Abstract
Acrylamide (ACR) is a water-soluble chemical that is commonly used in chemical and cosmetic manufacture. Many studies have been carried out to investigate the neurotoxicity mechanisms of ACR, resulting in oxidative stress and nerve damages. One of the commonly used traditional Chinese medicines is notoginsenoside R1 (NR1). However, its mitochondrial-mediated apoptotic effect caused in ACR-induced neurotoxicity has not been reported. Our results have shown that NR1 resisted the neurotoxicity induced by ACR by upregulating the levels of thioredoxin-1 (Trx-1) in Rat adrenal chromaffin cell tumor (PC12) cells. NR1 inhibited the increase in levels of Bax, caspase-9, and caspase-3, which was instigated by ACR. Moreover, NR1 inhibited the decrease in levels of B-cell lymphoma 2 and Trx-1 induced by ACR. The downregulation of Trx-1 aggravated the mitochondrial-mediated apoptosis and increased the expression of the above molecules, which was induced by ACR. In contrast, overexpression of Trx-1 attenuated the mitochondrial-mediated apoptosis and inhibited the expression of the mentioned molecules induced by ACR. Our results suggested that NR1 protected ACR-induced mitochondrial apoptosis by upregulating Trx-1.
Introduction
Acrylamide (ACR) is a water-soluble vinyl monomer. It is widely used as industrial raw materials in polyacrylamide production, water purification, cosmetics production, and paper industry. 1 ACR also has an important application in laboratory research, used in gel electrophoretic separation of molecules. Because of its wide application, people experience high levels of exposure. 2 Exposure to ACR has been shown to cause neurotoxicity, genotoxicity, developmental toxicity, and carcinogenicity in experimental models. 3 Certain clinical symptoms that are associated with neurotoxicity triggered by chronic low-level exposure to ACR are weight loss, gait changes, ataxia, muscle weakness, and numbness in the limbs and bones. 4 –7
Notoginsenoside R1 (NR1) is a powerful free radical scavenger. It has antioxidant properties and can suppress adhesion molecules and chemokines. 8 Panax notoginseng, a kind of traditional Chinese medicine, is used for the treatment of cardiovascular disease 9,10 and acute ischemic stroke. 11,12 Extractions of P. notoginseng have been widely applied in clinical situations in China. 13
Zeng et al. and Yu et al. have reported that NR1 resisted ischemia or hydrogen peroxide injuries by regulating oxidative stress. 14,15 Besides, NR1 significantly reduced the expression of tumor necrosis factor-alpha in human arterial smooth muscle cells, which is caused by hydrogen peroxide. 16 Hydrogen peroxide is widely used to induce reactive oxygen species (ROS) within a short time. NR1 also provides neuroprotective effects by inhibiting the production of ROS in SH-SY5Y cells. 17 In other words, NR1 can resist the damage induced by ROS in human being.
ROS and nitrogen species change cellular responses through various mechanisms. At low levels, they behave as signaling molecules, whereas at high levels, they damage organelles, especially the mitochondria. Oxidative damage and associated mitochondrial dysfunction can result in energy depletion, accumulation of cytotoxic mediators, and cell death. Besides, a significant fraction of endogenous B-cell lymphoma 2 (Bcl-2) family proteins, including Bax and Bcl-2, is associated with mitochondrial apoptosis. 18 Caspase activation is a crucial and complex early event in the commitment of a cell to undergo apoptosis. 19 Lots of studies have proved that caspase-3 and caspase-9 are involved in the mitochondrial apoptosis pathway.
Thioredoxin-1 (Trx-1) has various biological activities, such as regulating cellular redox balance, activating transcription factors, and protecting neurons. 20,21 Previous studies have shown that NR1 could increase the expression of Trx-1 in vivo. 22
Although NR1 has been showed to protect against various neurological diseases, the underlying mechanisms remain unclear. In this study, we explored the roles of NR1 in ACR-induced neurotoxicity and the pathways related to mitochondrial apoptosis. Our results indicated that NR1 plays a protective role in ACR-induced neurotoxicity. This protective effect may be dependent, at least in part, by suppressing mitochondrial apoptosis. Therefore, it is important to explore the effect and mechanism of NR1 as a novel therapeutics for ACR-induced neurotoxicity.
Materials and methods
Reagents
Trx-1 rabbit polyclonal antibody (14999-1-AP; 1:1000) and β-actin rabbit polyclonal antibody (20536-1-AP; 1:1000) were purchased from ProteinTech (Wuhan, China). Bax rabbit polyclonal antibody (50599-2-lg; 1:1000) was purchased from ProteinTech. Bcl-2 rabbit polyclonal antibody (GTX100064; 1:1000) was purchased from GeneTex (Irvine, California, USA). Caspase-3 rabbit polyclonal antibody (#9661) was purchased from Cell Signaling Technology (Danvers, Massachusetts, USA). Caspase-9 mouse monoclonal antibody (#9508) was purchased from Cell Signaling Technology. ACR (A501033, purity ≥ 98%) was purchased from Sangon Biotech (Shanghai, China). NR1 (IN0240, purity ≥ 98%) was purchased from Solarbio life sciences (Beijing, China). Trx-1 small interfering RNA (siRNA), and rat Trx-1 (NM_053800.3) plasmid were chemically synthesized by Shanghai GeneChem Corporation, Ltd (Shanghai, China).
Cell culture
PC12 cells (poorly differentiated) of the rat pheochromocytoma tumor cell line were purchased from Kunming Institute of Zoology (Kunming, China) and were maintained in RPMI-1640 medium supplemented with 10% heat-inactivated horse serum, 5% heat-inactivated fetal bovine serum, and antibiotics (100 U/ml penicillin and 100 μg/ml streptomycin) at 37°C in a humid atmosphere containing 5% CO2. PC12 cells are widely used in the study of nervous system diseases, for example, Parkinson’s disease, 23 neurotoxicity, 24 Alzheimer’s disease, 25 and so on. Upon exposure to nerve growth factor (NGF), PC12 cells undergo neuronal differentiation. NGF-treated PC12 cells release several neurotransmitters including dopamine, noradrenaline, and acetylcholine.
Cell viability assay
The Cell Counting Kit-8 (Dojindo Laboratories, Kumamoto, Japan) was performed to quantify the proliferation of PC12 cells. Cells were cultured at the concentration of 5000/well in 96-well plates. PC12 cells were treated with a wide range of concentrations of NR1 (0.000, 0.0125, 0.025, 0.05, and 0.1 mg/ml) and ACR (0, 1.25, 2.5, 5, and 10 mM) for 24 h in 96-well plates. After incubated for 24 h, Cell Counting Kit-8 (CCK-8) was added to each well and incubated for 1 h. The absorbance of each well was read at 450 nm. To test the neuroprotective effect of NR1 at low doses against ACR-induced cell damage, PC12 cells were treated with indicated concentrations of NR1 and ACR for 24 h. And then, CCK-8 was added to each well and incubated for 1 h. The absorbance of each well was read at 450 nm. In this manner, the experiment was performed in triplicates.
Trx-1 siRNA preparation and cell transfection
The sequences of Trx-1 siRNA and negative control siRNA were as follows: Trx-1 siRNA sense: 5′-GUCAAAUGCAUGCCAACAUtt-3′ and antisense: 5′-AUGUUGGCAUGCAUUUGACtt-3′. Trx-1 siRNA was diluted to 20 μM with a universal buffer.
PC12 cells were plated in six-well plates at a density of 1 × 105 cells/well and were allowed to adhere for 12 h. The contents of 5 μl siRNA and 5 μl lipofectamine™ 2000/well were diluted separately in serum-free Opti-MEM for the final volume of 50 μl, gently mixed, and incubated for 5 min at room temperature. Then, the diluted siRNA solution and the diluted lipofectamine 2000 were mixed gently and incubated for 20 min at room temperature. The diluted siRNA/lipofectamine 2000 complex was added to the six-well plates. After transfection with siRNA for 24 h, the cells were stimulated with ACR and NR1 and then harvested for assay.
Trx-1 overexpression preparation and cell transfection
Rat Trx-1 (NM_053800.3) plasmid (4 μg) and 10 μl lipofectamine 2000/well were diluted separately in serum-free Opti-MEM for a final volume of 250 μl, gently mixed, and incubated for 5 min at room temperature. Then, the diluted plasmid solution and the diluted lipofectamine 2000 were mixed gently and incubated for 20 min at room temperature. The diluted plasmid/lipofectamine 2000 complex was added to the six-well plates. After transfection with Rat Trx-1 plasmid for 24 h, the cells were stimulated with ACR and NR1 and then harvested for assay.
Western blot analysis
Protein lysates were prepared using the solubilizing solution (20 mM Tris–HCl (pH 7.4), 150 mM NaCl, 1% NP-40, 1 mM ethylenediaminetetraacetic acid, 1 mM phenylmethanesulfonyl fluoride, 1 mM ethylene glycol tetraacetic acid, 1% Triton X-100, 2.5 mM sodium pyrophosphate, 1 mM Na3VO4, 1 mM β-glycerol phosphate, and 1 mg/ml leupeptin). Protein concentration was determined by a Bio-Rad protein assay reagent (Hercules, California, USA). Equal quantities of proteins were separated by 12% (for caspase-9) or 15% (for Bax, Bcl-2, and Trx-1) sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to a polyvinylidene difluoride membrane (Millipore, Billerica, Massachusetts, USA). The membrane was soaked in 10% skimmed milk (in phosphate-buffered saline, pH 7.2, containing Tween 20 0.1%) overnight at 4°C and then incubated with primary antibody followed by peroxidase-conjugated anti-mouse or anti-rabbit immunoglobulin G (Kirkegaard & Perry Lab Inc, Gaithersburg, Maryland, USA). The epitope was visualized using an ECL Western blot detection kit (Millipore). Densitometry analysis was performed using ImageJ software (Version 1.38X).
Measurement of mitochondrial transmembrane potential
The cells were seeded in six-well plates and then treated with ACR or NR1 at different concentrations for 24 h. Matrix metalloproteinase (MMP) was measured using the Tetraethylbenzimidazolylcarbocyanine iodide (JC-1) assay (Solarbio) according to the manufacturer’s protocol. JC-1 is a membrane-permeant lipophilic dye that exists as J-aggregates in the mitochondrial matrix (red fluorescence) and as monomers in the cytoplasm (green fluorescence). After incubation with JC-1 dye for 20 min at 37°C and washed with incubation buffer for two times, the ratio of green to red was analyzed by fluorescence cytometry. Cells containing forming J-aggregates had high MMP and showed red fluorescence (FL2). Cells with low MMP were those in which JC-1 maintained monomeric form and showing green fluorescence (FL1).
Measurement of intracellular ROS
The production of ROS was measured by a commercial intracellular ROS kit (Solarbio). Every 5 × 106 cells were suspended in 1 ml of dichloro-dihydro-fluorescein diacetate (DCFH-DA) (10 μmol/l) and incubated for 20 min at 37°C in the dark. After the cells were washed three times with FBS-free RPMI-1640 medium, the fluorescence was analyzed by fluorescence cytometry.
Statistical analysis
Data were expressed as mean ± SE values. Statistical analysis was performed using SPSS software (Version 23.0). A one-way analysis of variance followed by a Bonferroni post hoc analysis was used to identify differences between the treated groups. The p-value less than 0.05 was considered statistically significant.
Results
NR1 ameliorated ACR-induced ROS in PC12 cells
The probe DCFH-DA could be oxidized into the fluorescent compound DCF, which indirectly reflected the level of ROS in PC12 cells. As shown in Figure 1(a) and (b), DCFH-DA fluorescence intensity was distinctly strengthened by ACR, indicating that the excessive accumulation of ROS. Also, the results showed that NR1 could weaken the intracellular ROS level and suppress the oxidative stress induced by ACR.

NR1 suppressed ACR-induced ROS in PC12 cells. (a) ROS production was markedly inhibited by NR1. (b) Quantitative analysis of ROS. Each bar represents the mean ± SE (n = 3 independent experiments). n.s. > 0.05; *p < 0.05; **p < 0.01; ***p < 0.001 statistically significant. NR1: notoginsenoside R1; ACR: acrylamide; ROS: reactive oxygen species; SE: standard error.
NR1 ameliorated ACR-induced disruption of MMP in PC12 cells
The reduction of MMP reflects mitochondrial dysfunction, which is usually used for predicting early apoptosis. JC-1 is a specific fluorescent dye that accumulates in energized mitochondria. Our results showed that a significant MMP loss was found in the ACR group; however, NR1 ameliorated ACR-induced disruption of MMP in PC12 cells (Figure 2). These results indicated that NR1 could alleviate ACR-induced MMP loss in PC12 cells.

NR1 ameliorated ACR-induced disruption of MMP in PC12 cells. (a) ACR-induced MMP depolarization in PC12 cells characterized by transformation of JC-1 dye aggregated to JC-1 monomers; however, NR1 ameliorated ACR-induced disruption of MMP in PC12 cells. (b) Quantitative analysis of MMP. Each bar represents the mean ± SE (n = 3 independent experiments). n.s. > 0.05; *p < 0.05; **p < 0.01; ***p < 0.001 statistically significant. NR1: notoginsenoside R1; ACR: acrylamide; MMP: matrix metalloproteinases; JC-1: tetraethylbenzimidazolylcarbocyanine iodide; SE: standard error.
Effects of NR1 or ACR on PC12 cell viability and Trx-1 expression
To examine the dose-response, PC12 cells were treated with NR1 at concentrations ranging from 0.0125 mg/ml to 0.1 mg/ml for 24 h. Moreover, PC12 cells were treated with ACR at concentrations ranging from 1.25 mM to 10 mM for 24 h. The cell viability was determined by using a CCK-8 assay. We found that cell viability and Trx-1 expression were increased by treatment with increasing concentration of NR1 (0, 0.0125, 0.025, 0.05, and 0.1 mg/ml; Figure 3(a) and (b)). We also found that cell viability and Trx-1 expression were decreased by treatment with increasing concentration of ACR (0, 1.25, 2.5, 5, and 10 mM; Figure 3(c) and (d)). We chose the concentration of 0.05 mg/ml NR1 and 5 mM ACR in the following experiments.

Effects of NR1 or ACR on PC12 cell viability and Trx-1 expression. (a) Effects of NR1 on PC12 cell viability. (b) Effects of NR1 on PC12 Trx-1 expression. (c) Effects of ACR on PC12 cell viability. (d) Effects of ACR on PC12 Trx-1 expression. Each bar represents the mean ± SE (n = 3 independent experiments). n.s. > 0.05; *p < 0.05; **p < 0.01; ***p < 0.001 statistically significant. NR1: notoginsenoside R1; ACR: acrylamide; Trx-1: thioredoxin-1; SE: standard error.
NR1 suppressed ACR-induced mitochondrial apoptosis in PC12 cells
It’s well known that cell apoptosis is generally mediated by mitochondria, endoplasmic reticulum, or death receptors. Previous studies have shown that ACR could induce mitochondrial dysfunction 26 and ultimately lead to cell death. Moreover, lots of studies have proved that NR1 plays an important role in anti-inflammatory, hemostatic, hypolipidemic, hepatoprotective, renoprotective, and estrogen-like activities 27,28 ; however, there is no study reporting that NR1 resists ACR-induced neurotoxicity. In our study, the results showed that NR1 inhibited the cytotoxicity of ACR (Figure 4(a)). We also showed that the decrease of Trx-1 expression induced by ACR was inhibited by NR1 (Figure 4(b)). We also investigated the mitochondrial apoptosis pathway-associated proteins Bcl-2, Bax, caspase-9, and caspase-3. We found that the NR1 could inhibit Bcl-2 expression induced by ACR was inhibited by NR1 (Figure 4(c)). The increase of Bax, caspase-9, and caspase-3 expression induced by ACR was inhibited by NR1 (Figure 4(d) to (f)). These data suggested that NR1 resisted ACR-induced mitochondrial apoptosis in PC12 cells.

Treatment with NR1 suppressed ACR-induced mitochondrial apoptosis in PC12 cells. (a) Cell viability was measured by the CCK-8 assay after treatment with NR1 and ACR. (b) The decrease of Trx-1 expression induced by ACR was inhibited by NR1. (c) The decrease of Bcl-2 expression induced by ACR was inhibited by NR1. (d) The increase of Bax expression induced by ACR was inhibited by NR1. (e) The increase of caspase-9 expression induced by ACR was inhibited by NR1. (f) The increase of caspase-3 expression induced by ACR was inhibited by NR1. Each bar represents the mean ± SE (n = 3 independent experiments). n.s. > 0.05; *p < 0.05; **p < 0.01; ***p < 0.001 statistically significant. NR1: notoginsenoside R1; ACR: acrylamide; Trx-1: thioredoxin-1; CCK-8: Cell Counting Kit-8; SE: standard error.
Trx-1 overexpression attenuated ACR-induced mitochondrial apoptosis in PC12 cells
Our results have demonstrated that NR1 could resist ACR-induced cell apoptosis; however, the specific mechanism is still unclear. Previous studies proved that NR1 could increase the expression of Trx-1. 14,29 Besides, Trx-1 has various biological activities, such as regulating redox, activating transcription factors, and protecting mice from morphine addiction. 20,21,30 Therefore, we further examined the effect of Trx-1 overexpression on ACR-induced mitochondrial apoptosis in PC12. The PC12 cells were transfected with rat Trx-1 plasmid. As shown in Figure 5(a), Trx-1 was overexpressed in the PC12 cells transfected with rat Trx-1 plasmid and it also reduced the downregulation of Trx-1 levels induced by ACR. We also found that the decrease of Bcl-2 expression induced by ACR was inhibited by Trx-1 overexpression (Figure 5(b)). The increase of Bax, caspase-9, and caspase-3 expression induced by ACR was repressed by Trx-1 overexpression. These results suggested that Trx-1 overexpression attenuated ACR-induced mitochondrial apoptosis by inhibiting expression of Bcl-2, Bax, caspase-9, and caspase-3 (Figure 5(c) to (e)).

Trx-1 overexpression attenuated ACR-induced mitochondrial apoptosis in PC12 cells. (a) The decrease of Trx-1 expression induced by ACR was inhibited by Trx-1 overexpression. (b) The decrease of Bcl-2 expression induced by ACR was inhibited by Trx-1 overexpression. (c) The increase of Bax expression induced by ACR was inhibited by Trx-1 overexpression. (d) The increase of caspase-9 expression induced by ACR was inhibited by Trx-1 overexpression. (e) The increase of caspase-3 expression induced by ACR was inhibited by Trx-1 overexpression. Each bar represents the mean ± SE (n = 3 independent experiments). n.s. > 0.05; *p < 0.05; **p < 0.01; ***p < 0.001 statistically significant. ACR: acrylamide; Trx-1: thioredoxin-1; SE: standard error.
Trx-1 siRNA aggravated ACR-induced mitochondrial apoptosis in PC12 cells
We investigated the effect of downregulation of Trx-1 expression by siRNA on ACR-induced mitochondrial apoptosis. Our results showed that the expression of Trx-1 was reduced by siRNA in the PC12. And the expression of Trx-1 was further decreased by Trx-1 siRNA and ACR (Figure 6(a)). Moreover, the expression of Bcl-2 was further reduced after Trx-1 siRNA treatment followed by ACR treatment (Figure 6(b)). We also found that the expression of Bax, caspase-9, and caspase-3 was further increased after Trx-1 siRNA treatment followed by ACR treatment. These data suggested that Trx-1 siRNA aggravated ACR-induced mitochondrial apoptosis in PC12cells via induction of Bcl-2, Bax, caspase-9, and caspase-3 (Figure 6(c) to (e)).
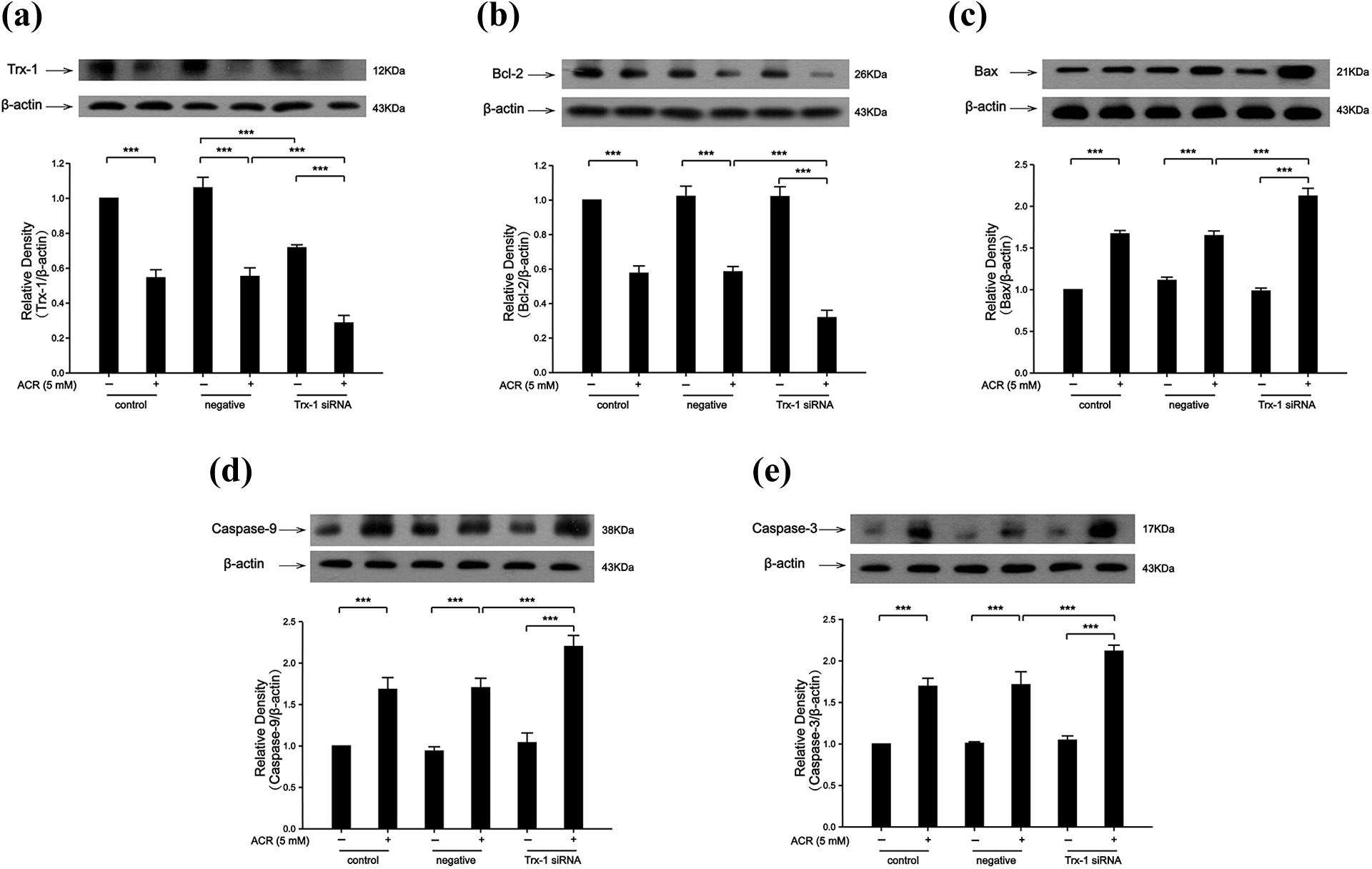
Trx-1 siRNA aggravated ACR-induced mitochondrial apoptosis in PC12 cells. (a) The expression of Trx-1 was further decreased after Trx-1 siRNA treatment followed by ACR treatment. (b) The expression of Bcl-2 was further decreased after Trx-1 siRNA treatment followed by ACR treatment. (c) The expression of Bax was further increased after Trx-1 siRNA treatment followed by ACR treatment. (d) The expression of caspase-9 was further increased after Trx-1 siRNA treatment followed by ACR treatment. (e) The expression of caspase-3 was further increased after Trx-1 siRNA treatment followed by ACR treatment. Each bar represents the mean ± SE (n = 3 independent experiments). n.s. > 0.05; *p < 0.05; **p < 0.01; ***p < 0.001 statistically significant. ACR: acrylamide; Trx-1: thioredoxin-1; siRNA: small interfering RNA; SE: standard error.
Discussion
ACR can be formed by the Maillard reaction during the thermal processing of food rich in carbohydrates and proteins. 31 High level of ACR has been reported in roasted coffee beans, breakfast cereals, crisps, and chips. 31 Many studies have shown that ACR neurological toxicities can induce carcinogenesis in multiple organs in animals. 32,33 Therefore, it is necessary to investigate the mechanism of ACR-induced apoptosis.
Numerous studies have implied that mitochondrial apoptosis is involved in neuronal death in ACR-induced neurotoxicity. 34 –36 In this present study, we proved that NR1 could resist ACR-induced neurotoxicity by regulating the mitochondrial apoptosis pathway. It is also the first time that NR1 has been proved to inhibit ACR-induced neurotoxicity. Previous studies also showed that NR1 was effective in treating many diseases, such as cerebral ischemia, 37 Parkinson’s disease, 38 and cancer. 39 Moreover, many studies proved that NR1 could increase the expression of Trx-1, which is consistent with our results.
Panax notoginseng has been a key component in Chinese herbal medicine for more than 600 years. 29 Gui et al. suggested that P. notoginseng (450 mg once a day) improved the clinical symptoms, increased relative cerebral blood flow, and decreased the relative mean transit time in the region of interest in elderly patients with lacunar infarction. 40 Intravenous infusion of peripheral nervous system (500 mg/day, once daily) was very effective in the treatment of unreconstructable critical limb ischemia in human. 41 In addition, Lang et al. proved that P. notoginseng (280 mg, 250 ml) could treat early diabetic nephropathy. 42 The content of NR1 in P. notoginseng was 2.74%. 43 Therefore, the concentration of P. notoginseng was similar to our results in clinical treatment.
It is reported that hepatitis C virus infection induces Bax-triggered mitochondrial-mediated apoptosis. 44 This pathway is mediated by the opening of the outer membrane pore of the mitochondria, including Bcl-2 family members: Bak and Bax. Bax is known to heterodimerize with Bcl-2 and a preset ratio of Bcl-2/Bax is thought to be critical in determining survival in certain types of cells following specific apoptotic stimuli. 45 This pathway leads to the activation of caspase-9 through activation of caspase-3 and eventually leads to programmed cell death. 46 Our data showed that the decrease of Bcl-2 expression induced by ACR was inhibited by NR1. The increase of Bax, caspase-9, and caspase-3 expression induced by ACR was inhibited by NR1 (Figures 2 and 4). These data suggested that NR1 resisted ACR-induced mitochondrial apoptosis in PC12 cells. Trx-1 played an important role in regulating various diseases. Trx-1 suppressed cytochrome c release from mitochondria most likely by the induction of Bcl-2. 47 Tian et al. proved that overexpression of Trx-1 in HepG(2) cells led to the inhibition of As2O3-induced cytochrome c release, caspase activation, and apoptosis and downregulation of Trx-1 expression by RNAi-sensitized HepG(2) cells to As2O3-induced apoptosis. This suggested that Trx-1 played a central role in regulating apoptosis. 48 Our data showed that the decrease of Bcl-2 expression induced by ACR was repressed by Trx-1 overexpression. The increase of Bax, caspase-9, and caspase-3 expression induced by ACR was inhibited by Trx-1 overexpression (Figure 5). Moreover, the expression of Bcl-2 was further decreased after Trx-1 siRNA treatment followed by ACR treatment. The expression of Bax, caspase-9, and caspase-3 was further increased after Trx-1 siRNA treatment followed by ACR treatment (Figure 6). These data suggested that NR1 resisted ACR-induced neurotoxicity by upregulating the expression of Trx-1.
Conclusion
In summary, NR1 plays a neuroprotective role in ACR-induced by suppressing mitochondrial apoptosis by regulating the expression of Trx-1, Bax, Bcl-2, caspase-3, and caspase-9. Therefore, NR1 can be used as a therapeutic drug to resist the neurotoxicity induced by ACR.
Footnotes
Author contributions
The authors declare no competing financial interests. XL was responsible for the study concept and design. WW, LH, and YH did the experiment. LH and ERT drafted the manuscript. WW and XL provided a critical revision of the manuscript for important intellectual content. All authors read and approved the final version. WW and LH were contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by grants from the Science and Technology Strategic Cooperation Project of the Luzhou People’s Government and Southwest Medical University (No. 2019LZXNYDJ34) and the undergraduate innovation and entrepreneurship training program (S201910727096).
