Abstract
Carbon tetrachloride (CCL4) is often employed in the production of chlorofluorocarbons, petroleum refining, oil and rubber processing, and laboratory applications. Oral, subcutaneous, and inhalation exposure to CCL4 in animal studies have been shown to be capable of leading to various types of cancer (benign and malignant, liver, breast, and adrenal gland tumors). The present study also evaluated the protective role of infliximab (INF) against the deleterious effects of CCL4 on the intestinal system. Twenty-four male Sprague-Dawley rats were randomly assigned into three groups, control (n = 8), CCL4 (n = 8), and CCL4 + INF (n = 8). The control group received 1 mL isotonic saline solution only via intraperitoneal (i.p.) injection. The CCL4 group received a single i.p. dose of 2 mL/kg CCL4. The CCL4 + INF group received a single i.p. dose of 7 mg/kg INF followed 24 h later by a single dose of 2 mL/kg CCL4. All rats were euthanized 2 days following drug administration. CCL4 group samples also exhibited diffuse loss of enterocytes, vascular congestion, neutrophil infiltration, an extension of the subepithelial space and significant epithelial lifting along the length of the villi with a few denuded villous tips. In addition, CCL4 treatment increased intestinal malondialdehyde (MDA) level and caspase-3 positivity. On the other hand, INF decreased MDA levels, caspase-3 positivity, and loss of villous. Our findings suggest that CCL4 appears to exert a highly deleterious effect on the intestinal mucosa. On the other hand, INF is effective in preventing this CCL4-induced intestinal injury by reducing oxidative stress and apoptosis.
Introduction
Carbon tetrachloride (CCL4) is a transparent, colorless, heavy liquid with an odor resembling that of ether. 1 It is often employed in the production of chlorofluorocarbons, petroleum refining, oil and rubber processing, and laboratory applications. 2 Human exposure usually occurs through inhalation, ingestion, or the dermal route and the general population will most probably be exposed to CCL4 through the air or drinking water. 1,2 Exposure through the digestive tract is also possible via contaminated water or foodstuffs. 3
Experimental animal models suggest that CCL4 is a human carcinogen. 1,3,4 Oral, subcutaneous, and inhalation exposure to CCL4 in animal studies have been shown to be capable of leading to various types of cancer (benign and malignant, liver, breast, and adrenal gland tumors). 3,5 Nonetheless, epidemiological evidence of a clear link between CCL4 and human cancer types is lacking. However, studies have reported an increased incidence of non-Hodgkin’s lymphoma and leukemia in studies involving subjects exposed to CCL4, such as industrial and laboratory staff and individuals working with pesticides. 2,6 –8
The effects of CCL4 on various organs, and particularly the liver, have been studied in animal models. 3,5,8 It has been shown to be metabolized to its metabolites trichloromethyl (CCL3) and trichloromethyl peroxide (CCL3O2) radicals by cytochrome P450 enzyme. 9 These free radicals then cause lipid peroxidation in cell membrane. 9 Malondialdehyde (MDA) is a product of this lipid peroxidation, and high levels are an indicator of tissue damage. 10 MDA is a product of lipid peroxidation, and superoxide dismutase (SOD) is a scavenger enzyme of superoxide, and both are employed as indirect indicators of tissue damage. 11 –13 This may indicate a different tissue damage mechanism of CCL4 in intestinal tissue, or may perhaps be attributed to low study population numbers. 1,4,7,13,14 Administration of CCL4 also stimulates the caspase pathway and regulates apoptosis. 15,16 Caspase-3, a terminal and irreversible stage of the caspase cascades, is used as a biomarker of apoptosis. 17 –19 Animal studies have revealed CCL4-induced alterations in animal models of the liver, kidney, lung, and spleen. 4,9,13,20 –23 Such studies have shown that CCL4 causes oxidative stress in a variety of organs and exhibits a broad range of toxicity. 12,13,17,23 Its toxic effects are due to its metabolites trichloromethyl free radical and trichloromethyl peroxyl radical, and also trigger the release of tumor necrosis factor-alpha (TNF-α) and interleukin-1 beta. 8,12,15,22 CCL4 also induces tissue injury by enhancing lipid peroxidation, increasing MDA levels, and activating caspases. 13,17 8-Hydroxy-2′-deoxyguanosine (8-OHdG) has been used in a biomarker for the measurement of endogenous oxidative DNA damage. 24,25 To the best of our knowledge, there has been no previous research into the direct effects of CCL4 on the intestinal system. Although CCL4 has been reported to aggravate carcinogenesis in the intestinal system, it has not been directly linked to intestinal carcinogenesis. 26 CCL4 has also been implicated in rectal cancer resulting from occupational exposure in the human population. 14
Infliximab (INF) is a monoclonal antibody and TNF-α antagonist employed in the treatment of various inflammatory diseases. 27,28 INF has been reported to ameliorate inflammation-related tissue damage in various tissues by inhibiting the production of TNF-α cytokines and free oxygen radicals. 21,29
TNF-α has been proved to increase the production of cytokines and to directly stimulate the caspase pathway. Several studies have demonstrated the protective effects of INF, a TNF-α blocker, against CCL4-induced organ damage. 21,22 The present study also evaluated the protective role of INF against the deleterious effects of CCL4 on the intestinal system.
Materials and method
Twenty-four male Sprague-Dawley rats aged 3–5 months and weighing 243 ± 40 g were obtained from the University Animal Care and Research Unit. All animals received humane care in line with the criteria set out in the National Academy of Sciences “Guide for the Care and Use of Laboratory Animals” published by the National Institutes of Health. The study was approved by the University Animal Ethical Committee. The animals were housed at the University Faculty of Medicine Basic Medical Sciences Experimental Animal Application Unit in a 12-h light:12-h dark cycle, at a room temperature of 22 ± 3°C and 55–60% humidity.
After being allowed sufficient time to adapt to the laboratory conditions, the 24 rats were randomly assigned into three groups: control (n = 8), CCL4 (n = 8), and CCL4 + INF (n = 8). Prior to drug administration, anesthesia was induced using 50 mg/kg intraperitoneal (i.p.) ketamine hydrochloride (Ketalar ®, Eczacıbaşı Parke-Davis, Istanbul, Turkey) and 10 mg/kg i.p. Xylazine HCl (Alfazyn®, Alfasan International BV, Woerden, The Netherlands). The control group received 1 mL isotonic saline solution only via i.p. injection. The CCL4 group received a single i.p. dose of 2 mL/kg CCL4. 21 The CCL4 + INF group received a single i.p. dose of 7 mg/kg INF (Remicade, Schering-Plough (Brinny) Co., Innishannon, Ireland) followed 24 h later by a single dose of 2 mL/kg CCL4. 21,30 All rats were euthanized 2 days following drug administration. Intestinal tissues were extracted and stored at −80°C until analysis.
Histopathological analysis procedure
Small intestine tissues were subjected to fixation in 10% neutral formaldehyde for 36 h. Specimens were then dehydrated by being passed through 50%, 70%, 80%, 90%, 96%, and 100% alcohol series (two times for each) (Merck, Darmstadt, Germany). Next, the specimens were cleared in xylene (Merck) and blocked in paraffin (Merck) using routine laboratory methods. Sections were then analyzed under a light microscope (Olympus Corporation, Tokyo, Japan) and photographed with an Olympus DP20 camera (Olympus Corporation).
Immunohistochemistry analysis procedure
Caspase-3 (Abcam, ab4051 Rabbit polyclonal to active caspase-3, UK) and 8-OHdG (Elabsciences, Beijing, People’s Republic of China) were used to identify apoptotic cells in small intestine sections. The sections were first deparaffinized, treated with proteinase K solution (20 μg/mL in phosphate-buffered saline (PBS)), and immersed in 3% hydrogen peroxide and equilibration buffer. Small intestine sections were then incubated with caspase-3 (1:150, Abcam Rabbit polyclonal to active caspase-3). Sections were next incubated in anti-digoxigenin-peroxidase, and the reaction was visualized with 0.06% 3, 3-diaminobenzidine tetrahydrochloride (Sigma Chemical, St. Louis, Missouri, USA) in PBS. Finally, the sections were counterstained with Harris hematoxylin (Merck) and evaluated by two blinded histopathologists.
Semiquantitative analysis
Small intestine section damage was scored as described by Chiu 31 (Table 1). Twenty randomly selected areas on each section were assessed by two blinded histopathologists.
Grading of mucosal changes following CCL4 administration.a

CCL4: carbon tetrachloride.
a Based on the Kruskal–Wallis test.
bp < 0.05 versus the CCL4 group.
cp < 0.05 versus the CCL4 + infliximab group.
dp < 0.05 versus the control group.
Stereological analysis
Mean caspase-3 positive numerical cell densities were calculated on the Stereo Investigator (MicroBrightField 9.0, Colchester, Vermont, California, USA) software system. The stereological analysis involved the fractional method, a stereological method using an unbiased counting frame. Separate and distinct immunohistochemistry (IHC)-caspase 3-stained sections were determined by systematic random sampling, following the rules of space fragmentation with the step interval of the x- and y-axes. Measurements were then performed in 40 different selected areas in all groups, as described elsewhere by Mercantepe et al. 32
Biochemical investigations
Superoxide dismutase analysis
The SOD activity was measured using a colorimetric assay kit (Cayman Chemical Company, Ann Arbor, Michigan, USA). The procedure was performed in line with the instructions provided by the manufacturer. Absorbance was measured at a wavelength of 450 ηm using a plate reader. The SOD activity results were expressed as U/mL. The intra-assay and inter-assay coefficients of variation were 3.2% and 3.7%, respectively. The dynamic range for the SOD assay kit was 0.025–0.25 U/mL.
Malondialdehyde analysis
MDA forming as a result of lipid peroxidation was determined by measuring the concentration of the pink-colored complex that develops after heating with TBA solution. Following incubation, 900 μL was taken from the study tubes and added to 600 μL TCA solution and centrifuged at 3000 r/min for 10 min. After centrifugation, 900 μL supernatant was collected. To this was added 300 μL freshly prepared TBA solution, and the mixture was left to incubate in a boiling water bath for 15 min. 33
Statistical analysis
Numerical densities of caspase-3-positive cells were expressed as mean ± standard deviation. Differences in numerical data between the groups were assessed using one-way analysis of variance followed by a Tukey HSD test (p values <0.05 were regarded as significant). Values were determined as a mean ± standard deviation.
Assessment of histopathological status was based on damage scores expressed as median values (min–max). The Kruskal–Wallis test was used for intergroup analysis (p < 0.05 being considered significant). Statistical analysis of SOD and MDA values in all groups was performed on SPSS software (IBM SPSS Statistics 18.0, IBM Corporation, Somers, New York, USA).
Results
Light microscopy results
Light microscopic sections obtained from the intestinal tissue of the control group exhibited normal histological structural features. The level of damage for samples from the control group was evaluated as 0 (normal mucosa; following Chiu et al.) (Figure 1(a)). Intestinal tissue from the CCL4 group exhibited diffuse loss of epithelial cells and capillary congestion. CCL4 group samples also exhibited neutrophil infiltration and extension of the subepithelial space. The degree of structural damage in CCL4 group intestinal tissue samples was 4 (significant epithelial lifting along the length of the villi with a few denuded villous tips) (Figure 1(b)). Specimens from the CCL4 + INF group exhibited regular intact mucous membrane structures. However, the development of subepithelial (Gruenhagen) spaces from the lamina propria of the villi was also observed. The degree of structural intestinal damage in the CCL4 + INF treated group was 1 (development of subepithelial (Gruenhagen) spaces near the tips of the villi with capillary congestion) (Figure 1(c) and (d); Table 1).

Representative images of small intestine sections from all groups. H&E. (a) Control group: damage grade score 0; normal mucous membrane morphology (arrow). Lamina propria (Lp), lacteal (L), crypts of Lieberkühn (Lb), tunica muscularis (Tm) (×40). (b) CCL4 group: damage grade score 4; CCL4 group samples exhibited denuded villi with exposed lamina propria, capillary congestion (spiral arrow), neutrophil infiltration (blue arrow), and extension of the subepithelial space (tailed arrow). Moderate loss of epithelial integrity (arrow) (×20). (c) CCL4 group: damage grade score 4; capillary congestion (spiral arrow), neutrophil infiltration (blue arrow), and moderate loss of epithelial integrity (arrow) (×40). (d) CCL4 + infliximab group: damage grade score 2; intact mucous membrane structures (arrow). Development of subepithelial (Gruenhagen) spaces (tailed arrow) with occasional epithelial lifting (green arrow) from the lamina propria of the villi. Lamina propria (Lp) (×10). CCL4: carbon tetrachloride.
Immunohistochemical results
Sections from the control group were caspase-3 negative (Figure 2(a)). Intense caspase-3 positivity was observed in epithelial cells in the CCL4 group (Figure 2(b)). In contrast, sections from the CCL4 + INF group samples exhibited moderate epithelial cell positivity (Figure 2(c)).
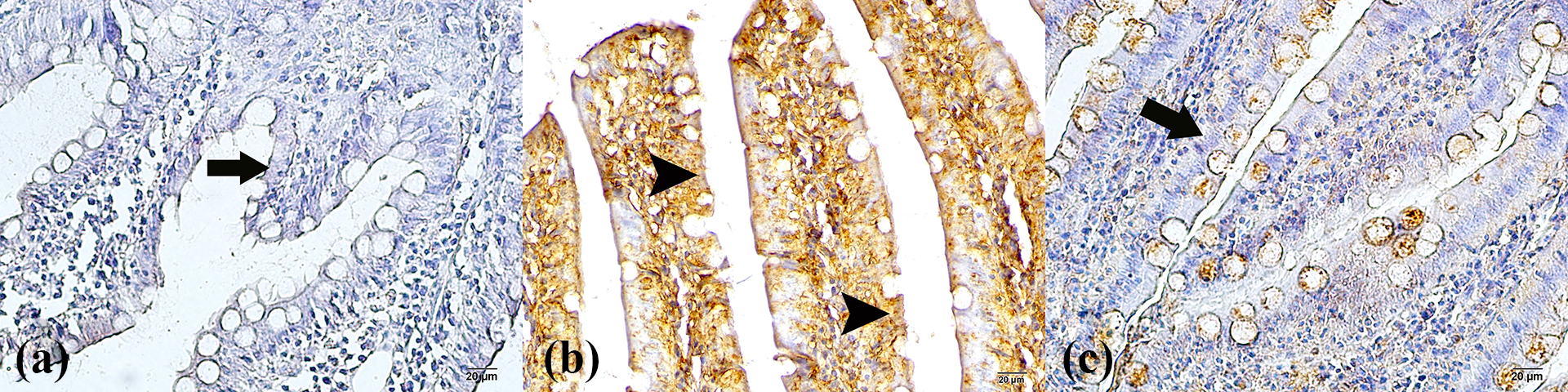
Immunohistochemical staining with caspase-3 and histopathological examination of the small intestine under light microscopy. (a) Control group: normal enterocytes (arrow) (×40). (b) CCL4 group: We show apoptotic enterocytes (arrowhead) with caspase-3 positivity (×40). (c) CCL4 + infliximab treated group: the typical enterocytes (arrow). Immunoperoxidase staining with anticaspase-3 antibody (×40). CCL4: carbon tetrachloride.
Semiquantitative results
Our results revealed greater caspase-3 and 8-OHdG positivity of the intestinal epithelial cell in the CCL4 group compared to the control group (p < 0.05). The CCL4 + INF group exhibited less staining for caspase-3 and 8-OHdG expressions than the CCL4 group. Caspase-3 and 8-OHdG staining of the intestinal epithelial cell in the CCL4 + INF group was similar to that of the control group (p > 0.05) (Table 2 and Figure 3).
Caspase-3 positive numerical density measurement (mm3) data.a

CCL4: carbon tetrachloride; 8-OHdG: 8-hydroxy-2′-deoxyguanosine.
a Tukey HSD test.
bp < 0.05 versus the control group.
cp < 0.05 versus the CCL4 group.

Immunohistochemical staining with 8-OHdG and histopathological examination of the small intestine under light microscopy. (a) Control group: normal enterocytes (arrow) (×40). (b) CCL4 group: 8-OHdG positive enterocytes (×40). (c) CCL4 + infliximab treated group: The typical enterocytes (arrow). CCL4: carbon tetrachloride; 8-OHdG: 8-hydroxy-2′-deoxyguanosine.
The results also revealed a higher damage score in the CCL4 group (score: 5) compared to the control group (score: 0) (p < 0.05). The CCL4 + INF group exhibited a lower damage score (score: 2) than the CCL4 group (score: 5) (p < 0.05).
Biochemical results
Effects of INF on levels of SOD and MDA, a product of lipid peroxidation, are shown in Table 3. MDA levels were significantly higher (p > 0.05) in the CCL4 group compared to the control group. In addition, INF treatment reduced MDA levels compared to the CCL4 group. SOD levels in the control group were not significantly different from those in the CCL4 and CCL4 + INF groups (p > 0.05) (Table 3).
Biochemical analysis results.

CCL4: carbon tetrachloride; MDA: malondialdehyde; SOD: superoxide dismutase; INF: infliximab.
ap < 0.05 versus the CCL4 group.
bp < 0.05 versus the CCL4 + INF group.
cp < 0.05 versus the control group.
Discussion
CCL4 is an organic solvent with carcinogenic and toxic effects used for a range of industrial purposes. 1 Previous studies have reported that CCL4 causes fibrosis, necrosis, inflammation, vascular congestion, hemorrhage, intracellular clusters of fat vesicles, and degenerative nodules in tissues, especially the liver and kidneys. 9,23,34 In one of the few studies of the effects of CCL4 on intestinal damage, Cosgun et al. observed intestinal epithelial shedding, villus and Lieberkühn crypt losses, intense inflammation, and hemorrhage. 35 Similarly, we observed denuded villi with exposed lamina propria and loss of epithelial integrity. We also observed capillary congestion and neutrophil infiltration, together with the moderate loss of epithelial integrity and extension of the subepithelial space.
Although the mechanism by which CCL4 causes damage in tissues is unclear, studies have reported that it leads to oxidative stress by increasing free oxygen radical production. 7,9,20 Yousefi-Manesh et al. reported that CCL4 increased MDA and reduced SOD levels in hepatic tissue. 9 In contrast, Cosgun et al. reported that CCL4 increased both MDA and SOD levels in small intestine tissue. 35 Similarly, in the present study, we observed an increase in MDA levels. However, CCL4 toxicity resulted in no difference in intestinal SOD levels.
Previous studies have reported that 8-OHdG expression increases in oxidative stress DNA damage caused by ROS. 24,25,32,36,37 Deniz and coworkers reported that CCL4 increased 8-OHdG expression in their studies. 18 Similarly, in our study, we observed that CCL4 causes an increase in 8-OHdG positivity in enterocytes.
Li et al. investigated the effects of CCL4 on hepatic tissue and showed that it led to apoptosis in hepatocytes. 15 Although there have been no studies of the effects of CCL4 on caspase-3 expression in small intestinal enterocytes, Deniz et al. investigated the effects of CCL4 on hepatic tissue and reported that CCL4 caused caspase-3 expression in hepatocytes. 18 Similarly, we determined that CCL4 increased caspase-3 positivity in enterocytes.
Studies have also reported that in addition to oxidative stress, CCL4 also accompanies apoptosis in tissues. 15,18 Al-Sayed et al. reported that CCL4 increased MDA levels and caspase-3 expression. 38 Similarly, in the present study, we observed that CCL4 increased MDA levels and caspase-3 expression in enterocytes.
Bahcecioglu et al. reported that INF, an antibody against TNF-α, reduced inflammation and fibrosis. 22 Similarly, in the present study, we observed that INF reduced inflammation and fibrosis caused by CCL4. In addition, it reduced intestinal epithelium, villus and Lieberkühn crypt losses caused by CCL4. Pergel et al. investigated ischemia and reperfusion-related intestinal damage and reported that INF reduced intestinal MDA levels. 39 Similarly, we observed that INF reduced intestinal MDA level elevation caused by CCL4. Aydin et al. reported that INF exhibited no effect on increased caspase-3 expression in enterocytes associated with cisplatin toxicity. 30 In contrast, Kurt et al. reported that INF inhibited Methotrexate intoxication-related apoptosis in lung tissue by reducing caspase-3 expression. 21 Similarly, in our study, we determined that INF reduced caspase-3 positivity observed in enterocytes in association with CCL4 toxicity.
In conclusion, CCL4 appears to exert a highly deleterious effect on the intestinal mucosa through oxidative stress and apoptosis. However, INF is effective in preventing this CCL4-induced intestinal injury through decreased MDA levels and caspase-3 expression. We think that this study may be of assistance to further research into the effects of oxidative/antioxidative enzymes and inflammatory cytokines.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
