Abstract
Melamine contamination is a recent public health problem emerging globally. Present study aimed to detect the rate of melamine presence in human milk in a cohort study and to evaluate any possible differences in maternal–infant pair characteristics such as breastfeeding status, crying and sleep problems of infants, maternal postpartum depression, maternal–infant bonding, infant and maternal anthropometry, and maternal complete blood count caused by the melamine exposure. Mothers of infants born in Şanlıurfa were invited to participate in ‘Urfa Child Cohort Survey’. Overall, two breast milk samples were taken between 5 days and 15 days postpartum and between 4 weeks and 10 weeks. Randox Food’s InfiniPlex array was used to analyse the presence of melamine. Melamine was detected in 32.2% and 24.4% of the first and the second milk samples; 16.7% of mothers had two positive samples. z Scores for birth weight and z scores for height for age were detected to be significantly lower in cases with two positive samples compared to cases with negative samples. Mean maternal white blood cells counts were found to be lower in cases with repeated melamine contact. Melamine might have a detrimental effect on birth weight, infant height and maternal blood count. Further studies should be done to detect environmental contamination in different regions and countries.
Introduction
Melamine appears everywhere, namely, nitrogenous fertilizer mixtures, paints, paper coatings, plastics, textile finishes and wrinkle-free textile. 1 Cyromazine, an insecticide widely used in the cultivation of vegetables, is converted to melamine in the body of animal and human. 1 –3 Due to a high nitrogen content, melamine has also been reported to be illegally added into several foods including dairy products, candies, cakes, biscuits, chocolates, wheat gluten, pet foods, nutritional supplements and eggs in order to fortify its protein content. 2,4 –6 This is defined as melamine adulteration (MA) and MA in baby formula resulted in an outbreak of renal calculi, renal failure and death in infants in China in 2008. 7 The effects of MA have also been evaluated in experimental animals including mice, rats, fish, chicken, cats, dogs and pig models. 8 In rodent models, maternal exposures led to the transfer of melamine to serum, amniotic fluid and foetal tissues including brain, heart, kidney, liver, lungs as well as into the milk. 9 Also, when cows and goats were fed the melamine-added feeds, melamine was detected in their milk. 6,10,11 Therefore, besides the intake of contaminated food, infants may come into contact with melamine antenatally through a placental barrier or postnatally through breastfeeding. To date, limited data are available on assessing the transfer of melamine into human milk. 12 –14 Although the exposure level is very little compared to self-consumption of contaminated food, melamine exposure to pregnant and lactating women can be a potential risk to the offspring during critical periods of development. 14 Since foetuses and infants are more vulnerable to the effects of environmental pollution, any possible route of melamine transfer should be prevented. 1,3,6
There are several studies about MA and renal disorders, however, its extra-renal effects are not well researched. 3,7,8,15 To best of our knowledge, there is no published report available on the effects of MA on haematopoietic parameters in human. Being at a global risk for MA, the research into mother–infant pair’s common problems with unknown pathogenesis such as postpartum depression, breastfeeding problems, and sleep and crying characteristics can also contribute in melamine risk assessment. So far, data regarding the effects of melamine on infant’s anthropometric evaluation are very scarce. 15
Therefore, this study was aimed to assess the presence of melamine in breast milk and evaluate any possible differences in maternal–infant pair characteristics such as breastfeeding status, crying and sleep problems of infants, maternal postpartum depression, maternal–infant bonding, infant and maternal anthropometry, and maternal complete blood count in cases with melamine exposure. We hypothesized that the maternal exposure of MA could be detected easily with breast milk and would influence the well-being of maternal–infant pairs in the early postpartum period.
Materials and methods
Study design and participants
Subjects of the study were recruited from the Özel Şan Med Hospital in Şanlıurfa, Turkey. Mothers giving birth between May 2017 and April 2018 were invited to participate in ‘Urfa Child Cohort Survey’. Of those invited in the survey, 90 mother–infant pairs with complete follow-up schedule and two breast milk samples at different visits were selected for this study. Visit 1 (V1): All voluntary mother–infant pairs were evaluated between 5 days and 15 days post-delivery, and they answered the questionnaire concerning mother–infant health. Items on maternal sociodemographic and health properties included maternal age, educational level, working status and health status (hypertension, diabetes, anaemia, drug use, smoking exposure status, urolithiasis, weight gain during pregnancy and weight at birth). The criterion for smoking exposure status was (1) maternal smoking, (2) environmental tobacco smoke exposure without maternal smoking and (3) no known smoke exposure for the last 3 months. In the questionnaire, items on infant characteristics included the presence of assisted reproduction, gestational length, type of delivery, birth order, gender, birth weight, head circumference at birth, any health problem including hyperbilirubinemia and urolithiasis, presence of any breastfeeding-related problem and feeding type. Visit 2 (V2): When infants were 4–10 weeks of age, mother–infant pairs were re-evaluated by a second questionnaire, which covered infant’s crying properties, feeding and sleep-related problems, and maternal smoking status for the last months.
Mothers and infants were weighed, and height and head circumference of infants were measured at V1 and V2.
Outcomes of the study
The outcomes examined in this study were breastfeeding status, colicky crying and sleep problems of infants, postpartum depression, maternal–infant bonding, infant and maternal anthropometry and maternal complete blood count.
Breastfeeding status were evaluated according to ‘early initiation of breastfeeding within 1 h of delivery’, ‘delayed onset of lactogenesis which was defined as maternal perception of onset of noticeable fullness after 72 h postpartum’, ‘exclusive breastfeeding that is the infant only receives breast milk with vitamin–mineral supplement, without any additional food or drink, not even water’ and ‘breastfeeding problems were accepted as presence of anyone of cracked nipples and insufficient milk during follow-up visits’.
Length of crying episodes in a day and weekly episodes were asked. Inconsolable infant crying lasting for >2 h per day and >2 days/week was defined as ‘colicky crying’ in the protocol.
Sleep-related problems were defined as ‘absence or presence’ according to maternal perception.
To assess mother–infant bonding, Postpartum Bonding Questionnaire (PBQ) with self-rating 25-item questionnaire scored on a 6-point Likert scale was used. 16,17 A high score indicates worse bonding.
Edinburgh Postnatal Depression Scale (EPDS) composed of self-rating 10-item questionnaire with a 4-point Likert scale was used to identify postpartum depression. 18
Maternal body mass index was calculated using weight and height at each visit. For each visit, z scores of head circumference for age, weight for age, length for age and body mass index for age (HCZ, WAZ, LAZ and BAZ, respectively) of infants were calculated with World Health Organization Multicentre Growth Reference Study. 19
Maternal data on complete blood count (haemoglobin value, haematocrit value, mean corpuscular volume, white blood cells and thrombocyte count) which were evaluated at 1 month postpartum were taken from hospital file.
Samples collection and analysis
At both visits (V1 and V2), breast milk samples were obtained from the participants via manual pumping. Samples were taken into polyethylene tubes, and immediately after collection, they were kept frozen (−20°C) until analysed. InfiniPlex kit for Milk array (EV4076, Lot number: 9034, Randox Food Diagnostics, Crumlin, UK) was used to analyse the presence of melamine in the milk according to the manufacturer’s instructions. 20 The detection limits for melamine and cyromazine contamination are 0.200 and 0.027 mg/kg milk.
Statistical analyses
Data were analysed using SPSS-Windows 23.0 (SPSS Inc, Chicago, Illinois, USA).
According to the status of melamine detection in visits, three ‘melamine-exposed groups’ were designed: not detected at all (M0), in one visit (M1) and in both visits (M2).
Shapiro–Wilk test was used to evaluate the normality of continuous variables in melamine exposure groups. Data with skewed distribution were shown as median and interquartile values and Kruskal–Wallis H test was applied to compare groups.
Normally distributed data were given as mean, standard deviation (SD) and 95% confidence interval (CI) and one-way analysis of variance was used to compare groups. Anthropometric measurements were compared with ‘General Linear Models, Univariate Analysis of Variance’ after controlling for confounding factors at relevant visit data. ‘Birth WAZ and HCZ data’ was compared in melamine-exposed groups with adjustment of gestational length (weeks) and maternal smoking exposure. ‘Infant’s anthropometry on admission (V1)’ with adjustment of adjusted for gestational length (weeks), maternal smoking exposure, feeding type of infant (exclusive breastfed vs. mixed feeding), infant’s age and gender. ‘Infant’s anthropometry on V2’ adjusted for gestational length (weeks), feeding type (exclusive breastfed vs. mixed feeding), smoking exposure (no known exposure, environmental exposure vs. maternal smoking), infant’s age (days) at V2 and gender.
The percentages in melamine-exposed groups were calculated and the χ2 test was used to compare proportions.
Only parameters having the p value less than 0.1 were included in further analysis. Multiple logistic regression with backward stepwise condition was performed to detect the effect of melamine exposure (M1 vs. M0 and M2 vs. M0), gestational length (weeks), birth type (caesarean delivery vs. normal delivery), early initiation of breastfeeding (<1 h vs. other) and birth order (1 vs. ≥1 child) on delayed onset of lactogenesis.
Statistical significance level was selected as p < 0.05.
Results
General characteristics of mother–infant pairs
In the study, 36.7% of the participants were primiparous and mean (±SD) maternal age calculated was 27.9 (±5.3) years. Mean (±SD) gestational length recorded was 38.3 (±1.5) weeks (Table 1). Of all, 18.9% of mothers stated they had some health problems (number) during gestational period, [gestational diabetes (3), hypothyroidism (2), hypertension (1), iron deficiency anaemia (5), oedema on their legs (2), urinary tract infection (3) and upper respiratory infections (2)].
Background characteristics of mother–infant pairs, n = 90.

CI: confidence interval; HCZ-0: z scores of head circumference for age taken at birth; WAZ-0: z scores of weight for age taken at birth.
Mean (±SD) birth weight of infants calculated was 3132 (±412) g. Six infants had a problem at birth: four with respiratory distress syndrome, two with neonatal transient tachypnoea. Meconium aspiration was also present in one of two tachypnea cases. Physical examination of the infants at V1 showed normal health status, except four cases of hyperbilirubinemia (with no need of phototherapy), two cases of mild omphalitis. At V2, one case of developmental dysplasia of hip, four cases of upper respiratory infections and one case of hyperphenylalaninemia were detected.
Early initiation of breastfeeding was performed in 53.3% of mother–infant pairs. One in five reported delayed lactogenesis after delivery (Table 1). One-third had breastfeeding problems at V1. The proportions of exclusively breastfed infants were 62.2% at V1 and 45.6% at V2 (Table 2).
Characteristics of infants (age, anthropometric parameters, health problems, crying and sleep problems), mothers (smoking, scores of EPDS and PBQ, BMI and blood count) and breastfeeding (problems and melamine content) at visit 1 and visit 2, n = 90.

EPDS: Edinburgh Postnatal Depression Scale; PBQ: Postpartum Bonding Questionnaire; BMI: body mass index; visit 1: admission visit; visit 2; follow-up visit; CI: confidence interval; WAZ: z scores of weight for age; HCZ: z scores of head circumference for age; LAZ: z scores of length for age; BAZ: z scores of BMI for age; EPDS: Edinburgh Postnatal Depression Scale; PBQ: Postpartum Bonding Questionnaire.
a Detection limits were 0.200 for melamine and 0.027 mg/kg-milk for cyromazine.
Melamine detection and mother–infant characteristics
Melamine was detected in 32.2% and 24.4% of the first and the second samples, respectively (Table 2). Of the mothers enrolled in the study, 60% had no melamine present in either of the samples while 16.7% of mothers had two positive samples.
Maternal age, level of education, working status, health problems, presence of assisted reproduction for pregnancy, birth order, gestational length, mode of delivery and infant’s gender did not differ among melamine-exposed groups (Table 3). Maternal anthropometric evaluations were also similar for all groups (Tables 4 and 5).
Background characteristics of mother–infant pairs according to melamine-detection groups.

CI: confidence interval; M0: absence of melamine in the samples; M1: one positive sample; M2: two positive samples; BMI-b: body mass index taken at delivery; HCZ-b: z scores of head circumference for age taken at birth; WAZ-b: z scores of weight for age taken at birth.
a,b,cValues with different letter in the same row are different (p < 0.05).
d Adjusted for gestational length, maternal smoking exposure.
Characteristics of infants (age, anthropometric parameters), mothers (smoking and BMI) and breastfeeding (problems) at visit 1 (admission visit) according to melamine-detection groups.

M0: absence of melamine in the samples; M1: one positive sample; M2: two positive samples; CI: confidence interval; anthropometric measurement taken at the first visit (admission visit), BMI-1: body mass index; HCZ-1: z scores of head circumference for age; WAZ-1: z scores of weight for age; LAZ-1: z scores of length for age; BAZ-1: z scores of body mass index for age.
a,b,cValues with different letter in the same row are different (p < 0.05).
d Adjusted for gestational length, maternal smoking exposure, feeding type of infant, infant’s age and gender.
Characteristics of infants (age, anthropometric parameters, crying and sleep problems), mothers (smoking, scores of EPDS and PBQ, BMI and blood count) and breastfeeding (problems and melamine content) at visit 2 (V2; follow-up visit) by melamine-detection groups.
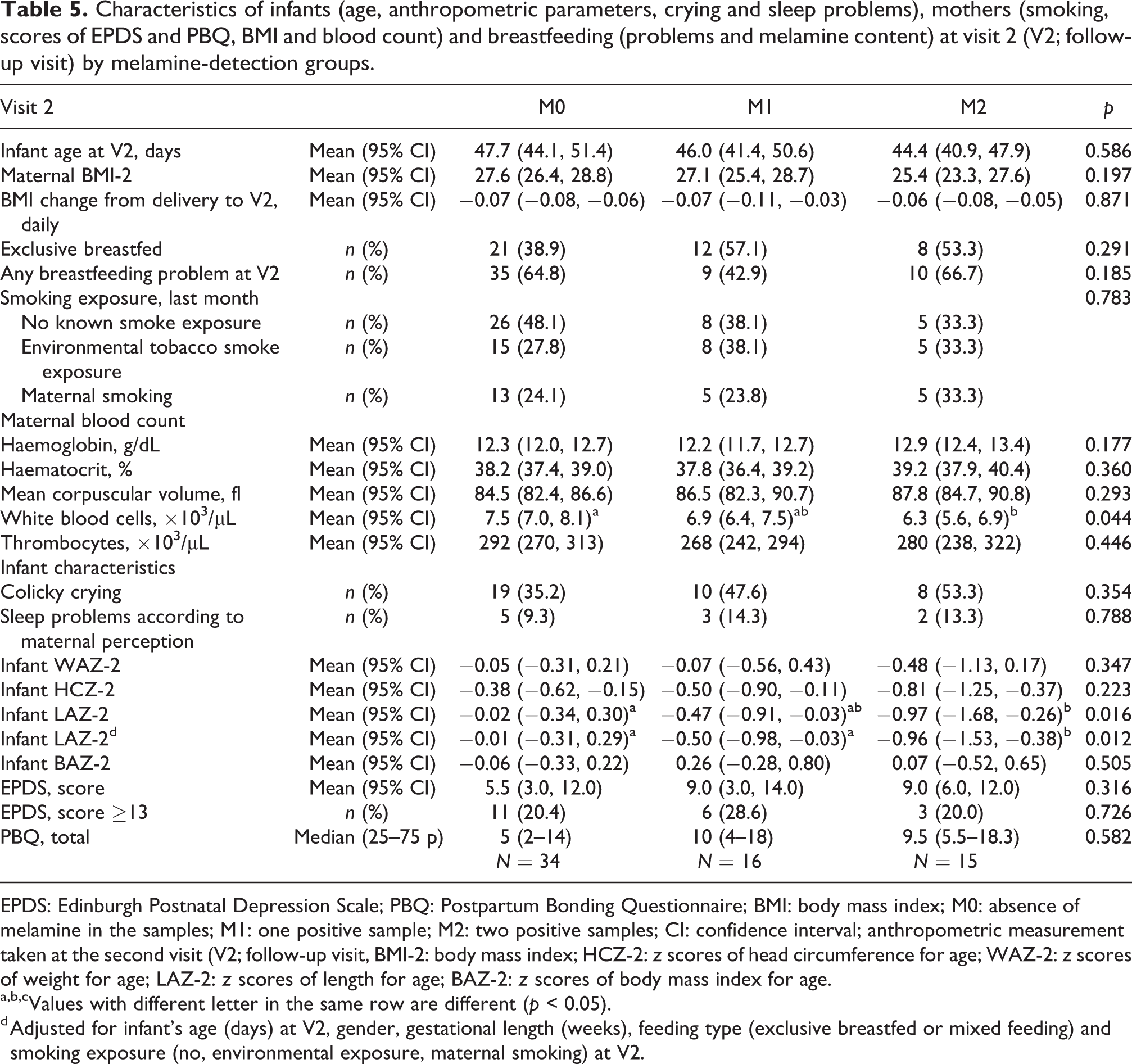
EPDS: Edinburgh Postnatal Depression Scale; PBQ: Postpartum Bonding Questionnaire; BMI: body mass index; M0: absence of melamine in the samples; M1: one positive sample; M2: two positive samples; CI: confidence interval; anthropometric measurement taken at the second visit (V2; follow-up visit, BMI-2: body mass index; HCZ-2: z scores of head circumference for age; WAZ-2: z scores of weight for age; LAZ-2: z scores of length for age; BAZ-2: z scores of body mass index for age.
a,b,cValues with different letter in the same row are different (p < 0.05).
d Adjusted for infant’s age (days) at V2, gender, gestational length (weeks), feeding type (exclusive breastfed or mixed feeding) and smoking exposure (no, environmental exposure, maternal smoking) at V2.
When maternal complete blood count values were compared, leucocytes count of M2 cases was found to be significantly lower than other groups (p = 0.044, Table 5).
The detection rate of delayed onset of lactogenesis in M2 was slightly higher compared to the counterparts (p = 0.097, Table 3). However, multiple logistic regression with stepwise method revealed only birth order (1 vs. ≥1 child) affected delayed lactation (odds ratio = 0.15, %95 CI = 0.05–0.47) among factors including melamine-exposed groups, gestational length, birth type and early initiation of breastfeeding. There were no differences present in the frequency of being exclusively breastfed among groups at both visits.
WAZ scores of infants at birth (WAZ-b) were detected to be significantly lower in M2 compared to M0 (Table 3). Similarly, low WAZ-b was observed in M2 after controlling gestational length, presence of maternal health problems and maternal smoking exposure. However, WAZ-1 and WAZ-2 scores were found to be similar in melamine-detection groups (Tables 4 and 5). On the other hand, low HAZ-1 and HAZ-2 scores of infants were recorded in the M2 group compared to M0 (Tables 4 and 5). Adjusting gestational length, mode of delivery (vaginal vs. caesarean delivery), infant gender (male vs. female), age (days), maternal and neonatal health problems (presence vs. absence), maternal smoke exposure (environmental and maternal smoke vs. absence) and feeding type of infant (exclusive breastfed vs. mixed) at both visits had no impact on the results. Melamine contamination did not affect HCZ and BAZ scores of infants during the follow-up period (Tables 4 and 5).
Proportions of colicky crying, sleep problems according to maternal perception were similar in all groups. Melamine presence did not alter maternal EPDS and PBQ scores (Table 5).
Discussion
Melamine transfer to human milk
This study showed that 40.0% of breast milk from mothers in Şanlıurfa, Turkey, had melamine and/or cyromazine contamination at the level of ≥0.200 mg/kg and/or ≥0.027 mg/kg, respectively. Exposure to melamine might change depending on region and country. 13,14 One study with the same methodology reported melamine contamination in 3.6% (3/83) of breast milk samples in Eskişehir, Turkey. 13 So far, the previous study in Ankara, Turkey showed that 20.8% of 77 milk samples were positive for melamine presence (range = 10.6–76.4 ng/L). 12 Compared to studies in Turkey, a recent study in the United States reported the sum of melamine and its three derivatives in breast milk at a lower concentration (0.176–10.0 ng/mL). 14 However, melamine detection in breast milk has raised concerns about the environment surrounding of mother–infant pairs. Interestingly, melamine and its derivatives were detected in 341 samples of indoor dust collected from 12 countries; highest in the United States and lowest in India. 21 Previously, 300 samples of milk and dairy products from Ankara, capital of Turkey, were analysed for the presence of melamine and detected in 44%, 8% and 2% of yogurt, milk powder and cheese samples, respectively. 22
Melamine is not metabolized or bio-transformed in the body. It is eliminated by the kidney and has been detected in urine. 23 The pharmacokinetics of melamine revealed that half-life and clearance time were depended on the species; it was as short as 90–120 h in animals like pigs, chickens and rats. 6,10,11 In our study, 16.7% of mothers had two positive samples showing continuous or repeated exposure of melamine. During the lactation period, maternal exposure to melamine, either through contaminated foods or use of dishes, can result in melamine transfer to the infants via milk. 14
Health concerns and melamine
Previous studies revealed that prenatal and postnatal exposures to melamine caused altered birth weight and crown-to-rump length in rat models. 9,24 One study reported high percentages of delayed growth (under height and underweight) in melamine-related urinary stones group than the control group. 15 In the present study, cases of melamine contact in two periods (M2 group) had lower z scores of birth weight than others and showed disruptions in height for age during the follow-up period. However, none of the mother–infant pairs had a history of urolithiasis. On the other hand, the previous study in the United States reported no differences among cases in the melamine compound’s according to birth weight. 14 This might be due to low dose exposure of mother–infant pairs in the United States. There is no published study to evaluate growth in the exclusively breastfed infant. Further studies in a large sample size are necessary to detect the effect of melamine in infant growth.
Previous studies showed that prenatal melamine exposure resulted in neurophysiological changes and neurotoxicity with the underlying mechanisms including synaptic plasticity, oxidative stress and the cholinergic system. 25 Considering the possibility of neurobehavioural effect, postpartum maternal depression and maternal–infant bonding, infants’ sleep and crying characteristics were evaluated, however, no interaction was detected with melamine exposure. However, it does not mean that there are no interactions and it is possible to observe such interactions in high-level exposure models. This hypothesis can be analysed in high exposure areas. On the other hand, unexplained crying especially when urinating had been observed in infants affected by the melamine-contaminated infant formula in China. 26 Renal damage might have a role in these crying.
As expected, some environmental chemicals might affect breastfeeding success. Our previous study showed that ‘delayed onset of lactogenesis’ was detected 3.33 times higher in mothers who had the highest quarter level of zearalenone in their milks than counterparts. 27 In the present study, delayed lactogenesis was slightly higher in mothers who had repeated melamine exposure, which is not found to be statistically significant. Further studies with large sample size are necessary to evaluate the effect of melamine exposure on breastfeeding problems.
Studies also showed changes in white blood cells, haemoglobin and haematocrit value of tilapia caused by melamine in feed 28 and altered white and red blood cell counts in mice with melamine exposure. 29 The present study also showed decreased white blood cells counts in the group with repeated melamine contact. Further research is needed to evaluate the effect of different levels of melamine contamination on haematologic parameters.
Strengths and limitations
The present study, for the first time, evaluated the impact of melamine exposure (in two periods) on maternal psychopathologic, haematologic and anthropometric parameters as well as infants’ anthropometric and sleep–crying characteristics.
As a limitation, the contamination level of melamine in breast milk was not evaluated. However, the primary aim of our study was the screening of melamine contamination of human milk. We used Randox’s patented InfiniPlex Biochip Array Technology, a standard competitive immunoassay format, specifically developed to ensure a 98% compliance with all currently monitored residues by the European Union. 20 Screening immunoassay methods typically provide results in less time and is cost-effective than other selective techniques, but they have no power of the methods based on high-performance liquid chromatography and mass spectrometry to unambiguously identify melamine and related compounds. 30,31 Present study showed that population in Urfa, Southeast Anatolia, had a high risk for melamine biohazards and there is a need to further investigate this area with detailed pharmacokinetic and pharmacodynamic studies in long-term follow-up cohort studies. Another limitation is the small sample size, 90 mothers with 180 breast milk samples. However, this study is a descriptive study with a convenience sample. As a strength of the study, two samples from each case were also taken with a 1-month interval, showing repeated exposure.
Conclusions
Women might have an exposure of melamine throughout their life including lactation period and melamine can pass through milk. Incidents of melamine contamination have become an international problem, risking the public health all over the world. This study with a convenient sample did not detect any effect on maternal psychopathologies, maternal bonding and infantile crying and sleeping problems. On the other hand, maternal repeated melamine exposure and transfer through breast milk lead to a negative impact on the height of her infant. Similarly, lower infant birth weight of mothers with repeated exposure could support this hypothesis. In addition, the mean value of maternal white blood cells in mothers with repeated exposure was lower than cases with no exposure. Therefore, melamine exposure of lactating women should be prevented. Breast milk screening can easily evaluate the environmental risks for mother–infant pairs. Further studies including environmental history and food sampling will be helpful in revealing the source of melamine contamination.
Footnotes
Acknowledgement
The authors are grateful to lactating mothers supplying breast milk.
Data accessiblity
Research data can be requested from the corresponding author.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The funding organization(s) played no role in the study design; in the collection, analysis and interpretation of data; in the writing of the report or in the decision to submit the report for publication.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Hacettepe University, School of Medicine Institutional Review Board (project number: GO 17/687) approved the protocol.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Hacettepe University Scientific Research Projects Coordination Unit [Project No. THD-2017-15742].
Informed consent
Informed consent was obtained from parents included in the study.
