Abstract
Cancer continues to be a major cause of mortality globally. Zebrafish present suitable models for studying the mechanisms of genotoxic carcinogens. The aim of this study was to investigate the interaction between oxidant–antioxidant status, apoptosis and immunity in zebrafish that were exposed to three different genotoxic carcinogens methylnitrosourea, dimethylbenzanthracene, benzoapyrene and methylnitrosourea + dimethylbenzanthracene starting from early embryogenesis for 30 days. Lipid peroxidation, nitric oxide levels, superoxide dismutase and glutathione-S-transferase activities and mRNA levels of apoptosis genes p53, bax, casp3a, casp2 and immunity genes fas, tnfα and ifnγ1 were evaluated. The disruption of the oxidant–antioxidant balance accompanied by altered expressions of apoptotic and immunity related genes were observed in different levels according to the carcinogen applied. Noteworthy, ifnγ expressions decreased in all carcinogen-exposed groups. Our results will provide basic data for further carcinogenesis research in zebrafish models.
Introduction
Carcinogenesis is a multi-stage phenomenon which consists of initiation, promotion and progression stages where genetic and epigenetic pathways lead to the hyperproliferation and malignant conversion of healthy cells. 1 Treatment of cancer – especially at the metastatic stage – is very difficult and sometimes impossible in several circumstances. Hence, understanding pathways of carcinogenesis would provide precious data for future prevention and treatment of this common and deadly disease. In this study, we aimed to investigate the interaction between oxidant–antioxidant status, apoptosis and immunity in zebrafish that were exposed to three different genotoxic carcinogens starting from early embryogenesis for 30 days. As genotoxic carcinogens, we selected 7,12-dimethylbenz[a]anthracene (DMBA), N-methyl-N-nitrosourea (MNU) and benzo(a)pyrene (BaP) because they are prominent carcinogens existing in the environment. 2 DMBA is a policyclic aromatic hydrocarbon (PAH) and MNU is an alkylating agent which is also endogenously produced from creatinine metabolism and is mutagenic, genotoxic and teratogenic. 2,3 One of the main differences between the DMBA and MNU is in their activation. DMBA needs to be metabolized by P450 enzymes (cytochrome P450 A1 and cytochrome P450 B1) to become genotoxic, while MNU do not require metabolic activation. 1,4 Hence, the actions of DMBA are generally delayed in comparison to the biological actions of MNU. 2 Both MNU and DMBA convert the parent molecule into an active carbonium cation and subsequently alkylate the macromolecules including DNA. 2 MNU acts as a tumour initiator via transferring methyl groups to DNA, whereas DMBA is involved both in the initiation and in the promotion of tumourigenesis. 4 The chemical carcinogens DMBA and MNU were determined in the 1960s and 1970s, respectively, to induce rat breast cancer and became the most frequently employed breast carcinogens. 5 Rat mammary carcinomas display many features similar to human breast carcinoma, including histological progression and ovarian hormone dependence. 5,6 BaP is a toxicant in cigarette smoke that exerts genotoxicity and carcinogenicity and recognized as a human carcinogen by the International Agency for Research on Cancer. 7,8 In this study, due to their physiological, biochemical and genetical resemblances to human, we employed a zebrafish model to evaluate the effects of three genotoxic carcinogens DMBA, MNU and BaP and measured levels of lipid peroxidation (LPO), nitric oxide (NO) and activities of superoxide dismutase (SOD) and glutathione-S-transferase (GST). Using quantitative reverse transcription polymerase chain reaction (RT-PCR), we also determined mRNA levels of apoptosis genes including p53, bax, casp3a and casp2. Lastly, we determined mRNA levels of immunity genes including fas, tnfα and ifnγ1.
Materials and methods
Animals and treatment
Wild-type male and female AB/AB strain zebrafish were housed in ZebTEC (Tecniplast, Italy) aquarium rack system at 27 ± 1°C under a 14/10 h light/dark cycle. The experimental procedures were approved by the Institutional Animal Care and Use Committee of Marmara University. Zebrafish embryos were randomly divided into five groups as control, MNU, DMBA, BaP and MNU + DMBA, and each group consisted of 200 embryos.
The control group was exposed to a solution of 0.01% DMSO. The MNU group was exposed to MNU (20.6 mg/L MNU dissolved in 0.01% DMSO, Sigma, St Louis, USA) for 1 h. The DMBA group was exposed to DMBA (0.5 mg/L DMBA dissolved in 0.01% DMSO) for 1 day and the BaP group was exposed to BaP (10 mg/L BaP dissolved in 0.01% DMSO) for 1 week. The MNU + DMBA group was exposed first to DMBA and then to MNU similar to their respective groups. MNU, DMBA and BaP dosages were selected based on the previous studies 9 –11 and based on our preliminary studies.
At the end of 30 days fish were anaesthetized and were prepared as replicate pools for biochemical and RT-PCR analyses. The supernatant was used for the determination of biochemical parameters.
Biochemical analyses
Total protein level was measured according to the method of Lowry to express the results of the parameters per protein. 12 The method of Yagi was used to measure malondialdehyde (MDA) level which is the end product of LPO, as thiobarbituric acid reactive substances. 13 NO levels were measured according to the method of Miranda et al. 14 SOD activities were determined based on the ability of SOD to increase the effect of riboflavin-sensitized photo-oxidation of o-dianisidine and the results were expressed in U/mg protein. 15 GST catalyzes the conjugation of GSH and the activity of GST is measured by spectrophotometer at 340 nm. 16
RT-PCR methodology and utilized primers
RNA was isolated from zebrafish using Rneasy Mini Kit and Qiacube (Qiagen, Hilden, Germany). RT2 Profiler PCR Arrays (Qiagen) was used to synthesize single-stranded cDNA from 1 μg of total RNA. DNA Master SYBR Green kit (Qiagen) was used to perform PCRs. The expression of ifnγ1; tnfα; fas; casp3a; casp2 and p53 were evaluated by quantitative RT-PCR using the Qiagen Rotor Gene-Q Light Cycler instrument. Primers used are given in Table 1. β-actin was used as the housekeeping gene. Relative transcript levels were calculated using the delta-delta Ct method by normalizing the values to the housekeeping gene. 17
Forward and reverse primers used for the RT-RCR analyzes.

Statistical analysis
Statistical analysis was performed by using GraphPad Prism 5.0 (GraphPad Software, San Diego, California, USA). The data obtained were given as the mean ± standard deviation. To compare the groups, Kruskal–Wallis test was used which was followed by Dunn’s multiple comparison tests. The p values of <0.05 was regarded as significant.
Results
Representative images of the zebrafish embryos of the control, BaP, DMBA, MNU and MNU + DMBA groups at 7 dpf are given in Figure 1. Pericardial oedema and yolk sac oedema were observed in the BaP and DMBA exposed embryos respectively, whereas axial malformation was observed both in MNU and MNU + DMBA groups. The highest levels of LPO and NO and the highest activations of SOD and GST are observed in zebrafish exposed to MNU with significant difference from the control group (Figures 2 and 3). bax expressions are slightly yet significantly lowered in MNU-treated zebrafish in comparison to controls (Figure 4). p53 expressions are slightly and insignificantly lowered in the MNU exposed group (Figure 4). casp-3a expressions are significantly higher in MNU-treated groups in comparison to controls, yet lower than the DMBA and BaP groups (significant in comparison to BaP). Strikingly, highest levels of casp-2 are observed in MNU-treated groups. fas expressions were prominently high in the MNU-exposed group (Figure 5). tnfα expressions were also increased in MNU-exposed groups in comparison to controls, yet at lower levels than groups exposed to other carcinogens. Lastly, ifnγ1 expressions are significantly decreased in MNU-exposed groups similar to the features observed with other carcinogens (Figure 6).

Representative images of the zebrafish embryos of the (a) control, (b) BaP, (c) DMBA, (d) MNU, and (e) MNU + DMBA groups at 7 dpf. Pericardial oedema and yolk sac oedema were observed in the BaP and DMBA exposed embryos, respectively, axial malformation was observed both in MNU and MNU + DMBA groups. Morphology of the embryos were monitored and documented under a stereomicroscope (Zeiss Discovery V8, Germany) Scale bar: 200 μm. BaP: benzo(a)pyrene; DMBA: 7,12-dimethylbenz[a]anthracene; MNU: N-methyl-N-nitrosourea.

(a and b) Results for oxidant parameters of the control, MNU, DMBA, BaP and MNU + DMBA groups. Replicate pools of 30 dpf zebrafish (n = 5, 100 individuals per pool) were used. Values are given as mean ± standard deviation. *p < 0.05 significantly different from the control group a p < 0.05 significantly different from the MNU group, b p < 0.05 significantly different from the DMBA group and c p < 0.05 significantly different from the BAP group. LPO: lipid peroxidation; NO: nitric oxide; BaP: benzo(a)pyrene; DMBA: 7,12-dimethylbenz[a]anthracene; MNU: N-methyl-N-nitrosourea.
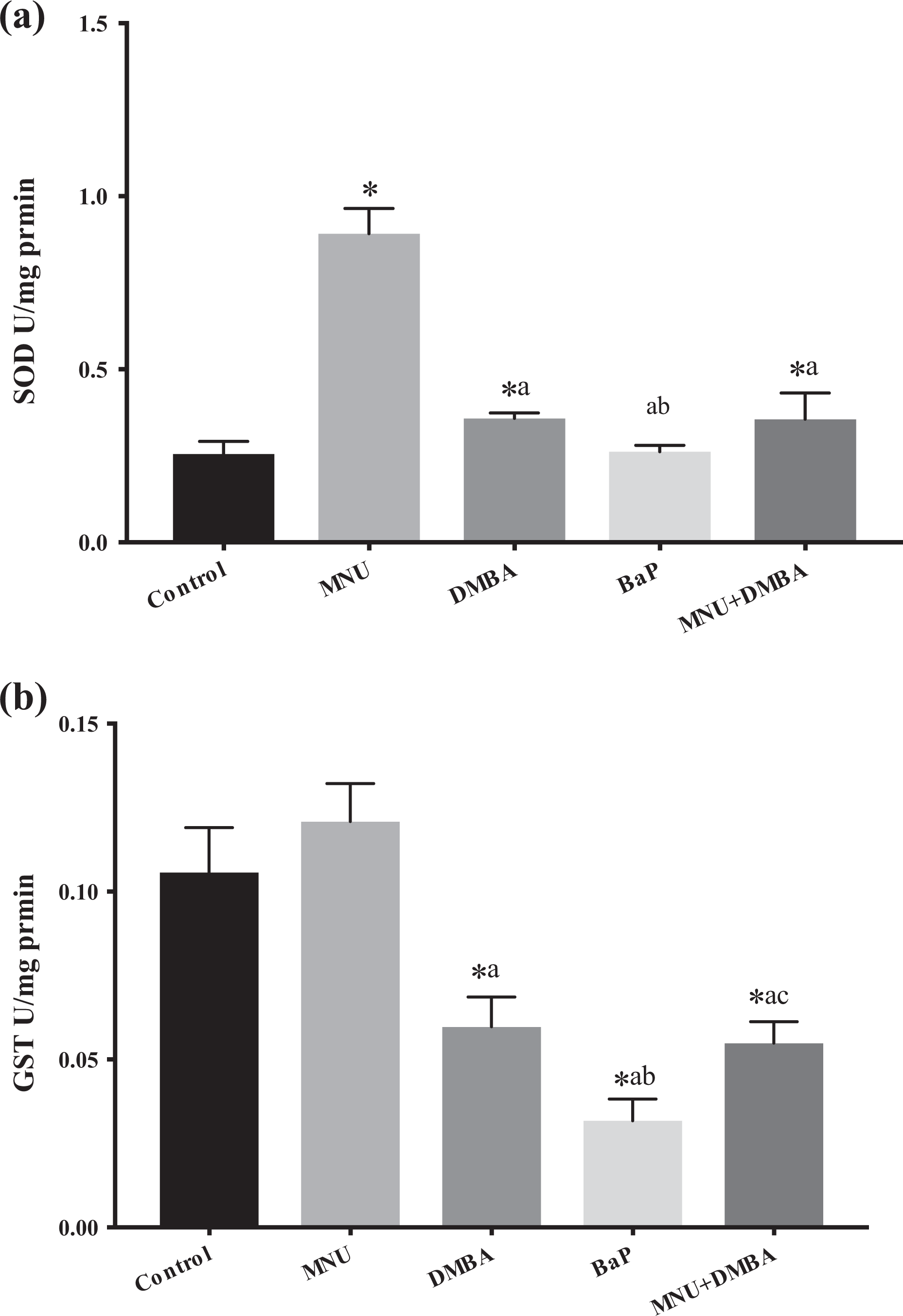
(a and b) Results for antioxidant parameters of the control, MNU, DMBA, BaP and MNU + DMBA groups. Replicate pools of 30 dpf zebrafish (n = 5, 100 individuals per pool) were used. Values are given as mean ± standard deviation. a p < 0.05 significantly different from the MNU group, b p < 0.05 significantly different from the DMBA group and c p < 0.05 significantly different from the BaP group. SOD: superoxide dismutase; GST: glutathione S-transferase; BaP: benzo(a)pyrene; DMBA: 7,12-dimethylbenz[a]anthracene; MNU: N-methyl-N-nitrosourea.

Bar graph presentation of fold change of (a) bax and (b) p53 transcript quantified by RT-PCR. All RT-PCR results are normalized to β-actin, the housekeeping gene and expressed as change from their respective controls. The average values were obtained from three experiments. Data presented are mean ± standard deviation. Significant difference is indicated by asterisk, *p < 0.05 significantly different from the control group. a p < 0.05 significantly different from the MNU group and b p < 0.05 significantly different from the DMBA group. RT-PCR: reverse transcription polymerase chain reaction; DMBA: 7,12-dimethylbenz[a]anthracene; MNU: N-methyl-N-nitrosourea.

Bar graph presentation of fold change of (a) casp3a and (b) casp2 transcript quantified by RT-PCR. All RT-PCR results are normalized to β-actin, the housekeeping gene and expressed as change from their respective controls. The average values were obtained from three experiments. Data presented are mean ± standard deviation. Significant difference is indicated by asterisk, *p < 0.05 significantly different from the control group. a p < 0.05 significantly different from the MNU group, b p < 0.05 significantly different from the DMBA group and c p < 0.05 significantly different from the BaP group. RT-PCR: reverse transcription polymerase chain reaction; BaP: benzo(a)pyrene; DMBA: 7,12-dimethylbenz[a]anthracene; MNU: N-methyl-N-nitrosourea.
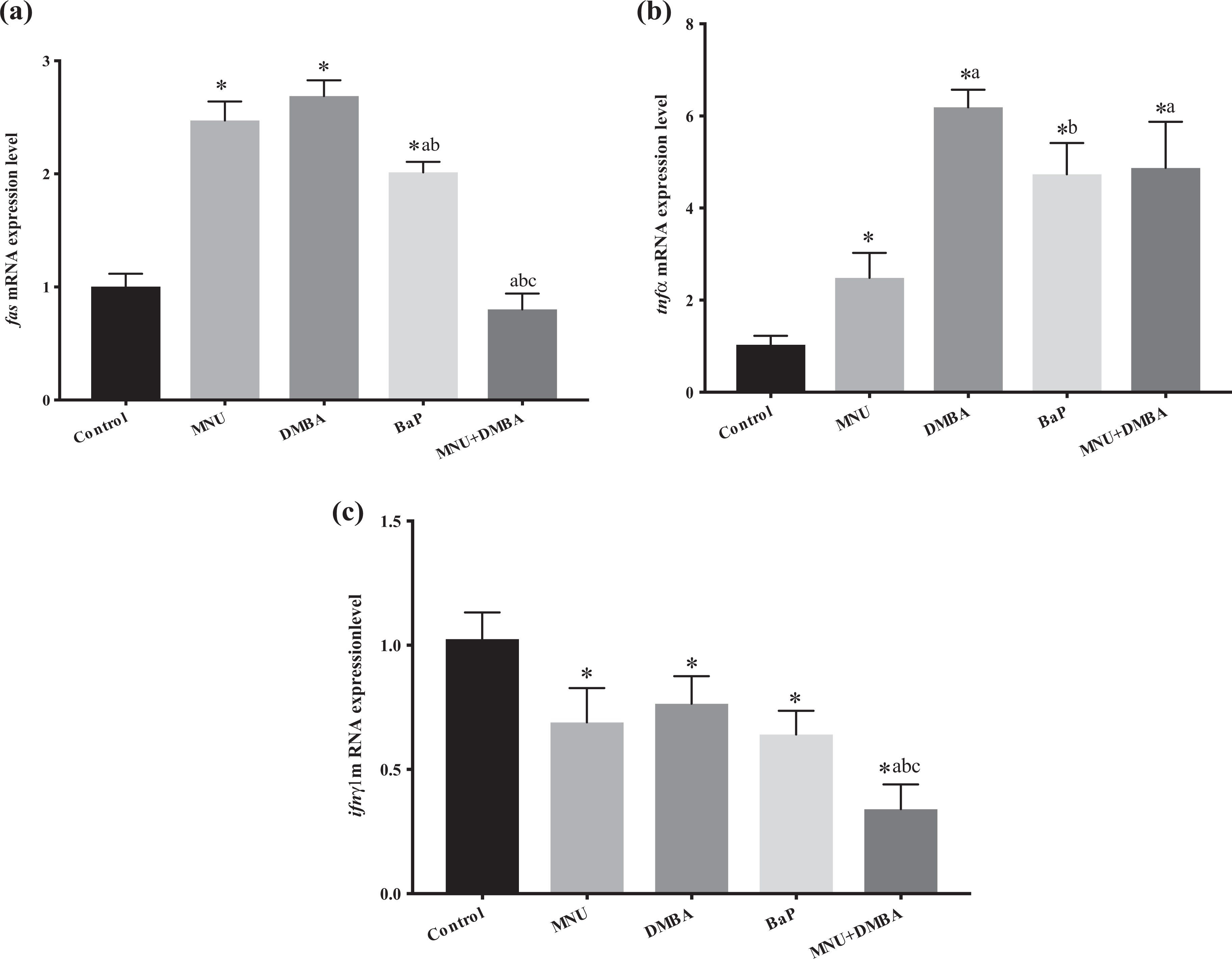
Bar graph presentation of fold change of (a) fas, (b) tnfα and (c) ifnγ1 transcript quantified by RT-PCR. All RT-PCR results are normalized to β-actin, the housekeeping gene and expressed as change from their respective controls. The average values were obtained from three experiments. Data presented are mean ± standard deviation. Significant difference is indicated by asterisk, *p < 0.05 significantly different from the control group. a p < 0.05 significantly different from the MNU group, b p < 0.05 significantly different from the DMBA group and c p < 0.05 significantly different from the BaP group. RT-PCR: reverse transcription polymerase chain reaction; BaP: benzo(a)pyrene; DMBA: 7,12-dimethylbenz[a]anthracene.
LPO and NO are enhanced in zebrafish exposed to DMBA with significant difference from the control group (Figure 2). SOD activities were slightly yet significantly higher in the DMBA-treated group. Peculiarly, GST activities are lowered in zebrafish exposed to DMBA (Figure 3). bax expressions slightly decreased and p53 expressions slightly increased in comparison to controls but both changes are significant (Figure 4). casp-3a and casp-2 expressions are increased in the DMBA-treated group in comparison to controls (Figure 5). Noteworthy, highest expressions of fas and tnfα are observed in the DMBA-treated group suggesting strong activation of extrinsic apoptotic cascades. Similar to other carcinogen-exposed groups, ifnγ1 expressions are lowered in comparison to controls (Figure 6).
In the BaP group, increased LPO was observed but this increase was significantly lower than MNU and DMBA-treated groups (Figure 2). NO production increased in the BaP group but this increase was significantly lower than MNU- and DMBA-treated groups (Figure 2). Peculiarly, SOD activity was lowest in the BaP treated group, significantly lower than MNU- and DMBA-treated groups; also noteworthy, GST activities were lowest in zebrafish exposed to BaP significantly lower than control and MNU groups (Figure 3). bax expressions are dramatically increased in group exposed to BaP at significantly higher levels in comparison to controls, MNU- and DMBA-treated groups. p53 expressions were also prominently high, significantly higher than controls and MNU-treated group. casp-3a expressions are highest among all groups, significantly higher than controls, MNU- and DMBA-treated groups (Figure 4). casp-2 expressions are also increased in comparison to controls yet this increase was slightly (insignificantly) lower than MNU- and DMBA-treated groups (Figure 5). fas expressions significantly increased in comparison to controls, yet these levels were significantly lower than MNU- and DMBA-treated groups (Figure 6). tnfα expressions increased which were significantly higher than control and MNU-exposed groups. Similar to other carcinogen-exposed groups, ifnγ1 expressions are lowered in comparison to controls (Figure 6).
Very peculiarly, LPO and NO production are lowest in this group. LPO levels are significantly lower in comparison to controls, MNU-, DMBA- and BaP-treated groups. NO production is significantly lower in comparison to controls, MNU-, DMBA- and BaP-treated groups (Figure 2). SOD activities are slightly yet significantly higher than controls, yet significantly lower than MNU-treated group. In zebrafish exposed to MNU + DMBA, GST activities were significantly lower than control, MNU- and BaP-treated groups (Figure 3). bax expressions were significantly lower than controls and DMBA-exposed groups, while p53 expressions were dramatically high; significantly higher than controls, MNU- and DMBA-exposed groups (Figure 4). Peculiarly, casp3a expressions were lowest in the MNU + DMBA treated groups, significantly lower than the control, MNU- and BaP-treated groups. casp-2 expressions were slightly but significantly higher in MNU + DMBA treated groups, yet these levels are significantly lower than MNU- and DMBA-exposed groups (Figure 5). fas expressions were slightly and insignificantly lower than controls (Figure 6). tnfα expressions increased which were significantly higher than control and BaP-exposed groups. Noteworthy, ifnγ1 expressions are lowest and significantly lower than controls, MNU-, DMBA- and BaP-exposed groups (Figure 6).
Discussion
LPO is the oxidative lipid degradation and end products of LPO may act mutagenic and carcinogenic.
18
–20
In our study, all carcinogens (MNU, DMBA and BaP) enhanced LPO, which was mostly pronounced in the MNU group. Peculiarly, LPO levels were lowest in the MNU and DMBA co-treated group. NO levels increased in MNU and DMBA groups. NO is an endogenous free radical which is produced enzymatically from the terminal guanidino-nitrogen of
SOD is a necessary antioxidant defense in nearly all cells exposed to oxygen, it catalyzes the partitioning (dismutation) of the superoxide (O2 −) radical into either molecular oxygen (O2) or hydrogen peroxide (H2O2). 27 BaP has been shown to be toxic to rat neutrophils and BaP exposure induced SOD activity. 21 On the other hand, reduced SOD activity was reported in BaP exposed rat liver. 28 This difference may associate with the duration of exposure. At acute exposure, SOD activities may increase to reduce oxygen radical injury, while its levels may deplete at chronic exposure to oxidant carcinogens. In our study, MNU treatment induced highest SOD activity, which may suggest that the highest formation of superoxide radicals occurred with MNU exposure. In the MNU + DMBA group, decreased SOD activity was observed in comparison to the MNU group, either due to reduced oxidative injury or due to apoptotic elimination of cells with very high genotoxic damage leaving cells with lesser lipid peroxidating and nitrosative damage.
GSTs make xenobiotic molecules more water soluble for detoxification. 29 DMBA increased GST-π activities in rat ovaries 30 and GST-α activities in rat mammary gland, 31 but decreased GST activity in plasma and mammary tissues once mammary tumors were established. 32 GST has been shown to be involved in the detoxification of BaP in fish 33 and GST activity increased in hippocampus of BaP-exposed rats. 34 In our study, DMBA, BaP and MNU + DMBA prominently reduced GST activity in zebrafish, while MNU alone slightly increased GST activity. These findings may reflect that strong carcinogens easily deplete GST in zebrafish.
P53 acts as ‘a genomic gatekeeper’ to maintain the integrity of the genome by arresting cell cycle and triggering DNA repair following a DNA-damaging insult. Therefore, P53 expression is a DNA damage biomarker. 35 –38 DMBA exposure in mice stimulated the expression of both p53 and p21WAF1 indicating a DNA injury and in p53-deficient mice, DMBA-induced skin papillomas exerted malignant conversion. 36 In DMBA-induced hamster buccal-pouch carcinogenesis, p53 and iNOS genes were activated in the buccal mucosa. 37 The important role of p53 against genomic damage, enhanced genotoxicity of MNU in p53-semideficient state and upregulation of p53 with the progression towards malignancy have been shown in DMBA-induced breast cancer in rats. 38,39 In another study preventive role of green tea extracts has been shown in BaP-induced lung carcinoma in rats, in association with stimulation of p53 levels. 40 Zebrafish also harbours the genes of the p53-mediated damage recognition pathway. 41 In our study, BaP strongly induced p53 expressions, indicating the highest DNA damage caused by BaP. In combined exposure to MNU and DMBA, p53 expressions were even higher. We conclude that BaP and MNU + DMBA combination induced robust p53 dependent apoptosis which left cells behind carrying lower lipid peroxidative and nitrosative stress.
Apoptosis has versatile functions including eradication of cells with genomic damage to prevent carcinogenesis. 42 –45 In our study, MNU slightly depleted bax expressions, which may contribute to its procarcinogenic effect. The most dramatical bax increase was in the BaP group, suggesting that the organismal response against BaP-genotoxicity may associate with induction of mitochondrial intrinsic apoptosis pathways. This robust apoptotic cell elimination may explain relatively lower lipid peroxidative and nitrosative injuries in comparison to DMBA and MNU in surviving cells. In our study, all carcinogens significantly induced caspase 2, which was highest with MNU treatment. Interestingly, adding DMBA to MNU reduced casp2 expressions, which may be a pseudo-effect due to higher apoptotic elimination of cells with greater genomic damage. As an executioner caspase, caspase 3 is activated both by extrinsic (death ligand) and intrinsic (mitochondrial) apoptotic pathways. 46,47 In our study, enhanced casp3a expression was most pronounced in the BaP-treated group in accordance with strong induction of p53 and bax suggesting that an intrinsic apoptotic pathway involving p53/bax/caspase 3 cascades are specifically activated against BaP-induced genotoxicity.
TNF regulates many physiological immune responses, pathological conditions and inflammation. 46 IFNγ is a dimerized soluble cytokine with antiviral, immune regulatory and anticancer properties. 47 –49 In mice, IFNγ prevented MNU-induced limb teratogenesis. In turn, carcinogens decreased levels of IFNγ. 3 In our study, all carcinogens increased tnfα expressions and reduced ifnγ1 expressions. Highest tnfα expression was observed with DMBA-exposure likely indicating an endeavour of the organism to eliminate protumourigenic cell clones harbouring genomic damage. Expression of death receptor fas increased in all groups except the MNU + DMBA group. These results suggest that genotoxic injury induces a profound inflammatory signal and diverse carcinogens share a common pathway of immunotoxicity, including suppression of ifnγ in zebrafish.
Pathways of repair or die are activated by exposure to carcinogens. Accordingly in our study, in the MNU-treated groups, very potent redox injuries were compensated with very efficient SOD and GST responses likely because that MNU is a physiologically produced toxic catabolite. TNFα seems to involve partially against MNU exposure. In the MNU-exposed group, major involvement of Fas-caspase axis was witnessed with highest activation of caspase 2 and independence of P53 and Bax pathways. Hence, it can be deduced that both preventive/repair and eradication/killing strategies are strongly active against MNU-carcinogenesis. In DMBA exposure, limited involvement of SOD and impaired response of GST revealed inefficient antioxidant responses. Caspase 3 and caspase 2 activation occurred with strongest increase of Fas and TNFα. It is possible that Fas and TNFα may trigger extrinsic apoptotic eradication of cells with DNA damage with mechanisms additional to caspases and seemingly independent of P53 and Bax pathways. The limited and lowered (GST) antioxidant responses may have increased Fas/TNFα/caspase axis or alternatively, increased Fas/TNFα/caspase activation may have eradicated cells with higher oxidative and nitrosative injury leaving tissues with lower injury. In BaP exposure, highly impaired SOD and GST responses were witnessed with very strong activation of Bax/p53 pathway and caspases (most pronounced in caspase 3). Against BaP-induced genotoxicity, intrinsic apoptotic pathway seems to be more pronounced, but Fas and TNFα may also have been activated to eliminate cells with DNA injury. The limited and lowered antioxidant responses may have increased Bax/p53/caspase axis to eradicate cells with higher oxidative and nitrosative injury. In combined exposure to both MNU and DMBA, these two different carcinogens either neutralized their lipid peroxidative and nitrosative stress or very dominant p53 pathway activation eradicated cells with high lipid peroxidative and nitrosative stress. Early studies indeed suggest that some carcinogens may antagonize their carcinogenic effects. For instance, carcinogenesis was prominently inhibited by the administration of carcinogenic ENU (ethyl nitrosourea, on the 13th day) prior to MNU treatment (on the 15th day). 50 On the other hand, the combined application of DMBA and MNU enhanced the breast carcinogenic effect in rats. 6 It is possible that cells with high genotoxic injury may be eliminated without progression into different stages of carcinogenesis. 51,52
Zebrafish constitute simple and easily manipulable models which could fascinatingly mimic complex molecular interactions observed in higher vertebrates. We think that they could provide important and crucial information in regard to cascades of oncogenesis and pave to develop molecules which could efficiently block carcinogenesis. However, some limitations should be noted in our study. Protein expression studies, along with histological studies, are required to support our results. We aim to improve our research in terms of these deficiencies and conduct studies to test whether plant-derived efficient anticancer agents are capable to reverse carcinogenesis at the initiation phase in zebrafish, which would provide novel important data.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Marmara University Scientific Research Projects Commission, project no. SAG-C-YLP-090518-0220.
