Abstract
In the present study, Veronica multifida leaf extract and zinc acetate dihydrate were utilized to synthesize zinc oxide (ZnO) nanoparticles (NPs) eco-friendly and cost-effectively under different physical conditions. Soxhlet extractor was used for the preparation of aqueous plant extract. UV-Vis (ultraviolet–visible) spectrophotometer, X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), and transmission electron microscope (TEM) were used to characterize the ZnO NPs. UV-Vis spectrophotometer in the range of 200–800 nm was used to get information about the formation of ZnO NPs at different pH and temperatures. FTIR spectrum revealed the presence of functional groups in ZnO NPs. XRD, scanning electron microscope, and TEM analyses confirmed the crystal structure and average size of ZnO NPs. The antimicrobial activities of ZnO NPs were tested on microorganisms, that is, Escherichia coli ATCC 43895, Staphylococcus aureus ATCC 29213, Bacillus subtilis, Bacillus licheniformis, Pseudomonas aeruginosa, and Salmonella typhimurium. Moreover, antibiofilm activity of ZnO NPs was performed against P. aeruginosa and S. aureus ATCC 29213. ZnO NPs have shown effective antimicrobial and antibiofilm activities against tested microorganisms. The results elucidated that eco-friendly and cost-effectively produced ZnO NPs could be used as coating materials and in a wide range of industrial applications, such as pharmaceutical industries and cosmetics.
Introduction
Recent advances in nanotechnology, especially the preparation of nanoparticles (NPs) in various sizes and shapes, allow the development of new biological agents. 1,2 Various methods based on biological, chemical, physical, and their hybrids have been proposed for the synthesis of NPs. Most of the traditional methods for the synthesis of NPs are expensive. Besides that, especially the chemical and physical methods require the usage of chemicals that are toxic to humans and environmentally hazardous.
Therefore, their replacement with a variety of biological substances leading to little or no harm to the environment has been the center of interest of recent studies. 3 Over the last years, it has been shown that inorganic metal ions can be converted into metal NPs with the help of metabolites and proteins present in many biological systems, such as plants, bacteria, algae, and fungi through their reductive capacities. 4 Polyphenols, carbohydrates, terpenoids, and alkaloids (main metabolites found in plant biological system) play an important role in reducing metal ions to NPs and maintaining their stability. 5 Therefore, the use of plant extracts for the bioreduction of metal ions by adjusting the pH and the temperature of the reaction provides a selective control on the size and shape of NPs because of the varieties of their metabolites.
Besides, green synthesis method has some advantages over traditional methods. Firstly, green synthesis is simple and required short reaction time at low temperature. Secondly, the adsorption of toxic chemical on the surface of NPs during chemical methods leads to unfavorable effects in vivo applications. 6 On the other hand, NPs synthesized by green method are used mainly plants for reducing and stabilization agent instead of hazardous substances. Therefore, NPs synthesized by green method are potentially more biocompatible than those synthesized by chemical methods. 7 Finally, the synthesis process using green method is cost-effective over the chemical method.
NPs can be synthesized from a wide range of materials, thus, there are various nanomaterials, such as FeO, AlO, and zinc oxide (ZnO). ZnO is one of the most important metallic nanomaterials due to its distinctive optical, chemical sensing, semiconducting electrical conductivity, and piezoelectric properties. 8 Hence, it has been reported that ZnO NPs are used in food packaging, sunscreens, paints, and as coating materials. 9 Moreover, nanomaterials are a promising tool to combat pathogenic bacteria, biofilm formation, and resistant infectious diseases. 10
Pathogenic microorganisms construct biofilms containing polysaccharides, proteins, and DNA to protect themselves against inhibitory compounds. 11 The microbial biofilms formation might cause several important problems such as industrial fluid treating processes, 12 food contamination, 13 and the spread of infectious disease. 14 Numerous studies have indicated that ZnO NPs show toxic property on bacteria while having little side effect on human cells; therefore, they have been used as potential nanomaterials in agriculture, food industries, diagnostics, surgical devices, and nanomedicine based on antimicrobial agents. 15 –18
In this study, we used Veronica multifida leaf extract to synthesize eco-friendly and cost-effective ZnO NPs under different physical conditions. The synthesized ZnO NPs were characterized by UV-Vis spectrophotometer, X-ray diffraction (XRD), Fourier transform infrared (FTIR), and transmission electron microscope (TEM). After the characterization of ZnO NPs, the antimicrobial and antibiofilm activities of ZnO NPs were tested on gram-negative and gram-positive pathogenic strains to investigate its potential usage as coating materials.
Materials and methods
Preparation of plant extract
The plant leaves of V. multifida were collected from Karaman, Turkey, in spring. The aqueous plant extract was prepared by mixing 5 g of washed and dried fine powdered leaves with 250 mL deionized water and subjected to Soxhlet extractor for 3 days. After cooling to room temperature, the extract was filtered by Whatman no. 1 filter paper. The extract was stored at 4°C for further applications.
Biological synthesis of ZnO NPs
Biological synthesis of ZnO NPs using plant extract of V. multifida was carried out by dropwise addition to the 0.5 g zinc acetate solution, which was stirred at 1200 r min−1 and 50°C. The solution was kept under observation until the color changes to whitish yellow. The solution was then centrifuged at 9000 r min−1 for 15 min and the NPs were collected. The collected NPs were washed three times with deionized water and then centrifuged again. The synthesized ZnO NPs were kept at 4°C for characterization studies and further experiments. For temperature experiments, the synthesis was performed at 60°C, 80°C, and 100°C. For pH experiments, the pH of the zinc acetate solution was adjusted to pH 7 or 12 with 1 M sodium hydroxide prior to synthesis.
Characterization of ZnO NPs
UV-Vis spectrophotometer
ZnO NPs were preliminarily characterized using UV-Vis spectrophotometer. UV-Vis spectra of leaf extract and synthesized ZnO NPs were measured by UV-Vis spectrophotometer (Thermo MultiSkan Go, Finland) between 200-nm and 800-nm wavelength.
XRD analysis
XRD results of the ZnO NPs were determined in the scanning range of 20° and 80° by XRD (D8 ADVANCE with DAVINCI, Bruker, Germany) with copper K α radiation (k = 1.5406 Å).
FTIR spectra analysis
FTIR (Vertex 70 ATR-FTIR spectrometer, Bruker) was used to analyze the functional groups of ZnO NPs with detector at 4000–400 cm−1 resolution.
SEM analysis
Scanning electron microscopy (SEM) was used to analyze morphology and size of ZnO NPs. SEM analysis was carried out by field-emission scanning electron microscopy (FESEM; Hitachi SU-5000, Nederland). Sample was placed on a carbon-coated copper grid and scanned with FESEM. Furthermore, energy-dispersive spectroscopy (EDAX) analysis was performed to evaluate the elemental composition and purity of ZnO NPs with FESEM-EDAX (Hitachi SU-5000).
TEM analysis
TEM (Tecnai TM G2 Sipirit, FEI Company, Nederland) analysis was performed to evaluate the size of ZnO NPs.
Antimicrobial activity
Disc diffusion assay
Disc diffusion method 19 was applied to evaluate the antimicrobial activities of ZnO NPs synthesized using the leaf extract of V. multifida against microorganisms, that is, Escherichia coli ATCC 43895, Staphylococcus aureus ATCC 29213, Bacillus subtilis, Bacillus licheniformis, Pseudomonas aeruginosa, and Salmonella typhimurium. One hundred microliters of each overnight grown bacterial culture was adjusted to McFarland 0.5 turbidity (1.5 × 108 CFU mL−1) was inoculated on the Mueller-Hinton agar plates. The 6 mm of sterile discs poured with various concentrations of ZnO NPs (5, 2.5, 1, 0.5, and 0.25 mg mL−1) were applied to the surface of the inoculated agar plate within 15 min of inoculation. The leaf extract, penicillin G 10 μg/disc, gentamicin 10 μg/disc, and tetracycline 30 μg/disc were used as a control. Inoculations were kept at 35°C for 16–20 h, and the diameters of all inhibition zones were measured.
MIC and MBC assay
Minimum inhibitory concentration (MIC) 19 of ZnO NPs was determined for E. coli ATCC 43895, S. aureus ATCC 29213, B. subtilis, B. licheniformis, P. aeruginosa, and S. typhimurium. After adjusting the turbidity of overnight grown bacterial cultures to McFarland 0.5 turbidity, 100 μL of each was mixed with an equal volume of ZnO NP solution having different concentrations (5, 10, 25, and 50 μg mL−1) into the well of microplate, and the optical density (OD) was recorded at 595 nm by UV-Vis spectrophotometer after keeping at 35°C for 24 h. Following the determination of MIC value by visual observation, 50 μL of grown broth from the microplate wells was inoculated on Mueller-Hinton Broth (MHB) agar to assess the bactericidal/bacteriostatic effect of ZnO NPs.
Antibiofilm activity detection
Antibiofilm activity of the ZnO NPs was determined against S. aureus ATCC 29213 and P. aeruginosa using 96-well polystyrene plate method. 20 After adjusting the turbidity of overnight grown bacterial cultures to McFarland 0.5 turbidity, 100 μL of each was mixed with equal volume of ZnO NP solution having different concentrations (5, 10, 25, and 50 μg mL−1) into the well of microplate and incubated at 35°C for 24 h. Bacterial culture without ZnO NPs as control and sole nutrient broth as blank were inoculated into the wells. After incubation time, the microplates were washed three times with deionized water and adherent bacteria on the plate were subjected to 200 μL of 0.1% crystal violet solution (w/v) for 15 min. The excess crystal violet dye was detached by rinsing microplates three times with deionized water. After drying at room temperature, 200 μL of absolute ethanol was added to microplates for 15 min. OD was recorded at 595 nm by UV-Vis spectrophotometer (Thermo MultiSkan Go).
Results
Various techniques were used for the characterization of ZnO NPs to evaluate their properties, including size, shape, and functional groups. Furthermore, the antimicrobial and antibiofilm effects of ZnO NPs were determined against both gram-negative and gram-positive pathogenic strains.
ZnO NP characterization
First step of ZnO NP characterization was UV-Vis spectral scanning at the wavelength of 200–800 nm. The results of prepared ZnO NPs at different temperature are shown in Figure 1(a). Furthermore, UV-Vis spectra of the synthesized ZnO NPs with pH 7 and 12 values are displayed in Figure 1(b). The leaf extract was used as a control.

UV-Vis scanning spectrum of synthesized ZnO NPs: (a) different temperatures and (b) pH 7 and 12. UV-Vis: ultraviolet–visible; ZnO: zinc oxide; NP: nanoparticle.
As a second step of characterization, XRD patterns of the ZnO NPs were evaluated in the scanning range of 20° and 80°. The XRD spectrum of ZnO NPs was dealt with the standard The International Center for Diffraction Data (ICCD) file, as shown in Figure 2. 21 The XRD results of ZnO NPs exhibited diffraction peaks, including 002, 100, 101, 102, 103, 110, 112, 200, and 201 reflection planes of ZnO NPs with hexagonal phase. The average crystalline size of ZnO NPs corresponding to XRD peaks was calculated according to the Debye Scherrer’s formula and found as 29.5 for ZnO NPs at pH 12 and 11.5 for ZnO NPs at pH 7.

XRD pattern of synthesized ZnO NPs. ZnO: zinc oxide; NP: nanoparticle; XRD: X-ray diffraction.
Third step was the investigation of the possible functional groups involved in ZnO NPs by FTIR. The FTIR spectrum showed peaks at 3230 cm−1, 1637 cm−1, 1246 cm−1, 1394 cm−1, 1088 cm−1, 1034 cm−1, 837 cm−1, 709 cm−1, and 480 cm−1 (Figure 3). The broad peak at 3230 cm−1 might be ascribed the O–H stretch, hydroxyl group, and H-bonded. 22 The 2114 cm−1 peak was resulted from the vibrations of alkyne –C≡C– stretching 23 and the 1394 cm−1 peak correspond to amines. The peaks at 1637 cm−1 assigned to the stretching bands of C=O functional groups. 24 A standard peak of ZnO was recorded around 480 cm−1. 25 The vibrations of alkene C–H stretching caused the peak at 1246 cm−1. The 1034 cm−1 peak shows a stretch of aliphatic amines. 26 The 1088 cm−1, 837 cm−1, and 709 cm−1 peaks corresponded to C–N stretching of amine group. 27

FTIR spectra of synthesized ZnO NPs. ZnO: zinc oxide; NP: nanoparticle; FTIR: Fourier transform infrared.
As a final step, TEM was used to analyze the size of ZnO NPs as a last step of characterization. The TEM image of synthesized ZnO NPs was given in Figure 4(a). The size of ZnO NPs was computed in the range of 10–100 nm. The aggregation of ZnO NPs adversely affected the interpretation of size and shape of NPs. Moreover, SEM image provided information about the morphology of ZnO NPs that were irregular to nearly hexagonal and quasi-spherical (Figure 4(b)). Moreover, the purity of ZnO NPs was analyzed by SEM-EDAX. The results of EDAX analysis represented two picks, which were zinc and oxygen elements. EDAX results confirmed that there are no impurities in ZnO NPs composed of 52.75% oxygen and 47.25% zinc (Figure 4(c)).
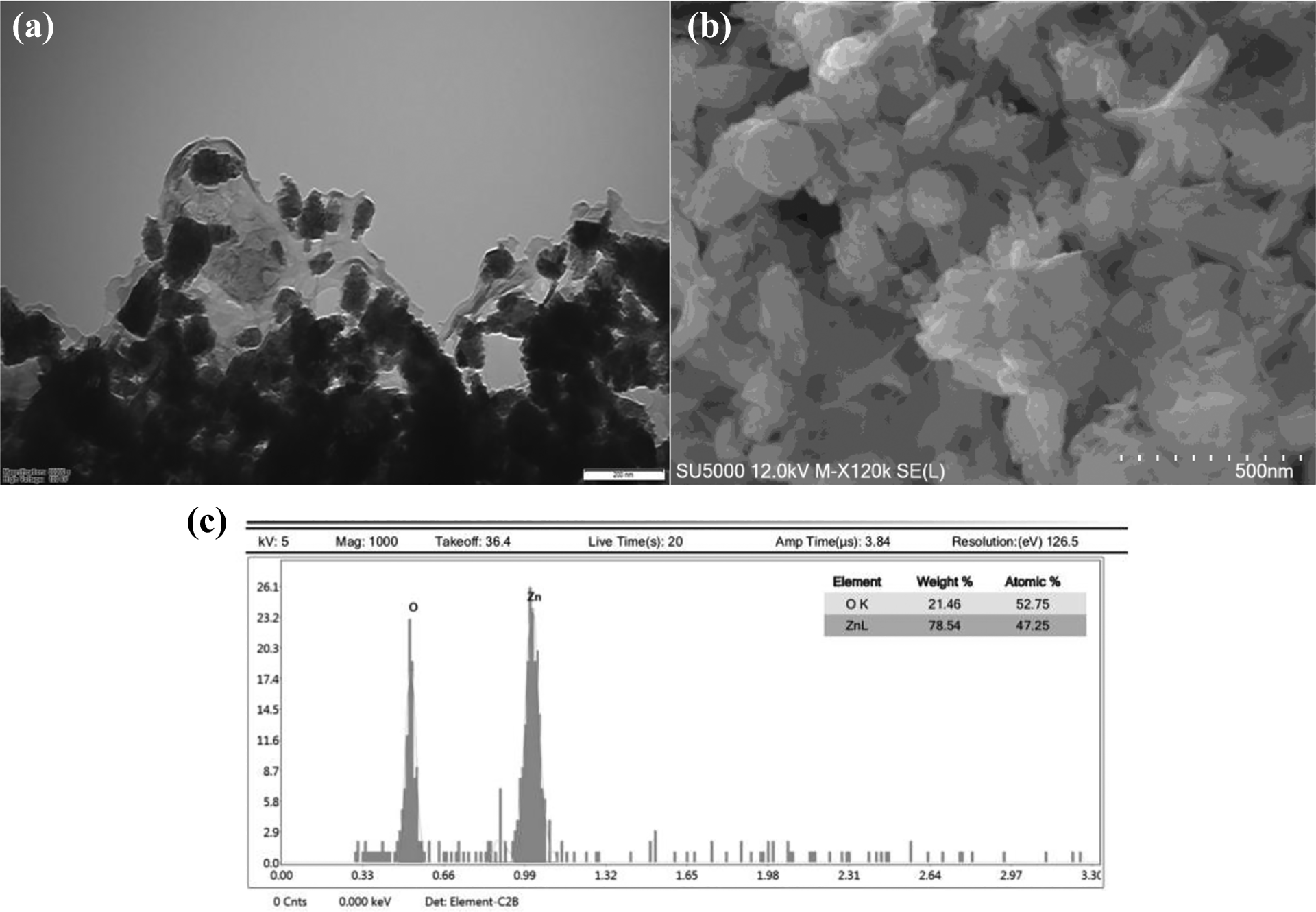
Electron microscopic images of ZnO NPs: (a) TEM image of ZnO NPs, (b) SEM image of ZnO NPs, and (c) SEM-EDAX image of ZnO NPs. ZnO: zinc oxide; NP: nanoparticle; TEM: transmission electron microscope; SEM: scanning electron microscope; EDAX: energy-dispersive spectroscopy.
Antimicrobial activity
The antimicrobial activity of ZnO NPs was tested on E. coli ATCC 43895, S. aureus ATCC 29213, B. subtilis, B. licheniformis, P. aeruginosa, and S. typhimurium by disc diffusion method. Promising antimicrobial activity was observed at 2.5 mg mL−1 concentration of the ZnO NPs against all tested bacteria (Table 1). The diameters of inhibition zones of ZnO NPs were verified between 8 mm and 15 mm (Table 1). MIC values for tested bacteria excluding E.coli and B. subtilis were determined at performed range of ZnO NPs. Furthermore, OD values of six microorganisms treated with ZnO NPs were elucidated that ZnO NPs inhibited successfully the growth of tested microorganisms (Figure 5). Minimum bactericidal concentration (MBC) assay was performed following MIC assay showed that ZnO NPs at applied concentrations have a strong bacteriostatic effect, not bactericidal on both gram-positive and gram-negative bacteria. Higher concentration of ZnO NPs is required for bactericidal effect against applied microorganisms.
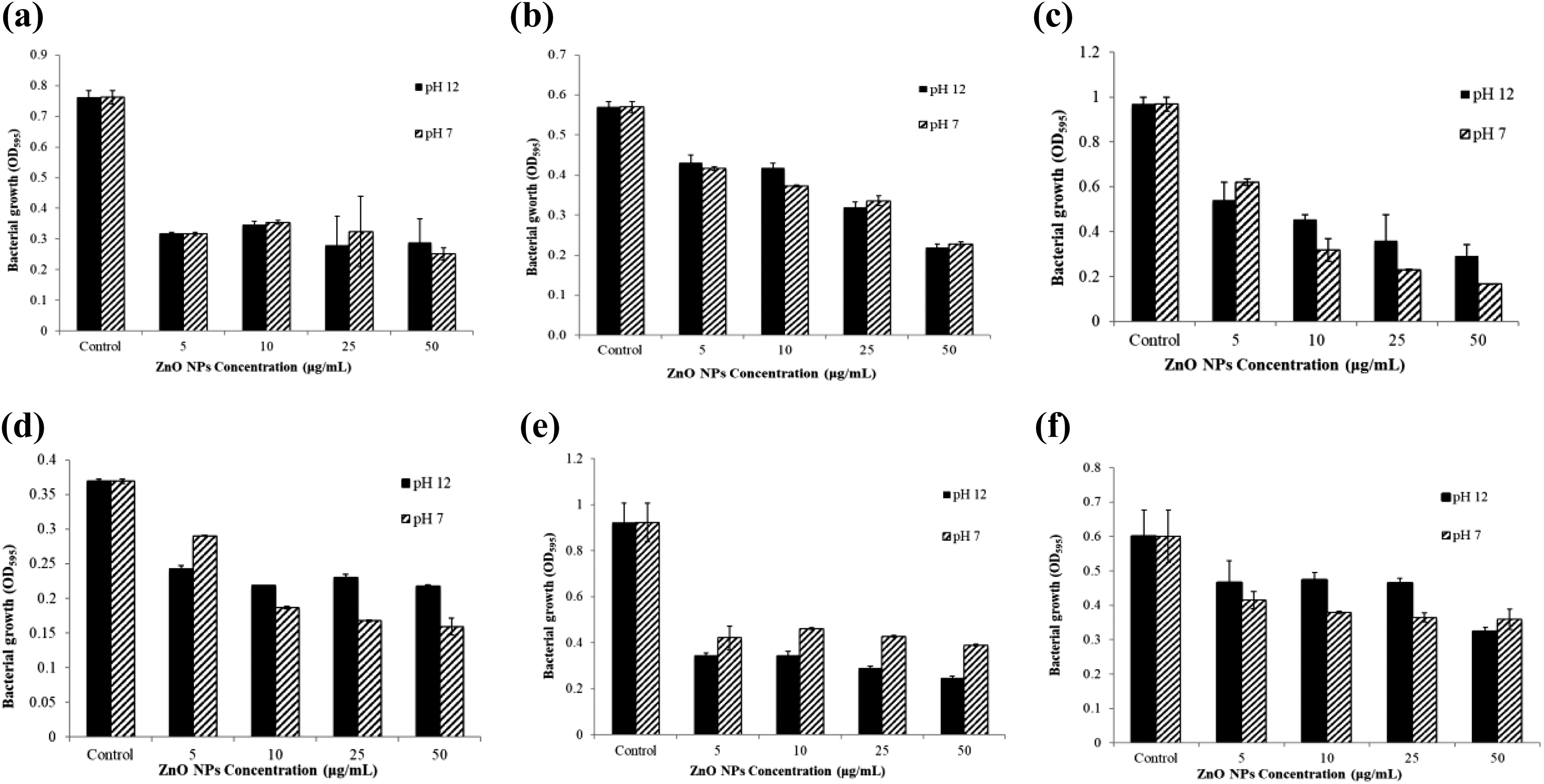
Effect of ZnO NPs on bacterial growth: (a) Pseudomonas aeruginosa, (b) Staphylococcus aureus, (c) Bacillus licheniformis, (d) Bacillus subtilis, (e) Salmonella typhimurium, and (f) Escherichia coli. ZnO: zinc oxide; NP: nanoparticle.
Antimicrobial activity of ZnO NPs.

NP: nanoparticle; —: no antimicrobial activity; ZnO: zinc oxide; TE: tetracycline; CN: gentamicin; P: penicillin G; MIC: minimum inhibitory concentration.
Biofilm inhibition assay
The biofilm inhibition effect of the ZnO NPs was determined against S. aureus ATCC 29213 and P. aeruginosa by crystal violet method as shown in Figure 6. ZnO NPs efficiently inhibited the formation of biofilm by both microorganisms in a dose-dependent manner (5, 10, 25, and 50 μg mL−1).

Antibiofilm activity of ZnO NPs: (a) Staphylococcus aureus ATCC 29213 and (b) Pseudomonas aeruginosa. ZnO: zinc oxide; NP: nanoparticle.
Discussion
UV-Vis spectrophotometer, XRD, FTIR, and TEM were used to assess the structure of ZnO NPs. In our study, based on UV-Vis spectrophotometer, the absorption peak of the ZnO NPs at pH 12 was around 360 nm, whereas the absorption peak for ZnO NPs at pH 7 was recorded as 240 nm (Figure 1(b)). It has been indicated that ZnO NPs have characteristic peaks between 330 nm and 370 nm at UV-Vis spectrophotometer. 28 Moreover, other studies recorded UV–Vis spectra of ZnO NPs as 281 and 314 nm. 29,30 In the present study, ZnO NPs at pH 7 gave the absorption peak of 240 nm differently from recent studies. XRD result of ZnO NPs at pH 7 supported the formation of ZnO NPs at this lower pH (Figure 2). In addition, during the synthesis process, creamish white colored precipitation proving the formation of NPs was observed. The formation of ZnO NPs at pH 12 showed more intense peak than pH 7 according to XRD analysis (Figure 2). Finally, FTIR spectroscopic analysis revealed that ZnO NPs at pH 12 formed stronger picks at 480 cm−1, 709 cm−1, 1034 cm−1, and 1088 cm−1 than pH 7 (Figure 3).
Disc diffusion, MIC, and MBC assays were performed for the evaluation of antimicrobial activity of ZnO NPs against tested bacteria in addition to their antibiofilm activity. Bacterial biofilms, an extracellular matrix including DNA, polysaccharides, and proteins, were produced by bacterial groups to maintain a complex construction and adhere to living or nonliving surfaces. 31 Studies on biofilms showed that bacterial biofilm has a potential role in the formation of antibiotic resistance of pathogens. 32 Therefore, we tested the antibiofilm effect of ZnO NPs besides the antimicrobial activity of ZnO NPs for investigation of its potential usage as coating materials.
MIC values of ZnO NPs against all tested bacteria except E. coli and B. subtilis were at the applied range of concentration. MIC values for E. coli and B. subtilis were determined as higher than 50 μg mL−1. On the other hand, B. licheniformis needed an MIC value of as low as 10 μg mL−1. When MBC assay tested on microorganisms determined MIC values, it was revealed that ZnO NPs did not have bactericidal effect on microorganisms but bacteriostatic effect at applied concentrations of ZnO NPs. We can infer that higher concentrations of ZnO NPs are required for bactericidal results.
Poovizhi and Krishnaveni indicated that the MIC value of the green synthesized ZnO NPs was measured as 50, 25, and 12.5 μg mL−1 against S. aureus, P. aeruginosa, E. coli, respectively. 33 Another study indicated that the MIC value of ZnO NPs for S. aureus was 625 μg mL−1. 34 In our study, we found that MIC values of ZnO NPs at pH 7 were >50 μg mL−1 for E. coli and B. subtilis, 50 μg mL−1 for S. aureus and P. aeruginosa, 10 μg mL−1 for B. licheniformis, and 50 μg mL−1 for S. typhimurium. Moreover, MIC values of ZnO NPs at pH 12 were verified against E. coli, S. aureus, B. subtilis, B. licheniformis, P. aeruginosa, and S. typhimurium, which were >50, 50, >50, 25, 25, and 25 μg mL−1 sequentially.
It has been noted that ZnO NPs have strong antimicrobial activity against various pathogenic microorganisms, such as E. coli, P. aeruginosa, Campylobacter jejuni, etc. 35 Furthermore, the spherical shape of ZnO NP was synthesized by Azadirachta indica leaf extract with size ranging from 9.6 nm to 25.5 nm, showing good antibacterial effect against Streptococcus pyogenes, S. aureus, and E. coli. 36 ZnO NPs may inhibit bacterial cell growth by disrupting bacterial membrane structure and accumulating in the cytoplasm. 37
S. aureus and P. aeruginosa were used for antibiofilm assay because of their high biofilm formation capacity 14 as also shown in Figure 6. The antibiofilm assay results showed that ZnO NPs efficiently inhibited the formation of biofilm by both microorganisms in a dose-dependent manner. Biofilm inhibition (87–91%) was achieved for both microorganisms with ZnO NPs. Concentration of 10 μg mL−1 ZnO NPs at pH 7 inhibited 88% of S. aureus biofilm formation, while it was 50 μg mL−1 ZnO NPs at pH 12 to inhibit 87% biofilm formation. For S. aureus, ZnO NPs synthesized at low pH exhibited better antibiofilm activity. It can be due to the effect of the NP size since ZnO NPs synthesized at pH 7 showed smaller size distribution than at pH 12 based on XRD results. On the other hand, for P. aeruginosa, the highest biofilm inhibition concentration of ZnO NPs was determined as 50 μg mL−1 for both conditions (NPs synthesized at pH 7 and pH 12).
In general, the studies show that ZnO NPs might be used as antibacterial agents in lotions, mouthwashes, and surface coatings on different materials for protecting from microorganisms to attach and colonize on their surfaces.
Conclusion
NPs based on their physical and chemical properties can be used in different industrial applications; thus, NP research is of great scientific interest. Moreover, their green synthesis can provide ecologically friendly production besides economic profits. In the present study, we synthesized ZnO NPs with the leaf extract of V. multifida. These ZnO NPs effectively inhibited biofilm formation of S. aureus and P. aeruginosa. Furthermore, it was detected that ZnO NPs had considerable antimicrobial activity against both gram-positive and gram-negative bacteria. Therefore, the results revealed that ZnO NPs could be used as a coating material in pharmaceutical industries, cosmetics, and other industries.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Karamanoglu Mehmetbey University Coordinatorship of Scientific Research Project [Grant Number: 21-M-17].
