Abstract
Diabetes mellitus (DM) is a complex metabolic disease and it is also closely associated with a reduction in fertility in male patients. The purpose of the present study was to investigate the antidiabetic effect of carvacrol (CRV), as a potent antioxidant, on the numbers of germ cells and Sertoli cells in testicular tissue, and the messenger RNA (mRNA) and protein expression of some genes involved in spermatogenesis, including luteinizing hormone/choriogonadotropin receptor (LHCGR), follicle-stimulating hormone receptor (FSHR), and steroidogenic factor 1 (SF-1), as well as hormones such as luteinizing hormone (LH), follicle-stimulating hormone (FSH), testosterone (T), and insulin. Adult male Wistar rats (n = 32) were randomly divided into four groups (eight animals per group), including healthy control that received 0.2% Tween 80, diabetic control group, the diabetic group treated orally with CRV (75 mg/kg), and CRV group that received orally CRV (75 mg/kg). The duration of the treatment period lasted 8 weeks. In the diabetic group, the numbers of Sertoli cells and germ cells were significantly decreased, while the treatment with CRV prevented the degree of the damage to the cells mentioned earlier. CRV administration elevated the concentrations of insulin, T, FSH, and LH. Moreover, treatment with CRV significantly enhanced the levels of the mRNA and protein expression of SF-1, LHCGR, and FSHR. According to the obtained results, CRV administration could prevent the deleterious effects of DM on testicular germ cells, and it increases the levels of hormones and some essential genes, such as SF-1, LHCGR, and FSHR, involved in the process of spermatogenesis.
Introduction
Diabetes mellitus (DM) is a major endocrine and metabolic disorder in the world and has been considered as one of the most threatening health problems to current global health. 1 There are two types of DM, including DM type 1 (DM1), which results from severe insulin deficiency because of the loss of pancreatic β-cells by the immune system, and DM type 2, which stems from relatively reduced insulin secretion and insensitivity of the cells to circulating insulin. 1,2
One of the most common complications of diabetes reported in men is sexual dysfunction. 1 DM causes detrimental changes in the reproductive system and reduces the fertility rate in humans and animals by at least two different mechanisms, including oxidative stress and endocrine disorders. 3,4 It has been shown that diabetes can influence the male reproductive system such as alterations in spermatogenesis, testicular tissue structure, sperm quality, the level of testosterone (T), and the rate of gonadotropin secretion. 5 It has been proposed that the deleterious effects of diabetes on testicular function are likely due to the lack of insulin. 5
Insulin could regulate the hypothalamic–pituitary–gonadal (HPG) axis. 5,6 In a normally functioning HPG axis, gonadotropin-releasing hormone (GnRH) is secreted from the hypothalamus, leading to the stimulation of the pituitary gland to release some hormones, namely luteinizing hormone (LH) and follicle-stimulating hormone (FSH). 5,6 The lack of a proper insulin signaling in the brain reduces the HPG axis function, which, in turn, decreases the hormonal outputs (i.e., LH and FSH from the anterior lobe of the pituitary gland) which are necessary for the maintenance of the population of Leydig cells and Sertoli cells to ensure the process of spermatogenesis in the seminiferous tubules, 5 –7 but exact mechanisms underlying this phenomenon are not fully understood.
In recent years, much attention has been focused on herbal nutrition and compounds for the control of diabetes complications since synthetic agents have numerous adverse effects on human health in comparison with herbal medicine. 8,9
Carvacrol (CRV, 2-methyl-5-isopropylphenol) is a predominant monoterpene phenolic agent found in the essential oil of some species which belong to the family of Lamiaceae 10,11 and has a broad range of biological and pharmacological properties, including antioxidant, antidiabetic, antibacterial, antifungal, anticancer, antiapoptotic, anti-inflammatory, and hepatoprotective activities. 10,12,13 Shoorei et al. demonstrated that CRV administration could decrease the amount of reactive oxygen species (ROS) and the intensity of Bax immunoreactive staining and enhance the activity of antioxidant enzymes in the testicle of diabetic rats. 12 Emerging evidence indicates that the antioxidant potential of CRV is due to the presence of the hydroxyl radical (OH·), covalently attached to the aromatic ring. 10
This study was designed to evaluate the effect of CRV on testicular function as well as the messenger RNA (mRNA) and protein expression of steroidogenic factor 1 (SF-1), luteinizing hormone/choriogonadotropin receptor (LHCGR), and follicle-stimulating hormone receptor (FSHR) genes, which are important in hormonal balance and spermatogenesis process, in testicular tissues of type 1 diabetic male rats because there are no studies carried out in this area so far.
Materials and methods
Animals and experimental design
Adult male Wistar rats (n = 32, weighing 250–300 g) were used in this experimental study. The animals had free access to standard murine food and tap water and housed in a temperature-controlled room with a 12-hour light/12-hour dark cycle. The experimental procedures of the current research were in line with the Guidelines for the Care and Use of Laboratory Animals and approved in the Ethics Committee of the Tabriz University of Medical Sciences with an ethical code no: IR.TBZMED.VCR.REC1397.020.
Rats were randomly assigned to four groups as follows: G1 (healthy control group) in which rats daily received 0.2% Tween 80 (Sigma-Aldrich, Steinheim, Germany) dissolved in distilled water at the same volume as the treated groups for 8 weeks 14 ; G2 (diabetic control group) in which they were daily received normal saline at the same volume as the treated groups for 8 weeks 12 ; G3 (CRV-treated diabetic group) in which diabetic rats were daily treated with CRV (purity >97%; Sigma-Aldrich) at a dose of 75 mg/kg for 8 weeks 12 ; and G4 (CRV-treated control group) in which rats were daily treated with CRV at a dose of 75 mg/kg for 8 weeks. 12
DM1 was induced by the intraperitoneal injection of streptozotocin solution (0.01 M citrate buffer [pH = 4.5]) at a dose of 50 mg/kg. 15 After 3 days, the concentration of the blood glucose was measured in rats, and they were considered diabetic if the level of glucose was higher than 250 mg/dL. 15 The dosage of CRV was chosen based on our previous study 12 and the other study. 16 CRV treatment was started 10 days after the induction of diabetes model. 12
All animals, at the end of the experiment period, were anesthetized by the administration of ketamine/xylazine (10/1; Sigma Aldrich). 12 After anesthesia, for the measurement of hormonal concentrations, blood samples were taken from the hearts of animals and then centrifuged at 3000 r/min for 5 min. The isolated serum samples were subsequently frozen at −80°C until use. Moreover, for the histopathological and molecular assessments, the left testicles were fixed in Bouin’s solution (Sigma-Aldrich) for 48 h and the right testicles were frozen at −80°C until analysis. In the present study, all experiments were performed in triplicate.
Morphological analysis
Fixed testicular tissue samples dehydrated in an ascending graded series of ethanol, cleared in xylene, and embedded in paraffin. Samples sectioned at the thickness of 5 µm, and finally, the obtained slides stained with hematoxylin and eosin according to the protocol introduced in our previous work for counting testicular tissue cells. 12 Next, the numbers of Sertoli cells and germ cells were quantified. The number of Sertoli cells per tubule (only those showing typical morphological nuclear features and evident nucleolus) was enumerated in 30 tubules/animal under a light microscope at 400× magnification (Carl Zeiss, Oberkochen, Germany) based on the study performed by Caneguim et al. 17 Moreover, the frequency of spermatogonial cells, primary spermatocytes, and spermatid cells (round) was counted in 10 tubules/animal. 18
Measurement of insulin concentration
The concentration of insulin in serum samples isolated from rats was calculated by the enzyme-linked immunosorbent assay (ELISA) method specified for rat insulin (cat. no: 10-1250-01, sensitivity = 0.15 μg/L, assay range: 0.15–5.5 μg/L; Mercodia AB, Uppsala, Sweden) based on the absorbance at 450 nm according to the manufacturer’s instructions.
Assessment of LH, FSH, and T levels
The serum level of T was determined using an enzymatic immunoassay test (cat. no: DE1559, sensitivity = 0.08 ng/mL, assay range: 0.08–16 ng/mL; Demeditec Diagnostics, Kiel, Germany) based on the absorbance at 450 nm according to the manufacturer’s instructions. The intra-assay and interassay coefficients (coefficients of variability [%CV]) for T were found to be 5.4% and 7.4%, respectively. Moreover, the concentrations of serum FSH (cat. no: ZB101825R9648, sensitivity = 0.12 mIU/mL, assay range: 0.12–64 mIU/mL) and LH (cat. no: ZB10179SR9648, sensitivity = 0.05 mIU/mL, assay range: 0.05–40 mIU/mL) were quantified by the commercial ELISA kits (Zellbio GmbH, Ulm, Germany) based on the absorbance at 450 nm according to the manufacturer’s instructions.
Real-time polymerase chain reaction
The total RNA of testicles (right) was extracted utilizing the TRIzol reagent kit (Invitrogen, Paisley, UK), according to our previous work. 12 Next, the extracted RNA samples were treated with DNase I (Invitrogen Life Technologies, Carlsbad, CA, USA), to eliminate any trace of DNA contamination. 12 Afterward, the absorbance of the purified RNA contents was recorded at the optical density (OD) 260/280 ratio (1.8 < OD260/OD280 < 2.0), 19 eluted with DNase/RNase free water, and adjusted to a concentration of 0.7 μg/mL. After that, the complementary DNA (cDNA) was synthesized in a total volume of 20 μL by means of M-MuLV reverse transcriptase (Thermo Scientific First-Strand cDNA Synthesis Kit, Waltham, USA). Table 1 indicates the sequences of the designed primers that belong to each target gene. 12,18 The quantitative real-time polymerase chain reaction (qRT-PCR) was performed on an Applied Biosystems 7900HT thermocycler (Applied Biosystems, Inc., Cheshire, UK), as previously described. 12 The qRT-PCR reactions were carried out in a volume of 20 µL, containing 10 μL SYBR Green master mix (Sigma-Aldrich), 2 µL primers (1 µL for forward and 1 µL for reverse primer), 1 μL cDNA, and 7 μL DNase-/RNase-free water. The thermocycling conditions were similar to our previous studies. 12,20 Finally, the relative quantitative analysis of the target genes was calculated based on the 2–ΔΔCt method.
Table 1. Primer sequences of SF-1, LHCGR, FSHR, and GAPDH.

SF-1: steroidogenic factor-1; LHCGR: luteinizing hormone/choriogonadotropin receptor; FSHR: follicle-stimulating hormone receptor; GADPH: glyceraldehyde-3-phosphate dehydrogenase.
Western blot analysis
The right testicle of each animal was lysed and homogenized in radioimmunoprecipitation assay (RIPA) buffer lysis buffer (Sigma-Aldrich). Next, the protein concentrations were measured using the Bradford protein assay.
The extracted protein contents were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis and then transferred onto polyvinylidene difluoride membranes. Afterward, the membrane was blocked with 5% StartingBlock Blocking Buffer (Thermo Scientific, Pittsburgh, PA, USA) for 24 h. When the blocking step was terminated, the membrane was incubated with the primary antibodies against anti-SF-1 (1:1000; Santa Cruz Biotechnology, Inc., Santa Cruz, California, USA), LHCGR (diluted at 1:500; ProteinTech, USA), FSHR (1:1000; Abcam, Cambridge, UK), and GAPDH (1:25,000; Abcam) at 4°C 21 –23 for 24 h. The membrane was rinsed in phosphate-buffered saline (PBS)-Tween (005% Tween in PBS) three times (each time 5 min) and probed with horseradish peroxidase-conjugated antigoat or antirabbit secondary antibody (immunoglobulin G, diluted at 1:5000; Abcam) at room temperature for 1 h. 21 Finally, the protein bands were visualized using the enhanced chemiluminescence reagent kit (ECL-Plus; GE Healthcare), according to the manufacturer’s instructions.
Statistical analysis
Statistical significance was determined by analysis of variance followed by Tukey’s post hoc test using SPSS software (Version 18, SPSS Inc., Chicago, IL, USA). Value of p < 0.05 was considered as the level of significance.
Results
Histopathological evaluation
The histological assessment of the testicles in the diabetic control group showed the abnormal morphological structures, such as seminiferous tubule damage and atrophy, as well as irregularities and degenerations in germ cells, spermatocytes, spermatogonia, and spermatids (Figure 1(b)). The results showed that the administration of CRV markedly reduced the degree of degeneration in the seminiferous tubules of the CRV-treated diabetic group when compared with the diabetic control group (Figure 1(c)). As shown in Figure 1(a) and (d), testicular tissues obtained from the healthy control and CRV-treated control groups (G1 and G4) showed that the epithelial tissue of the seminiferous tubules was histologically normal.

Hematoxylin and eosin staining of testicular tissues. (a) The healthy control group, which daily received Tween 80 (75 mg/kg, orally) for 8 weeks, shows typical testicular architecture. (b) The diabetic group, which daily received normal saline (75 mg/kg, orally) for 8 weeks, shows severe testicular damage. (c) Carvacrol-treated diabetic group, which daily received carvacrol (75 mg/kg, orally) for 8 weeks, shows that the seminiferous tubules improved. (d) Healthy-treated control group, which daily received carvacrol (75 mg/kg, orally) for 8 weeks, shows standard testicular architecture. Yellow, red, black, and blue arrows show spermatogonia, primary spermatocytes, spherical spermatids, and Sertoli cells, respectively. Scale bar: ×400 (20 μm)
In this experimental study, the numbers of Sertoli cells and germ cells, including primary spermatocytes, spermatogonial cells, and round-shaped spermatids, are also mentioned in Table 2. According to Table 2, the number of Sertoli cells in the diabetic control group was remarkably decreased when compared with the healthy control group (p < 0.05). Also, a significant difference was also observed in the number of Sertoli cells between the diabetic control and CRV-treated diabetic groups (p < 0.05), while there was no significant difference in the number of Sertoli cells between the healthy control and CRV-treated control groups (p > 0.05). Our findings showed that the number of germ cells, including primary spermatocytes, spermatogonial cells, and round-shape spermatids, was substantially lower in the diabetic control group in comparison with the healthy control group (p < 0.05). In the diabetic group treated with CRV, the number of germ cells mentioned earlier was remarkably higher compared with the diabetic control group (p < 0.05). No significant difference was detected between the G1 and G4 groups when the number of germ cells was considered (p > 0.05).
The number of germ cells, spermatogonial cells, primary spermatocytes, and spermatid cells as well as the number of Sertoli cells in all groups of the study.

CRV: carvacrol. SD: standard deviation.
a A significant difference compared to the control group (p < 0.05).
b A significant difference compared to the diabetic control group (p < 0.05).
Hormonal evaluation
The results indicated that the serum concentration of insulin, on the last day of the experiment, was significantly diminished in the diabetic control group compared with the healthy control group (p < 0.02; Table 3). Moreover, the administration of CRV markedly increased the serum levels of insulin in the CRV-treated control group when compared with the diabetic control group (p < 0.03). No statistically significant difference was observed between the healthy control and CRV-treated control groups when insulin concentration was measured (p > 0.05).
The serum concentrations of hormones, including insulin, T, LH, and FSH.

CRV: carvacrol; T: testosterone; LH: luteinizing hormone; FSH: follicle-stimulating hormone; SD: standard deviation. aA significant difference compared to the control group (p < 0.02).
b A significant difference compared to the diabetic control group (p ≤ 0.03).
According to Table 3, the serum levels of T, LH, and FSH were considerably reduced in the diabetic control group compared with the healthy control group (p < 0.02). CRV administration significantly elevated the serum concentrations of mentioned hormones in the CRV-treated diabetic group in comparison with the diabetic control group (p ≤ 0.03). The statistical analysis showed no significant difference in the levels of the above hormones between the G1 and G4 groups (p > 0.05).
Evaluation of mRNA and protein expression of FSHR, LHCGR, and SF-1
In the current study, the qRT-PCR and Western blot analyses were carried out to evaluate the effect of diabetes and CRV on the expression of FSHR, LHCGR, and SF-1 at the gene and protein levels in testicular tissues of adult male rats.
According to Figure 2, the induction of diabetes in male rats significantly lowered the FSHR expression at both mRNA and protein levels in comparison with the healthy control group (p ≤ 0.001). In the CRV-treated diabetic group, the expression of FSHR was significantly upregulated compared with the diabetic control group (p < 0.003). There was no significant difference in the expression rate of FSHR between the healthy control and CRV-treated control groups (p > 0.05).
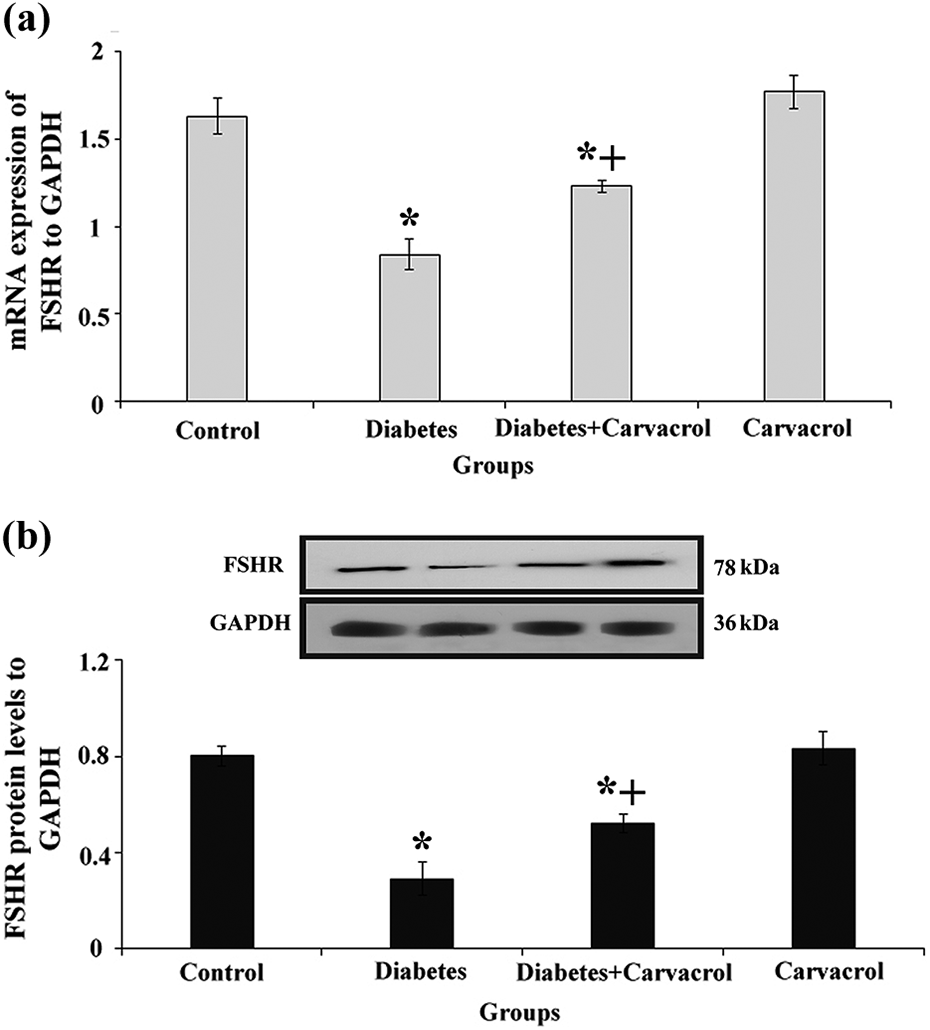
Relative levels of mRNA and protein expression of FSHR gene. (a) and (b) The mRNA and protein expression levels of FSHR which were normalized against the level of the internal control, endogenous glyceraldehyde-3-phosphate dehydrogenase (GAPDH), respectively, are shown. The asterisk “*” shows a significant difference compared to the control group (p ≤ 0.001). The symbol “+” shows a significant difference compared to the diabetic control group (p < 0.003). mRNA: messenger RNA; FSHR: follicle-stimulating hormone receptor.
As shown in Figure 3, in the diabetic control group, the mRNA and protein levels of LHCGR were significantly decreased when compared with the healthy control group (p ≤ 0.001). The results indicated that the treatment of rats with CRV significantly upregulated the expression of LHCGR in the CRV-treated diabetic group when compared with the diabetic control group (p < 0.002). As expected, no significant difference was found between the healthy control and CRV-treated control groups when the expression of LHCGR was analyzed (p > 0.05).

Relative levels of mRNA and protein expression of LHCGR gene. (a) and (b) The mRNA and protein expression levels of LHCGR which were normalized against the level of the internal control, endogenous GAPDH, respectively, are shown. The asterisk “*” shows a significant difference compared to the control group (p ≤ 0.001). The symbol “+” shows a significant difference compared to the diabetic control group (p < 0.002). mRNA: messenger RNA; LHCGR: luteinizing hormone/choriogonadotropin receptor.
Figure 4 shows the levels of mRNA and protein expression of SF-1. As displayed in Figure 4, there was a significant difference between diabetic control and healthy control groups (p < 0.001). Regarding the results obtained from testicular tissues of all experimental groups, the administration of CRV significantly upregulated the expression of SF-1 in the CRV-treated diabetic group compared with the diabetic control group (p < 0.002).

Relative levels of mRNA and protein expression of SF-1 gene. (a) and (b) The mRNA and protein expression levels of SF-1, which were normalized against the level of the internal control, endogenous GAPDH, respectively, are shown. The asterisk “*” shows a significant difference compared to the control group (p < 0.001). The symbol “+” shows a significant difference compared to the diabetic control group (p < 0.002). mRNA: messenger RNA; SF-1: steroidogenic factor 1.
No statistically significant difference was observed between the CRV-treated control and healthy control groups when the expression of SF-1 was assessed (p > 0.05).
Discussion
Nowadays, the number of individuals who suffer from DM is rapidly rising. It has been reported that DM is responsible for the causation of various complications within the human/animal body, such as the overproduction of ROS. 4 Also, it has detrimental effects on male fertility, especially on spermatogenesis, by causing damages to the male germ cells or affecting the HPG axis. 4 Antioxidant therapy could regulate a fine-tuned balance between prooxidant and antioxidant agents and therefore decrease oxidative damages. CRV, as a powerful antioxidant molecule, possesses several biological and pharmacological properties and also reduces oxidative damage in testicular tissues of animals induced by DM. 11,12 However, the precise mechanism underlying how CRV influences different hormones and a group of genes involved in the spermatogenesis process remained elusive.
Insulin massively secretes from pancreatic β-cells and partially from Sertoli cells. 24 In animal models of DM1, streptozotocin enters the pancreatic β-cells through the glucose transporter 2 and increases the generation of free radical agents and DNA fragmentation, leading to the induction of cell death in β-cells. 12 Moreover, overproduction of ROS caused by DM1 activates apoptotic signaling pathways within Sertoli cells that finally affect the survival of these cells. 25,26 Hence, the source of insulin production would be lost, and the levels of insulin would be decreased.
One study reported that CRV was capable of ameliorating the rate of injury in pancreatic β-cells, induced by H2O2. 27 Therefore, CRV administration possibly through lowering the amount of ROS could mitigate the degree of damage to the pancreatic β-cells and could finally prevent the reduction of the serum insulin concentration (Figure 5). However, we strongly suggest that further studies should be performed to exactly show whether the effect of CRV in this regard is due to a rescue in pancreatic β-cells or a suppression of oxidative agents.

The oxidative stress induced by type 1 diabetes mellitus destroys pancreatic β-cells; consequently, the level of insulin secretion from the pancreas is reduced. Insulin plays a key role in the secretion of gonadotropin hormones (LH and FSH) from the anterior lobe of hypophysis by binding to its receptor located on the hypothalamus. Therefore, when insulin secretion is reduced, gonadotropin levels of hormones also decrease. On the other hand, endogenous and exogenous generated ROS activate apoptosis-related proteins, leading to damage to the germ cells, Leydig cells, and Sertoli cells. Moreover, diabetes mellitus type 1 (DMT1) possibly leads to degeneration in FSHR and LHCGR because both mRNA and protein expression levels of them decrease. Therefore, after decreasing the levels of FSH and FSHR, germ cell apoptosis increases because it had been reported that FSH can confine the massive wave of germ cell apoptosis in the early stages of spermatogenesis. On the other hand, after decreasing the levels of LH, LHCGR, and SF-1, the secretion of T from Leydig cells decreases, too. Hence, positive feedback is formed in the HPG axis that partially enhances the secretion of FSH and LH (two short white arrows). Furthermore, by reducing the expressions of FSHR and SF-1, negative feedback is formed to prevent the production of gonadotropin hormones (two big red arrows). Treatment with CRV, possibly through lowering the amount of ROS, could mitigate the degree of damage to the pancreatic β-cells and could finally prevent the reduction of the concentrations of serum insulin. Moreover, the administration of CRV could also enhance the hormonal concentrations—T, LH, and FSH—and both mRNA and protein expression of FSHR, LHCGR, as well as SF-1. CRV: carvacrol; T: testosterone; LH: luteinizing hormone; FSH: follicle-stimulating hormone; SF-1: steroidogenic factor-1; LHCGR: luteinizing hormone/choriogonadotropin receptor; FSHR: follicle-stimulating hormone receptor; ROS: reactive oxygen species; mRNA: messenger RNA.
The histological evaluations revealed that diabetes caused degeneration in testicular tissue and also decreased the numbers of germ cells and Sertoli cells, while treatment with CRV preserved the reservoir of germ cells/Sertoli cells and attenuated the rate of testicular degeneration. DM1 by activating apoptotic pathways within the germ cells ultimately leads to testicular degeneration. 28 –30 One study reported that CRV can downregulate cisplatin-induced reproductive injuries and decrease the rate of degeneration in seminiferous tubules that may be due to the antioxidant nature of this molecule. 16
In the present study, CRV administration enhances the expression levels of SF-1, LHCGR, and FSHR genes and enhances the levels of hormones (i.e., T, LH, and FSH) in the diabetic rats.
SF-1, known as an adrenal-4-binding protein, is expressed in pituitary gonadotropes, in the ventromedial part of the hypothalamus, and in Leydig cells (responsible for the secretion of T hormone) in male gender. 31 SF-1 expression enhances the steroidogenic activities through cytochrome P450 components and regulates the function of the hypothalamic–pituitary–steroidogenic organ axis. 31,32 Therefore, the reduction in the levels of T may be owing to a decrease in the expression of the SF-1 gene (Figure 5); yet, the exact mechanism involved in this scenario has not been elucidated and it should be further investigated. Interestingly, one study showed that the concentration of T was increased in male rats treated with essential oil of Satureja khuzestanica (225 mg/kg, for 45 days), which contains a high amount of p-cymene and CRV. 33
On the other hand, LH by binding to its receptor (i.e., LHCGR which belongs to a subgroup of class A [rhodopsin-like] G-protein-coupled superfamily and expresses on Leydig cells) is capable of triggering these cells to produce androgens, including T, which is required for spermatogenesis. 30 LHCGRs can also activate the signaling pathways, such as protein kinase B or the extracellular signal-regulated kinase 1/2 cascade, which are necessary for survival/proliferation of Leydig cells as well as androgen synthesis, such as T. 28,30 We suggest that the decrease in the level of serum T in diabetic rats may also be due to the pathways activated by the stimulation of LHCGR; therefore, to illuminate how the signaling mechanisms work, mentioned pathways must be further examined in in vitro and in vivo situations.
Moreover, in our study, although the number of Leydig cells did not count, the number of Sertoli cells significantly decreased in the diabetic group. Our results are in accordance with the study that reported the numbers of Leydig cells and Sertoli cells are decreased in the diabetic condition because of the activation of apoptosis through the Fas/Fas Ligand (FasL) signaling pathway. 25 CRV could decrease the rate of apoptosis in mentioned cells possibly through its antiapoptotic property and could improve T concentrations and spermatogenesis process.
Under the pathological conditions of DM1, as a result of insulin insufficiency, the hypothalamic–pituitary axis function is affected, leading to a decrease in LH and FSH secretions 34 ; however, the exact mechanisms underlying this phenomenon are not fully understood. In our study, a reduction in the serum levels of LH and FSH may rely on SF-1 expression, as some pieces of evidence showed that SF-1 is an upstream regulator of a group of genes that encode the GnRH receptor and the b-subunits of LH and FSH hormones. 35,36 Another study reported that the levels of LH-β subunit, LH immunoreactivity, and LH concentration are remarkably reduced in SF-1 knockout mice. 35 Reduction of LH could indirectly influence the production of T in testicles because the biosynthesis of T is merely related to the stimulation of Leydig cells mediated by LH functions on these types of cells. 37 However, it seems that a decrease in LH concentration in the serum samples of diabetic rats may stem from a reduction in SF-1 expression in pituitary gonadotropes, which are precursor cells producing gonadotropins that regulate the reproductive function. We suggest the expression of SF-1 gene must be further studied in pituitary gonadotropes of diabetic rats to elucidate how its expression could affect the HPG axis function. However, in this study, CRV enhanced the decreased concentration of LH possibly through reducing the rate of damages in pancreatic β-cells, resulting in the elevation of insulin concentration, then upon an improvement in insulin levels, it would be capable of binding to its receptor expressing on the hypothalamus, and therefore, it can modulate the HPG axis function to ensure the regular synthesis of LH or FSH.
FSH, as one of the most significant pituitary glycoprotein, by binding to its receptor (i.e., FSHR which is expressed on Sertoli cells) along with T, has a regulatory role in spermatogenesis and spermiogenesis. 38 The early stages of spermatogenesis until the development of round-shaped spermatids are influenced by FSH and it can confine the massive wave of germ cell apoptosis in these stages, while the effect of T could be enhanced thereafter. 38 Moreover, FSH is necessary for germ cell survival and development by affecting Sertoli cells at the transcriptional and metabolic levels providing the hormonal and nutritional environment. 39,40 FSHR-knockout mice have a lower number of germ cells in comparison with normal mice. 41 Therefore, a decrease in the number of germ cells (primary spermatocytes, spermatogonial cells, and round-shaped spermatids) in our study may pertain to the insufficient amount of FSH or to the reduced FSHR expression. On the other hand, previous research indicated that diabetes can cause injuries to Sertoli cells through increasing the rate of oxidative stress, and it also triggers apoptosis in these cells, leading to a decrease in the number of FSHR on Sertoli cells. We propose that upon a reduction in the expression of FSHR at both mRNA and protein levels, negative feedback could be activated in the HPG axis to decrease the synthesis of FSH (Figure 5).
Notably, it has been implicated that both SF-1 and FSHR are the integral components of the HPG axis. 42 It has been reported that SF-1 partially expresses in Sertoli cells and stimulates the FSHR transcription through binding to several low-affinity binding sites within the upstream region of the FSHR promoter, and it interacts with the upstream stimulatory factor proteins which are bound to the E box. 43 SF-1 can directly influence the production of FSH in the pituitary, and it is capable of regulating the FSHR function and activating the protein kinase A, which is the major downstream regulator of the FSH signaling pathway in the gonad. 31,41 In a normal condition, the elevated levels of FSH in the presence of SF-1 dramatically stimulate the synthesis of the inhibin protein, generated by gonads. 41 This would then send negative feedback signals to the pituitary to decrease the synthesis of FSH for the maintenance of the hormonal balance 5 (Figure 5). Therefore, in our study, it seems that reduced expression of SF-1 can influence the regulation of the FSHR, subsequently, leads to a reduction in the level of FSH.
Conclusion
In summary, the results of the present study showed that the serum levels of LH, FSH, T, and insulin, the expression levels of SF-1, LHCGR, and FSHR genes, and the numbers of testicular germ cells and Sertoli cells were decreased in diabetic rats, while the treatment with CRV attenuated the deleterious effects of DM on the above parameters. After decreasing the serum level of T, a positive feedback loop leads to the activation of the hypothalamus to the secretion of GnRH, which subsequently enhances the levels of LH and FSH partially (Figure 5). Probably, because of degeneration of Leydig/Sertoli cells, caused by ROS, and also possibly by damaging their receptors (i.e., LHCGR and FSHR, respectively) in the diabetic condition, elevated LH and FSH lead to the formation of a negative feedback loop, which deactivates hypothalamus to the secretion of GnRH, which finally decreases the serum levels of FHS and LH (Figure 5). Therefore, DM can influence the HPG axis function to deliver the negative feedback signal to the brain to suppress the biosynthesis of gonadotropic hormones.
Study limitations and suggestions
Due to the lack of sufficient budget, we were not able to use antibodies or specific staining to indicate the cell types and to evaluate the molecular signaling pathways involved in the pathogenesis of diabetes and their impacts on the reproductive system. The SF-1 expression must be further examined in the brain (in the pituitary and hypothalamus glands) of diabetic rats to shed light on our understanding of this gene in the HPG axis function. On the other hand, to investigate the steroidogenesis in the diabetic condition under CRV treatment, in vitro studies using respective cell types should be considered in the future to support underlying mechanisms.
Footnotes
Acknowledgments
Author contributions
AAK, AK, and MM participated in study design and statistical analysis. HS and MS participated in data collection and evaluation. HS, S-HA-E, and AAH conducted molecular, PR-qPCR, Western blot, and hormonal analyses.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received a grant (No. 59341) for a PhD thesis of Mr. Hamed Shoorei carried out in Women’s Reproductive Health Research Center, Tabriz University of Medical Sciences, Tabriz, Iran.
