Abstract
Cadmium is a well recognized carcinogenic, cytotoxic and mutagenic transition metal. Recent evidence suggests that the proteins participating in the DNA repair systems, especially in excision and mismatch repair (MMR), are sensitive targets of cadmium toxicity. Microsatellite instability (MSI) is regarded as one of the phenotypes of defective DNA MMR and, consequently, as a marker of high risk for cancer. The purpose of this work is to determine whether cadmium, in the form of cadmium chloride (CdCl2), may induce microsatellite mutations in murine testes. For this study, 2-month-old male ICR-CD1 mice were treated by a single subcutaneous injection of 1, 2 and 3 mg CdCl2/kg body weight and killed after 35 days. A panel of six microsatellite markers, previously reported as being the most sensitive in detecting MSI in murine tumours, was used in this study. The results show that CdCl2 in the doses of 2 and 3 mg/kg induced a decrease in the testis weight and severe histopathologic changes with complete disorganization of testicular structure and evidences of severe necrosis. In addition, the animals exposed to the lowest CdCl2 dose presented MSI in the testis. The results indicate the existence of MSI in at least two nuclear loci suggesting putative genotoxic effects induced by cadmium.
Introduction
Cadmium is a heavy metal and an environmental contaminant, with long environmental persistence and estimated mean half life in humans of 13.6 to 23.5 years. 1 General population may be chronically exposed to cadmium by contaminated food or water intake and by inhalation. 2 As tobacco is usually cultivated in cadmium contaminated soils, tobacco smoke is one of the most common forms of exposure to cadmium. Cadmium concentration in tobacco leaves may vary from 0 to 6.78 μg/g dry weight, and each cigarette may contain 1 to 2 μg of cadmium. 3
Studies about the effects of cadmium in animals indicate that the testis is more sensitive to cadmium toxicity than other organs such as liver or kidney. 4 , 5 The testicular sensitivity to cadmium toxicity may vary among mouse strains. For example, AJ and C3H/HEJ strains are considered resistant, while 129J, DBA/2 J and CD1 strains are considered sensitive to cadmium. 6 Resistance to cadmium toxicity was also associated with the presence of the autosomic recessive gene Cdm, localized in mice 7 chromosome 3.7 Cadmium (Cd2+) ion is similar to other ions such as Fe2+, Cu2+, Mn2+, Co2+, Ni2+ and Pb2+ and may enter cells through ion channels. 8 More recently, Dalton and coworkers 9 identified a specific ion transporter protein, ZIP8, codified by the Slc39a8 gene, which facilitates cadmium accumulation in testis vascular endothelium. This protein is expressed in testicular endothelial cells of sensitive strains and is absent in the resistant strains. 9
Cadmium is classified as a known human carcinogen by the International Agency for the Research on Cancer. 10 Studies in animals also have shown that the administration of cadmium resulted in tumour development in multiple organs and tissues. 11 Nevertheless, cadmium exerts low direct genotoxicity, that is, it does not induce direct DNA damage. It was suggested that the cadmium may induce carcinogenesis through multiple mechanisms such as aberrant gene expression, cadherin disruption, oxidative stress, interference with apoptosis and inhibition of DNA repair (for a review see Refs. 12, 13).
The acquisition of a mutant phenotype allows cells to accumulate mutations required for tumour initiation and progression. Tumours with defective DNA mismatch repair (MMR) acquire deletion and insertion mutations at high frequency, in particular within microsatellite repeat sequences, a molecular phenotype referred to as microsatellite instability (MSI). 14 , 15 Microsatellites, also known as short tandem repeats or simple sequence repeats (SSRs), are short repeated in tandem DNA sequences that involve a repetitive unit of 1–6 bp forming series with lengths of up to 100 nucleotides. Microsatellites are distributed throughout the eukaryotic genomes and are found mainly in noncoding regions, while only about 8% are located in coding regions. Replication of these repeats is error prone due to slippage of DNA polymerase during synthesis, leading to insertion/deletion loops. Repair of these loops is performed by DNA MMR proteins MLH1, MSH2, MSH3, MSH6, PMS1 and PMS2. 16 , 17 Absence or low activity of MMR proteins results in losses and gains of dinucleotide repeats in microsatellites leading to MSI. 18 Thus, MSI can be considered a marker of high risk for cancer. 19 It was previously demonstrated that low doses of cadmium inhibit MMR in yeast and in human cell extracts. This effect is mediated through the inhibition of MSH2–MSH3 20 and MSH2–MSH6 protein complexes. 20 , 21 Banerjee and Flores-Rozas 22 suggest that cadmium inhibits adenosine triphosphate (ATP) binding and hydrolysis activities of MSH2–MSH6. Wieland et al also found that MSH2–MSH6 inhibition occurs through a nonspecific mechanism involving binding of cadmium ions to multiple sites leading to change in protein conformation and loss of function. 21
Despite the association of cadmium to the inhibition of MMR, there are few studies reporting MSI in vivo in human or animal models. Slebos and co-workers 23 evaluated microsatellite mutations in three hypermutable tetranucleotide markers in the human fibrosarcoma cell line HT108 after exposure to cadmium ions. Recently, El-Ghor et al 24 also evaluated MSI in three microsatellite loci (D6mit3, D9mit2 and D15Mgh1) in rats exposed to cadmium. In mice, however, few microsatellites are reported in literature: the commonly used microsatellites for murine tumours are the CAn repeats, for example, D1Mit4, D1Mit36, D3Mit10, D14Mit15, D10Mit2 and D17Mit123. 25 , 26 Kabbarah et al 25 discovered a new panel of microsatellites (TG27, TA27, GA29, CT25CA27, A33 and A27 repeats) that showed more sensitivity in detecting MSI than the six commonly used CAn repeats.
Our previous studies showed that the cadmium at low doses (2 and 3 mg cadmium chloride (CdCl2)/kg body weight (bw)) induced severe histological damage in the testis of mice. 27 Also, sperm function and genetic integrity were affected at these cadmium doses. 28
In the present work, the effects of CdCl2 exposure on the genomic stability of murine testes were evaluated, 35 days after a single administration, using a panel of microsatellites that proved to be sensitive for detecting MSI in murine samples.
Materials and methods
Animals and treatment
Eight weeks old male ICR-CD1 mice were provided by Harlan Interfauna Ibérica SA, Barcelona, Spain. Animals were housed in a constant temperature (22 ± 2°C) and relative humidity (40–60%) vivarium on a 12 h/12 h, light/dark cycle. Water and food were provided ad libitum. Mice were allowed to acclimate for 1 week before experimental use.
Animal experiments were conducted in accordance with the institutional guidelines for ethics in animal experimentation (Directive 86/609/EEC).
Three groups of five mice each were administered with a single subcutaneous injection of 1, 2 and 3 mg CdCl2/kg bw. A control group of five mice was injected with the vehicle (0.9% NaCl). Mice were killed by cervical dislocation 35 days after exposure.
Weight and histological analysis
Mice were killed and the right side testes were removed and weighed. Organ samples were then fixed in Bouin’s solution, dehydrated and embedded in paraffin wax. Transverse sections of 5 μm thickness were prepared using a microtome Leitz 1512 (Leitz, Wetzlar, Germany), stained with haematoxylin and eosin and examined histologically using an Olympus BX 41 microscope (Olympus, Tokyo, Japan).
Microsatellite analysis
For MSI analysis, samples of the left side testes (20–30 mg) were kept at −80°C until DNA extraction. Total genomic DNA was extracted from mouse testis samples with the DNeasy® Tissue kit (QIAGEN, Germany) following the manufacturer’s instructions. DNA concentration and purity were estimated by electrophoresis on a 0.8% (w/v) agarose gel stained with ethidium bromide (EB), comparing with a standard molecular weight marker (lambda Hind III, NEB, UK).
Microsatellite mutations were detected using a panel of six molecular markers developed by Kabbarah et al (Table 1). 25 These markers proved to be highly sensitive in detecting MSI in murine tumours. 25 The forward primers were fluorescently labelled (ABI dyes: 6-FAM or HEX).
Characteristics of the SSRs used: SSR key, locus, motif of cloned allele and primer sequences 25

F: forward primer; R: reverse primer; SSR: simple sequence repeat.
Polymerase chain reactions (PCR) were performed in a 10μL volume containing 1–20 ng of genomic DNA, 1.5 mM MgCl2 reaction buffer (Roche Diagnostics Corp., USA), 0.8 μM of each primer and 0.4 U of Taq. DNA polymerase (Roche Diagnostics Corp., USA). DNA amplification was carried out in a Thermo Hybaid PX2 PCR Thermal Cycler according to the cycling profile described by Kabbarah et al. 25
After the PCR amplification, the PCR products were separated by electrophoresis in 2% agarose gels, stained with EB and visualized on a UV transilluminator G:Box (Syngene, Cambridge, UK). The software GeneSnap was used for image acquisition and analysis. Fragment analysis reactions were prepared by mixing 1 μL of each PCR product with 0.5 μL of GeneScan internal size standard labelled with ROX (Applied Biosystems, USA) and 25 μL of deionized formamide and the mixture was briefly vortexed. PCR products were visualized by capillary electrophoresis on an ABI Prism 310 Genetic Analyser (PE Biosystems). Fragment sizes were calculated automatically to two decimal places using the Local Southern Method option of the GeneScan v.3.1 software.
From the electropherogram analyses, samples were classified as MSI-high, if MSI level was higher or equal to 40%, or MSI-low, if MSI levels were lower than 40%. All the remaining samples, not showing MSI, were considered as microsatellite stable (MSS). This classification was done according to the guidelines suggested by the National Cancer Institute (NCI) Workshop-Revised Bethesda Guidelines.29,30
Statistical analysis
Weight results are expressed as mean ± SD. Statistical comparisons were performed using SigmaStat for Windows (Version 3.1, SPSS Inc., USA). Comparisons between the weights of control and CdCl2-treated groups were performed using one-way analysis of variance, followed by the Tukey’s test. The level of statistical significance was set at p ≤ 0.05.
Results
Body and testis weights
CdCl2, in the doses tested in this assay, did not affect body weight of mice killed 35 days after administration (Table 2). On the other hand, testis weight was highly affected and a significant decrease in the absolute and relative testis weight was found in mice exposed to either 2 or 3 mg CdCl2/kg bw (p < 0.001). More specifically, relative testis weight decreased 53.3 and 83.3% in mice exposed to 2 and 3 mg CdCl2/kg bw, respectively.
Effect of CdCl2 on mice body and testis weights and on testis/body weight ratioa

CdCl2: cadmium chloride.
aValues represent mean ± SD
bFor the same parameter, these values indicate significant difference between the control and 1, 2 and 3 mg CdCl2/kg bw-treated samples at p < 0.001
Histology
Figure 1 shows the effects of CdCl2 exposure on testis histology. Testis was highly affected by CdCl2 exposure, particularly in the doses of 2 and 3 mg CdCl2/kg bw. For the dose of 1 mg CdCl2/kg bw, no histopathologic changes were detected in the analysed testes. The higher concentrations of CdCl2 (2 and 3 mg/kg bw) provoked severe alterations in the testis, namely severe necrosis with complete disorganization of the testicular structure, disruption and loss of the germinative epithelium and the centrally located spermatids in the seminiferous tubules.

Representative histological sections of (a) control testis showing normal spermatogenesis and well-organized distribution of cells in the seminiferous epithelium (SC: Sertoli cell; es: elongating spermatid; sp: spermatocyte) and the interstitial Leydig cells (LC); sections of testis following cadmium chloride (CdCl2) treatment 35 days after exposure to (b) 1 mg CdCl2/kg bw showing histology similar to control, (c) 2 mg CdCl2/kg bw and (d) 3 mg CdCl2/kg bw showing testicular necrosis (
Microsatellite analysis
PCR products were analysed by capillary electrophoresis and fragment sizes from testes of control and CdCl2 exposed mice were scored and compared. From the six markers tested, only five (Mus 1, Mus 3, Mus 4, Mus 5 and Mus 6) gave consistent and successful amplifications. Therefore, marker Mus 2 was not considered for the analysis. Uniform microsatellite patterns were found for all markers in the control group mice. The allele sizes of the SSRs analysed in the control group mice were 137, 141, 140, 141 and 123 for Mus 1, Mus 3, Mus 4, Mus 5 and Mus 6 markers, respectively (Table 3). As an example of the results obtained with the capillary electrophoresis apparatus, a set of electropherograms is presented in Figure 2(a) and (b), showing the amplification products of PCR of markers Mus 1 and Mus 5, respectively.

Examples of microsatellite instability observed in mouse samples exposed to cadmium chloride (CdCl2). Size of the predominant allele (shown in solid) of control samples was compared with those of CdCl2 exposed samples. Deletions of 2 and 1 bp were detected in markers Mus 1 (a) and Mus 5 (b), respectively. Top scale indicates fragment size in nucleotides. Left scale indicates fluorescence intensity measured in relative fluorescence units. Samples T1–T5 represent testis samples from mice exposed to 1 mg of CdCl2/kg bw.
Allele size (bp) of the control samples of testis

Allelic patterns of cadmium exposed samples were compared with those of control samples and were scored as MSI-positive, if one or more novel alleles (mutations) were found. Table 4 presents the samples that were scored as MSI-positive. All samples from the group of mice exposed to 1 mg CdCl2/kg bw were MSI-positive. Mus 1 detected 2 bp deletions in four of five samples and Mus 5 detected 1 bp deletion in three samples. Interindividual variations in MSI were observed for both Mus 1 and Mus 5. The MSI-positive samples were run twice to verify that it was not an artefact.
Analysis of samples displaying MSI (mutations) with the tested simple sequence repeata
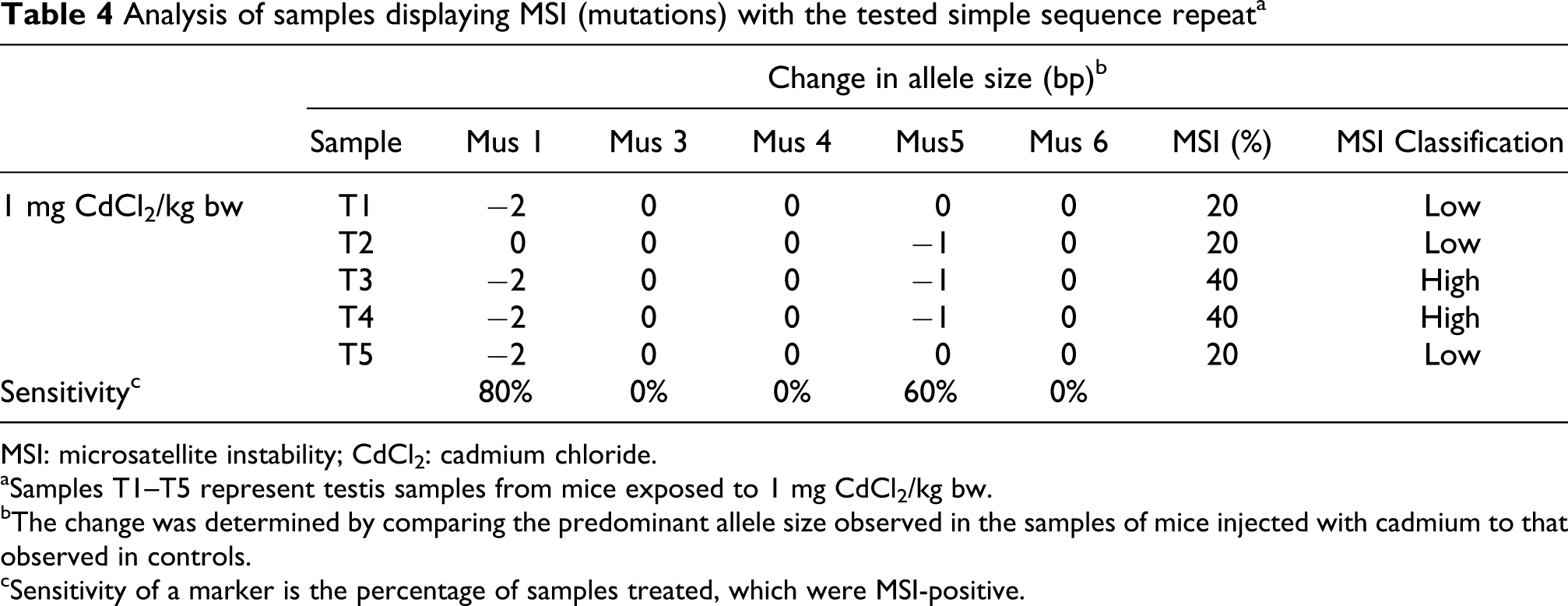
MSI: microsatellite instability; CdCl2: cadmium chloride.
aSamples T1–T5 represent testis samples from mice exposed to 1 mg CdCl2/kg bw.
bThe change was determined by comparing the predominant allele size observed in the samples of mice injected with cadmium to that observed in controls.
cSensitivity of a marker is the percentage of samples treated, which were MSI-positive.
Two of the MSI-positive samples (T3 and T4, exposed to 1 mg CdCl2/kg bw) can be classified as MSI-high (MSI ≥ 40%), whereas the others are termed MSI-low (MSI < 40%). All the remaining samples, from the groups of mice exposed to 2 and 3 mg CdCl2/kg bw did not show MSI and, thus, were considered as MSS. This classification was done according to guidelines suggested by the NCI Workshop-Revised Bethesda Guidelines.29,30
Discussion
In the past years, microsatellite polymorphisms have assumed to play a major role in genomic analysis, with applications as diverse as positional cloning, tumour biology, human evolution, medical diagnostics and forensic identification, and are also gaining interest in toxicological studies as markers of genetic instability.
Our previous studies showed that 24 h after a single subcutaneous dose of 2 and 3 mg CdCl2/kg bw, mice testes were severely injured in the presence of haemorrhage, interstitial oedema and disruption of germinative epithelium. 27 CdCl2 administration also induced an increase in absolute and relative testis weight 24 h after exposure. 27 The present work showed that after one spermatogenic cycle these lesions were not reversible and even progressed to the degeneration of the seminiferous tubules, together with germ cell depletion and testicular necrosis. These lesions are the result of the multiple action of cadmium on the testis: (i) irreversible lesions induced by cadmium on the blood–testis barrier (for a review see Refs. 31, 32); (ii) induced increase in vascular permeability leading to oedema and haemorrhages, with consequent anoxia. 33 Thus, testicular necrosis occurred as a result of ischemia induced by increased permeability of blood vessels, increase of intratesticular pressure and decreased blood flow to the testis. These necrotic and degenerative lesions induced by CdCl2 in the testis also explain its weight decrease and were previously described by Nordberg. 34 A decrease in testis weight was also detected in rats 35 and mice 36 after a prolonged exposure to cadmium.
Our previous work has also shown that cadmium rapidly accumulates in the testis after a single dose administration. 28 In that study, mice injected with 1, 2 and 3 mg CdCl2/kg bw and killed 24 h after administration, presented cadmium levels in the testis of 185, 226 and 304 times to those of controls. 28 Cadmium presents high affinity to metallothioneins, small cystein rich molecules that sequester and accumulate metals and contribute to its inactivation and detoxification. The liver is particularly efficient in the production of metallothioneins, while the testis has low ability to produce these molecules, 5 which explains the highest sensitivity to cadmium. This high accumulation of cadmium in the testis plus the low capacity of detoxification also contributes to this high level of injury.
In the present work, exposure to a single dose of 1 mg CdCl2/kg bw changed the MMR mechanism and induced MSI in the testis. Although the markers tested showed different sensitivities, two of them (Mus 1 and Mus 5) allowed the detection of MSI. Mus 1 showed the highest level of sensitivity, since it detected MSI in four of five samples analysed, while Mus 5 detected MSI in three samples. Interindividual variations in MSI were observed for both Mus 1 and Mus 5. As the animals were of the same age and were maintained exactly in the same conditions, we conclude that these differences are due to natural interindividual variability. Despite the severe histopathologic symptoms induced by 2 and 3 mg CdCl2/kg bw in mice testis, no MSI was detected as measured by the selected panel of microsatellite markers under the in vivo conditions tested. This effect may be explained by the tissue necrosis found in both cadmium doses. Furthermore, our previous work 27 has shown that after 24 h, the doses of 2 and 3 mg CdCl2/kg bw induced complete disruption of germinal epithelium, haemorrhage and interstitial oedema, leading to cell death. Under these circumstances, DNA replication does not occur. As MSI occurs due to both the errors during DNA replication and subsequent absence of repair by MMR enzymes, 19 it is proposed that without DNA replication, these mutations will not occur, supporting the findings of the present work. Overall, our data suggest that at doses of 2 and 3 mg/kg bw, cadmium toxicity resulted in cell death that may have precluded MSI formation.
Recent studies performed by El-Ghor and co-workers 37 focusing on the effects of cadmium in rats using a panel of three microsatellite loci showed that the acute exposure did not induce MSI in blood cells, while after subchronic exposure, MSI was observed in all loci. Slebos and co-workers 38 also showed that the exposure of a human fibrosarcoma cell line (HT1080-C1) to cadmium leads to an increase in MSI. The results of these authors provide evidence for the presence of low levels of cadmium in human cells suggesting that the cadmium is involved in the inhibition of DNA repair. Zhou and coworkers tested a lower CdCl2 dose (5 μmol/kg ≅ 0.926 mg/kg bw) and did not find histological changes in the testes; however, they found that the inhibition of the expression of some genes are related with DNA repair. 39 Another study performed by Jin and coworkers found that the chronic exposure to low, nonlethal doses of CdCl2 (3 and 5 μM) caused a substantial increase in mutability (about 2000-fold) and may inhibit MMR. 40 MMR is involved in the correction of replication mistakes, that if being unrepaired have tumorigenic and mutagenic potential. The cadmium-induced inhibition of DNA repair may be due to its effects on enzymes responsible for genome stability, DNA repair and DNA damage signalling. Several of these enzymes are the members of the zinc finger family of proteins, 41 and as it is known, cadmium competes with zinc and can substitute zinc in those enzymes, interfering with their efficiency as DNA repair enzymes.41,42 Beyond mutations in MMR genes, MMR can also be affected through an epigenetic process in which DNA MMR can be inactivated by the hypermethylation of the promoter that silences hMLH1 transcription. 43 Supporting this hypothesis are the studies of Jiang and coworkers, 44 which showed that the long-term exposure to low levels of cadmium is able to increase the levels of DNA methylation.
The present study demonstrates the efficacy of a panel of dinucleotide markers for the detection of MSI in mouse testis. MSI was detected in mouse testis exposed to low cadmium doses in two of five tested markers. In fact, two samples were classified as MSI-high as they presented changes in two of five markers tested. Therefore, the initial hypothesis that cadmium induced MSI in mice testis was confirmed. The presence of this mutant phenotype in the samples of testis of CdCl2 exposed mice may be an indicative of an increased risk for tumour development.
In conclusion, MSI evaluation should be considered as an important approach in the evaluation of metal induced genotoxicity not only in the mice but also in other mammals including humans.
Footnotes
Funding
The work of H. Oliveira was supported by FCT (grant reference SFRH/BPD/48853/2008). This work was supported in part by Centre for Research in Ceramics and Composite Materials from Aveiro University (Portugal).
