Abstract
The purpose of this study is to histologically and immunohistochemically determine the changes created by grayanotoxin-III (GTX-III), which is a sodium channel neurotoxin, on heart tissues in different dosages. Rats were randomly divided into 10 groups to determine the acute and chronic effects of GTX-III. While the rats in groups 1 and 6 were control rats, the rats in groups 2–5 (1, 2, 4, and 8 μg/kg bw GTX-III) received a single dose of intraperitoneal GTX-III, and the rats in groups 7–10 received GTX-III every day for 3 weeks. As a result of the trial, in the heart tissues, histopathological changes were determined by hematoxylin–eosin staining, interleukin-1 (IL-1β), interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), and brain natriuretic peptide (BNP) were determined by the avidin–biotin peroxidase method, and apoptosis was examined by immunohistochemistry (IHC) analysis and the terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling (TUNEL) staining method. In the immunohistochemistry sense, while the BNP level in the AGTX-III groups did not vary significantly, an increase in dosage significantly increased the IL-6, IL-1β, and TNF-α levels in comparison to the control groups. In their comparison to the control groups, the BNP levels increase and the IL-6 and IL-1β levels decreased in the CGTX-III groups. TUNEL analysis revealed that apoptosis increased in both the acute and chronic groups.
Keywords
Introduction
Grayanotoxin (GTX) is effective especially on the ion channels on the cell membrane. It was reported that the toxic effect of GTX is a result of its interaction with voltage-gated sodium channels. Direct bonding of GTX to the sodium channels on the cell membrane leads to an increase in the sodium permeability of the membrane, and as a consequence of this, long-term depolarization occurs on the cell membrane. 1 GTX leads to presynaptic permeation of Ca+2 through voltage-gated Ca+2 channels by the activation of Na+ channels. The effects of GTX on the cell membrane are observed on both skeletal and cardiac muscles, as well as cells in the peripheral and central nervous systems. 2
The effects of GTX usually emerge as a result of consumption of GTX-containing honey. Rhododendron honey (mad honey) is a type of honey that is formed as a result of the collection and maturation of the nectars of the plants
Materials and methods
Formation of the experiment groups
Standard GTX-III was supplied as GTX-III Hemi (ethyl acetate) from Sigma-Aldrich (St Louis, Missouri, USA). The study was carried out on 100 male rats of the
Groups on the acute effects
Group 1: Control: no GTX-III administration, 1 ml saline
Group 2: 0.1 µl/kg bw GTX-III
Group 3: 0.2 µl/kg bw GTX-III
Group 4: 0.4 µl/kg bw GTX-III
Group 5: 0.8 µl/kg bw GTX-III
Each group included 10 animals, and GTX-III was administered i.p. by a single dose. 6,16,17
Groups on the chronic effects
Each group included 10 animals, and GTX-III dosages were administered i.p. for 3 weeks.
18
Group 6: Control: no GTX-III administration, 1 ml saline Group 7: 0.1 µl/kg bw GTX-III Group 8: 0.2 µl/kg bw GTX-III Group 9: 0.4 µl/kg bw GTX-III Group 10: 0.8 µl/kg bw GTX-III
All i.p. injections were made at the same hour of the day. Studies reported that the effects of poisoning in humans were observed after 1 h. 4 Therefore, after 1 h following the administration in the acute experiment groups 6,16,17 and after 3 weeks for the chronic experiment groups, 18 the heart tissues of the rats were collected after anesthesia by ketamine hydrochloride (50 mg/kg intramuscular (i.m.)/i.p.) and 2% xylazine hydrochloride (10 mg/kg i.m./i.p.).
Histological analyses
The tissue samples were fixed by a 10% formaldehyde solution to be used in the histological analyses, routine tissue examination stages were applied on the tissues, and they were embedded in paraffin blocks. Cross-sections of the paraffin blocks with thicknesses of 5–6 µm were stained by hematoxylin–eosin (H&E) and examined under an Olympus BX51 light microscope.
Immunohistochemistry analyses
In order to determine the differences in the expression of IL-1β, IL-6, TNF-α, and BNP in heart tissues, marking was carried out with the avidin–biotin (AB) peroxidase method. For this, the 5–6-µm cross-sections that were obtained were kept for a night at 60°C, cut first, then rehydrated by passing through graded alcohol series, they were washed for 5 min three times by a phosphate-buffered saline (PBS) and boiled in a microwave with a 5% citrate buffer at 600 W 3 × 5 for reuptake of antigens. The cross-sections, that were left in the same buffer solution for 20 min at room temperature and washed again with PBS, were treated for 5 min with 3% hydrogen peroxide to prevent endogenous peroxidase, and an avidin-biotin complex (ABC) staining system kit was used for further processes. Large Volume Ultra V Block was applied to the cross-sections to block non-specific staining, then we washed again with PBS at room temperature for 20 minutes. Immediately after this, the cross-sections were kept at +4°C for one night after dripping primary antibodies (separately for IL-1β, IL-6, TNF-α, and BNP) on them and incubated for 20 min the next day. As a negative control, PBS was used in the place of primary antibodies. After the washing process, the cross-sections were incubated for 30 min with biotinylated secondary antibodies, and the washing process was repeated. Moreover, the cross-sections that were treated with AB enzyme marker for 30 min were washed, and after they were treated for 5 min with the peroxidase substrate in the kit then showed diaminobenzidine characteristics in order to make the immunoreactivities apparent, they were washed with deionized H2O, and the cross-sections that were counterstained by Gill hematoxylin were washed a few times with deionized H2O. As the final step, the cross-sections that were passed through xylene by replacing the water with increasing alcohol gradients were closed with Entellan. The images that were obtained by a DP71 digital camera under an Olympus BX51 light microscope were analyzed by using the Image J 1.48v software in terms of expression differences.
Apoptosis (TUNEL)
In situ terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling (TUNEL) immunofluorescent staining was used for detection of DNA damage. 19 Using a Roche (Mannheim, Germany) in situ Cell Detection Apoptosis Fluorescein Kit, the apoptotic cells in the cross-sections obtained from the subjects were determined. The staining process was carried out based on the procedure of the kit. After the 5-µm thick heart tissue cross-sections were deparaffinized and rehydrated, they were washed two times for 5 min by PBS, and then, kept in a 0.01 M 5% sodium citrate buffer in a microwave at 350 W for 5 min for antigen reuptake. They were then left at room temperature for 10 min for cooling. The tissues that were then washed by PBS two times for 5 min were placed into a humidity chamber with the TUNEL reaction mixture that was in the kit at 37°C, incubated for 60 min in the stove, and then, counterstaining was made on the tissues that were washed by PBS two times for 5 min using 4′,6-diamidino-2-phenylindole. The tissues that were closed with a closing solution containing glycerol were imaged under an Olympus BX51 fluorescent microscope. For the apoptotic index, with 40× magnification, apoptotic cells were counted in the Image J software from 15 different areas.
Statistical analysis
The SPSS 22 software was used for the statistical analyses. For intergroup comparisons of the obtained expression values and TUNEL positive cell count results, the parametric test of one-way analysis of variance was used. Post hoc Tukey’s test was used for multiple comparisons.
Results
Histological analysis results
As a result of H&E staining, the changes in the heart tissues were analyzed. In the acute and chronic 0.1, 0.2, 0.4, and 0.8 µl/kg bw dosage groups, hemorrhage was observed in addition to irregular cell placement (Figure 1).

H&E staining results. Hemorrhage is indicated by arrow. Irregular cell placement is indicated by arrow stars. Magnification: ×400. H&E: hematoxylin–eosin. a) control b) 0,1 c) 0,2 d) 0,4 e) 0,8 microliter of the acute groups; f) control g) 0,1 h) 0,2 i) 0,4 j) 0,8 microliter of the chronic groups.
Immunohistochemistry analysis results
When the BNP immunoreactivity in the acute groups was examined, there was no significant difference in comparison to the control group at low doses (0.1 and 0.2 µl/kg bw), while there was a significant reduction in comparison to the other groups in groups where the dosages of 0.4 and 0.8 µl/kg bw were applied (
Immunohistochemistry data on the heart tissues of the groups that were given acute GTX-III.a

GTX-III: grayanotoxin-III; BNP: brain natriuretic peptide; IL-6: interleukin-6; TNF-α: tumor necrosis factor-
a The data are expressed as mean ± standard deviation.

Immunoreactivity values in the groups that were given acute GTX-III. Immunoreactive areas are indicated by arrows. Magnification: ×400. GTX-III: grayanotoxin-III. a) Immunoreactivity values of BNP and IL-6, b) Immunoreactivity values of TNF alpha and IL-1 beta in the groups that were given acute GTX-III.
Considering the immunoreactivity values of the groups that were given GTX-III chronically, BNP immunoreactivity in the 0.2 µl/kg bw dosage group increased significantly in comparison to the control group (
Immunohistochemistry data on the heart tissues of the groups that were given chronic GTX-III.a

GTX-III: grayanotoxin-III; BNP: brain natriuretic peptide; IL-6: interleukin-6; TNF-α: tumor necrosis factor-
a The data are expressed as mean ± standard deviation.
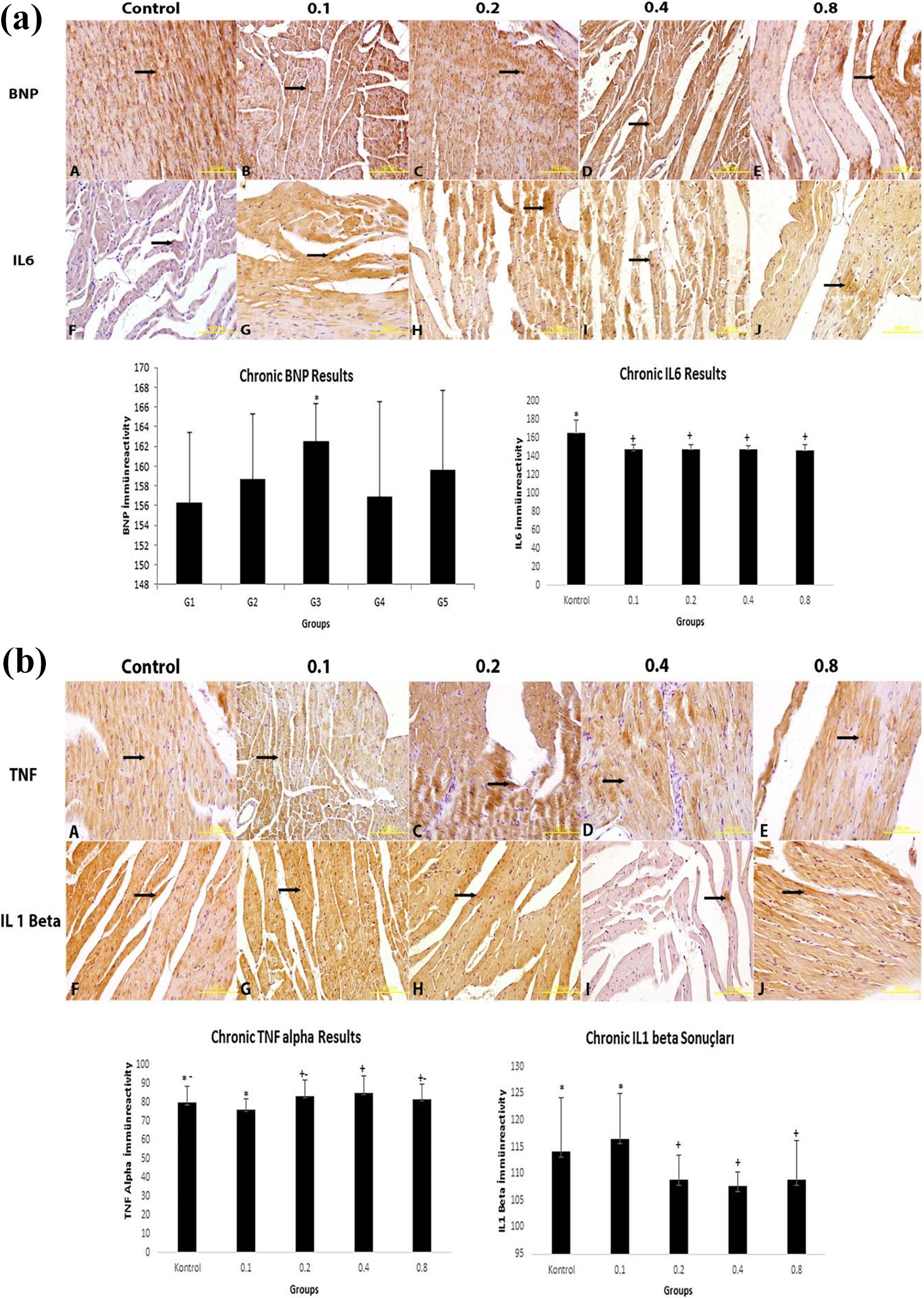
Immunoreactivity values in the groups that were given chronic GTX-III. Immunoreactive areas are indicated by arrows. Magnification: ×400. GTX-III: grayanotoxin-III. a) Immunoreactivity values of BNP and IL-6, b) Immunoreactivity values of TNF alpha and IL-1 beta in the groups that were given chronic GTX-III.
TUNEL analysis
When TUNEL data are evaluated, it is seen that GTX III increases apoptosis in both acute and chronic groups compared to the control group. The rate of apoptosis in chronic groups increased significantly compared to those in the acute group. According to the acute TUNEL data, there was a significant increase in the apoptotic cells in the group that was given 0.8 µl/kg bw GTX-III in comparison to the control group (
TUNEL data of the groups that were given acute and chronic GTX-III in different dosages.

GTX-III: grayanotoxin-III; TUNEL: terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling.
a The data are expressed as mean ± standard deviation.
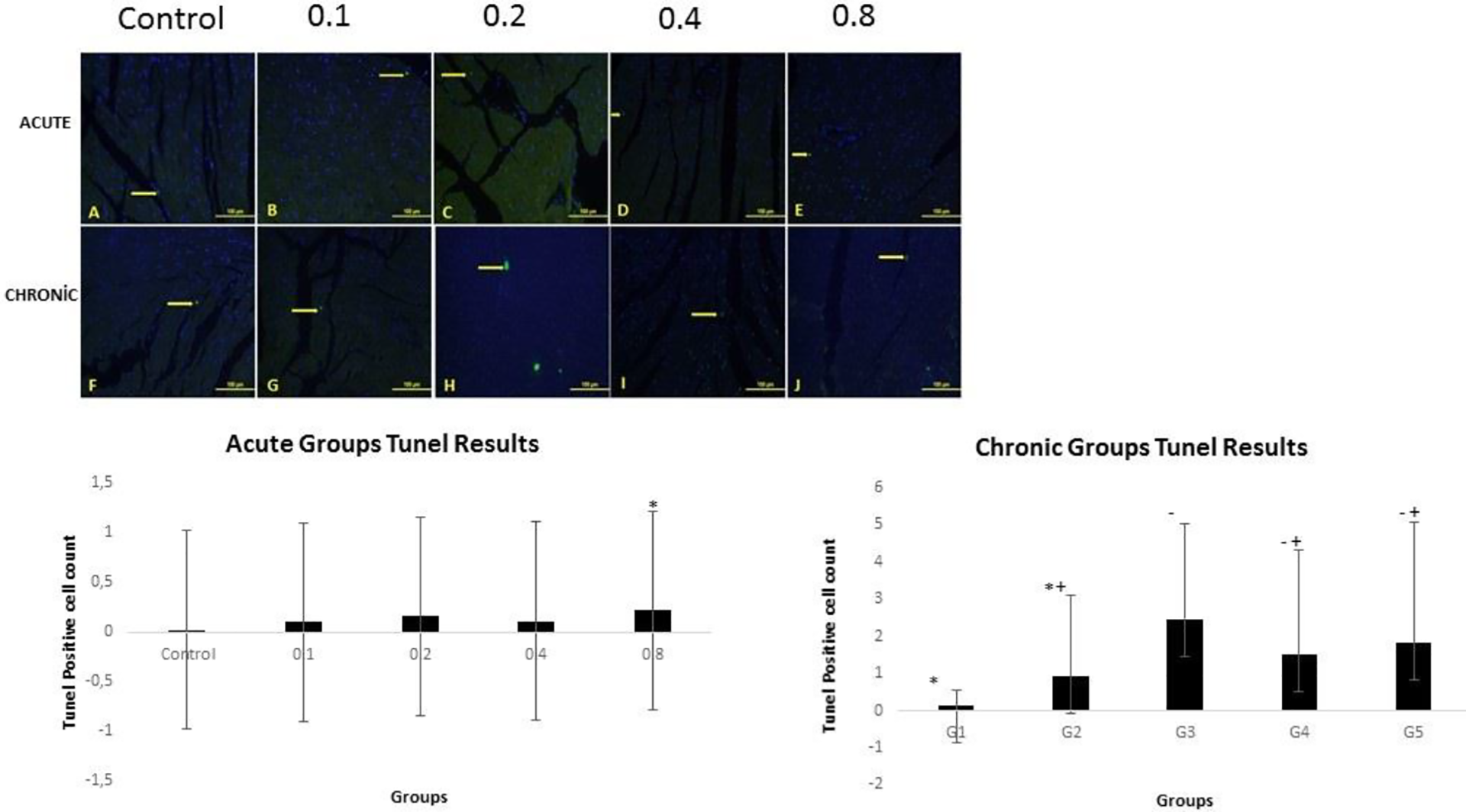
TUNEL data of the groups that were given acute and chronic GTX-III in different dosages. TUNEL: terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling; GTX-III: grayanotoxin-III.
Discussion
Despite their toxicities, rhododendron species are used in traditional systems including Chinese and Indian medicine and folk medicines in Europe and the Americas.
20
Studies on rhododendron extract and plant parts showed its anti-inflammatory, analgesic, antioxidant, antimicrobial, antidiabetic, insecticidal, and cytostatic biologic activities.
7,21,22
GTX is found not only in parts of the plant but also in the pollen and nectar of the plant. Nevertheless, rhododendron honey which is obtained by collecting the nectars of the plants
Although the dose-dependent beneficial effects of plant parts of honey from the nectar of the plant containing GTX have been reported, GTX poisoning takes place rather by the consumption of this honey. There have been several case presentations regarding honey poisoning as a result of the consumption of GTX-containing honey. 7,12 It was reported that the effects of mad honey poisoning on the cardiovascular system emerged in the form of bradycardia or atrioventricular block and cardiac rhythm disorders. 10,11 It was stated that these effects occur as a result of sodium channel activation, cell depolarization, and therefore, the stimulation of the vagal nervous system caused by GTX. 27 According to preclinical research, the voltage-gated sodium channels of neurons are some of the obvious targets of GTX. Bradycardia usually occurs as a result of the consumption of foods that contain GTX. Previous studies reported that the mad honey that is produced in the Black Sea Region in Turkey led to cardiac complications by blocking the sodium channels on the cell membrane. 4 Excessive consumption of rhododendron honey may be observed with cardiac side effects such as bradycardia and severe hypotension, and the modification of Na and K-ATPase channels is shown as the most prominent reason for this.
As a result of histopathologically analyzing the heart tissues of rats that were given acute and chronic GTX-III in different dosages in this study, no significant difference was found in all groups in comparison to the control group except for irregular cell placement and hemorrhage. While the effects of GTX variants are dose-dependent, it was observed that the dosages that were applied did not cause a significant difference on the tissue level.
Na/K modification, which is also known as “cardiotoxicity,” may be measured based on the levels of blood serum bioindicators such as Aspartate amino transferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), creatine kinase (CK), creatine kinase-myocardial bound (CK-MB), troponin T, troponin I, atrial natriuretic peptide (ANP), and BNP. As an important biomarker, BNP concentrations in the plasma increase in cardiac hypertrophy or heart failure based on increased hemodynamic stress or ventricular gap increase. 28 –30 BNP is secreted as a response to increase in ventricular wall stress such as ventricular hypertrophy, and it may be detected at high concentrations in some conditions seen in cardiac ischemia and severe heart failure. 31 In our study, while BNP immunoreactivity did not show a significant change in the groups that were given acute GTX-III, it increased significantly in the groups that were given GTX-III chronically.
In the study by Gao et al., 32 BNP increased in rat myocardial tissue throughout cardiac dysfunction as a result of acute cardiac dysfunction. Korashy et al. 33 determined in vivo and in vitro in rat cardiomyocytes that gefitinib-induced cardiac hypertrophy and cardiotoxicity through the pathways of apoptotic cell death and oxidative stress with increased BNP levels. These data support the idea that increased reactive oxygen species (ROS) formation also increases myocyte apoptosis and toxicity. 34 Indeed, excessive production of ROS may cause not only direct organ damage but also the simultaneous inflammation of inflammatory reactions. Secretion of proinflammatory cytokines and chemokines including the TNF-α, IL-1β, and IL-6 which are the most important factors mediating inflammatory response also trigger the congenital response to tissue damage. 35 Including TNF-α and IL-6, proinflammatory cytokines play a role in the initiation and regulation of inflammatory response. When this study immunohistochemically analyzed the IL-1β, IL-6, and TNF-α levels, while there was a significant increase in the groups that were acutely given GTX-III in comparison to the control group, IL-1β and IL-6 decreased in the groups that were chronically given GTX-III in comparison to the control group, and TNF-α immunoreactivity increased just as BNP. In this case, it is seen that GTX-III varied in relation to its dose-dependent increases in inflammation and oxidative stress, but these changes were significant in the groups that were given acute GTX-III and insignificant in the groups that were given chronic GTX-III.
Apoptosis has a significant role in cardiovascular diseases. 36 –38 It increases cellular oxidative stress which is the main cause of mitochondrial dysfunctions such as ROS increase, saturation of the antioxidant system, and adenosine triphosphate (ATP) exhaustion. Mitochondrial dysfunction leads cells toward programmed cell death. 39 Previous studies demonstrated that an increase in apoptosis accompanied a decrease in cardiac functions. 40,41 It is known that many cardiac events are associated with oxidative stress and apoptosis. 42 Mitochondrial apoptosis induction related to ROS plays a significant role in the mechanisms of cardiac diseases. ROS induces secretion of cyt-c from the mitochondria and starts apoptosis. Recent studies showed that apoptosis plays a role in cardiac hypertrophy and heart failure in cardiomyocytes. 43 In another study in which we evaluated apoptosis in testicular tissue, we demonstrated that GTX III increased apoptosis. 44 Our study found that apoptosis increased in comparison to the control group in the groups that were given both acute and chronic GTX-III.
Consequently, GTX III intake causes cardiac tissue damage and apoptosis in both acute and chronic groups. This outcome may be explained by oxidative stress that is probably induced by GTX-III intake. However, more detailed studies are needed. The data obtained will shed light on further studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Yozgat Bozok University Project Coordination Application and Research Center (6602b-TF/18-181).
