Abstract
Benign prostatic hyperplasia (BPH) is an important key health concern for aging men. Polyphenolic compounds have been found to possess important roles in the inhibition of numerous ailments that involve reactive oxygen species and inflammation. Diosmin is a citrus flavone that possesses antioxidant, anti-inflammatory, antiproliferative, and anticancer activities, so based on these properties of diosmin, we decided to evaluate its effect on testosterone propionate (TP)-induced BPH. A total of 30 Wistar rats were randomly assigned to five groups having six animals in each. This study was of 28 days in which TP (5 mg kg−1) was administered to induce BPH in the last 10 days of the study. It was found that diosmin at the doses of 20 and 40 mg kg−1 significantly reduced malondialdehyde and xanthine oxidase formation in a dose-dependent manner; however, it replenished catalase, glutathione (GSH), and GSH-dependent enzymes, that is, glutathione peroxidase, glutathione reductase, and glutathione-S-transferase significantly against TP-induced BPH. Further, immunohistochemical study showed that diosmin alleviated inflammatory markers (nuclear factor kappa-light-chain-enhancer of activated B cells, cyclooxygenase-2, and interleukin-6). It was also found that diosmin downregulated the expression of androgen receptor and decreased the prostate-specific antigen concentration dose-dependently, significantly against TP-induced BPH. Diosmin also restored histoarchitecture of the prostate in a dose-dependent manner. Findings from the present study revealed the protective role of diosmin against TP-induced BPH in Wistar rats.
Introduction
Benign prostatic hyperplasia (BPH) is a multifactorial disease involving endocrine, environmental, and genetic factors. No specific gene mutations are reported in BPH, DNA methylation is reduced in BPH. 1 It is a nonmalignant proliferation of the prostate epithelium and stroma within the transition zone, which presents as enlargement of the prostate that contributes to a group of voiding complications that can range from troublesome to significantly impacting quality of life in older men. 2,3 The major function of the prostate is to propel its secretions into the seminal fluids upon ejaculation. 3,4 Endogenous testosterone is crucial for the development and function of the prostate throughout life and the cells of the gland to proliferate and to survive. Enzyme 5α-reductase is an important enzyme that converts testosterone into dihydrotestosterone (DHT). 5 In addition, the serum level of the prostate-specific antigen (PSA) is elevated in BPH patients compared with unaffected men since PSA production is promoted by binding DHT to the androgen receptor (AR). 6,7 The important characteristics involved are gush in the smooth muscle tone and nonmalignant enlargement of the gland. 8 This may lead to recurrent urinary withholding, urinary tract infections, and erratic postrenal failure. 9,10
Meanwhile, oxidative stress (OS) is one of the mechanisms that trigger the chain of reactions involved in the development of prostatic hyperplasia. 11,12 Many studies have confirmed that reactive oxygen species (ROS) and other oxidative factors are associated with prostatic hyperplasia. 13,14 Transcription factor, nuclear factor erythroid-2-related factor 2 (Nrf2), is the key gene that regulates the expression of numerous antioxidant proteins. The cyclooxygenase-2 (COX-2)/Nrf2 pathway is one of the most important signal transduction pathways of anti-inflammation and antioxidation. 14
Flavonoids are phenolic compounds and therefore act as powerful metal chelators and free radical scavengers. 15 It has been reported that flavanoids play a potent role in ROS scavenging and have shown several budding health welfares. 16 Diosmin is one of such natural citrus flavone of hesperidin derivatives, which has been reported to have antioxidant, anti-inflammatory, antimutagenic, antiallergic effect, antidiabetic effect, and antiproliferative and anticancer activities. 17 –21 Moreover, it has been reported that diosmin increases the venous tone; improves lymphatic drainage; protects hepatic, renal, and cardiac injury; and reduces the capillary hyperpermeability, thereby leading to reduction in the release of inflammatory mediators. 22 –24 Keeping in view the above biological activities of diosmin, the present study was designed to make an attempt to evaluate the preventive efficacy of diosmin against testosterone propionate (TP)-induced prostatic injury in Wistar rats by modulating OS markers and inflammatory cytokines (nuclear factor kappa B (NF-κB), COX-2, and interleukin-6 (IL-6)).
Materials and method
Chemicals and reagents
Reduced glutathione (GSH), oxidized glutathione (GSSG), nicotinamide adenine dinucleotide phosphate reduced (NADPH), nicotinamide adenine dinucleotide phosphate oxidized, ethylenediaminetetraacetic acid (EDTA), thiobarbituric acid (TBA), pyrogallol, poly-
Animals
Male Wistar rats used in this study were obtained from Central Animal House Facility of Jamia Hamdard, New Delhi, India. All procedures using animals were reviewed and approved by the Institutional Animal Ethical Committee that is fully accredited by the Committee for Purpose of Control and Supervision on Experiments on Animals, Chennai, India. The rats were 8–9 weeks old at starting of the study (weights in the range of 175–200 g). They were housed in polypropylene cages in groups of six rats per cage, were kept in a room maintained at 25 ± 2°C with a 12-h light/12-h dark cycle, and were allowed to acclimatize for 1 week before the initiation of experiments. They were given free access to standard laboratory animal feed (Hindustan Lever Ltd, Mumbai, India) and water ad libitum.
Experimental design
Wistar rats were divided into five groups with six animals in each group, receiving the following treatment: diosmin was administered orally at two doses based on reports 25 and the dose of TP was selected, with little modification, based on previous reports. 26,27 Group I: Served as control and was administered normal saline orally (10 ml kg−1) and olive oil intraperitoneally (i.p.) at the dose of 5 ml kg−1 for the last 10 days. Group II: Served as disease group and received dose of TP (5 mg kg−1 in olive oil, i.p.) for last 10 days (19th day to 28th day). Group III: Pretreated with diosmin at the dose of 20 mg kg−1 body weight (bw) from day 1 to day 28 and TP (5 mg kg−1 in olive oil) was given on 18th day after 1 h of the pretreatment with diosmin. Group IV: Pretreated with diosmin at the dose of 40 mg kg−1 from day 1 to day 28 and TP (5 mg kg−1 in olive oil) was given on 18th day after 1 h of the pretreatment with diosmin. Group V: Received only higher dose of diosmin (40 mg kg−1) up to 28 days. All the animals were euthanized under mild anesthesia 24 h after the last treatment, blood was collected, and the prostate was taken out for examination of various biochemical and histological parameters.
Tissue processing
Postmitochondrial supernatant preparation and estimation of different parameters
Ventral prostate (VP) was removed quickly and perfused with ice-cold saline (0.9% sodium chloride). The VP (10% w/v) was homogenized in chilled phosphate buffer (0.1 M, pH 7.4) using a Potter Elvehjem Homogenizer (Thomas Scientific Swedesboro, New Jersey, USA). The homogenate was filtered through muslin cloth and was centrifuged at 3000 r min−1 for 10 min at 4°C in a Remi Cooling Centrifuge (C-24 DL) to separate the nuclear debris. The aliquot so obtained was centrifuged at 12,000 r min−1 for 20 min at 4°C to obtain postmitochondrial supernatant (PMS), which was used as a source of various enzyme assays.
Assay for lipid peroxidation
The assay for membrane lipid peroxidation (LPO) was done by the method of Wright et al. 28 with some modifications. The reaction mixture in a total volume of 3.0 ml contained 1.0 ml tissue homogenate, 1.0 ml TCA (10%), and 1.0 ml TBA (0.67%). All the test tubes were placed in a boiling water bath for a period of 45 min. The tubes were shifted to an ice bath and then centrifuged at 2500 × g for 10 min. The amount of malondialdehyde (MDA) formed in each of the samples was assessed by measuring the optical density of the supernatant at 532 nm. The results were expressed as the nmol MDA formed/g tissue by using a molar extinction coefficient of 1.56 × 105 M−1 cm−1.
Assay for xanthine oxidase
The activity of xanthine oxidase (XO) was assayed by the method of Athar et al. 29 The reaction mixture consisted of 0.2 ml PMS, which was incubated for 5 min at 37°C with 0.8 ml phosphate buffer (0.1 M, pH 7.4). The reaction was started by adding 0.1 ml xanthine (9 mM) and kept at 37°C for 20 min. The reaction was terminated by the addition of 0.5 ml ice-cold perchloric acid (10% v/v). After 10 min, 2.4 ml of distilled water was added and centrifuged at 4000 r min−1 for 10 min and mg uric acid formed/min/mg protein was recorded at 290 nm.
Assay for catalase
Catalase (CAT) activity was assayed by the method of Claiborne. 30 Briefly, the reaction mixture consists of 1.95 ml phosphate buffer (0.1 M, pH 7.4), 1.0 ml hydrogen peroxide (0.019 M), and 0.05 ml PMS in a final volume of 3 ml. Changes in absorbance were recorded at 240 nm. CAT activity was calculated as nmol H2O2 consumed/min/mg protein.
Assay for reduced GSH
GSH content of the prostate was determined by the method of Jollow et al. 31 Briefly, 1 ml of PMS (10%) was mixed with 1.0 ml of sulfosalicylic acid (4%). The samples were incubated at 4°C for 1 h and then centrifuged at 12,000 × g for 15 min at 4°C. The assay mixture (3 ml) consists of 0.4 ml supernatant, 2.2 ml phosphate buffer (0.1 M, pH 7.4), and 0.4 ml DTNB (4 mg ml−1). The yellow color developed was read immediately at 412 nm. The GSH concentration was calculated as nmol GSH/g tissue.
Assay for glutathione peroxidase
The glutathione peroxidase (GPx) activity was measured by the method of Mohandas et al. 32 The reaction mixture consists of 1.44 ml phosphate buffer (0.1 M, pH 7.4), 0.1 ml EDTA (1 mM), 0.1 ml sodium azide (1.0 mM), 0.05 ml GR (1 eu ml−1), 0.05 ml GSH (1.0 mM), 0.1 ml NADPH (0.2 mM), 0.01 ml H2O2 (0.25 mM), and 0.1 ml PMS (10%) in a total volume of 2.0 ml. Disappearance of NADPH at 340 nm was recorded. The enzyme activity was calculated as nmol NADPH oxidized/min/mg protein using molar extinction coefficient of 6.22 × 103 M−1 cm−1.
Assay for GR
The GR activity was measured by the method of Carlberg and Mannervik. 33 The assay system consists of 1.65 ml phosphate buffer (0.1 M, pH 7.6), 0.1 ml EDTA (0.5 mM), 0.05 ml GSSG (1.0 mM), 0.1 ml NADPH (0.1 mM), and 0.1 ml PMS (10%) in a total volume of 2.0 ml. The enzyme activity was recorded at 25°C by measuring the disappearance of NADPH at 340 nm and was calculated as nmol NADPH oxidized/min/mg protein using molar extinction coefficient of 6.22 × 103 M−1 cm−1.
Assay for glutathione-S-transferase
The assay for activity was estimated by the method of Habig et al. 34 The assay consisted of 2·5 ml phosphate buffer (0·1 M, pH 6·5), 0·2 ml GSH (1 mM), 0·2 ml CDNB (1 mM), and 0·1 ml of the cytosolic fraction (10%) in a total volume of 3·0 ml. Changes in absorbance were recorded at 340 nm, and enzymatic activity was calculated as nmol CDNB conjugate formed/min per mg protein using a molar extinction coefficient of 9·6 ×103 M−1 cm−1.
Protein estimation
Protein content in all samples was estimated by the method of Lowry et al. 35 using BSA as standard.
Histopathology of prostate
For histopathology study, the VP was removed and immediately fixed in freshly prepared 10% neutral-buffered formalin at 4°C. Then, the prostate was embedded in paraffin wax. A vertical section of the prostate (5-µm thick) was cut and stained with hematoxylin and eosin (H&E). The prostate acini and stroma were observed as an indicator of histological changes with a microscope (Olympus BX51, Olympus Corporation, Tokyo, Japan) at 10× and 40× magnification at least in six different regions.
Immunohistochemical staining
The VP tissues were fixed in formalin and paraffinized. Sections of 5 µm thickness were cut onto poly-
PSA level in serum
PSA level in serum was measured by enzyme-linked immunosorbent assay (ELISA) kits for rat PSA ELISA kit (Cloud-clone Corp., Katy, Texas, USA). Standards and samples prepared according to the manufacturer’s protocol were added to the precoating with target-specific capture antibodies microtiter plates. The optical density of each sample was determined using a microplate reader at 450 nm.
Statistics
The differences between the groups were analyzed using ordinary one-way analysis of variance followed by Tukey’s multiple comparisons test. Results were expressed as mean ± standard error of the mean. The p value <0.05 was considered statistically significant. GraphPad Prism 6 was used for the statistical analysis.
Results
Effect of diosmin on LPO
There was a significant (***p < 0.001) augmentation in the level of MDA formation in TP-induced BPH group (GII) in the prostate tissue when compared with the control group (GI). We observed that the treatment with diosmin at the dose 1 (20 mg kg−1) (in GIII) led to decline in the MDA level (### p < 0.001) and also at dose 2 of diosmin (40 mg kg−1) (in GIV), there was a significant decrease (### p < 0.001) in MDA of membrane integrity in the prostate tissue against TP administered (5 mg kg−1 bw, i.p.). There was no significant difference found in the MDA level between higher dose of diosmin-treated group (GV) with the control group (GI) (Figure 1).
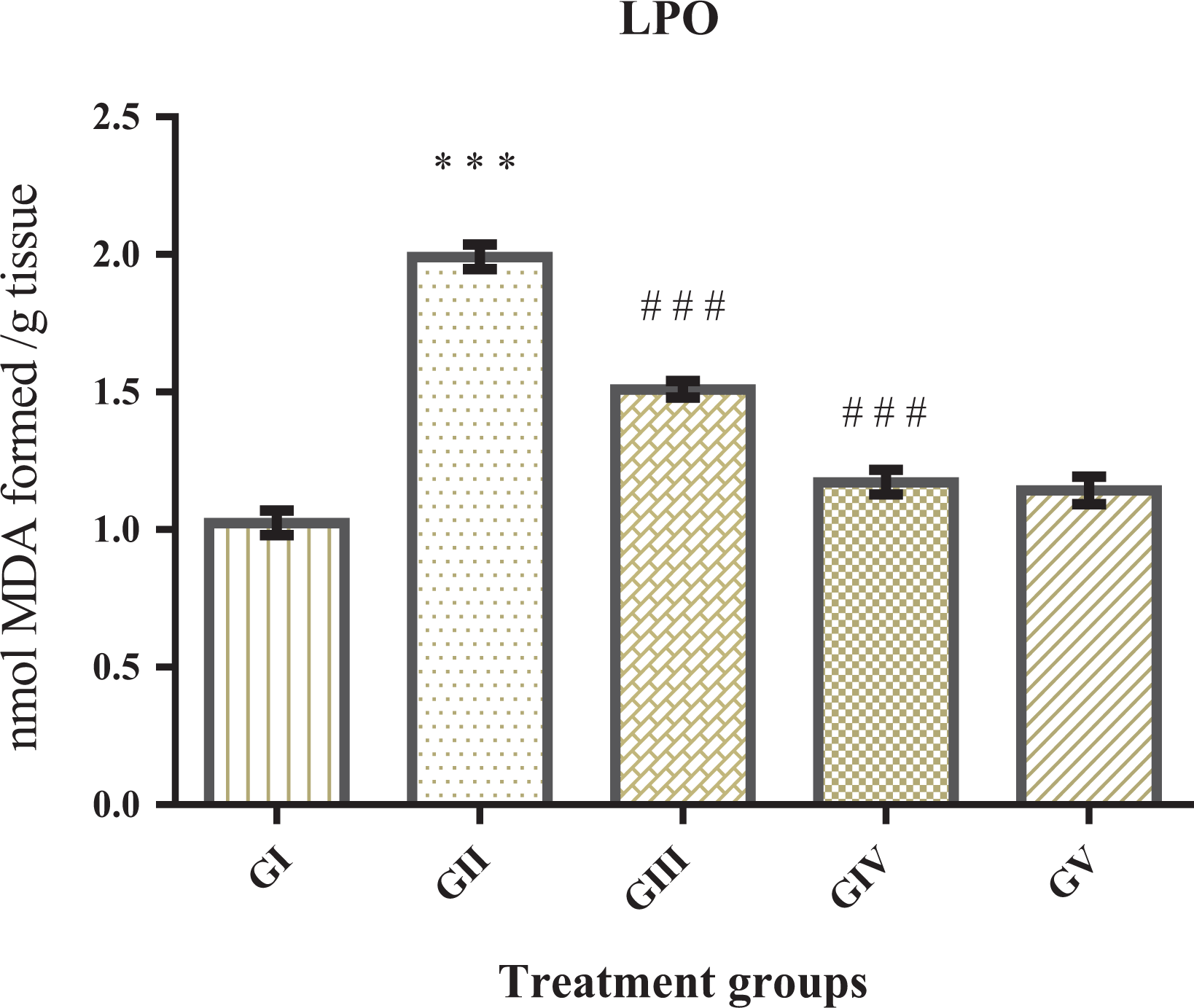
Effect of diosmin on LPO, results represent mean ± SE of six animals per group. This result showed significant augmented MDA level in TP-induced group (GII) (***p < 0.001) when compared to control (GI). Treatment with diosmin at both the doses significantly prevented TP induced increase in MDA levels (### p < 0.001). There was no significant difference between GV (only diosmin dose 2) and GI. LPO: lipid peroxidation; MDA: malondialdehyde; TP: testosterone propionate.
Effect of diosmin on XO activity
There was a significant (***p < 0.001) augmentation in the level of uric acid formation in TP-induced group (GII) in the prostate tissue when compared with controls (GI). We observed that the treatment with diosmin at both the doses in GIII and GIV led to a significant decline in uric acid level in prostate tissues (### p < 0.01 and ### p < 0.001) when against TP-induced BPH group GII. There was no significant difference found in the uric acid level between GV (higher dose of diosmin only i.e. 40 mg kg−1 bw) and control (Figure 2).

Effect of diosmin on XO, results represent mean ± SE of six animals per group. The result showed the significant elevation of uric acid formation (XO) in GII (***p < 0.001) in comparison to GI. However, the treatment with diosmin at both the doses (in GIII and GIV) prevented TP-induced uric acid formation significantly (### p < 0.001). There was no significant difference between GV and GI. TP: testosterone propionate; XO: xanthine oxidase; SE: standard error.
Effect of diosmin on CAT activity
It was found that the activity of antioxidant enzyme CAT was significantly depleted in TP-induced group (***p < 0.001) as compared to the control group. However, the treatment with diosmin against TP administration significantly restored the activity of this enzyme (### p < 0.001 and ### p < 0.001). However, there was no significant difference in the activity of CAT between GV and GI (Figure 3).

Effect of diosmin on CAT, results represent mean ± SE of six animals per group. Effect of diosmin on CAT activity in μmoles of H2O2 consumed/min/mg protein. TP-induced BPH group (GII) showed an increase in the level of CAT (***p < 0.001). Diosmin at both the doses showed a significant increase in the level of CAT (***p < 0.001) when compared to GII. There was no significant difference between GV and GI. TP: testosterone propionate; SE: standard error; CAT: catalase; BPH: benign prostatic hyperplasia.
Effect of diosmin on GSH level
The level of GSH was depleted significantly in GII (***p < 0.001) as compared to control (GI). The GIII with diosmin at the dose of 20 mg kg−1 showed a significant increase in the level of GSH (## p < 0.01) when compared to GII. Restoration in the level of GSH was seen in GIV at its high dose of diosmin (40 mg kg−1) (### p < 0.001]. There was a significant (p < 0.001) elevation of GSH at the high dose of diosmin (40 mg kg−1) in GV when compared to control (Figure 4).

Effect of diosmin on GSH, results represent mean ± SE of six animals per group. Effect of diosmin on μmoles of GSH formed/g tissue. GI: control group; TP-induced BPH (GII) showed a decrease in the level of GSH (***p < 0.001). Diosmin at both the doses significantly replenished the GSH level in GIII (## p < 0.01) and GIV (### p < 0.001). There was a significant difference observed in GV as compared to GI (***p < 0.001). TP: testosterone propionate; BPH: benign prostatic hyperplasia; SE: standard error; GSH: glutathione.
Effect of diosmin on GPx, GR, and GST activity
GSH-dependent enzymes, namely GPx, GR, and glutathione-S-transferase (GST), were significantly exhausted (***p < 0.001) in TP-induced BPH group as compared to control group. Diosmin administration at the dose of 20 mg kg−1 bw (GIII) significantly replenished GPx (## p < 0.01), GR (### p < 0.001), and GST (## p < 0.05), though (GIV) higher dose of diosmin (40 mg kg−1 bw) significantly restored GPx, GR (### p < 0.001, ### p < 0.001) as well as GST (### p < 0.001) when compared to GII. However, these enzymes at higher dose of diosmin (GV) showed significantly increased activity (p < 0.5, p < 0.5, and p < 0.01), respectively, as compared to the control group (Figures 5 to 7).
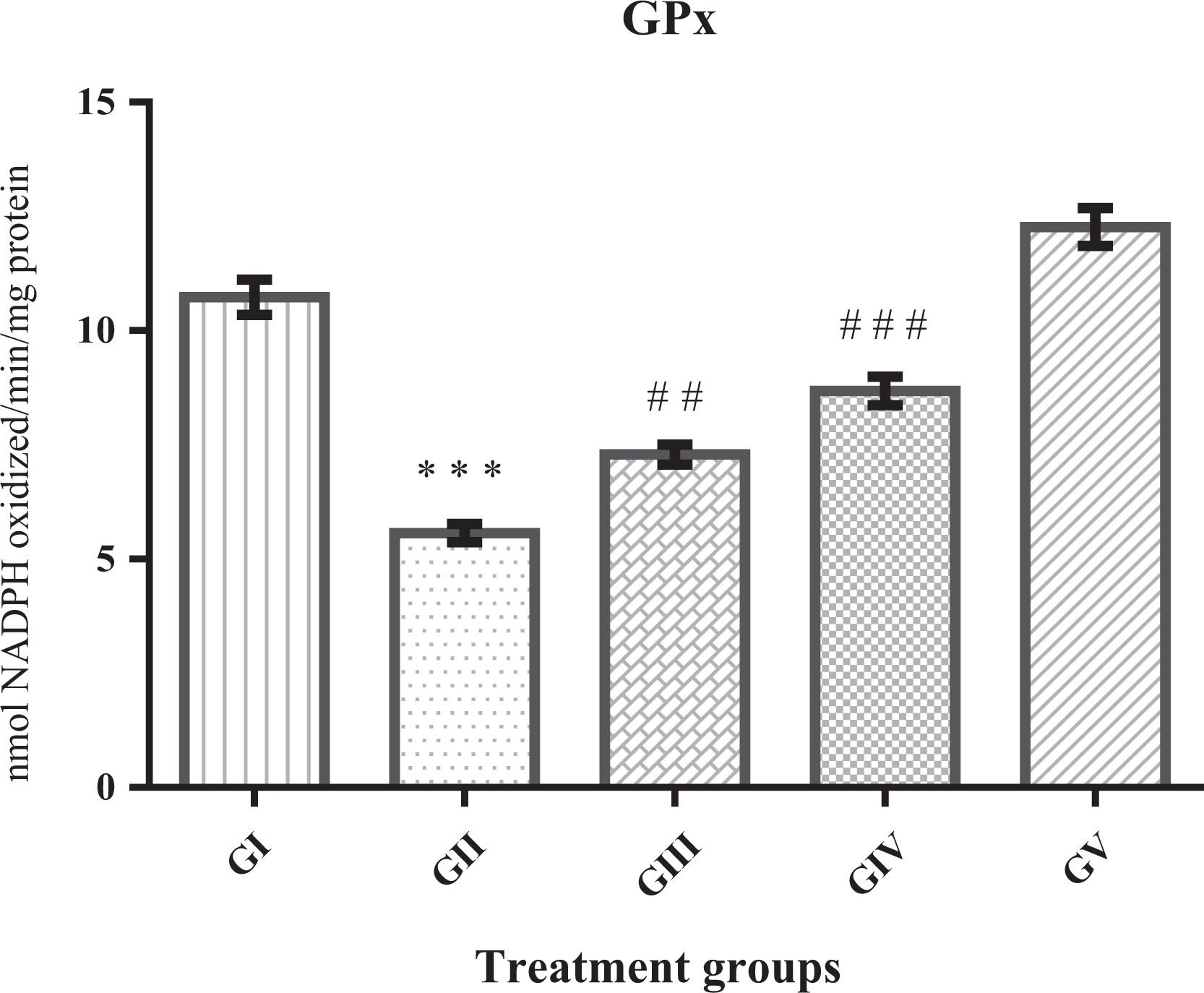
Effect of diosmin on GPx, results represent mean ± SE of six animals per group. Result obtained here shows significantly depleted GPx in GII (***p < 0.001). However, treatment with diosmin at both the doses elevated GPx activity against TP-induced BPH, significantly as GIII (## p < 0.01) and GIII (### p < 0.001). There was a significant elevation (p < 0.05) in GV when compared to GI. TP: testosterone propionate; BPH: benign prostatic hyperplasia; GPx: glutathione peroxidase; SE: standard error.
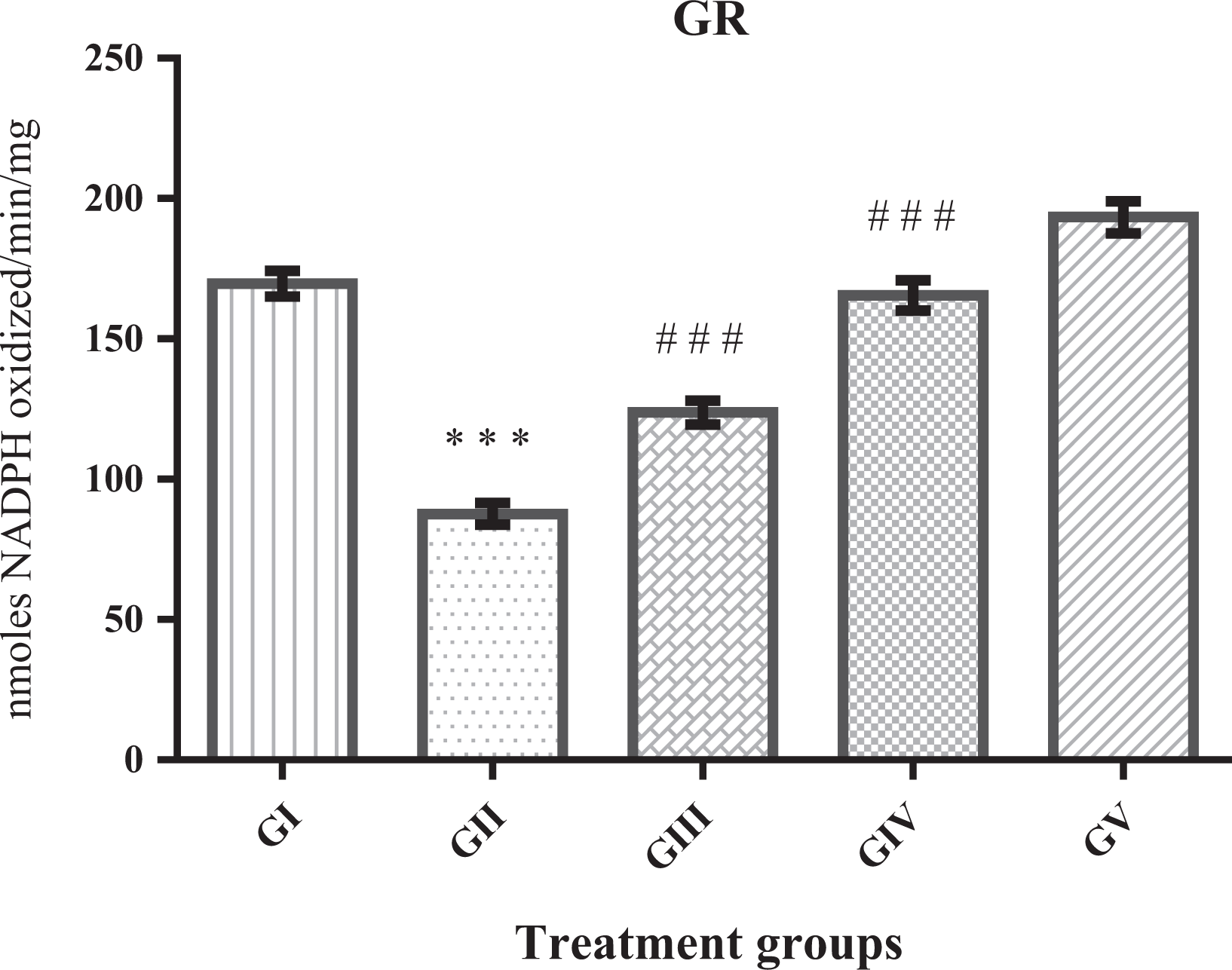
Effect of diosmin on GR, results represent mean ± SE of six animals per group. Result obtained here shows a significant depletion in GII (***p < 0.001) when compared to GI. However, administration of diosmin at both the doses elevated GR activity against TP-induced BPH significantly GIII (### p < 0.001) and GIV (### p < 0.001). There was a significant elevation (p < 0.05) in GV when compared to GI. TP: testosterone propionate; BPH: benign prostatic hyperplasia; SE: standard error; GR: glutathione reductase

Effect of diosmin on GST, results represent mean ± SE of six animals per group. This result obtained here shows a significant depletion in GPx activity in GII (***p < 0.001) when compared to GI. However, administration of diosmin at both the doses significantly elevated GPx activity against TP significantly in GIII (## p < 0.01) and in GIV (### p < 0.001). There was a significant elevation in GV (p < 0.01) when compared to GI. TP: testosterone propionate; GPx: glutathione peroxidase; SE: standard error; GST: glutathione-S-transferase.
Effect of diosmin on histological changes of the prostate tissues in TP-induced BPH rats
The H&E staining analysis showed histology of the prostate of the control group is normal in epithelial thickness, lumen area, and papillary fronds (Figure 8(a) and (b)). However, in the TP-induced BPH group, the epithelial layer and the number of papillary fronds were increased significantly but the lumen area was decreased. Due to oral administration of diosmin at both the doses (III: 20 mg kg−1 and IV: 40 mg kg−1), the epithelial thickening was found reduced, the number of papillary fronds was decreased, and the lumen area was enlarged in dose-dependent manner. No significant pathological features were observed in the control and diosmin only treatment group (epithelial layer, lumen area, and papillary frond).
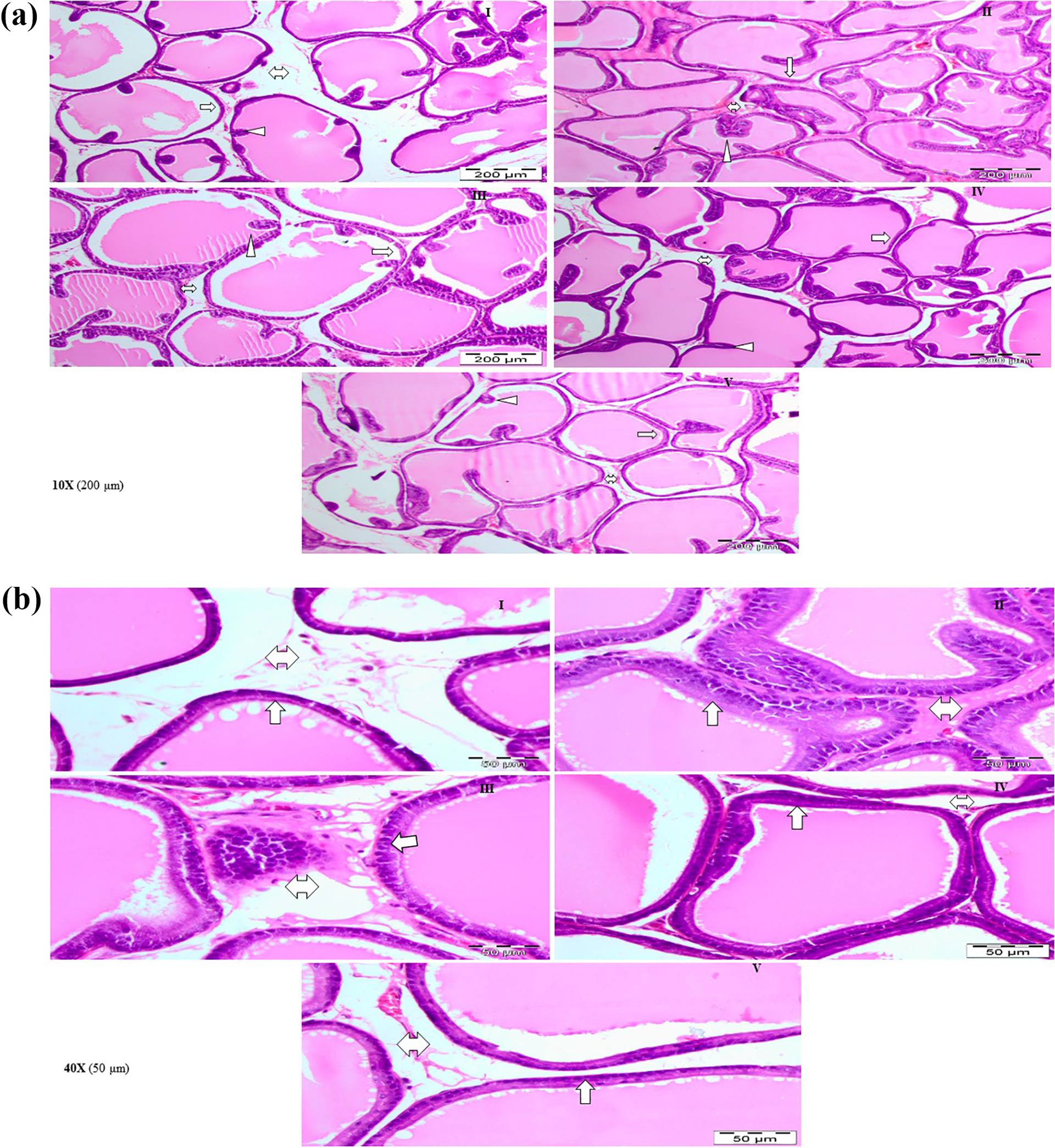
(a) Histopathology of the prostate gland, representative photomicrograph of H&E stained prostate tissues (×10). (b) Histopathology of the prostate gland, representative photomicrograph of H&E stained prostate tissues (×40). I: Section of the control group showed a normal morphological structure of the prostate. II: Prostate section of TP (5 mg kg−1) induced BPH group shows enlarged epithelial layer, decrease in lumen area, and an increase in papillary fonds. III: Diosmin at 20 mg kg−1 against TP-induced hyperplasia shows lesser enlargement of the epithelial layer, increase in lumen area, and lesser papillary fonds than TP-induced group. IV: Diosmin at 40 mg kg−1 against TP-induced hyperplasia shows nearly restored histoarchitecture of the prostate. V: Diosmin at 40 mg kg−1 shows normal histoarchitecture as control. TP: testosterone propionate; BPH: benign prostatic hyperplasia; H&E: hematoxylin and eosin.
Effect of diosmin on immunohistochemical changes in BPH rats
There were higher expressions of COX-2 (Figure 9), NF-κB (Figure 10), IL-6 (Figure 11), and AR (Figure 12) in TP-induced BPH group animals (GII) than the control group (GI). The animals which received diosmin at a dose of 20 mg kg−1 bw (GIII) and diosmin at the dose of 40 mg kg−1 bw (GIV) against TP-induced BPH showed low expression.

Photomicrograph depicting immunohistochemistry (IHC) staining of COX-2 (representative tissue, n = 6 animals per group). Control group is GI. There was a marked increase in the expression of COX-2 expression in TP-induced BPH group (GII) rats when compared to control group (GI). Diosmin treatment at both the doses (GIII and GIV) decreased the expression of COX-2 against TP-induced BPH (×40 magnification). TP: testosterone propionate; BPH: benign prostatic hyperplasia; COX-2: cyclooxygenase-2.
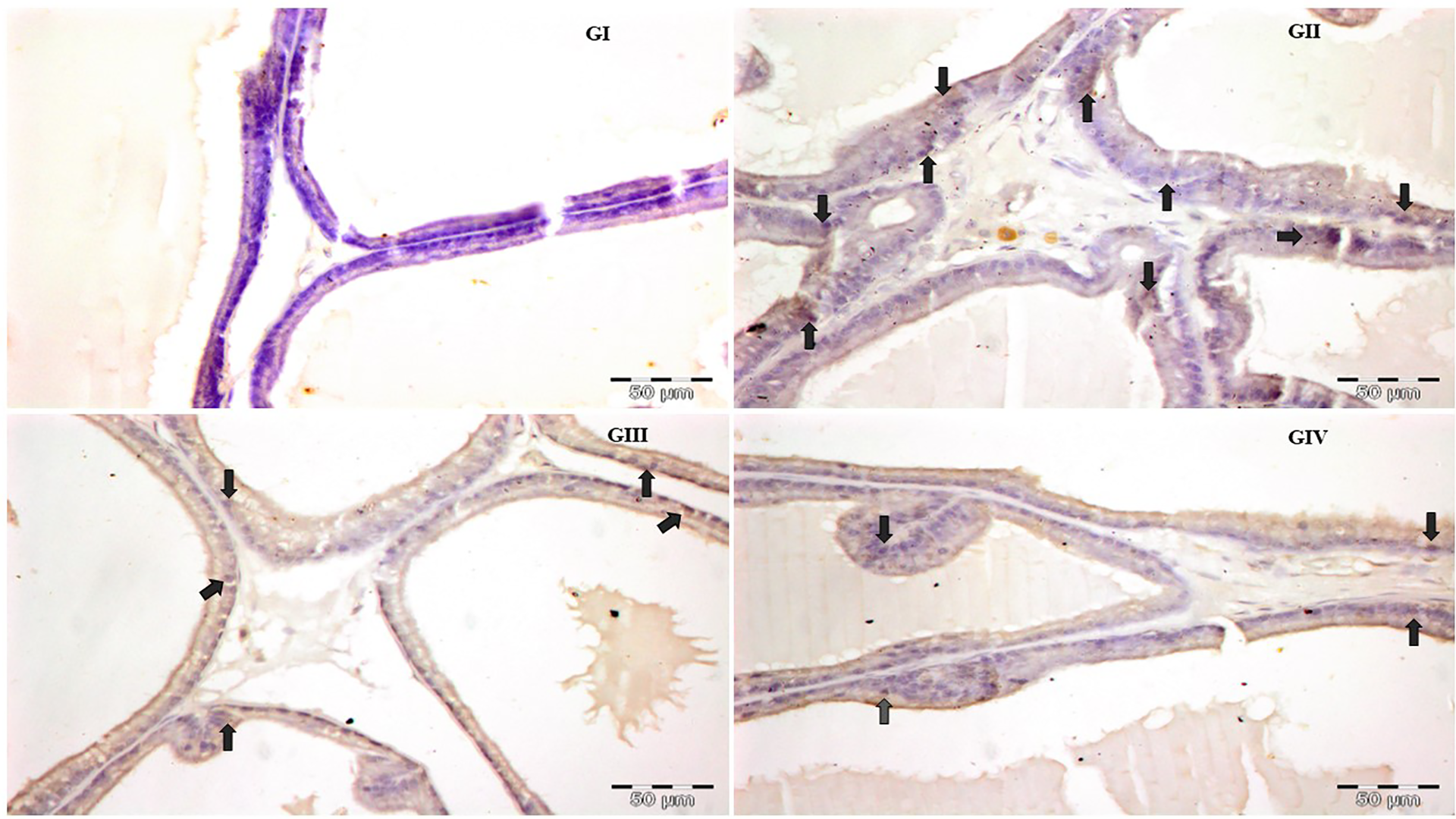
Photomicrograph depicting IHC staining of NF-κB (representative tissue, n = 6 animals per group). There was a higher expression of NF-κB expression in TP-induced group (GII) animals when compared to GI. Diosmin treatment (GIII and GIV) decreased the expression of NF-κB at both doses (×40 magnification). TP: testosterone propionate; NF-κB: nuclear factor kappa B.

Photomicrograph depicting IHC staining of IL-6 (representative tissue, n = 6 animals per group). There was a higher expression of IL-6 expression in TP-induced group (GII) animals when compared to GI. Diosmin treatment (GIII and GIV) decreased the expression of IL-6 at both the doses against TP-induced BPH (×40 magnification). TP: testosterone propionate; BPH: benign prostatic hyperplasia; IL-6: interleukin-6.

Photomicrograph depicting IHC staining of AR (representative tissue, n = 6 animals per group). There was a marked increase in the expression of AR expression TP-induced group rats (GII) when compared to GI. Diosmin administration at both the doses (GIII and GIV) decreased the expression of AR against TP-induced BPH (×40 magnification). TP: testosterone propionate; BPH: benign prostatic hyperplasia; AR: androgen receptor.
Effect of diosmin on PSA concentration in BPH rats
The serum level of PSA concentration was significantly higher in the TP-administered group (5 mg kg−1, i.p.) (TP) (***p < 0.001) than in the control group. Concentration of serum PSA in the TP + D1 (TP 5 mg kg−1 + diosmin 20 mg kg−1) as well as in the TP + D2 (TP 5 mg kg−1 + diosmin 40 mg kg−1) groups were significantly lower than that in the TP group (### p < 0.001). However, there was no significant difference found between control and diosmin (40 mg kg−1) only group (D2) (Figure 13).

Control group received saline while TP group received i.p. injection of TP at 5 mg kg−1 that shows a significant elevation in PSA concentration when compared with control, whereas TP + D1 and TP + D2 received an oral dose of diosmin at 20 and 40 mg kg−1, respectively, along with TP 5 mg kg−1 and these group shows significantly dose-dependently decrease in the concentration of PSA when compared to TP. D2 received only diosmin 20 mg kg−1 and shows no significant difference with control. TP: testosterone propionate; PSA: prostate-specific antigen; i.p.: intraperitoneally.
Discussion
One of the most prevalent urological diseases occurs via enlargement of the prostate through the proliferation of the stromal and epithelial elements leading to BPH (alternatively prostatic hyperplasia). Moreover, upregulation of PSA secretion also has been reported to be a contributor in BPH cases. 38
There are reports in the literature, which explain that OS plays a vital role in the development of BPH. 11,39 TP-induced prostatic hyperplasia due to ROS generation is one of the reasons, which drives to OS, hyperproliferation, and augmentation of redundant ROS production. 40,41 Androgens have been shown to elevate prostatic cellular metabolism, acquiring massive generation of free radicals accompanied with large consumption of antioxidant enzymes. 42,43 When the inherent generation of ROS transcends cellular antioxidant protection in the biological system, the cellular component becomes damaged and leads to the abnormal proliferation of prostate cells. Eventually, OS results in histological changes as well as cellular dysfunction. 44 –46 Several investigators are working toward the reduction of such anomalies induced by TP with the use of plant products, which act as powerful scavengers, one of which is diosmin, a member of the flavonoid family. Flavonoid glycosides decrease cancer cell number, proliferative activity, and act as an antioxidant and detoxifying agent. 47
In the present study, the protective effects of diosmin observed may be correlated with the amelioration of OS in the prostate gland of TP-treated rats. LPO is a marker of OS and several studies have reported remarkable elevation in the level of MDA, a LPO product. 48 Elimination of free radicals in biological systems is accomplished over enzymatic and nonenzymatic antioxidants, which act as the most important shield against free radicals. We examined the activity of LPO, XO, CAT, GSH, and GSH-dependent enzymes against TP-induced rats. It was observed that there is an increase in the level of MDA in TP-administered Wistar rats as compared to the control but were significantly decreased the level of MDA in diosmin administered groups dose-dependently. It was also observed that XO activity was elevated in TP-treated rats as compared to the control group, however, the treatment with diosmin at both the doses significantly declined XO levels in both the groups.
The GST enzyme detoxifies ROS via catalyzing the conjugation with GSH. At the same time, GSH depletion and decrease of GST were seen in TP-treated rats when compared to the control. Treatment with diosmin at both the doses restored GSH level and GST activity significantly, in both the groups, which decipher its protective effects on TP. 38,48
Other enzymatic antioxidants, that is, CAT, GPx, and GR, were also evaluated. CAT converts hydrogen peroxide to water and oxygen, thus reducing oxidative burden. In the present study, rats administered with TP showed a decrease in enzyme activity of CAT. While diosmin treatment enhanced CAT activity in a dose-dependent manner. GPx possesses a great preference for lipid hydroperoxides, and it is expressed in almost all mammalian cells, nonetheless at very lesser levels. In the present study, there was a significant decrease in GPx in TP-administered rats. Administration of diosmin replenished GPx levels at both the doses significantly. GR catalyzes the reduction of glutathione disulfide to sulfhydryl form GSH by utilizing a flavin adenine dinucleotide (FAD) prosthetic group and NADPH. Depletion in GR was also found in TP-administered rats, however, the treatment with diosmin elevated GR levels at both the doses significantly.
Further, this study is supported by histological evaluation of rat prostate, which noticeably exhibited histopathological alterations, such as shrinkage in the cytoplasm and the lumen area by cell proliferation as well as polyp formation were observed in TP-induced rats. However, the typical histologic pattern of hyperplasia decreased with increasing concentrations in the diosmin administered group. This is possibly due to the antioxidant, anti-inflammatory, and chemopreventive properties of diosmin.
Inflammation is extremely prevalent in the prostates of aging men. 49 Prostatic inflammation involves both an inflammatory cell infiltrate and local tissue responses. There is a variety of inflammatory mediators, such as cytokines, histamine, growth factors, and ROS may produce these effects. 50 This inflammation plays a crucial role in the pathogenesis of BPH, and inflammatory infiltrates have been observed in the majority of pathologic specimens. 1,51,52 Role of transcriptional factor NF-κB has long been reported to be an important complex that mediates the expression of many inflammation-related genes After activation, NF-κB could lead to upregulation of proinflammatory cytokines including IL-6 by swiftly translocated to the nucleus. 53,54 In BPH progression, the multifunctional cytokine IL-6 plays an important role as the reactant that is secreted progressively in epithelial and stromal cells of the prostate. 55,56 This IL-6 could activate important inflammatory-related signaling pathways; this increased level of proinflammatory cytokines may upregulate the level of COX-2 in macrophagic and epithelial cells in the prostate. 57,58 The overexpression of COX-2 would induce to generate proinflammatory prostaglandins, which may promote cell proliferation and suppress the cell apoptosis to be associated with proliferative inflammatory lesions. 58 It is evident from many studies that the role of inflammatory infiltrates and their mediators is in the development of prostatic hyperplasia. Furthermore, histopathological observations show that inflammation can be associated with the development of prostatic hyperplasia. This commends that inflammation probably be a causative agent in the pathogenesis of prostatic hyperplasia. In the present study, immunohistochemical analysis of prostatic tissues from prostate hyperplasic rats establishes overexpressions of inflammatory markers (COX-2, NF-κB, and IL-6) and of AR.
One of the characteristic features of androgen-responsive genes expressed in the prostate gland is the PSA. 6,59 PSA is broadly recognized as a hallmark of prostate cancer even though a little amount of PSA is usually found in the blood of healthy men. 6,60 The transcriptional action of PSA is enhanced by the coupling of DHT to AR leading to its interaction with androgen response element (ARE) in the promoter region of PSA as found in BPH cases having high DHT activity. 61,62 This event results in the rise of PSA levels in prostate cancer patients as well as in the serum of BPH patients. 7 Therefore, alleviation in PSA levels may be a representative of the mitigation of BPH. We also studied how diosmin treatment against TP-induced hyperplasia could decrease the level of PSA. Our results deciphered that the administration of diosmin resulted in a decrease in the amount of PSA in serum and the AR expression in the prostate tissue of BPH rats, thereby resulting in attenuation of the androgen signal transduction pathway in prostate cells by diosmin treatment via regulation of PSA. In this study, diosmin effectively suppressed the expression of AR and the production of PSA.
Conclusion
This study has shown the protective role of diosmin against OS and ameliorated the level of LPO, XO, CAT, GSH, GPx, GR, and GST. It has also shown that diosmin replenishes the level of PSA. Anti-inflammatory activity of diosmin was also reported in this study. Based on this, we can say that diosmin has effectively protected the BPH in Wistar rats. This study supports the possibility for diosmin to be investigated as a potential agent for the treatment of prostatic and inflammatory diseases.
Footnotes
Acknowledgments
The authors are extremely thankful to late Dr AK Tiwari, then In-charge, Central Animal House Facility (CAHF), Jamia Hamdard, New Delhi, for his kind support in performing the histopathological studies. We also acknowledge the University Grant Commission (UGC), New Delhi, India, for providing UGC-BSR-fellowship to the first author AV.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
