Abstract
Tissue-dependent oestrogenic and anti-oestrogenic activity of polycyclic aromatic hydrocarbons (PAHs) has been suggested. In this study, the effect of two PAH mixtures, M1 composed of all 16 priority pollutants and M2 composed of five (noted in the highest levels) compounds, on follicle-stimulating hormone receptor (FSHR) expression, basal or FSH-induced oestradiol (E2) secretion and aromatase cytochrome P450 (P450arom) protein expression, by non-luteinised human granulosa cell line (HGrC1) was determined. In addition, the consequences of gene silencing of oestrogen receptor alfa (siESR1), oestrogen receptor beta (siESR2) and a G protein-coupled receptor (siGPER1) on the above parameters were described. Neither PAH mixture had an effect on basal FSHR protein expression; however, both mixtures increased FSH-induced FSHR expression. Decreased E2 secretion and P450arom expression was also demonstrated. In both basal and FSH treated cells, siESR1 and siGPER1 reversed the inhibitory effect of the mixtures on E2 secretion; however, in siESR2 cells, the inhibitory effect was still observed. This study showed that both classic ESR1 and GPER1 were involved in the inhibitory effect of both PAH mixtures on E2 secretion and confirmed that expression of P450arom could be downregulated through the aryl hydrocarbon receptor and additionally through the ESR2.
Introduction
Epidemiological studies have shown a link between health problems for offspring of couples who live in contaminated environments, compared to the progeny of those who live in uncontaminated environments. 1 –4 Sixteen polycyclic aromatic hydrocarbons (PAHs) are included among the 129 priority pollutants declared by the US Environmental Protection Agency. 5 Reports have found high levels of PAHs in umbilical cord blood and maternal blood. 6
Many endocrine disrupting compounds (EDCs) can interact with the female reproductive system and lead to endocrine disruption in the ovary. EDCs can alter endocrine function through altering the expression and/or activity of enzymes required for synthesis and/or catabolism of ovarian sex steroids and alter the expression of hormone receptors and/or their ability to bind their endogenous ligands. There are a broad group of compounds, including PAHs, 7 which can interfere with hormone systems and exert harmful effects on reproduction and immune function. The effects of PAHs on reproductive tissues could be a reflection of their multiple modes of action, such as activation of aryl hydrocarbon receptor (AHR)-dependent metabolism of PAHs, and their ability to affect oestrogen receptors (ERs), influencing endogenous steroid hormone secretion in ER-sensitive cells. Furthermore, crosstalk has been observed among AHR and nuclear receptors, though ER signalling has been more fully studied. The precise molecular mechanisms for this crosstalk are unclear, and may be a combination of several different mechanisms, including alterations in oestrogen synthesis/metabolism and influence on aromatase activity. 8,9 PAHs may act as anti-oestrogens by binding with the AHR receptor, 10 –12 leading to the induction of AHR-responsive genes that result in a broad spectrum of anti-oestrogenic responses. The main support for this scenario comes from studies indicating that abnormal cell proliferation and reduced oestradiol (E2) production occur after activation or lack of the AHR signalling pathway.
With functional characterisation of the two classical ERs, it has generally been accepted that the diverse physiological functions of oestrogen could be entirely ascribed to the combined effects of oestradiol receptor alfa (ESR1) and beta (ESR2) protein expression. 13 However, reports have also described oestrogen-binding properties and cellular/physiological effects that were not easily explained by transcriptional activation mediated by soluble ERs. 14 Such receptors have been postulated to mediate aspects of cellular oestrogen function, including traditional transcriptional signalling, as well as novel non-genomic, rapid signalling. 15 These non-genomic signalling events include pathways that are traditionally thought to originate from transmembrane growth factor receptors and G protein-coupled receptors (GPER). Despite the limited data currently available, the influence of PAHs on steroidogenesis is evident, and thus, it is reasonable to predict that studies aimed at understanding their mechanisms of action are forthcoming.
Taking into consideration that granulosa cells produce E2, we hypothesised that the anti-oestrogenic effect of the PAH mixtures on E2 secretion was association, not only with the well-known interaction between AHR and classic ERs (ESR1 and ESR2) but also with GPER1. To test this hypothesis, we determined the effect of PAH mixtures on basal or FSH-stimulated E2 secretion and, by gene silencing, confirmed the involvement of ESR1/2 and also indicated GPER1 in this action.
Materials and methods
Reagents and antibodies
Dulbecco’s modified eagle’s medium (DMEM), foetal bovine serum (FBS; heat inactivated),
Cell lines and treatment
Human granulosa cell line (HGrC1) was a gift from Dr Ikara Iwase (Nagoya University, Japan) and was cultured according to the protocol described by Bayasula et al.
16
Non-luteinised HGrC1 expresses enzymes related to steroidogenesis, such as steroidogenic acute regulatory protein (stAR), aromatase and follicle-stimulating hormone receptor (FSHR). It produces oestrogen in response to FSH stimulation; however, the cells do not possess luteinising hormone (LH) receptors and are not capable of undergoing luteinisation, similar to the growth characteristics of mitotic granulosa cells of preantral/early antral follicles.
16
The cell line was routinely cultured in DMEM with 10% FBS and 2 mM
siRNA transfection
For siRNA transfection, cells were seeded into 96-well (7.5 × 103 cells/well) and 48-well (15 × 103 cells/well) culture plates (Nunc, Roskilde, Denmark). siRNAs were introduced to HGrC1 cells using DharmaFECT 3 transfection reagent (GE Healthcare Dharmacon Inc.), according to the manufacturer’s instructions. Briefly, cells were incubated in the indicated culture medium. After 24 h, the medium was removed and cells were rinsed once with phosphate-buffered saline (PBS). Cells were then cultured in a serum-free medium containing DharmaFECT 3 transfection reagent (mock transfection), DharmaFECT 3 transfection reagent plus the non-targeting (control) siRNA or DharmaFECT 3 transfection reagent plus the targeting siRNAs, at a concentration of 100 nM for 24 h. SMARTpool siRNAs targeting human AHR, ESR1, ESR2 and GPER1 sequences (ON-TARGETplus human siRNA SMARTpool) were obtained from GE Healthcare Dharmacon. The culture medium was replaced with fresh medium after 24 h. The efficacy of gene knockdown was evaluated by real-time polymerase chain reaction (PCR) analysis.
qPCR analysis
For real-time PCR (qPCR) analysis, the cells were seeded into 96-well culture plates at a density of 7.5 × 103 cells/well HGrC1. After siRNA transfection, the next day, the medium was changed and the cells were treated with appropriate PAH mixtures for 48 h. After incubation, the cells were washed with ice-cold PBS, and the plates were frozen at −20°C until needed for the performance of PCR analysis. Total RNA isolated from the control cells, as well as those treated with siRNAs, were used for cDNA synthesis using the TaqMan Gene Expression Cells-to-CT kit (Applied Biosystems, Foster City, California, USA), according to the manufacturer’s instructions. The purity and quantity of the RNA and cDNA were determined using spectrophotometry at two optical densities, 260 nm and 280 nm. The lysis solution contained DNase I to digest contaminating genomic DNA. The resulting cDNA was analysed by qPCR using the StepOnePlus Real-Time PCR System (Applied Biosystems) and TaqMan Gene Expression Assays, together with TaqMan Gene Expression Master Mix and ROX reference dye (Applied Biosystems). A PCR was performed using a final volume of 20 µl, including 50 ng/reaction cDNA. The thermal cycling conditions were as follows: 50°C for 2 min, 95°C for 10 min and then 40 cycles of 95°C for 15 s and 60°C for 60 s. Duplicate control cDNA-free samples were prepared for each gene. The TaqMan Gene Expression Assays used for qPCR were AHR (assay no. Hs00169233), ESR1 (assay no. Hs00174860), ESR2 (assay no. Hs01100353) and GPER1 (assay no. Hs01922715). The relative expression of genes (RQ), determined by the 2−ΔΔCq method, was normalised against the endogenous reference gene, glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and to 18S ribosomal RNA (18S rRNA). Both GAPDH and 18S rRNA were stably expressed with minimal variability across samples, so in the experiment, we decided to normalise results for GAPDH.
Western blotting
For western blot analysis, the cells were seeded into 48-well culture plates at a density of 15 × 103 cells/well HGrC1. After incubation, the cells were washed with ice-cold PBS and lysed with Laemmli buffer (Laemmli Lysis buffer, cat. no. 38733, Sigma), sonicated and centrifuged at 1500 × g for 15 min at 4°C and stored at −20°C for western blot analysis. Protein expression was assessed and quantified using standard procedures. Samples were separated by sodium dodecyl sulphate-polyacrylamide gel electrophoresis, using Mini-Protean® TGX™ gels and the Mini-Protean II Electrophoresis Cell (BioRad, Hercules, California, USA). Proteins were transferred to polyvinylidene difluoride membranes and then incubated at 4°C overnight, with antibodies, all diluted to 1:200. Antibodies against FSHR (cat. no. sc-9828), ERα (cat. no. sc-542), ERβ (cat. no. sc-53494), GPR30 (cat. no. sc-48525) and P450arom (cat. no. sc-374176), as well as a horseradish peroxidase-conjugated secondary antibody and western blotting luminol reagent, were obtained from Santa Cruz Biotechnology (Dallas, Texas, USA). Membranes were incubated with horseradish peroxidase-conjugated antibody diluted to 1:5000. Protein-binding antibodies were detected by chemiluminescence using luminol reagent, visualised using a ChemiDoc-It Imaging System (UVP, LLC, Cambridge, UK) and quantified using ImageJ analysis software (US National Institute of Health, Bethesda, Maryland, USA). They were then stripped and probed with antibodies to β-actin.
Hormone secretion
To determine the secretion of oestradiol (E2), HGrC1 were cultured in DMEM medium containing 10% FBS with testosterone (100 nM) as a control and additionally with FSH (100 ng/ml) for 48 h. The secretion of E2 was determined by ELISA (DRG Instruments GmbH) according to the manufacturer’s instructions. Absorbance was measured with an ELx800 microplate reader (BioTek Instruments, Winooski, Vermont, USA) using a 450-nm filter. Analysis was performed using KC JUNIOR software (BioTek Instruments).
Statistical analysis
Each treatment was conducted in quadruplicate and each experiment was repeated three times (n = 3). All experimental results were presented as a mean ± standard error of the mean (SEM). All statistical analyses were performed using GraphPad Prism 5. Data were analysed by one-way analysis of variance, followed by Tukey’s test. Statistical significance is indicated in the figures by *p < 0.05, **p < 0.01 and ***p < 0.001 and different letters; with a < b; p < 0.05.
Results
Expression of AHR, ESR1, ESR2 and GPER1 mRNA after gene silencing
The AHR mRNA levels were significantly lower in cells transfected with AHR siRNA (0.4 ± 0.02 RQ), compared with the control (1 ± 0.01 RQ; p < 0.001) and those transfected with the non-specific siRNA (siNT; 0.89 ± 0.14 RQ). ESR1 and ESR2 expressions were efficiently knocked down following treatment of the cells with ESR1 and ESR2 siRNA (0.38 ± 0.05 and 0.36 ± 0.15 RQ, respectively) compared with the control (1 ± 0.07 RQ) and non-specific siRNA (0.94 ± 0.16 RQ; p < 0.001). GPER1 mRNA expression was significantly decreased by siRNA silencing (0.39 ± 0.015 RQ) when compared with the control (1.08 ± 0.09 RQ) and non-specific siRNA (siNT; 1.15 ± 0.02 RQ; p < 0.001). In each case, the expression of all silenced genes in mixtures of treated cells was 60% lower than in the control and those transfected with the non-specific siRNA (p < 0.001; Figure 1(a) to (d)).

AHR (a), ESR1 (b), ESR2 (c) and GPER1 (d) mRNA expression in control (C), non-targeting control (siNT), siRNA transfected and M1 and M2 treated cells after siRNA silencing. Statistically significant differences between control and treated cells are indicated by **p < 0.01 and ***p < 0.001. Experiments were performed three times (n = 3). The graphs are representative of the three independent experiments. AHR: aryl hydrocarbon receptor; ESR1: oestradiol receptor alfa; ESR2: oestradiol receptor beta; GPER: G protein-coupled receptors; siRNA: small interfering RNA.
Effect of PAH mixtures on the expression of FSHR
FSH decreased expression of its own receptor, FSHR (1.2-fold). Neither mixture had an effect on basal expression; however, both mixtures increased FSHR expression in FSH-stimulated cells (1.2-fold in M1 and M2 treated cells; Figure 2).

Effect of PAH mixtures M1 and M2 on basal (C) and FSH-stimulated (FSH) FSHR protein expression. Protein levels were densitometrically scanned and normalised against the β-actin (42 kDa) signal. Statistically significant differences between basal (C) and FSH-treated cells (FSH) are indicated with different letters, with a < b. Statistically significant differences between mixture treated cells in control and mixture treated cells in FSH-stimulated cells are indicated by *p < 0.05. Experiments were performed three times (n = 3). The blot is representative of the three independent experiments. PAH: polycyclic aromatic hydrocarbon; FSHR: follicle-stimulating hormone receptor.
Effect of PAH mixtures on basal and FSH-stimulated ESR1, ESR2 and GPER1 protein expression
M1 and M2 increased basal (3.1-, 2.2-fold, respectively) and FSH-stimulated (3.1-, 4.6-fold, respectively) ESR1 protein expression. Neither M1 nor M2 had an effect on ESR2 and GPER1 protein expression (Figure 3).
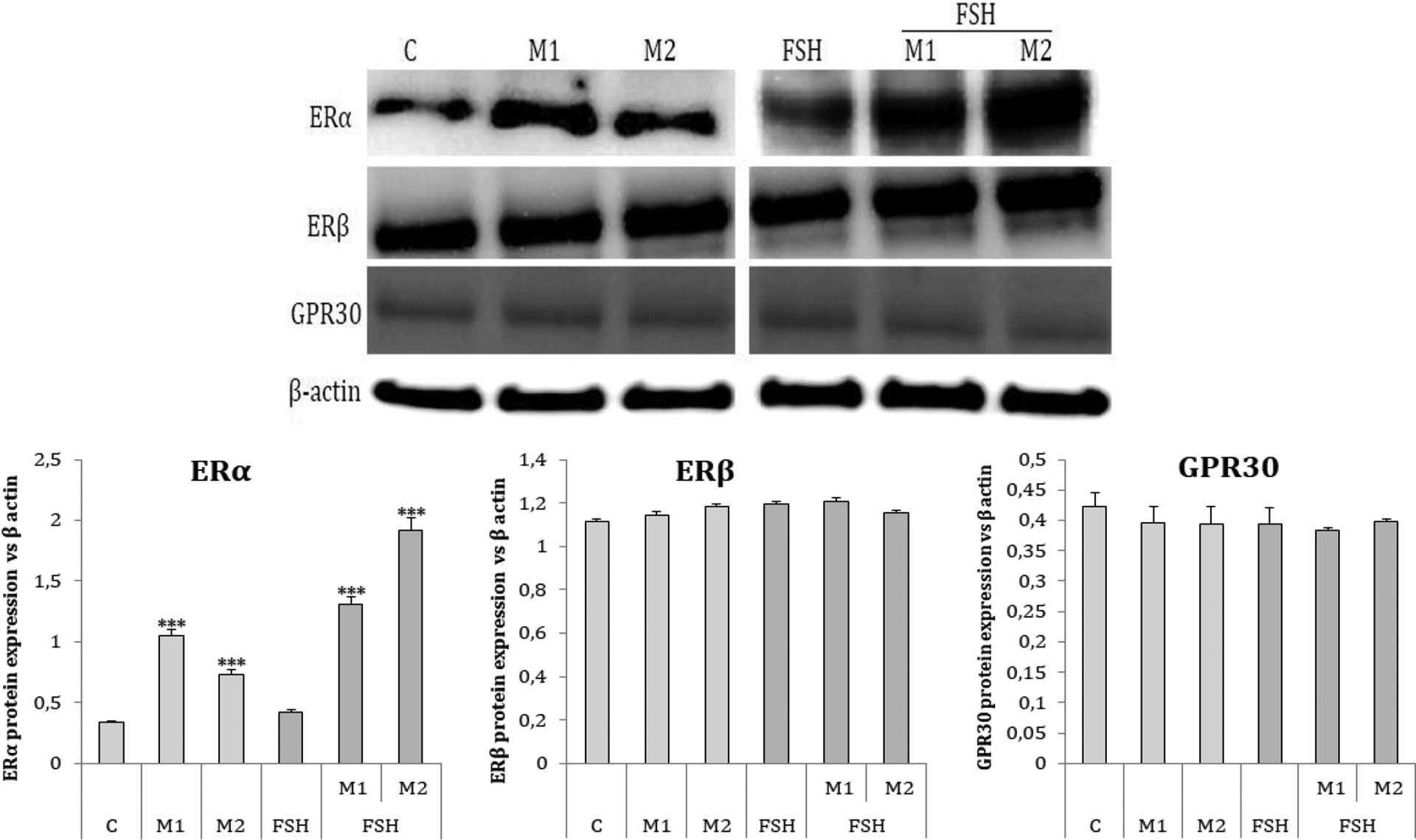
Effect of PAH mixtures M1 and M2 on basal (C) and FSH-stimulated (FSH) ERα, ERβ and GPR30 protein expression. Protein levels were densitometrically scanned and normalised against the β-actin (42 kDa) signal. Statistically significant differences between mixture treated cells in control and mixture treated cells in FSH-stimulated cells are indicated by ***p < 0.001. Experiments were performed three times (n = 3). The blot is representative of the three independent experiments. PAH: polycyclic aromatic hydrocarbon; ER: oestrogen receptor; GPER: G protein-coupled receptors.
Effect of PAH mixtures on basal and FSH-stimulated E2 secretion and P450arom protein expression
FSH had a stimulatory effect on oestradiol secretion (106.84 pg/ml vs. 75.05 pg/ml in control) and P450arom expression (1.4-fold). Both M1 and M2 decreased basal (50.8 and 50.1 pg/ml in M1 and M2 treated cells, respectively vs. 75.05 in control) and FSH-stimulated (74.0 and 78.0 pg/ml in M1 and M2 treated cells, respectively vs. 106.84 in FSH treated cells) oestradiol secretion (Figure 4(a)) and P450arom expression (1.3-fold in control and 5-fold in FSH treated cells; Figure 4(b)).
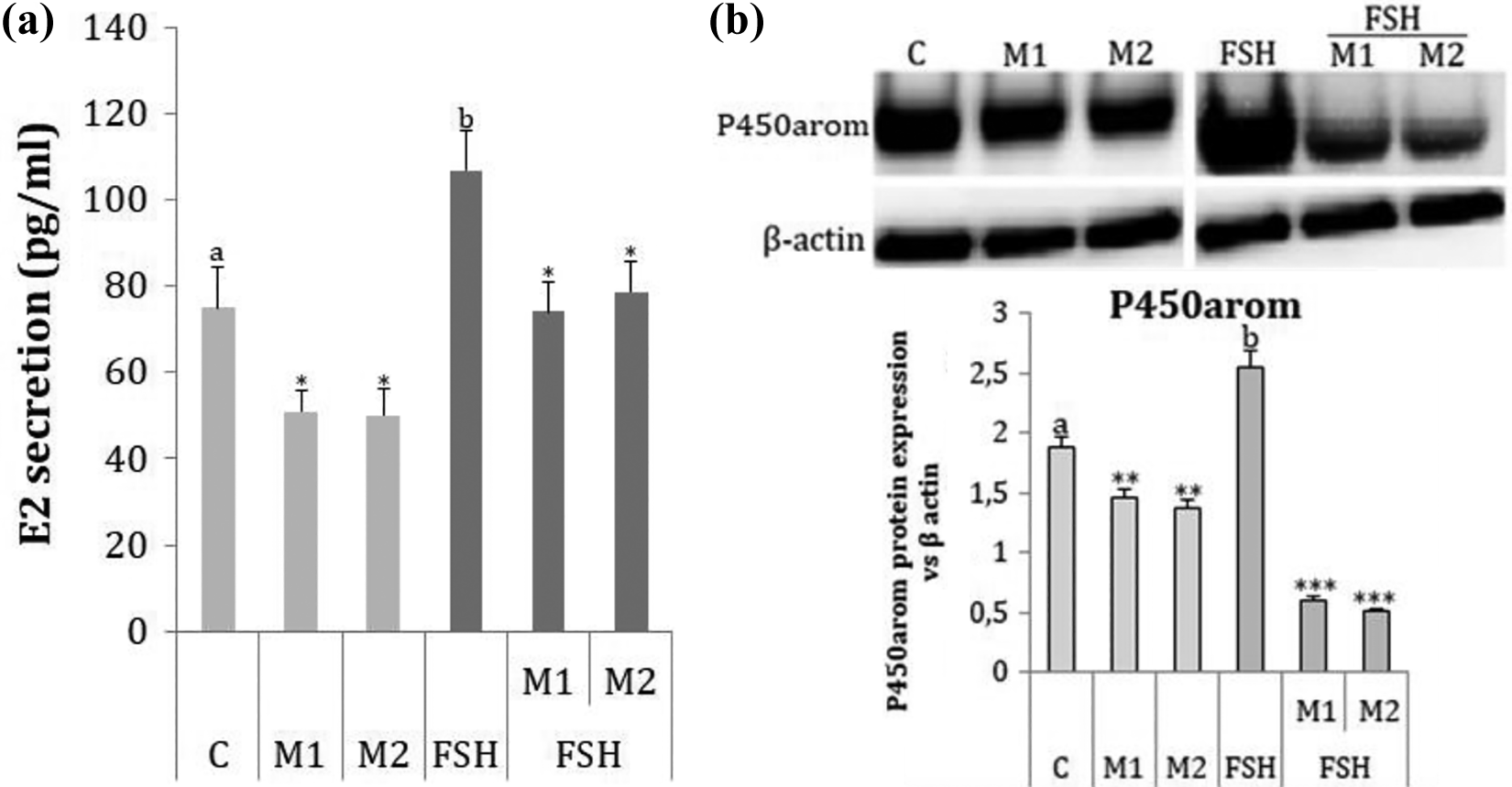
Effect of M1 and M2 mixtures on basal (C) and FSH-stimulated (FSH) E2 secretion (a) and P450arom protein expression (b). Protein levels were densitometrically scanned and normalised against the β-actin (42 kDa) signal. Statistically significant differences between basal (C) and FSH-treated cells (FSH) are indicated with different letters, with a < b. Statistically significant differences between mixture treated cells in control and mixture treated cells in FSH-stimulated cells are indicated by *p < 0.05, **p < 0.01 and ***p < 0.001. Experiments were performed three times (n = 3). FSH: follicle-stimulating hormone.
Effect of ESR1, ESR2 and GPER1 gene silencing on E2 secretion
In both basal and FSH treated cells, siESR1 and siGPER1 reversed the inhibitory effect of M1 and M2 on E2 secretion. By contrast, the inhibitory effect of the mixtures was still evident in siESR2 cells (Figure 5).

Effect of PAH mixture M1 (a) and M2 (b) on basal (C) and FSH-stimulated (FSH) E2 secretion after siESR1/siESR2/siGPER1 gene silencing. Differences between M1/M2 versus siESR1, siESR2, siGPER1 in control and FSH-treated cells are indicated by *p < 0.05, **p < 0.01. Experiments were performed three times (n = 3). PAH: polycyclic aromatic hydrocarbon; FSH: follicle-stimulating hormone; ESR1: oestradiol receptor alfa; ESR2: oestradiol receptor beta; GPER: G protein-coupled receptors.
Effect of AHR, ESR1, ESR2 and GPER1 gene silencing on P450arom protein expression
In both basal and FSH treated cells, siAHR, siESR2 and siGPER1 reversed the inhibitory effect of M1 and M2 on P450arom protein expression. By contrast, the inhibitory effect of the mixtures was still evident in siESR1 cells (Figure 6).
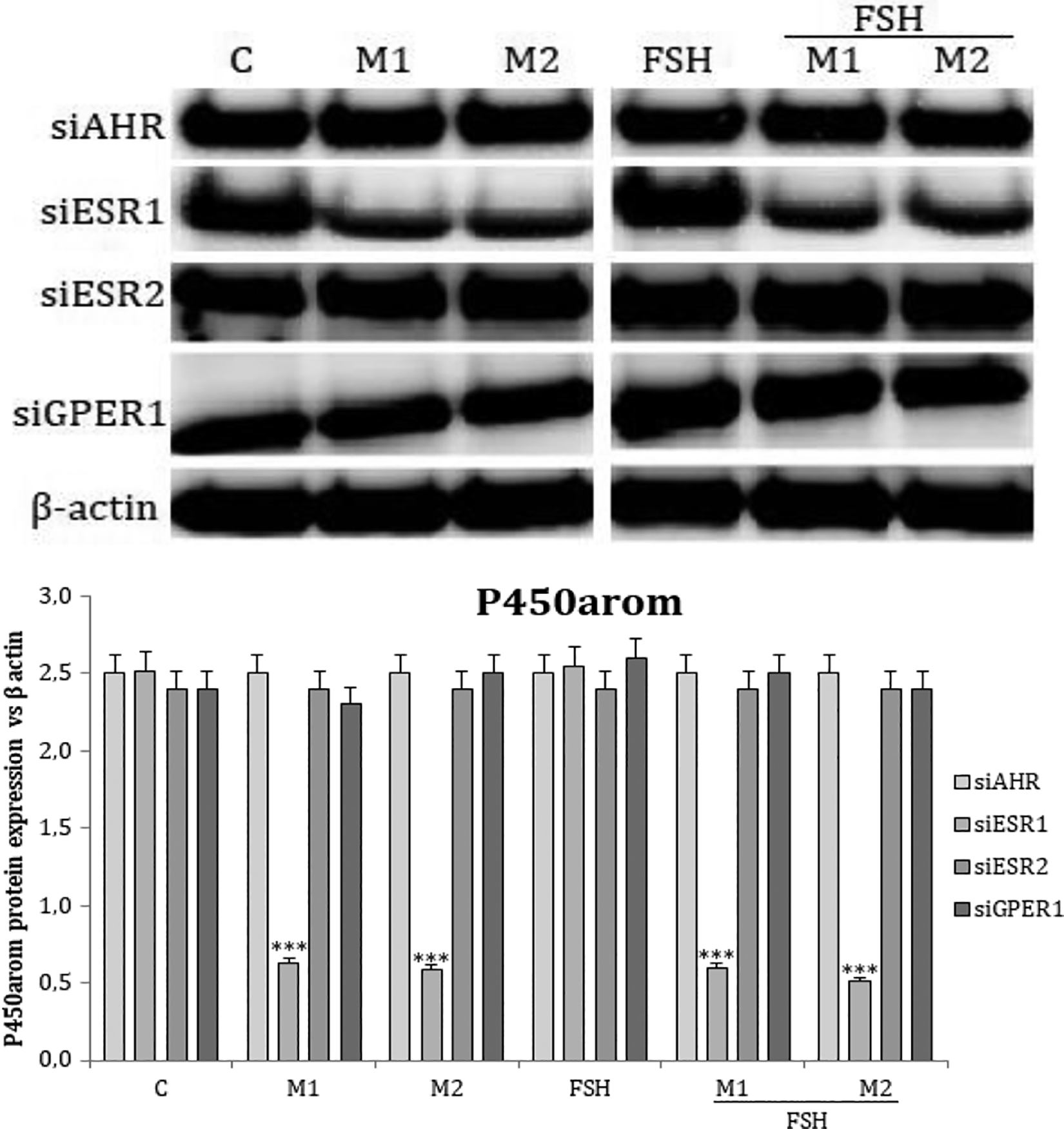
Effect of PAH mixtures M1 and M2 on basal (C) and FSH stimulated (FSH) of P450arom protein expression after siAHR/siESR1/siESR2/siGPER1 gene silencing. Protein levels were densitometrically scanned and normalised against the β-actin (42 kDa) signal. Differences between M1/M2 versus siAHR, siESR1, siESR2, siGPER1 in control and FSH-treated cells are indicated by ***p < 0.001. Experiments were performed three times (n = 3). The blot is representative of the three independent experiments. PAH: polycyclic aromatic hydrocarbon; AHR: aryl hydrocarbon receptor; FSH: follicle-stimulating hormone; P450arom: aromatase cytochrome P450; ESR1: oestradiol receptor alfa; ESR2: oestradiol receptor beta; GPER: G protein-coupled receptors
Discussion
The available data on the toxicity of PAH mixtures are very sparse and data concerning direct action on ovarian function is not presently available. The presented data show that PAH mixtures, in real-life levels, had no effect on FSHR protein expression in granulosa cells collected from antral follicles. However, they had an inhibitory effect on basal and FSH-stimulated E2 secretion by a direct impact on P450arom, an enzyme involved in the conversion of testosterone to E2. FSHRs are almost exclusively expressed in granulosa cells, and FSH action is probably most clearly reflected in the intrafollicular hormone milieu of the antral follicles. This study shows that FSH decreased expression of its own receptor FSHR. Whether or not FSH regulates its own receptor has been controversial, with earlier reports of both up- and downregulation of ligand binding after treatment with FSH. 18 These differences might partly be related to the species and model used, the stage of folliculogenesis being examined and the time of assaying after treatment with FSH. Steady-state levels of all four FSHR transcripts are increased in rat ovaries after in vivo equine chorionic gonadotropin stimulation, 19,20 are associated with an increase in FSH binding 21 and subsequently declined with the addition of a luteinising dose of hCG. 19,21 The influence of FSH is dose and time dependent. A single large dose of FSH downregulated the number of FSH-binding sites and also FSHR mRNA levels. 22 In vitro studies, using granulosa cells from oestrogen-treated immature rats, showed a transient decrease in FSHR mRNA 2–6 h after FSH treatment and then levels recovered. 23 Observations made 12–60 h after FSH treatment show maintenance of FSHR mRNA steady-state levels at pre-treatment levels, preventing the decline observed in the controls that did not receive FSH. 24,25 In addition, FSH repressed FSHR mRNA in Sertoli cells, which are the equivalent of granulosa cells in the testis. 26 Griswold et al. 27 showed that FSHR mRNA levels decreased by 50% after 5 h of FSH stimulation. In the current study, PAH mixtures had no effect on basal FSHR protein expression. Hoffmann and Oris 28 determined the effect of benzo[a]pyrene (BaP) on the transcription of genes involved in zebrafish reproduction, including gonadotropins FSH and LH, steroidogenic enzymes cytochrome P450 family (CYP) 11A1, CYP17, CYP19A1, CYP19A2 and 20β-hydroxysteroid dehydrogenase (20β-HSD), ERβ and vitellogenin. They showed no effects on the expression of FSH or LH, results which are consistent with the current study. By contrast, Archibong et al. 29 found that plasma FSH concentrations among BaP-exposed rats were significantly higher at all stages of the oestrous cycle, compared to the unexposed control.
We showed that the inhibitory effect on E2 secretion by human granulosa cells was associated with a direct impact on P450arom expression. PAHs in the environment are regarded as endocrine disruptors, which interfere with the homeostasis of organisms by mimicking endogenous hormones and disrupting steroidogenic enzyme function, including aromatase and as a consequence, potentially altering normal reproductive function. However, not many studies have evaluated their ability to directly alter levels of ovarian steroids. PAH mixtures derived from freshwater or coastal sediments have been found to produce mainly oestrogen-like effects, although in some cases anti-oestrogenic responses are obtained. 30 Polar compounds contribute significantly to the overall oestrogen-like effect of sediment samples. The variability of effects of complex environmental mixtures of PAHs and related compounds on ER signalling could be linked to their distinct compositions. Therefore, mixtures containing PAHs, that primarily act as potent AHR agonists, are likely to suppress ER signalling, while other mixtures, containing primarily hydroxylated or other polar compounds, would exhibit oestrogen-like effects. Using four single tested PAHs (naphthalene, anthracene, phenanthrene and pyrene), occurring at the highest levels in both maternal blood and placental tissue, Drwal et al. 31 described that only naphthalene and pyrene decrease the production of E2 and P4 in placental JEG-3 cells. However, different activities of the individual compounds and their mixtures must be taken into account. Sevastyanova et al. 32 showed that exposure to PAHs always involves complex action of particular compounds that may induce synergistic or antagonistic effects. Nevertheless, environmental mixtures may also contain additional endocrine-active chemicals, which further modulate their overall effect on ER activity. 33 The topic of PAH-induced depletion of preantral follicles has been briefly reviewed elsewhere. 34 Most reports concern the harmful effects of BaP. When female scallops were exposed to BaP, the levels of the sex steroids, such as 17β-oestradiol, testosterone and progesterone in the ovaries, were significantly reduced in a dose-dependent manner, and the expression of oestrogen-metabolising enzymes, like CYP17, was also decreased, implying that steroidogenesis was limited following exposure to BaP. 35 In addition, adult female croaker fish that received BaP had significantly lower E2 and testosterone concentrations, and ovarian growth was decreased compared to the control. 36 Similarly, inhibition of ovarian steroidogenesis and decrease in E2 production were observed in flounder ovarian tissue due to an inhibitory effect of phenanthrene, BaP and chrysene on aromatase, compared to the control. 37 In vitro inhibition of aromatase activity and increasing doses of two polynuclear aromatic hydrocarbons significantly reduced follicular 17β-oestradiol production in Coho salmon ovarian tissue. 38 Patel et al. 39 showed that BaP can potentially disrupt steroid homeostasis and lead to reproductive deficits by significantly decreasing ovarian aromatase activity. Research on rat models showed that treatment with BaP inhibited follicular growth and decreased E2 in isolated preantral follicles 40 and also decreased aromatase mRNA and protein. 41 Despite the limited data currently available, a clear influence of PAHs on steroidogenesis is evident. Additionally, Banerjee et al. 42 suggest that StAR may be a key steroidogenic protein that is targeted by BaP or other PAHs in Leydig cells’ steroidogenesis. Vidal et al. 43 described that in vitro exposure to extracts of cigarette smoke produced a direct inhibition of granulosa cell aromatase activity. Tanko and Christiansen 44 proposed that increased catechol oestrogen formation is a mechanism for the anti-oestrogenic effect of smoking.
The receptor mechanism of PAH mixture action on oestradiol secretion was defined by analysing ESR1, ESR2 and GPER1 protein expression. Both mixtures increased ESR1 protein expression. In addition, the action of the mixture on E2 secretion after gene silencing was defined. For both basal- and FSH-treated cells, siESR1 and siGPER1 reversed the inhibitory effect of both mixtures on E2 secretion. By contrast, siESR2 did not reverse the inhibitory effect. Many EDCs interfere with female reproductive function by activating or antagonising ESRs. Different EDCs show distinct effects on the ovary, depending on their binding affinity to different ESRs. PAHs can interact with ERs, interfering with the oestrogen signalling pathways involved in important reproductive processes. Exposure to PAHs has been shown to suppress the oestrogen response element (ERE), increase E2 metabolism and downregulate ESR1 protein levels. 45 Additionally, ERs and AHR could be involved in the expression of aromatase. The gene for aromatase has both EREs and xenobiotic responsive elements in its promoter, 46 so the expression of aromatase can be downregulated by anti-oestrogens through classical ERs or by dioxin-like chemicals through the AHR receptor. Reduced aromatase activity by the AHR receptor and ERs is caused by BaP and dioxin-like PAHs. 47 There is extensive evidence showing that crosstalk between the ER and AHR systems leads to inhibition of oestrogenic signalling both in vitro and in vivo. 45 ERs primarily mediate the biological actions of endogenously produced oestrogens; however, due to their promiscuous ligand-binding activity, they also show affinity for environmental pollutants, such as certain PAHs, phthalates and pesticides, thus termed xenoestrogens. 3-Methylcholanthrene (3MC), a PAH AHR agonist, has been reported to activate AHR, such that it interacts with unliganded ERα, and the entire AHR/ERα complex is recruited to ER genes, suggesting that ER signalling is modulated by co-regulatory-like functions of AHR. 48 AHR agonists, such as 3MC, have recently been shown to directly activate ESR1 and are suggested to represent a new class of mixed AHR/ER agonists. 49 The most studied mode of actions of xenoestrogens is focused on their ability to bind and activate ERs in target tissues. 50 However, it is of note that the two ERs mediate distinct biological effects in many tissues, such as the mammary glands, bone, brain and vascular system, in both males and females. ESR1 and ESR2 form heterodimers, and these can modulate the activities of their respective homodimers. 51 ESR2 exhibits an antagonistic action on ESR1-mediated signalling, 52,53 highlighting the importance of the balance between the two ER subtypes in oestrogen action. Several studies have aimed to assess the impact of the AHR-mediated xenobiotic response pathway on ER signalling. 54,55 The ER is a ligand-activated transcription factor with two main ER subtypes (ESR1 and ESR2). They are activated by oestrogens such as 17β-oestradiol, which play an important physiological role in female and also in male reproduction. 56 The different affinities to ESR1 of single PAHs and their mixtures, as described by Fertuck et al., 57 should be taken into consideration. In the ovary, ESR1 is expressed primarily in theca and interstitial cells, and its main function is to regulate steroidogenesis in theca cells. 58 In contrast, ESR2 is predominantly expressed in granulosa cells, and its main roles are FSH-directed granulosa cell differentiation, follicle maturation and ovulation. 59
Therefore, we were interested in determining the ability of PAH mixtures to affect ESR2 protein expression and E2 secretion, by assessing the effect of ESR2 gene silencing. Neither PAH mixture had an effect on ESR2 protein expression, and ESR2 gene silencing did not reverse the inhibitory effect on E2 secretion; however, the inhibitory effect on P450arom expression was reversed, suggesting that the expression of aromatase can be downregulated by anti-oestrogens, not only through classical ESR1 46 but also ESR2. Additionally, siAHR reversed the inhibitory effect of both mixtures on P450arom expression. AHR agonists have been extensively investigated as anti-oestrogens in the rodent mammary gland and uterus, and in human breast cancer cell lines, 60 showing that this effect is complex and related to inhibitory crosstalk between AHR and the ERs. 61 –63 Receptor AHR mediates most of the toxic and endocrine-disruptive actions of aromatic compounds in the ovary; however, paradoxically, this receptor has also been shown to play important roles in normal female reproductive function. Gene-depletion studies in mice have suggested that the AHR controls folliculogenesis by inducing apoptosis, which leads to germ cell nest breakdown and formation of primordial follicles. 64 It also regulates ovarian follicular growth and steroidogenesis in the antral follicles. 65 –67 Strong evidence in mouse models, from three independent research groups, where the AHR gene has been deleted or ‘knocked out’, supports a role for AHR in regulating ovarian follicle growth, granulosa cell proliferation and E2 synthesis and metabolism. 65 The reversed inhibitory effects of mixtures on P450arom secretion after AHR gene silencing confirmed the participation of this receptor in the action of PAHs in granulosa cell function. The results of the current study are consistent with those of Chaloupka et al. 11 who, in determining the effects of several different single PAH congeners, suggested that PAH interaction with AHR induced a broad spectrum of anti-oestrogenic responses in MCF-7 cells.
Identifying GPER1 as the plasma membrane receptor for oestrogens provides a higher level of complexity to the mechanisms of action of these hormones. 68 GPER1 is able to bind 17β-oestradiol and allows fast non-genomic responses by oestrogens, such as the stimulation of mitogen-activated protein kinase pathways, adenylyl cyclase or proto-oncogene c-fos expression in breast cancer cell line SKBR3, which does not express the classical ERs. 69,70 Since GPER1 is expressed in a wide number of cell types, it could potentially mimic environmental oestrogen effects in a great number of tissues. The current study showed for the first time that, although it had no effect on GPER1 protein expression, siGPER1 gene silencing reversed the inhibitory effect of both PAH mixtures on E2 secretion, suggesting compounds, such as naphthalene, phenanthrene, anthracene, fluoranthene and pyrene, could activate the GPER1 receptor.
The main highlights of our data are as follows: (1) determination for the first time the potential action of 16 PAH mixtures (declared by the US Environmental Protection Agency) in real-life levels noted in women’s blood during delivery, on ovarian follicular steroidogenesis. We cannot know for sure if the effects in cells and even animal models represent what will happen in humans. No less than the fact that in real-world situations, environmental and even occupational exposures are rarely due to a single chemical, but complex chemical mixtures. This also applies to PAH mixtures occurring in air pollutants, so humans are exposed not only to BaP or other rarely tested single PAHs but to mixtures of many compounds from which the 16 markers are identified, and (2) this study revealed that activation of GPER1 should be taken into consideration as an additional receptor involved in anti-oestrogen action of environmental PAH mixtures. Although further studies are needed, our results indicate that GPER1 and its agonists could be considered as potential tools in reducing the anti-oestrogenic effects of PAHs in the ovary. The main weaknesses of our data are that these results are based on experiments performed on the granulosa cell line. Studies with the use of cells obtained from women in the first oestrogenic cycle after delivery would be necessary. Maternal exposure to PAHs before pregnancy and/or during lactation may be responsible for the disturbances of the oestrogenic cycle, for compromises of ovarian reserves of female offspring 71 or the probability of polycystic ovarian syndrome (PCOS). Determination of the level of 16 PAH markers in the blood of women with fertility problems or PCOS (manifested by the arrest of follicular increase at the level of the antral follicles) could help draw appropriate conclusions.
Footnotes
Author’s note
This article includes some data presented in Karolina Zajda’s PhD dissertation.
Author contributions
KZ performed the experiments and compiled the results. KZ and EG designed the experiments and wrote the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Science Centre, Poland, project number 2015/17/B/NZ7/02954 and partly by DS/MND/WBiNoZ/IZ/39/2017.
