Abstract
Cytochrome P450 4B1 (CYP4B1) plays crucial roles in biotransforming of xenobiotics. Its predominant extrahepatic expression has been associated with certain tissue-specific toxicities. However, the expressions of CYP4B1 in various cancers and hence their potential roles in cancer development were inclusive. In this work, existing knowledge on expression and regulation of CYP4B1 gene and protein, catalysis of CYP4B1, association of CYP4B1 with cancers, contradicting findings about human CYP4B1 activities as well as the employing CYP4B1 in suicide gene approach for cancer treatment were reviewed. To date, it appears that there is a wide spectrum of tissue distribution of CYP4B1 with lungs as the predominant sites. Several nuclear receptors are possibly responsible for regulating its gene expression. The involvement of CYP4B1 in cancer was considered via activation of procarcinogens and neovascularization. However, human CYP4B1 was found to be inactive due to a substitution of proline with serine at position 427. Suicide gene approach combining reengineered CYP4B1 and prodrug 4-ipomeanol (4-IPO) has shown a promising potential for targeted cancer therapy. Further studies should focus on the verification of human CYP4B1 catalytic activities. More compounds with similar structure as 4-IPO should be tested to identify more alternative agents for the suicide gene approach in cancer treatment.
Introduction
Cytochrome P450 (CYP) is a superfamily of enzyme accountable for phase I drug metabolism and it is involved in biological processes, such as steroid and cholesterol biosynthesis, fatty acid metabolism, and maintaining calcium homeostasis. 1 There are at least 57 humans and 3753 mammalian CYP genes. In humans, CYP1, CYP2, and CYP3 families are the main enzymes for drug metabolism, while CYP4 family is involved to a lesser extent. CYP4 to CYP51 families are mainly responsible for endogenous processes. 2 On the other hand, CYPs are able to convert chemicals, such as polycyclic aromatic hydrocarbons, amines, and nitrosamines to reactive metabolites, leading to carcinogenesis and/or organ damage. 3 CYP enzymes are subjective to induction and inhibition by various types of substances, which may influence drug responses. 4 The variances in alleles or polymorphisms contribute to interindividual differences in gene expression and/or CYPs enzyme activity, subsequently affecting the underlying susceptibility toward diseases and differences in pharmacokinetic processes. 4 Most CYP isoforms are primarily expressed in the liver, while some others such as CYP1B1, CYP2J2, CYP2W1, and CYP4Z1 were found mainly in extrahepatic organs. Dysregulated expression of these extrahepatic CYPs possibly contributed to carcinogenesis in expressed organs. 5
CYP4 family consistes of 22 subfamilies that have highly conserved residues within its active site. 6 CYP4 family is also known as microsomal ω-hydroxylase that metabolizes endogenous fatty acids, vitamin D, carcinogens, and eicosanoids. 7 As the only member of CYP4B subfamily, CYP4B1 was initially isolated from rabbit lung microsomes. 8 To date, the function of innate human CYP4B1 protein still remains obscure, and therefore, it is categorized as an orphan CYP enzyme. CYP4B1 was found to be expressed primarily in extrahepatic tissues with the highest expression in lungs. 9,10
The contribution of CYP4B1 in cancer is of particular interest as CYP4B1 gene expression was found to be altered and presumably associated with certain cancers. 7,11 –17 On the contrary, several studies reported that human CYP4B1 was inactive due to a loss of function substitution of proline with serine at position 427 (p.S427P). 10 Therefore, it remains dubious that the low levels of CYP4B1 have functional consequences in normal or cancerous cells. To our best knowledge, the earlier review paper on CYP4B1 was written back in the year of 2006, which documented the detailed nature of the CYP4B1, covering its structures and functions. 18 In this article, we focused on the updated findings of CYP4B1’s expression in tissues together with possible regulation pathways involved, its potential roles in carcinogenesis, contradictory literature of inactive human CYP4B1 enzyme, as well as its application in cancer treatment. This work is expected to provide a better understanding of CYP4B1’s structure in relation to its functions. These findings would be helpful in finding the answer to whether CYP4B1 still remains its research interest to be developed as an ideal target for cancer treatment.
Tissue expression of CYP4B1 protein and mRNA
Most CYPs are primarily expressed in the liver, while CYP4B1 is predominantly expressed in lungs. 19 Besides, CYP4B1 is also found in adipose tissues, adrenal gland, arteries, bladder, breast, esophagus, fallopian tube, heart, salivary gland, skeletal muscle, pituitary, prostate, skin, stomach, thyroid, cervix, uterus, vagina, and a small amount in brain, blood, kidney, liver, ovary, pancreas, and spleen (https://gtexportal.org/home/gene/CYP4B1). Imaoka et al. detected CYP4B1 protein in human bladder microsomes by immunoblotting. 11 Furthermore, the expression of CYP4B1 protein was detected in human esophageal mucosa on 25 histologically non-neoplastic surgical tissues. No specific antibody was available to validate the corresponding protein in immunoblot even though the presence of CYP4B1 messenger RNA (mRNA) was recognized. 20 Luu-The et al. quantified the mRNA expression level of phase 1 and 2 metabolizing enzymes among Episkin™ (artificial epidermis model), full thickness model from Episkin (FTM, Episkin™ that has fibroblasts and keratinocytes), total human skin, dermis, and epidermis. They found that CYP4B1 mRNA expression was present in all human skin categories and the artificial skin types investigated. 21 These data suggested that the human skin and in vitro artificial skin could serve as excellent models to study CYP4B1 in drug metabolisms and possible treatments for skin cancer or diseases to ascertain pharmacological effectiveness and to avoid toxicities. CYP4B1 genes were reported to be expressed predominantly on the right side of heart ventricle 22 and also expressed in breast cancer tissues and breast cancer cell lines, including MCF-7, MDAMB-231, and MCF-10a. 16,23 Roos et al. detected CYP4B1 mRNA in most of the urothelial cell cultures and urinary sediments from healthy subjects. 24
Regulation of CYP4B1 expression
It was recognized that majority of the regulation of CYP genes occurred at the transcriptional level. The general mechanism is similar to those of steroid nuclear receptors, such as pregnane X receptor, constitutive androstane receptor (CAR), and peroxisomal proliferator-activated receptors (PPARs). 25 Briefly, upon binding a cytosolic receptor by a ligand, a heterodimer is formed with a partner protein, which moves further to the nucleus. Subsequently, the heterodimer binds to a consensus site in the 5′-regulatory sequence, which is able to open the promoter region, enabling RNA polymerase to alter CYP gene expression level (see Figure 1).

The general pathway involved in regulating CYP gene expression. CYP: cytochrome P450.
CYP4B1 expression is subject to be altered. For instance, the expression of CYP4B1 was induced by androgens, as observed in kidney and bladder of male mouse or rat. 11 Several nuclear receptors were proposed to be responsible for regulating CYP4B1 gene expression. Mastyugin et al. reported that the isolated 3.4 kb fragment of CYP4B1 promoter had the binding sequence for hypoxia-inducible factor 1 (HIF-1), activator protein 1, and nuclear factor-kappa light chain enhancer of activated B cells (NF-κB), which are hypoxia-sensitive transcription factors. HIF-1 was likely to be readily activated upon hypoxic injury leading to hypoxia-induced CYP4B1 expression. 26 Satih et al. reported the downregulation of CYP4B1 expression in breast tumor cell lines after exposures to potential anti-inflammatory agents, such as genistein and daidzein, that are primary soy isoflavones, which were structurally similar to estrogens. 23 In normal murine breast epithelial cells, the collagen density of the breast cells regulated the expression of CYP4B1. 27 The aryl hydrocarbon receptor and HIF-1α agonists enhanced the production of CYP4B1 in high-density mammary cells possibly due to an increase of NF-κB activation. 27 PPARs might be involved in regulating CYP4B1 expression in corneal epithelium following injury. PPARs are known to be activated upon binding of eicosanoids. 28 Hypoxia and inflammation may induce CYP4B1, which synthesizes eicosanoids. This brings cascades of CYP4B1 induction via PPAR activation. For instance, clofibrate (an exogenous ligand for PPARs) triggered the 12-hydroxyeicosatetraenoic acid and 12(S)-hydroxy-8,10,14-eicosatrienoic acid (12(R)-HETrE) in corneal epithelium and increased CYP4B1 expression levels in rabbit corneal epithelium cells. 29 Computer analysis of the 3.41 kb promoter sequence of CYP4B1 gene from rabbit cornea found that the cis-acting elements were functionally linked to CYP proteins induction. 30 These sequences offered several binding sites for retinoic acid heterodimeric receptors, namely, retinoic acid receptor/retinoid X receptor (RAR/RXR), vitamin D receptor/RXR (VDR/RXR), and PPAR/RXR, suggesting retinoic acid played a role in CYP4B1 regulation.
Catalysis of CYP4B1
CYP is a superfamily of cysteinato-heme monooxygenases, which is responsible for biotransforming a large number of endogenous and exogenous substances via oxidative reactions. 1 CYPs are able to hydroxylize saturated carbon–hydrogen bonds, epoxidize double bonds as well as oxidize heteroatoms and aromatics. 31 Briefly, upon binding of substrate to CYP enzyme, in the presence of oxygen, CYP inserts one oxygen atom into the substrate and the other one to a water molecule. Meanwhile, reduced nicotinamide adenine dinucleotide phosphate (NADPH) is used to provide two electrons through a CYP reductase protein and/or cytochrome b5 (see Figure 2). CYPs have highly diversified substrate specificities derived from their substantial different structural elements with very few invariant residues within the three-dimensional fold (reviewed by Poulos and Johnson). 32
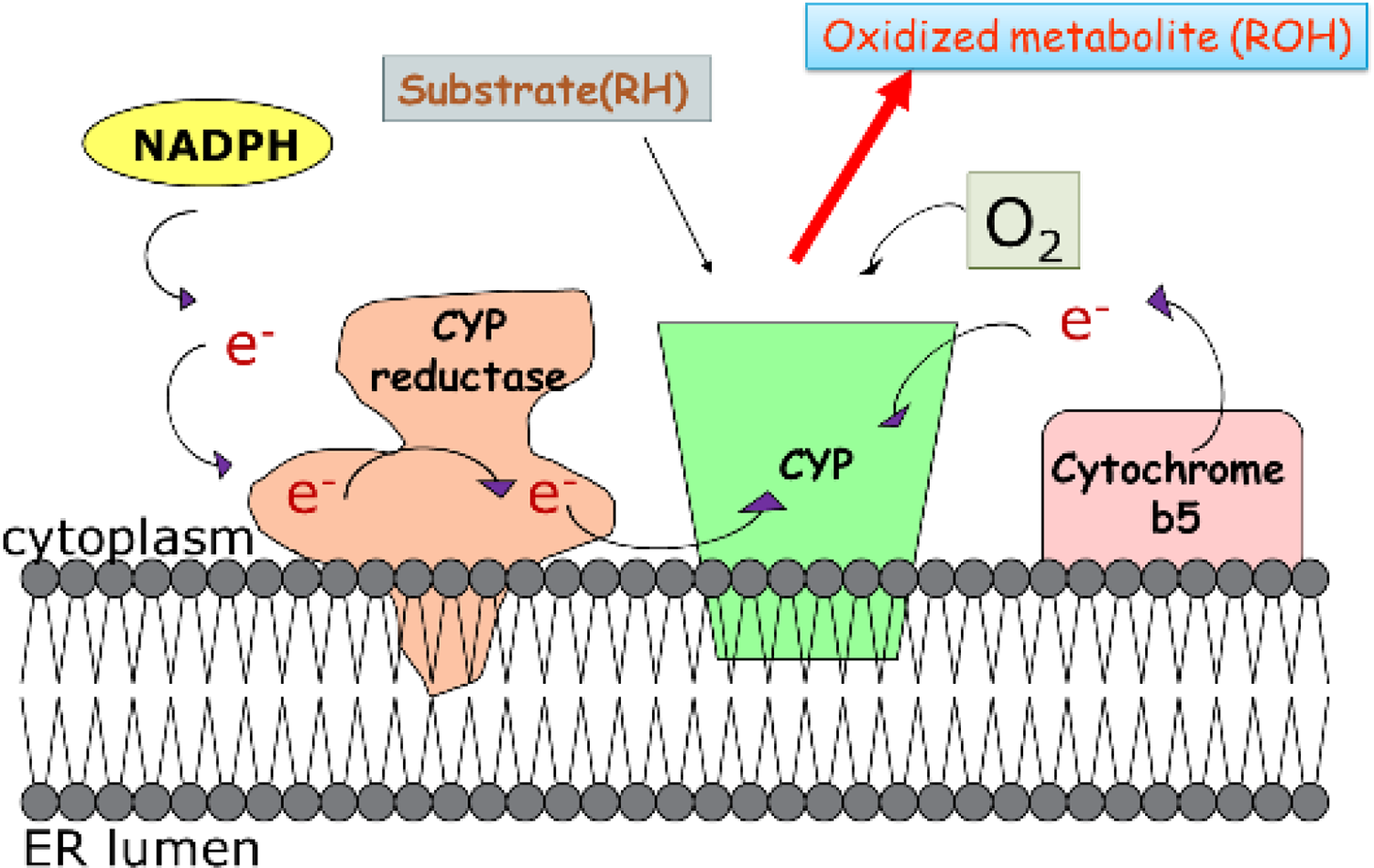
The general electron transference occurring in CYP-catalyzed oxidations. CYP: cytochrome P450.
Hsu et al. generated the crystal structure of rabbit CYP4B1 complexed with octane to reveal some structural adaptations to justify the preference toward ω-hydroxylation. 33 The active site of CYP4B1 was narrowed near the heme making the (ω−1)-hydrogens more protected from heme than ω-carbons and this promoted the region-selection of ω-hydroxylation process. 33 As a result of the narrow constraints, the reactive oxygen could access the primary C–H bonds more freely as compared to the secondary or tertiary C–H bonds. 33 Table 1 demonstrated the hydroxylation rate of CYP4B1 toward alkanes with various numbers of carbon atoms (n-heptane, n-octane, n-nonane, and n-decane) followed a general trend (ω > ω−1 > ω−2). Moreover, with increasing chain length, the hydroxylation decreased. The similar high preference for ω hydroxylation over ω−1 hydroxylation was observed in other CYP4 family enzymes. 34 The region selectivity of ω-hydroxylation was believed due to covalent linkage of the autocatalytically formed ester bond between the heme methyls and atoms of glutamic acid (Glu-310) side chains. 35 The ester bond contributed to side-chain rigidity, which prevented alternative conformations of the Glu-310 side chain that may affect substrate binding within the active site, and also making the active site less polar for hydrocarbon binding. 33 A very recent study by Jennings et al. revealed further insights on the features that contributed to ω-hydroxylation of octane in the absence of ester linkage by comparing E310A mutant complexed with octane with wild-type CYP4B1 structure. 36 This group employed an X-ray crystallography approach to comprehend the architecture of the active site, heme conformation, and substrate binding of the octane complex of the CYP4B1 E310A mutant. The E310A mutant was very similar to the wild-type CYP4B1 structure, containing ferum in the middle surrounded by four nitrogen, which was surrounded by rotated pyrrole rings of the heme. The ruffling of the heme, which was found to be induced by noncovalent interactions between the protein and heme, played a role in facilitating ester bond formation by engagement of 5-methyl in contact with Glu-310. 36 As shown in the E310A mutant structure, the process in which Leu-122 moved into space occupied by ester linkage could be reversed upon binding of larger substrates, which lead to an increase in ω−1 hydroxylation. With the increase in ω−1 hydroxylation in E310A mutant as compared to native CYP4B1, it was speculated that shorter alanine side chain and loss of ester bonds offered a more open active site that had been reduced in ω-hydroxylation preference. Jennings et al. concluded that the primary features that offer ω-selectivity in CYP4B1 oxidations were due to the interaction of noncovalent protein-heme ruffling and the rigid active site caused by covalent bonds between heme and protein. 36 However, these findings did not address how loss of ester bonds affects the active site dynamics and enhanced its conformational entropy of hydrocarbon.
Omega-hydroxylation region-selectivity of CYP4B1.

CYP4B1: cytochrome P450 B1.
CYP4B1 and cancers
Liver cancer
Based on The Cancer Genome Atlas (TCGA), data on liver cancers associated with CYP4B1 expression were collected based on 465 sample sizes from both men and women (https://www.cancer.gov/about-nci/organization/ccg/research/structural-genomics/tcga). CYP4B1 was not a prognostic biomarker in liver cancer. The reported median for CYP4B1 RNA expression in liver cancer was 0 fragments per kilobase million (FPKM) (https://www.proteinatlas.org/ENSG00000142973-CYP4B1/pathology). FPKM is a normalized estimation of gene expression based on RNA-seq data. Eun et al. determined the CYP4 expression profile in hepatocellular carcinoma (HCC) and its clinical relevance. 7 They obtained CYP4 mRNA expression records of 377 HCC cases from TCGA group. It was found that different CYP4 gene was associated with different gene expression levels. 7 The mRNA expression levels of CYP4B1 were analogous to the controls. High expression of other CYP4 genes, for instance, CYP4F2, CYP4F12, and CYP4V2, was reported to downregulate cell cycle pathways and thus, HCC with low CYP4 expression is linked to tumor proliferation. 7 However, whether CYP4B1 upregulated or downregulated in HCC was not clearly reported.
Bladder and urinary tract cancer
According to TCGA, among 877 samples of renal cancers, the mRNA expression of CYP4B1 was 250 FPKM, while out of 206 samples for urothelial cancers, there were more than 400 FPKM (https://www.proteinatlas.org/ENSG00000142973-CYP4B1/pathology). It was reported that patients suffering from bladder cancer had significantly higher CYP4B1 expression as compared to nonbladder cancer patients. This suggested that high CYP4B1 expression increased the chances of bioactivation of carcinogenic aromatic amines leading to higher risk of bladder cancer development. 11 In rodents, CYP4B1 was found to be responsible for activating procarcinogens in bladders. Moreover, Imaoka et al. reported that carcinogen 2-aminofluorene (2-AF) was a substrate of CYP4B1. 11 On the contrary, Lin et al. found that low CYP4B1 protein level was positively associated with poor prognosis of urothelial carcinomas of both urinary bladder and upper urinary tracts. 12
Corneal neovascularization
Upon injury of the corneal epithelium, the CYP4B1 enzymatic pathway was activated leading to the production of 12-HETrE, which was a potent angiogenic eicosanoid. 13 The silencing of CYP4B1 gene expression caused a decline in neovascularization. 13 It was also reported that vascular endothelial growth factor (VEGF) mRNA levels were reduced following CYP4B1 suppression, whereas CYP4B1 overexpression in corneal epithelial cells increased the VEGF mRNA levels. These findings suggested that CYP4B1 might be one of the proximal regulators of VEGF expression and contributed to corneal neovascularization. 13 Another study on patients with corneal neovascularization, who were treated with anti-VEGF antibody called bevacizumab, showed that treatment against VEGF could reduce neovascularization. 37 Overproduction of VEGF was observed in hypoxia and inflammation cases, and it was also observed in cancer cell proliferation and formation of corneal neovascularization. 37
Lung cancer
For lung cancers, as reported by TCGA that, in 994 samples, the mRNA expression of CYP4B1 was more than 350 FPKM (https://www.proteinatlas.org/ENSG00000142973-CYP4B1/pathology). Czerwinski et al. detected CYP4B1 in normal and neoplastic lung tissues, but the mRNA level was reduced by 2.4-fold in the tumor tissues compared to normal tissues. 14
Prostate cancer
According to TCGA, in 494 samples, the mRNA expression of CYP4B1 was more than 50 FPKM (https://www.proteinatlas.org/ENSG00000142973-CYP4B1/pathology). The presence or absence of CYP is crucial in determining the activation rate of carcinogen and therapeutic outcomes. One of the common therapy for prostate cancer is by suppressing CYP4B1 substrate, androgens, or androgen deprivation therapy. 38 It was observed that high occurrence of prostate cancer patients also developed bladder cancer. 39 The risk of developing bladder cancer and prostate cancer increased with an elevation of CYP4B1 expression. It was found that CYP4B1 mRNA was detected in 11 of 21 prostate cancer samples (60%) and 50% of the control samples. 15
Breast and ovarian cancer
TCGA reported that in 1075 samples, the mRNA expression of CYP4B1 was more than 200 FPKM (https://www.proteinatlas.org/ENSG00000142973-CYP4B1/pathology). Iscan et al. examined 13 sample pairs of breast tumor tissues and control samples. The findings demonstrated that 84.6% of breast tumor samples and 76.9% of control tissues were positive in CYP4B1 gene expression. 16 Moreover, higher level of CYP4B1 mRNA was found in recurrent serous ovarian cancer patients than that of cured ovarian cancer patients. 40 This suggested that the expression of CYP4B1 in cancerous tissues might disrupt chemotherapeutic drug metabolism and affect the curative process after treatment. 40
Adrenocortical tumorigenesis
In contrary to an upsurge of CYP4B1 mRNA and protein expression levels in most cancer tissues, CYP4B 1 expression was found to be 100% suppressed in both adrenocortical adenomas and adrenocortical carcinomas. 17 Loss of copy number, missense mutations, and deletions were predicted factors leading to CYP4B1 suppression and allowing malignant transformation. 17
CYP4B1 in carcinogenesis
Many carcinogens require metabolic activation and transformation into electrophilic molecules to bind to DNA and proteins to cause genetic damage. 11 Oxidative DNA damage could be induced by aromatic amines and their metabolites, as the further conversion of amino group of the arylamines leads to the formation of DNA adduct through covalent binding and/or oxidation. 41 These metabolic activations can be catalyzed by CYP enzymes. 42 CYP4B1 was found to be one of the CYP isoforms responsible for bioactivation of chemicals into precarcinogens, such as 2-aminoanthracene (2-AA), 2-AF, benzene, and valproic acid. 43 CYP4B1 in renal and bladder microsomes was also proposed to be responsible for activating carcinogens, including 3,2′-dimethyl-4-aminobiphenyl, 3,3′-dichlorobenzidine, and 2-naphthylamine. 44 In addition to carcinogen activation, induction of inflammation by CYP4B1 was also believed to lead to carcinogenesis. It was reported that retinoic acid enhanced the CYP4B1 gene expression and also increased the inflammatory 12-hydroxyeicosanoids production in corneal epithelia. 30 The alteration of CYP4B1 expression and activity of corneal epithelium was governed by heme oxygenase-1 produced eicosanoids that lead to inflammatory and neovascularization processes of the cornea. 45 Two pathways of how CYP4B1 promotes carcinogenesis are proposed including bioactivation of procarcinogens/carcinogens and induction of inflammation and neovascularization (see Figure 3). Initially, upon ligand binding onto nuclear receptors, heterodimers are formed and then it binds to the response element of CYP4B1. This causes upregulation or downregulation of the CYP4B1 gene expression. Upon activation of the NF-κB, the downstream VEGF is subsequently triggered to initiate neovascularization, increase of vessel permeability, upsurge of intratumor pressure, and reduction of host immune response; all these processes contribute to the onset of tumorigenesis. From the literature we have reviewed above, different types of cancer showed different CYP4B1 expression levels. The CYP4B1 mRNA expression level varied among different cancer types (see Table 2). This might indicate that CYP4B1 was the common enzyme involved across these cancers and its dysregulation lead to the development of different cancer types. The upregulation or downregulation of CYP4B1 in different cancers also suggested that tumor heterogeneity existed, as different cancer cells showed different genomic profiles, such as gene expression, metabolism, proliferation, and metastasis. 46 It seemed that CYP4B1 expression had certain links with cancer, however, whether cancer altered CYP4B1 expression or vice versa requires further investigation.

Proposed mechanisms that CYP4B1 were involved in carcinogenesis. CYP4B1: cytochrome P450 B1.
CYP4B1 mRNA expression in different types of cancer.

CYP4B1: cytochrome P450 B1; mRNA: messenger RNA.
Does the human CYP4B1 exists as an inactive form?
The CYP4B1 enzyme in humans is homologous to CYP4B1 in chimpanzee, mouse, rat, rabbit, cattle, wolf, red junglefowl, and African clawed frog. 47 Falzon et al. reported that the lung tumor cells were capable of bioactivate 4-IPO and had higher susceptibility to its cytotoxic effects. 48 Imaoka et al. claimed to have successfully expressed the active human CYP4B1 by constructing the fusion protein in yeast cells or by introducing the CYP4B1 gene into transgenic mice. 49 The transgenic mice contained human CYP4B1 protein that catalyzed ω-hydroxylation of lauric acid and 2-AF. 49 It was speculated that CYP4B1 was stabilized by the reductase and cytochrome b5 as well as the insertion of serine at position 207. 49
On the other hand, many more studies reported that the human CYP4B1 was incapable of catalyzing numerous compounds, including promutagen 2-AF 50 and 4-IPO, but rabbit CYP4B1 was found to be highly active. 14 Such species differences for substrate specificity were believed to result from a mutation at position 427, where proline was substituted with serine (p.S427P) in human CYP4B1. 51 This locus was later regarded as part of the CYP meander region, which was critical for heme binding and hence CYP enzyme activities. 52 In fact, at this locus, proline is invariant among all CYPs involved in xenobiotic metabolism. 53 Later, site-directed mutagenesis studies clearly demonstrated the importance of having serine at position 427 in human CYP4B1 and corresponding position (422) in rabbit CYP4B1, which allowed the incorporation of the heme prosthetic group in CYP4B1 proteins in expressing insect cells. 54,55 Clinical phase I and II studies revealed that the prodrug 4-IPO had no relevant degree of anticancer activity or lung toxicity, suggesting that the human CYP4B1 was unable to activate 4-IPO. 56 –58 Wiek et al. further substantiated the notion that the native human CYP4B1 with serine at position 427 was unstable, having short half-life in cells when being overexpressed in lentiviral vectors. 10 The differences in CYP4B1 activities of human and rabbit in 4-IPO activation might be due to protein half-life variances. 10 Moreover, Schmidt et al. demonstrated that the insertion of serine at position 207 did not rescue the CYP4B1 functional activity of native human CYP4B1 and rabbit CYP4B1. 6
Substrates of CYP4B1 and suicide gene for future cancer treatment
Among all the CYP4 isoforms, CYP4B1 was considered to have the widest substrate specificity covering metabolism of fatty acids, retinoic acids, hydrocarbons, unfunctionalized aromatic compounds, and arylamines. 10 The common features of CYP4B1 substrates include comparatively small, compact molecular structure, short-to-medium carbon chain lengths, and sterically unhindered position for oxidation. 9 Substrates of CYP4B1 were lauric acid, 2-AF, cumene, omega-6 polyunsaturated fatty acid, arachidonic acid, 2-AA, and 4-IPO. 49,59 –62
4-IPO was firstly isolated from sweet potato roots with fungus infection. 63 It has been well documented as a pulmonary toxin, which requires bioactivation by CYP4B1 to demonstrate its toxic effects in lung tissues. 56 CYP4B1 was found to be expressed in lung Clara cells and surface epithelial cells of the alveoli in many animal species. 48 As described previously, clinical trials testing 4-IPO for the targeted therapy of human nonsmall cell lung cancer and HCC were unsuccessful. 56,57 Gene mutation during the evolution of human CYP4B1 at position 427 (p.S427P) was believed to diminish its catalytic activity. 58 On the other hand, rabbit CYP4B1 has proline at corresponding position 422, which demonstrated high activity toward bioactivating 4-IPO. 10,58 Hence, a suicide gene approach of combining the 4-IPO with active rabbit CYP4B1 was firstly developed and tested in the treatment of brain tumor using both living cells and in vivo approach. 64,65 In principle, suicide genes are also known as prodrug transforming genes, which encode enzymes (CYP4B1) responsible for converting the nontoxic prodrug (4-IPO) substrates to toxic agents (furan epoxide) in tumor cells, leading to cell death, as shown in Figure 4. In human and rodent glioma cells, the rabbit CYP4B1 complementary DNA was ligated to the N-terminus of enhanced green fluorescent protein (EGFP) gene, which was subsequently expressed retrovirally. The tumor cells were killed efficiently after incubating with 4-IPO. 64 With regard to the in vivo study using Fisher 344 rats, 9 L gliosarcoma cells were implanted epidurally, which was followed by injection of recombinant HSV particles carrying CYP4B1-EGFP fusion protein to these tumors. Although without proper optimization of gene transfer conditions, the preliminary in vivo findings from Rainov’s study provided a feasible ex vivo approach to demonstrate the fluorescent-microscopic evaluation of tumors. 64 Similarly, chimeric fusion protein of CYP4B1-EGFP was overexpressed in three human HCC cell lines, namely, HuH-7, HepG2, and Hep3B. Cell death of these HCC cell lines was observed 48 h after 4-IPO administration. 66
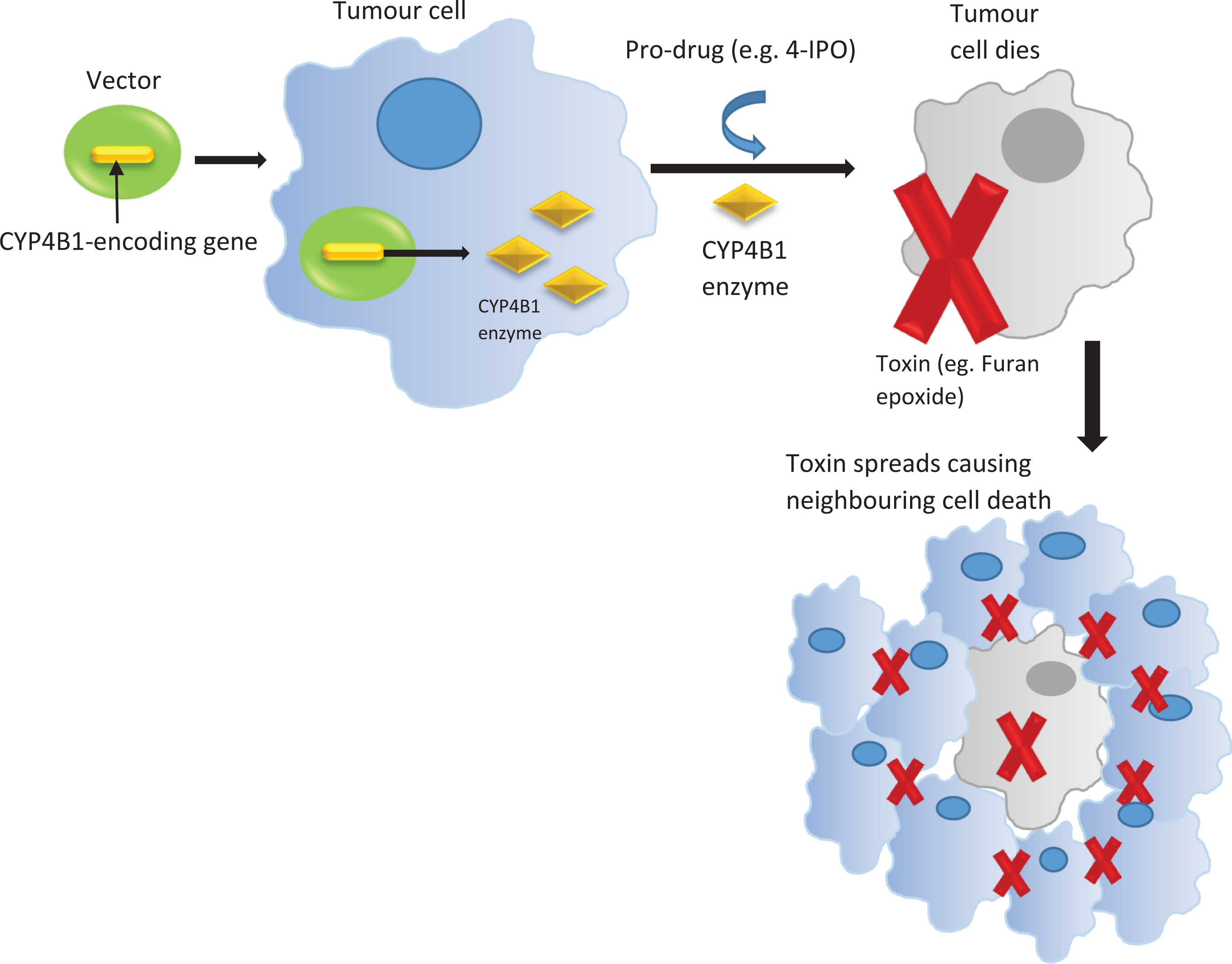
Suicide gene approach by employing CYP4B1 and prodrug. CYP4B1: cytochrome P450 B1.
Wiek et al. further developed this suicide gene approach using 4-IPO as the prodrug but replacing rabbit CYP4B1 with reengineered active human CYP4B1 enzyme in vitro. The reengineered CYP4B1 carried proline at position 427 together with another 12 single amino acid (glutamic acid, 170; threonine, 158; glutamic acid, 159; asparagine, 190; valine, 156; threonine, 202; arginine, 199; aspartic acid, 217; leucine, 226; glutamic acid, 130; arginine, 124; and leucine, 135) substitutions at corresponding positions similar to CYP4 family members to avoid immunogenicity. The reengineered CYP4B1 (CYP4B1P+12) was active in inducing 4-IPO-mediated apoptosis similar to the rabbit CYP4B1 when being introduced into human Hep G2 cells and primary human T-cells. 10 This suicide gene system with the combination of CYP4B1P+12 and 4-IPO was later optimized by employing clinically suited lentiviral vector to express CYP4B1P+12 and a magnetic-activated cell sorting (MACS) selection gene or a CAR. 67
Furthermore, perilla ketone (PK), isolated from the oil of Perilla frutescens, shares similar chemical structure with 4-IPO, which is also a well-known pneumotoxin. 68 Thus, Roellecke et al. developed PK as an alternative prodrug of 4-IPO to be used in this suicide gene system. 67 The following studies carried out by Roellecke group aimed to identify more new alternative prodrugs to be applied in the suicide gene system (HepG2 cells) expressing four different CYP4B1 enzymes, including the rabbit wild type (r-P422), the human wild type (h-S427), the human activated (h-P427), and human reengineered (CYP4B1P+12) proteins. 68 Nine furan analogs including 4-IPO, PK, 2-AA, 2-furylpentylketon (2-FPK), 2-pentylfuran (2-PenF), 2-hexylfuran (2-HexF), 2-heptylfuran (2-HepF), 3-methylindole (3-MI), and 2-AF were investigated. Firstly, 4-IPO, PK, and 2-AA showed significant and comparable cytotoxic activities in HepG2 cells expressing r-P422, h-P427, as well as CYP4B1P+12. Secondly, r-P422 was able to convert 2-FPK, 2-PenF, and 2-HexF efficiently to cytotoxic agents in HepG2 cells, while h-P427 was less active and the CYP4B1P+12 was inactive. Thirdly, cell deaths were observed when incubating with lower concentration of 3-MI in cells with r-P422 and CYP4B1P+12 but not with h-P427. Finally, 2-HepF and 2-AF resulted in nonspecific toxicities in the cells. 68 These findings suggested significant variations with regard to the substrate specificities among the CYP4B1 enzymes, which has been summarized in Table 3.
Compounds used together with CYP4B1 for suicide gene therapy.

CYP4B1: cytochrome P450 B1; r-P422: rabbit wild type; h-P427: human activated; h-P+12: human reengineered; 4-IPO: 4-ipomeanol; PK: perilla ketone; 2-FPK: 2-furylpentylketon; 2-PenF: 2-pentylfuran; 2-HexF: 2-hexylfuran; 2-HepF: 2-heptylfuran; 3-MI: 3-methylindole; 2-AF: 2-aminofluorene; 2-AA: 2-aminoanthracene; ++++: strong and comparable specific cytotoxicity; +++: quite efficient; ++: less active; +: almost inactive; −: no cell death and no specific toxicity.
Conclusion
The native human CYP4B1 was reported as an enzyme that might contribute to several cancer developments based on the enzyme mRNA level of expression. There were different claims on the roles of CYP4B1 in the initiation and progression of cancer. This review demonstrated the progress made in understanding the functions and roles of CYP4B1 with respect to its structural feature, regulation of gene expression, clinical impact, and its contribution to bioactivation of carcinogenic substances. Nonetheless, numerous studies reported that the native human CYP4B1 was inactive due to a mutation (p.S427P), although Imaoka et al. published contradictory findings. There is a need to further address the native human CYP4B1 activities in the future by employing different cell lines and animal models, as well as with the help of crystal structure of human CYP4B1. Otherwise, it is questionable to further investigate how the inactive and unstable CYP4B1 could be involved in cancer. Nevertheless, suicide gene approach was employed using reengineered CYP4B1, which was capable of converting the furan 4-IPO into a highly toxic DNA alkylating metabolite, leading to cancer cell apoptosis. This is a promising strategy to develop targeted therapies for cancer treatment. Efforts should be made to test on more compounds with similar structure as 4-IPO to identify more alternative agents for this suicide gene approach. Furthermore, clinical trials for cancer treatment are advised with optimized suicide gene system employing 4-IPOa or other prodrugs and reengineered CYP4B1. Genotyping among different populations should be conducted to identify any variant carrying serine at position 427, who potentially has higher risk of developing cancer via bioactivating procarcinogens by active CYP4B1 but may benefit from cancer treatment using prodrug, such as 4-IPO.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
