonidine, however, were associated with systemic side effects, including those of the neuropsychiatric spectrum. 8 Apraclonidine is an analog of clonidine, and it was believed that few central nervous system (CNS) events would occur since it was found to enter the CNS in lesser degree. 4 Brimonidine became the third of this group to be used topically for intraocular pressure therapy. It is also a 2-imidazoline derivative.
Clinical and experimental pharmacology
Much of the evolution of brimonidine use has followed a progressive increase in the science of adrenoreceptors. The brimonidine class of medications are indeed α-adrenoreceptor agonists, but there is differentiation of these receptors into α1- and α2- categories. The latter may be further subdivided or subtyped. 2 There are at least three considerably homologous subtypes in humans to which brimonidine may bind. Brimonidine was found to be more α2-adrenoreceptor selective (7–32 times) than either clonidine or apraclonidine. 4,9 α2-adrenoreceptors exist in both ocular tissues (iris, ciliary body, retina) and the CNS. 3,10 These are prejunctional receptors that may affect neurotransmitter release and negative feedback loops. 1 Brimonidine was deemed 1500–2000 times more α2- than α1- selective. 7,11 That said, it must be acknowledged that brimonidine is not purely α2- selective and that it nevertheless maintains measurable α1-agonist activity. Ocular α2-adrenoreceptor effects include conjunctival vasoconstriction, increased interpalpebral fissure, mydriasis, and decreasing corneal oxygen tension. 4,6,7 Initially, the effects of brimonidine were thought to be due mainly to its topical and peripheral activations. Brimonidine’s reduction of intraocular pressure is ascribed to both a mitigation in aqueous humor production and an increase in uveoscleral outflow. 4,6,7 The differential selectivity of brimonidine over clonidine and apraclonidine was thought to confer distinct ocular effects, and hence its preference for topical ophthalmological treatment prevailed. 4 With brimonidine and apraclonidine having less lipophilic properties than clonidine, it was also suggested that diminished penetration into the CNS would be associated with fewer side effects. 9
Topical administration of brimonidine yields aqueous humor concentrations up to 300 nM but nearly 100 times a lower concentration in the vitreous. 12 Other studies established an aqueous humor concentration variably from 5 mcg/L to 100 mcg/L approximately 1 h after a single drop instillation of a 0.1–0.15% solution. 7,13 Brimonidine may lower intraocular pressure within minutes, but it has a peak effect after approximately 2–3 h. The topical effect may have up to 14 h duration. The t ½ is 2–3 h, and there are blood levels obtained which are usually less than 1 mcg/L. There is considerable hepatic metabolism of the systemic drug, and urinary excretion of the metabolites follows. 1,4,7,10,11,14 Inter-person variability can be considerable. 12
When blood–brain barrier penetration occurs, the α-agonist class can stimulate CNS α-adrenoreceptors to cause systemic hypotension, bradycardia, and sedation. 4 Clonidine, for example, causes CNS effects through adrenoreceptor activation in the brain stem. 4 α2-adrenoreceptors are ubiquitous in many regions of the CNS. The subtypes of these receptors have more favored distribution in some CNS structures compared to others. 15
In humans and primates, there is a small but significant effect of reduction in intraocular pressure in the contralateral eye. 9,10 This has led to some to believe that brimonidine works mainly through its CNS presence. Studies in rabbits have demonstrated that topically applied brimonidine is found to penetrate the optic nerve and olfactory bulb. 16 At these latter sites, drug concentration is greater than it is in blood. High levels of drug were also found in the contralateral rabbit eye. The authors projected that such findings were supportive of CNS penetration with associated effects. They also proposed that absorption centrally was primarily achieved through the olfactory bulb after nasal absorption from ocular ductal flow to the nasal mucosa. 16 Animal model study has determined that systemic absorption is greater with a topical solution in contrast to topical gels. 17
Brimonidine can also be an agonist for peripheral α2-adrenoreceptors. 18 It is not evident at this time, however, as to whether an associated peripheral toxic event necessarily translates into adverse CNS events.
Other relevant CNS-related pharmacology
A number of other basic science studies have relevance to the concept that brimonidine may have CNS effects. In cats, topical application produced sedation for a variable period (30–120 min) of times. 19
Depression has been associated with an increased α2-adrenoreceptor activity in the brain. 20,21 Such determinations have utilized labeled brimonidine as the detector ligand. 20 Further characterization has particularly singled out the α2A-subtype in depression. 22,23
The α2-adrenoreceptor decreases in intensity in the aging brain. 24 The latter, in theory, could account for a lesser-than-expected CNS adversity among older age groups where intraocular pressure reduction therapy is more likely to be used.
Patient-dependent effects are also in part modulated by some of brimonidine’s putative neuroprotective effects. Brimonidine is said to upregulate neurotrophic factors and to have an antiapoptotic effect. 25 It may preserve optic neurons and enhance retinal transport in an animal model of eye injury and increased intraocular pressure. 26 Retinal cell protection in an animal model of optic nerve injury has also been detailed. 27 There may be multiple pathways, therefore, by which brimonidine protects glaucoma-induced degeneration. 28 In one study, brimonidine use improved visual fields. 29 Definitive proof of the same, however, in well-designed prospective human studies is lacking. These benefits would seem independent of how CNS α2-adrenoreceptor agonist activity would potentially lead to neuropsychiatric side effects.
Despite the advances in the understanding of α2-adrenoreceptor agonist activity and brimonidine, there is no definitive proof or highly plausible reason as to how CNS toxicity is actually produced. The science is further complicated by the effects of α2-adrenoreceptor agonists on ion channels and various cellular transporters. 30
Dose-response correlates
CNS side effects appear to have a dose-response correlate. In small mammals, topical application and oral ingestion lead to sedation and various other neurological complications. 19,31
Among humans, the dose-response effect is evidently manifested by many anecdotes of toxicity (Table 1). The case reports are variable for single or coadministration with other topical ophthalmic agents, but several of these citations note immediate systemic effects after administration of a single drop of brimonidine. The majority of reports describe patients of a very young age. Few citations have confirmed the associated reaction by a trial–retrial approach, but onset and resolution after drug administration and cessation has been evident in most circumstances. Toxicity from oral ingestion or other sites of topical application (e.g. dermatological) are consistent in the subsequent pathological profile that evolves. Cardiovascular dysfunction is not uncommonly experienced at the same time that neuropsychiatric events transpire. Some of these reports debate the value of naloxone in remedying the profound side effects. Resolution of adverse events occurs in short order which is consistent with the diminution of blood levels given the relatively short half-life of brimonidine. Although a dose-response effect is suggested by the pediatric experience, the detailed case studies in children nevertheless suggest considerable patient-to-patient variation. 36,39,41 In one prospective pediatric series, nearly three-quarters of all patients suffered sleepiness, and the side effects not uncommonly led to discontinuation of the topical agent. 52 Episodes of CNS depression, cyanosis, and dyspnea were also documented. It was evident that more side effects occurred for those with lower age and weight. A tabulation of toxicities in a poison control center also showed that children were at risk for brimonidine toxicity. 53
Case reports and small series for brimonidine toxicity among pediatric ages.

Singular reports of brimonidine toxicity among adults are uncommon, but a dose-response relationship is evident as well. 54,55 More tangible evidence, however, was found in a double-blind randomized trial of three topical brimonidine doses (0.08%, 0.2%, 0.5%) and placebo among adult volunteers. 56 The frequency of fatigue and drowsiness increased from 6.7% to 10.4% to 29.2% for the three brimonidine preparations in the latter study. The dose-related response for managing intraocular pressure is also accepted, and brimonidine must thus have a very narrow therapeutic index. 2
Given such dose-responsivity, and given therefore the potential for toxicity in young patients, it is not surprising that drug product monographs suggest that ophthalmic brimonidine (in doses of 0.1–0.2%) is contraindicated for children under 2 years of age and that the drug in these topical eye doses is not recommended for other pediatric ages under 18 years. Consideration for use in any pediatric patient must weigh the benefits and potential risks unique to that specific clinical context. In the event of a necessary prescription for a child while weighing all other options, careful monitoring for adverse events is imperative. The dose-response concept has also translated into advocating that brimonidine should not be used late in pregnancy. 57
Propositions of toxicity
Both clonidine and brimonidine in the oral form are associated with sleepiness and sedation; brimonidine seems less active in this regard. 8 Reviews of brimonidine use recognized the potential for side effects such as fatigue, somnolence, and drowsiness very early. 1,3,6
Table 2 lists various CNS side effects that have been cited. The vocabulary used to describe such events is not standardized and hence may overlap between studies. Some authors note that the timing for discontinuation of brimonidine and thereafter use of another topical drug was relatively short in comparison to switches from the other agents. 58 Use of brimonidine was also associated with poor adherence to prescribed dosing schedules. 59,60 When patients attended the emergency room with adverse reactions, the need to discontinue brimonidine was not uncommonly reported. 61
Reported neuropsychiatric events with the use of topical ophthalmic brimonidine.
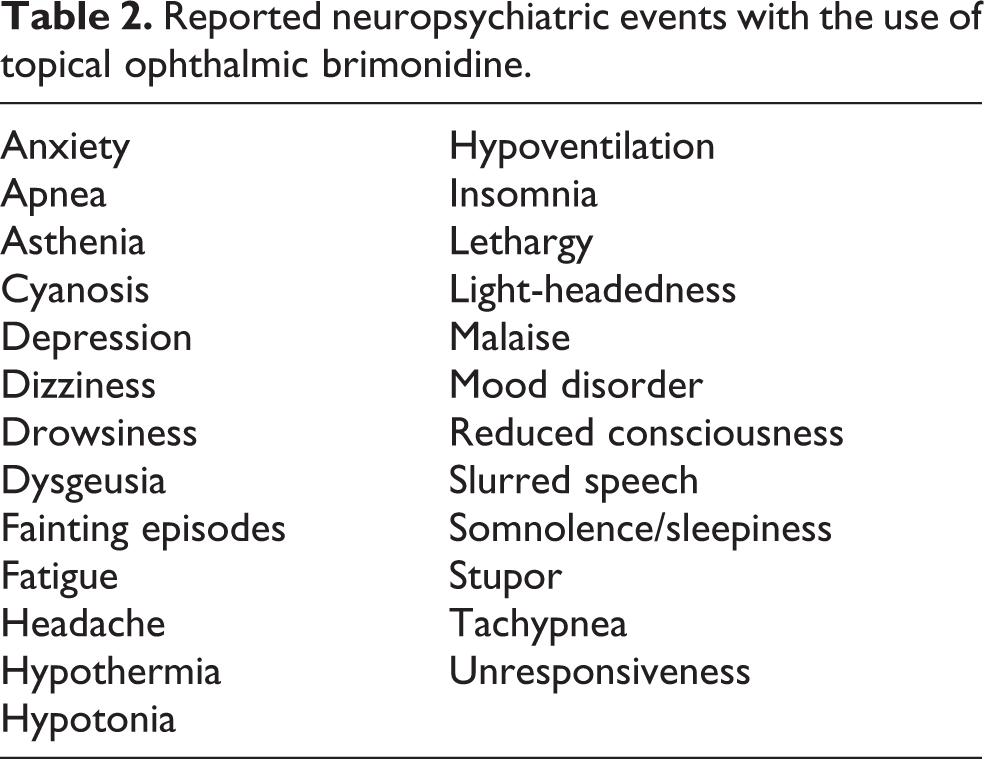
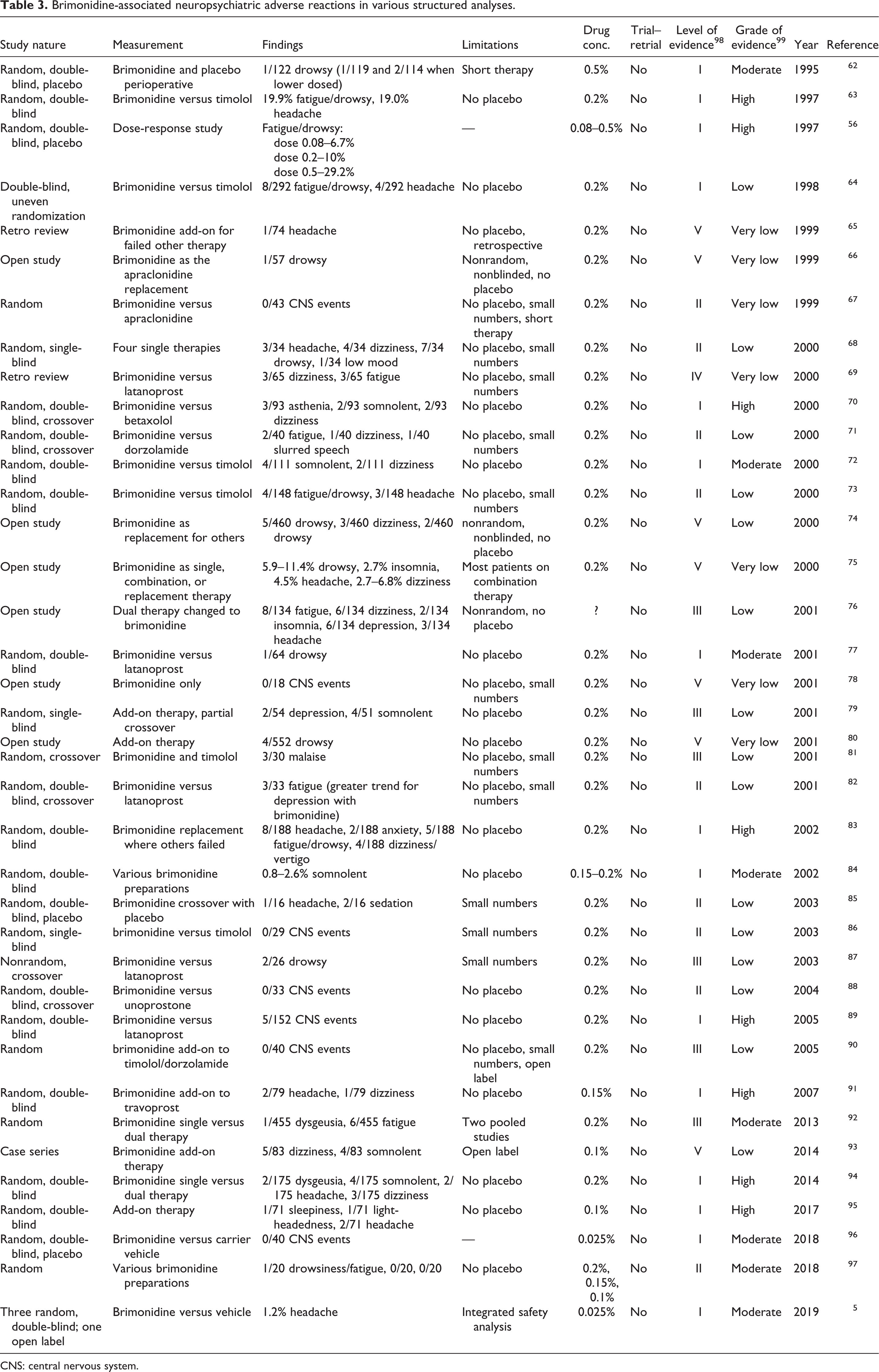
Table 3 details side effects ascribed to brimonidine in case series. Few of these studies are placebo-controlled, and many publications are complicated by the co-prescription of other topical therapies. The constitution of these studies is considerably heterogeneous, thus making the prospect of meta-analysis quite troublesome. Levels and grades of evidence are also quite variable, although the data can be summarized favoring an acceptance that brimonidine causes neuropsychiatric side effects. The types and frequencies of CNS events are also variable. No studies have validated causation by trial and retrial, but evidence for a dose-response is supported. It is clear that some patients withdraw from these studies due to the severity of adverse effects. Hwang et al. have recently found evidence of histopathologically confirmed lymphoproliferative activation in the conjunctivae of some patients treated with brimonidine, but it is not known how this finding may bear on toxicity generally. 100
Brimonidine-associated neuropsychiatric adverse reactions in various structured analyses.
CNS: central nervous system.
Although a dose-response for toxicity in younger age groups is suggested by the pattern of reported adverse events, molecular and physiological mechanisms that create any such toxicity are not sufficiently understood and are at this time mainly extrapolated from anticipated pharmacological responses. Theoretical considerations for dose-responsiveness regarding toxicity could conceivably include dose per body weight, differences in absorption or drug metabolism, differences in drug excretion, and variation in drug-receptor responsiveness. 47
Conclusion
A dose-response central effect for topical brimonidine has been found in both pediatric and adult populations. There is heterogeneity in the spectrum of these adverse effects. A CNS mechanism is credible but not fully proven. Despite the above, the medical literature is sparse for individual reports of neuropsychiatric complications among adults. Whereas brimonidine activates α2-adrenoreceptors, an age-related decrease in brain α2-adrenoreceptors has been found. The latter has the potential to mitigate the occurrence of CNS side effects in the elderly for whom topical brimonidine is more likely to be used in treating increased intraocular pressure. Judicious use of topical brimonidine seems prudent, and at-risk populations should be particularly reviewed after the onset of administration both acutely and chronically.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
