Abstract
Background:
Cadmium is an environmental pollutant which can induce the overproduction of free radicals while suppressing the antioxidant defense system. Curcumin is considered a free-radical scavenger and a potent antioxidant. This study was conducted to investigate the effect of curcumin on serum antioxidant enzymes and histopathological changes in mice treated with cadmium.
Methods:
In this experimental study, adult mice were divided into four groups, namely, control, cadmium chloride (5 mg kg−1), curcumin (100 mg kg−1), and curcumin+cadmium chloride. The animals received curcumin 24 h prior to cadmium chloride injection. After 24 h, blood samples were collected and used to assess the levels of malondialdehyde (MDA), antioxidant enzymes activity (catalase, superoxide dismutase, and glutathione peroxidase), total glutathione, total thiol, and hydrogen peroxide. Histopathological evaluation was also performed for testicular tissue.
Results:
Mice treated with cadmium showed a significant (p < 0.001) decrease in the activity of antioxidant enzymes, serum amounts of total glutathione and total thiol, and the diameter of seminiferous tubules compared to the control group. This pollutant also significantly (p < 0.001) increased serum levels of MDA and hydrogen peroxide and the lumen diameter of seminiferous tubules compared to the control group. In the curcumin+cadmium group, curcumin significantly (p < 0.001) reversed the adverse effects of cadmium, compared to the cadmium group. In addition, curcumin alone significantly (p < 0.001) increased serum glutathione peroxidase activity and thiol content compared to the control group.
Conclusion:
Curcumin, as a potent antioxidant, could compensate the adverse effects of cadmium on lipid and protein peroxidation, potentiated serum antioxidant defense system, and ameliorated some morphometrical parameters in the testis of cadmium-treated mice.
Introduction
Cadmium is one of the most toxic environmental pollutants, which is found at low concentrations in the Earth’s crust. 1,2 The major sources of cadmium contamination are industrial and agricultural processes. In addition, coal and mineral oil combustion, smelting, mining, alloy processing, paint industries, phosphate fertilizers industries/applications, and cigarette smoke contribute to cadmium contamination. 3 –5 Therefore, humans and animals may be exposed to this pollutant through ingestion of contaminated water and food as well as inhalation. 6 In humans, cadmium causes a wide variety of conditions such as osteoporosis, anemia, hepatotoxicity, nephrotoxicity, and carcinogenicity. 2,7 Experimental evidence showed that cadmium by inducing oxidative stress increases lipid peroxidation and decreases antioxidant enzyme activities such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx). 8,9
Since cadmium exerts its deleterious effects in the testis by induction of oxidative stress, 7,10,11 antioxidants can potentially prevent cadmium-induced effects. Curcumin, an active compound of turmeric, has a wide range of pharmacological and biological activities. 12 –14 Curcumin was shown to possess powerful radical-scavenging antioxidant properties and increase intracellular glutathione (GSH). 12,15 Curcumin can also reduce lipid peroxidation by enhancing the activities of antioxidant enzymes. 16
Considering cadmium-induced oxidative stress and the potent antioxidant properties of curcumin, the present study was conducted to investigate the possible protective effects of curcumin against the adverse effects of cadmium on testis histopathology and serum levels of oxidative stress markers and antioxidant enzymes activity in mice.
Methods
Animals and treatments
In this experimental study, adult male NMRI mice (32 ± 5 g) were purchased from Pasture Institute, Tehran, Iran. The animals were housed in plastic cages with 12-h light/12-dark cycles, at 24°C ± 2°C and water and food available were available ad libitum. The mice were divided into the following four groups (n = 6 per group): control, cadmium chloride (5 mg kg−1; Merck, Germany), curcumin (100 mg kg−1; Sigma, Louis, USA), and curcumin + cadmium chloride. The animals were first injected with a single intraperitoneal dose of curcumin; 17,18 after 24 h, the mice received cadmium chloride subcutaneously. 19 The experiments were approved by the local ethical committee at Arak University. Cadmium chloride and curcumin were dissolved in distilled water and dimethyl sulfoxide (DMSO), respectively. Since two different solvents were used, two groups were administered with either distilled water or DMSO; however, no significant difference was found between these two groups. So, the mice treated with distilled water alone were considered as the control group. Twenty-four hours after administration of cadmium, the animals were anesthetized by diethyl ether. Blood samples were collected from the heart and centrifuged; then, serum samples were prepared and stored at −80°C for biochemical analyses. Furthermore, the left testis was dissected and fixed for histopathological and histomorphometrical studies.
Tissue preparation and histopathological and histomorphometrical evaluations
The left testes were fixed in Modified Davidson’s fluid fixative (30% formaldehyde, 15% ethanol, 5% glacial acetic acid, and 50% distilled water) for 5 days. The fixed testes were cut into small slices, washed in phosphate-buffered saline (PBS, 3 × 5 min), and incubated in 20% sucrose in PBS at 4°C. Tissue samples were cut into 7-µm thick sections in a cryostat, mounted onto poly-
Assessment of serum MDA levels
Malondialdehyde (MDA) is a final product of oxidative stress-induced lipid peroxidation and is frequently used as an indicator of lipid peroxidation. The amount of serum MDA was measured using the thiobarbituric acid (TBA) assay according to the method described by Turki and Moayad Naji. 20 In brief, 1000 µL of a solution containing trichloroacetic acid (TCA, 15% w/v), TBA (0.375% w/v), and hydrochloric acid (HCl 0.25 N) was added to 500 µL of serum samples and heated for 15 min in a boiling water bath. The samples were then cooled and centrifuged at 1000 g for 10 min. The absorbance of the supernatant was determined using a spectrophotometer at 535 nm. The MDA concentration was calculated by TBA extinction coefficient (1.56 × 105M−1 cm−1) and expressed as nmol mL−1.
Assessment of antioxidant defense enzymes activity
CAT activity
CAT activity was determined using the method described by Aebi. 21 This method was developed based on the ability of the enzyme to decompose hydrogen peroxide (H2O2) into water and oxygen. Briefly, 2 mL of 50 mM potassium phosphate buffer (pH 7) and 1 mL of 50 mM H2O2 were added to 50 mL of serum samples and absorbance was ultimately measured by a spectrophotometer at 240 nm between 0 min and 3 min. The activity of CAT was calculated using the extinction coefficient of H2O2 (43.6 M−1 cm−1) and expressed as U mL−1. 19
SOD activity
SOD activity was assayed by the procedure described by Gavali. 22 Pyrogallol, which is rapidly autoxidized in aqueous solutions, is employed for the estimation of SOD activity. Briefly, 2.8 mL of Tris buffer containing 50 mM Tris buffer, 1 mM ethylenediaminetetraacetic acid (EDTA, pH 8.5), and 0.1 mL of 20 mM pyrogallol was added to 0.1 mL of serum samples. The absorbance was read by a spectrophotometer at 240 nm after 1.5 and 3.5 min; absorbance reading of control without serum was considered as A, and absorbance of sample with serum was considered as B. The activity of SOD was measured using A − B/A ×50 (100 ×10) formula and expressed as U mL−1. 22
GPx activity
The activity of GPx was estimated by the method explained by Rani and Unni KM Karthikeyan 23 with slight modifications. This method is based on GSH oxidation and reduction of H2O2 to water. In brief, 0.2 mL of 0.8 mM EDTA, 0.1 mL of 10 mM sodium azide, and 0.1 mL of 2.5 mM H2O2 were added to 0.2 mL of serum samples and incubated in a water bath at 37°C for 10 min. Then, 0.5 mL of 10% TCA was added to the reaction mixture and centrifuged at 2000 × g for 15 min. Next, 3 mL of 0.8 mM disodium hydrogen phosphate and 0.1 mL of 0.04% 2-2′-dinitro-5,5′-dithiodibenzoic acid (DTNB) were added to the solution and the color intensity was measured by a spectrophotometer at 420 nm. The activity of GPx was computed using the extinction coefficient of DTNB (13600 mol L−1) and expressed as U L−1.
Assessment of serum levels of total thiols
Total thiol concentration in the serum was measured using the reduction of DTNB reagent to create a yellow-colored complex. 24 Briefly, 250 µL Tris buffer (containing 0.25 M Tris and 20 mM EDTA; pH 8.2), 25 µL of 10 mM DTNB, and 1000 µL of absolute methanol were added to 25 µL of serum samples. After 15 min of incubation at room temperature, the samples were centrifuged at 4000 × g for 20 min and the absorbance of supernatant was measured by a spectrophotometer at 412 nm. The thiol content was determined using extinction coefficient of DTNB (13.6 mM) and expressed as mM. 25
Determination of serum GSH levels
GSH status is an important factor for maintenance of cell health and its low level could be an indicator of oxidative stress. GSH concentration was determined using a previously described method. 26 GSH reacts with DTNB, which in turn reacts with sulfhydryl groups, to develop a stable color. In brief, 200 μL of serum samples was mixed with 700 μL of Tris-HCl buffer (400 mM, pH 8.9) and 100 μL of DTNB reagent (2.5 mM DTNB dissolved in 40 mM Tris-HCl buffer, pH 8.9). After incubation at room temperature for 10 min, the optical density of the samples was measured at 412 nm. The concentration of GSH was calculated by extinction coefficient of DTNB (13.6 mM) and serum total GSH was expressed as μM.
Serum H2O2 measurement
H2O2 is one of the reactive oxygen species (ROS) which serves as a key intermediate for a number of oxidative stress conditions. Serum H2O2 level was estimated by the method described by Said et al. 27 In this assay, 100 μL of serum sample, 100 μL of potassium iodide (1 mM), and 100 μL of phosphate buffer (10 mM, pH = 7) were added successively and absorbance was read at 390 nm. The amount of H2O2 in serum samples was calculated using the regression equation (y = 0.001x + 0.023, R2 = 0.995; where y is the measured OD and x is the H2O2 concentration) obtained from the standard curve. H2O2 level was then expressed as μM.
Statistical analysis
Results were expressed as mean ± standard deviation for six animals per group. One-way analysis of variance followed by Tukey’s test was used to assess the statistical significances among the groups. A p < 0.05 was considered significant.
Results
Histopathological and histomorphometrical evaluation of the testis
Normal and uniform structure of seminiferous tubules was observed almost in all tubules in the control group (Figure 1(a)). In the cadmium chloride-treated group, degenerative changes were generally seen. Seminiferous tubule’s wall was shrunken, disorganized, and less compact; also, vacuolation of the seminiferous epithelium was observed. The cadmium chloride group, compared to the control group, indicated a statistically significant (p < 0.001) reduction in the mean diameter of seminiferous tubules and a significant (p < 0.001) increase in the diameter of seminiferous tubule’s lumen (Table 1 and Figure 1(a) and (b)). In the cadmium chloride + curcumin group, curcumin protected the testis against cadmium-induced histopathological damages (Figure 1(b) and (d)). Moreover, the findings of this study showed that pretreatment with curcumin can reverse the toxic effects of cadmium chloride on the mean diameter of seminiferous tubules and seminiferous tubule’s lumen (Table 1).

Testicular tissue in mice treated with cadmium chloride (5 mg kg−1) and curcumin (100 mg kg−1). Normal structure of seminiferous tubule with regular arrangement of germinal epithelium in: (a) control; (c) cadmium chloride + curcumin; and (d) curcumin groups. Tubular lumen was also filled with sperm in (b) cadmium chloride group: irregular and vacuolated (arrow) germinal epithelium, increased diameter of tubular lumen and lack of sperm compared to the control group. Heidenhain’s Azan staining. Magnification: ×400.
Testis histomorphometrical parameters in mice treated with cadmium chloride (5 mg kg−1) and curcumin (100 mg kg−1).a
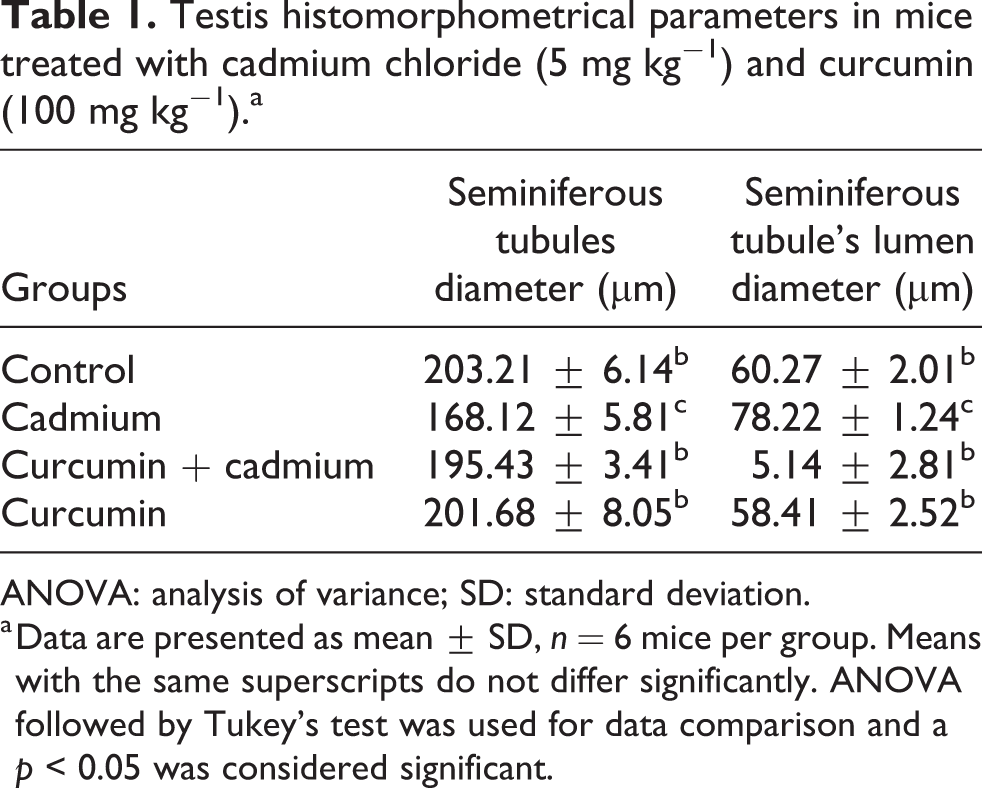
ANOVA: analysis of variance; SD: standard deviation.
a Data are presented as mean ± SD, n = 6 mice per group. Means with the same superscripts do not differ significantly. ANOVA followed by Tukey’s test was used for data comparison and a p < 0.05 was considered significant.
Evaluation of MDA concentration
Serum level of MDA in the cadmium chloride group was significantly (p < 0.001) higher than that of the control group. In the curcumin + cadmium chloride group, curcumin was able to significantly (p < 0.001) prevent the increment of MDA level compared to the cadmium chloride group. Animals treated with curcumin alone showed no significant difference in the MDA levels compared to the control group (Figure 2).

Level of serum MDA in mice treated with cadmium chloride (5 mg kg−1) and curcumin (100 mg kg−1). Data are presented as mean ± SD, n = 6 mice per group. Means with the same letters do not differ significantly. ANOVA followed by Tukey’s test was used for data comparison and a p < 0.05 was considered significant. ANOVA: analysis of variance; MDA: malondialdehyde; SD: standard deviation.
Evaluation of antioxidant defense enzymes activities
Serum activities of CAT, SOD, and GPx in the cadmium chloride group were significantly (p < 0.001) lower than those of the control group. In the curcumin + cadmium chloride group, curcumin could significantly (p < 0.001) restore serum CAT, SOD, and GPx activities compared to the cadmium chloride group. In mice treated with curcumin alone, the activities of GPx were significantly (p < 0.001) higher than those of the control group (Table 2).
Serum activities of antioxidant defense enzymes, CAT, SOD, and GPx in mice treated with cadmium chloride (5 mg kg−1) and curcumin (100 mg kg−1).a

ANOVA: analysis of variance; CAT: catalase; SOD: superoxide dismutase; GPx: glutathione peroxidase; SD: standard deviation.
a Data are presented as mean ± SD, n = 6 mice per group. Means with the same superscripts do not differ significantly. ANOVA followed by Tukey’s test was used for data comparison and a p < 0.05 was considered significant.
Evaluation of serum total thiols
Total thiols content was significantly (p < 0.001) lower in cadmium chloride group compared to the control group. A significant (p < 0.001) increase was found in the total thiols level in the cadmium chloride + curcumin group compared to cadmium chloride group. Additionally, mice treated with curcumin alone showed a significant (p < 0.001) increase in the total thiols level compared to the control group (Figure 3).

Serum total thiols levels in mice treated with cadmium chloride (5 mg kg−1) and curcumin (100 mg kg−1). Data are presented as mean ± SD, n = 6 mice per group. Means with the same letters do not differ significantly. ANOVA followed by Tukey’s test was used for data comparison and a p < 0.05 was considered significant. ANOVA: analysis of variance; SD: standard deviation.
Evaluation of serum GSH levels
In the cadmium chloride group, the total GSH level was significantly (p < 0.001) lower than that of the control group. Cadmium chloride + curcumin group showed a highly significant (p < 0.001) increase in serum GSH levels compared to the cadmium chloride group. However, no significant difference was found between the curcumin and the control group (Figure 4).

Serum total GSH levels in mice treated with cadmium chloride (5 mg kg−1) and curcumin (100 mg kg−1). Data are presented as mean ± SD, n = 6 mice per group. Means with the same letters do not differ significantly. ANOVA followed by Tukey’s test was used for data comparison and a p < 0.05 was considered significant. ANOVA: analysis of variance; GSH: glutathione; SD: standard deviation.
Evaluation of serum hydrogen peroxide
The level of serum hydrogen peroxide was significantly (p < 0.001) increased in the cadmium chloride group compared to the control group. Curcumin significantly (p < 0.001) ameliorated this elevation in the cadmium chloride + curcumin group compared to the cadmium chloride group. In addition, hydrogen peroxide concentration in the curcumin-only treated group was not significantly different from that of the control group (Figure 5).

Serum H2O2 levels in mice treated with cadmium chloride (5 mg kg−1) and curcumin (100 mg kg−1). Data are presented as mean ± SD, n = 6 mice per group. Means with the same letters do not differ significantly. ANOVA followed by Tukey’s test was used for data comparison and a p < 0.05 was considered significant. ANOVA: analysis of variance; H2O2: hydrogen peroxide; SD: standard deviation.
Discussion
In this study, cadmium chloride caused several histopathological and histomorphometrical damages in the testis by inducing oxidative stress while pretreatment with curcumin could protect the testis against toxic effects of this environmental pollutant. Histopathological changes such as disruption and vacuolation of seminiferous tubules and low sperm count (spermatozoa were not counted) in seminiferous tubules were observed in the cadmium chloride group, which were also shown in previous studies. 28 –31 Several mechanisms may mediate the toxic effects of cadmium chloride on the testis. Several line of studies have indicated that cadmium-induced histopathological insults are mediated through disruption of cell junctions, cytoskeleton, and the blood–testis barrier by modulation of specific signal transduction pathways and signaling molecules. 32,33 Furthermore, cadmium chloride by disruption of kinase and phosphatase activity can influence androgen function that is essential for the maintenance of spermatogenesis. 32 Moreover, cadmium is known as an endocrine disruptor that interferes with the synthesis or regulation of different hormones, which in turn may lead to testicular damage. 30 Due to the similarity between cadmium and calcium, cadmium is able to substitute for calcium. 30 Thus, another approach to combat cadmium-induced testis damage might be based on the interaction between cadmium and calcium-binding molecules such as calmodulin. 30 Another mechanism by which cadmium exerts its testicular toxicity is oxidative stress. 34 Since testicular tissue is rich in polyunsaturated fatty acids in membrane lipids, cadmium exposure may cause severe oxidative damage in this organ. 7,35 In biological systems, several molecules can be considered as the indicator/marker of occurrence of oxidative stress. Among these, MDA is an important indicator for membrane lipid peroxidation and elevated levels of MDA were documented following exposure to cadmium. 36 Also, cadmium chloride-induced oxidative stress may take place as a result of its binding to sulfhydryl groups of proteins and depleting GSH and other antioxidant agents. 35,37 GSH maintains protein thiols at an appropriate level 38 and reduced levels of GSH can cause ROS-mediated oxidative damage of proteins in testicular tissues. Thus, it is possible that cadmium exerts protein oxidative damage by decreasing the GSH levels. 37
In the present study, we hypothesized that severe histopathological and histomorphometrical changes induced by cadmium chloride result from oxidative stress and/or suppression of the antioxidant defense system. To test this hypothesis, we examined several specific indices of oxidative stress in the serum of mice treated with cadmium chloride. Significant increase in the MDA (as a marker of lipid peroxidation), H2O2 (as a ROS) and significant decrease in the activity of antioxidant defense enzymes (such as CAT, SOD and GPx), the amount of total glutathione as well as total thiol content could be a support for our hypothesis. In addition, our previous findings showed that cadmium significantly reduces total serum antioxidant power (as measured by FRAP) in mice. 39
The marked reduction of CAT activity might result from metal deficiency. Cadmium can interact with divalent cations, such as iron, and induce iron deficiency. 40 Iron is the structural component of CAT active site; thus, iron deficiency might be responsible for the reduction of CAT activity. 4 Therefore, cadmium causes a reduction in CAT activity which may result from decreased absorption of iron and inhibition of heme biosynthesis. 35 The interactions between cadmium and manganese (Mn), zinc (Zn), and copper (Cu) in SOD might be regarded as the mechanism underlying cadmium toxic effects on SOD activity. Cadmium can substitute for divalent ions (Zn2+, Cu2+, and Mn2+) present in many metalloenzymes and inhibit them. In addition, this pollutant has a high affinity for −SH groups in proteins, enzymes, and nucleic acids and may block their enzymatic activities. 35 There is a strong relationship between GPx activity and GSH concentration. In addition, selenium and zinc (cofactors for SOD) protect GSH. 5 Cadmium binds cysteine in GSH and deactivates GPx. 41 This heavy metal also interacts with selenium leading to disruption of GPx activity. 35 Since GPx is a selenium-containing enzyme, selenium depletion (by cadmium) may reduce GPx activity. 4,42 Decrement of total thiols is an important marker of oxidative stress effects on proteins. Thiols interact and can eventually neutralize hydroxyl radicals and nitric oxide metabolites. 43 Since cadmium induces the formation of free radicals and ROS, 44 it may also be involved in the reduction of serum thiol levels. Additionally, cadmium adverse effects on thiol production may be due to its high affinity for –SH groups. 35 Cadmium exposure results in a significant rise in the serum H2O2 concentration after 24 h. H2O2 is generated by NADPH oxidases or Fenton reaction. When oxidative stress is rather weak, antioxidant enzymes including CAT, SOD, and GPx are activated to scavenge ROS. However, SOD and GPx activities are decreased with increasing cadmium exposure time (approximately 24 h) 2 as indicated in the present study.
If oxidative stress is responsible for the hazardous effects of cadmium, an antioxidant may reverse the adverse effects of cadmium chloride. In this context, we treated the animals with a potent antioxidant, curcumin, prior to cadmium chloride administration, to provide further support for our hypothesis. Interestingly, curcumin not only compensated the toxic effects of cadmium chloride on testicular tissue and antioxidant defense enzymes, but also restored the levels of MDA, GPx activity, and total thiol in serum. Curcumin is a polyphenolic compound which is known as a potent antioxidant 45 and antiapoptotic agent. 31 The presence of the methoxy groups on phenolic rings further increases its activity. 45 Curcumin effectively scavenges free radicals 12,15,46 and thanks to its membrane-stabilizing ability, it can protect the membranes from lipid peroxidation. 47 Furthermore, our recent study revealed the protective effect of curcumin against sodium arsenite-induced kidney toxicity. 48 These results suggest that curcumin can improve the antioxidant defense system status, which in turn alleviates the toxic effects of cadmium chloride. For improving the testis structure, GPx activity and total thiol levels in the curcumin group may support the idea that this antioxidant induces its beneficial effects via boosting the antioxidant defense system. Taken together, it could be suggested that the presence of natural antioxidants in our diet can possibly strengthen the antioxidant defense system of the body to better protect us against oxidative stress induced by environmental pollutants.
Conclusion
The results of this study indicated that curcumin, which has putative antioxidant and free-radical scavenging properties, could counteract testicular tissue damage and serum oxidative stress induced by cadmium chloride.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported by a grant from the research and technology department of Arak University.
