Abstract
Zonisamide (ZNS) is an anticonvulsant which is used to treat the symptoms of epilepsy. Although it is frequently used during reproductive ages, studies that investigated the effects of ZNS on reproductive system are limited. Therefore, we aimed to assess the effects of ZNS on male reproductive system by oral administration to rats in 25, 50, and 100 mg/kg doses for 28 days. After the exposure period, sperm concentration, motility, morphology, and DNA damage, as biomarkers of reproductive toxic effects, were determined, and histopathological examination of testis was performed. In addition, levels of the hormones that play a role in the regulation of reproductive functions, such as follicle-stimulating hormone, luteinizing hormone (LH), and testosterone were measured and the levels of oxidative stress biomarkers that take part in the reproductive pathologies such as catalase, superoxide dismutase, glutathione, and malondialdehyde, were determined. Reproductive toxic effects related to ZNS administration were shown by the significant decrease of sperm concentration and normal sperm morphology in ZNS groups. Additionally, pathological findings were observed in the testicular tissues of ZNS-administered groups dose dependently. In addition, serum LH and testosterone levels were significantly decreased in the ZNS groups. Decreased catalase activities and increased malondialdehyde levels in ZNS groups were evaluated as oxidative stress findings in the testis tissue. It could be expressed that ZNS administration induced dose-dependent reproductive toxic effects in rats, and pathological findings associated with the reproductive system could be the result of that hormonal changes and testicular oxidative stress, which in turn might be considered as possible mechanisms of male reproductive toxicity.
Introduction
Determination of the adverse effects in different organs/systems during drug treatment is an important issue for drug safety. 1 Especially the evaluation of the adverse effects of drugs that are used in repeated doses is considered as a major area of this research. 2 Studies have shown that sperm quality of males was decreased in the last 60 years. 3 In the meantime, there is the fact that diversity and the amount of chemicals that humans are exposed have been increased. Therefore, it can be stated that chemicals including drugs can be the reason for toxic effects in the reproductive system of men. 4 In particular, drugs that are used at repeated doses can directly affect gonads or indirectly affect hypothalamic–pituitary–gonadal axis and, consequently, induce reproductive toxic effects in men. 5
Epilepsy is a neurological disorder characterized by the abnormal electrical activity of the brain, causes seizures or unusual behavior, sensations, and sometimes loss of consciousness. 6,7 It is one of the most common disorders of the brain affecting more than 70 million people worldwide. 8 Furthermore, it is not a completely treatable disease, but the abnormal electrical activity of the brain can be managed with antiepileptic drugs and other treatment strategies. 7,9,10 A complex interaction between epilepsy disease and reproductive function is shown in previous studies. In male patients, the frequently occurred sexual dysfunctions are decreased libido and erectile dysfunction. 11 –14 Endocrine dysfunction that occurs in epilepsy patients is considered as one of the important factors that can cause sexual dysfunction. 11,15 Furthermore, studies that investigated the relationship between sperm quality and epilepsy showed that sperm count and normal sperm morphology were decreased in patients. It is notable that infertility cases were increased in patients depending on the decrease of sperm quality. 16 –18 Additionally, antiepileptic drugs can also affect hormonal homeostasis and sperm quality, resulting in sexual dysfunction and infertility. 11,12,16,18 Zonisamide (ZNS), which has shown antiepileptic activity by blocking sodium and T-type calcium channels in central nervous system, is frequently used to control partial and generalized seizures. 19,20 In the patients under the ZNS treatment, reversible erectile dysfunction and decrease of libido have been reported. 21,22 In addition to functional toxic effects related to ZNS treatment, altered serum levels of reproductive hormones and abnormalities in sperm parameters were observed in different studies. 23,24
Epilepsy as a disease observed in reproductive ages can induce disturbances of reproductive functions as mentioned above, thus it is especially important to identify the reproductive toxic effects of the drugs used in the epilepsy treatment. Unlike the studies performed previously, this study was aimed at investigating the possible reproductive toxic effects of ZNS at repetitive pharmacological doses in male rats via the determination of sperm concentration, motility, morphology, DNA damage, and histological evaluation of testicular structure. In addition, possible mechanisms of action of the reproductive toxic effects were investigated by the determination of serum hormone levels, which play important roles in spermatogenesis and the oxidative status of testis tissue, which is extremely vulnerable to oxidative stress.
Materials and methods
Materials
The chemicals used were obtained from the following sources: urethane (Sigma-Aldrich Corporation, St Louis, Missouri, USA) and ZNS (Excegran® 100 mg, Zentiva Group, Prague, Czech Republic). Testosterone, follicle-stimulating hormone (FSH), luteinizing hormone (LH), glutathione (GSH), and malondialdehyde (MDA) levels along with superoxide dismutase (SOD) and catalase (CAT) activities were determined by enzyme-linked immunosorbent assay kits from Shanghai Sunred Biological Technology Co., Ltd (Shanghai, China).
Animals
Male Wistar rats weighing approximately 300 g and aged 10- to 12-week-old were obtained from Anadolu University Research Center for Animal Experiments. The rats were housed in laboratory conditions at a controlled temperature (24°C) and 12-h light–12-h dark cycle with free access to food and water ad libitum. Animals were acclimatized to the laboratory environment for at least 48 h before the experimental session. The experimental protocol was approved by the Local Ethical Committee on Animal Experimentation of Anadolu University, Eskişehir, Turkey (Approval Number: 2015-15).
The rats were assigned randomly into the following administration groups: ✓ Control group (C): animals received distilled water orally for 28 days (n = 10). ✓ 25 mg/kg ZNS-administered group (ZNS-25): animals received 25 mg/kg dose ZNS orally for 28 days (n = 10). ✓ 50 mg/kg ZNS-administered group (ZNS-50): animals received 50 mg/kg dose ZNS orally for 28 days (n = 10). ✓ 100 mg kg ZNS-administered group (ZNS-100): animals received 100 mg/kg dose ZNS orally for 28 days (n = 10).
The doses of ZNS were determined according to the previous studies. 25,26 Furthermore, clinical doses of ZNS are between 100 mg/day and 600 mg/day, 27 and the doses we have chosen were in accordance with the guidelines extrapolating human doses to animal doses. 28 All drugs were administered at a volume of 1 mL/100 g by dissolving in distilled water. The treatment period was in accordance with the guideline OECD 407: repeated dose oral toxicity study in rodents. 29
At the end of 24 h after the last dose, the rats were anesthetized by intraperitoneal injection of 1.5 g/kg urethane. 30 Blood samples were collected via cardiac puncture from the anesthetized rats. The left testis and epididymis were cleaned of blood in phosphate-buffered solution (PBS) (8 g/L sodium chloride, 0.2 g/L potassium chloride, 0.2 g/L monopotassium phosphate, 1.14 g/L disodium phosphate, pH 7.4) and weighed to determine relative organ weights.
Collection and evaluation of sperm samples
The cauda of the right epididymis was used to evaluate sperm parameters. Sperms were collected from the proximal cauda epididymis using the sperm isolation procedure as described by our previous studies. 31 –34
Evaluation of sperm parameters
Experimental procedures for assessment of sperm concentration, motility, and morphology were similar to our other studies. 31 –34 Additionally, sperm morphologic anomalies were evaluated as banana-shaped head, bent neck, headless, two-headed, broken tail, tailless, and bent tail (Figure 1). Detection of sperm DNA damage was performed using comet assay as described by our previous studies. 31 –34

Sperm morphology observed under ×60 magnification following zonisamide administration: (a) normal, (b) banana-shaped head, (c) bent neck, (d) headless, (e) two-headed, (f) broken tail, (g) tailless, (h) bent tail, and (i) banana-shaped head and bent tail (multiple anomalies).
Histological evaluation of the testicular tissues
The right testis tissues were sliced into small pieces (2 mm3) and then fixed in paraformaldehyde (4%) in phosphate buffer pH 7.2 for 2 h at 20–22°C for histological examinations. Histological examination for experimental procedures was conducted according to our other studies. 31 –34 Testicular injury and spermatogenesis were evaluated using Johnsen’s mean testicular biopsy score criteria. 35 A score of 1–10 was assigned to each tubule cross section according to the range from no cells to complete spermatogenesis. At the cellular level, three pathological viewpoints (spermatogonial swelling, cytoplasmic vacuolation, and detachment of basement membrane) were estimated on a semiquantitative scale from + to +++ according to their degree.
Evaluation of hormonal status
Blood samples from rats were kept at 2–8°C overnight, centrifuged for 15 min (at 4°C and at 1000 × g) and the serum was separated. The hormonal analyses were performed using commercially available kits and in accordance with the manufacturer’s instructions.
Evaluation of oxidative stress in testicular tissues
The left epididymis was used to determine the activities of SOD and CAT along with levels of GSH and MDA. The left testis was divided into equal parts and stored at −20°C after freezing in liquid nitrogen. Oxidative stress biomarkers in the testis were determined using commercially available kits and in accordance with the instructions of the manufacturer.
Statistical analysis
All data are expressed as the mean ± standard error. Statistical analyses of the groups were performed using the SigmaPlot Version 10 package program (Systat Software, San Jose, California, USA). In the sperm comet assay, one-way analysis of variance (ANOVA), followed by Dunnett’s T3 test as a post hoc test was performed. In the other experiments, one-way ANOVA, followed by Tukey’s test as a post hoc test was performed. The value of p < 0.05 was considered statistically significant.
Results
Relative weights of testis and epididymis of rats
When relative testis and epididymis weights were compared among all groups, no significant differences were observed (Table 1).
Relative weights of testis and epididymis of rats.a

ZNS-25: 25 mg/kg zonisamide-administered rats for 28 days group; ZNS-50: 50 mg/kg zonisamide-administered rats for 28 days group; ZNS-100: 100 mg/kg zonisamide-administered rats for 28 days group.
aAll data were expressed as mean ± standard error.
Sperm parameters of rats
The sperm concentration, motility, and morphology of the groups are presented in Table 2.
Sperm parameters of rats.a

ZNS-25: 25 mg/kg zonisamide-administered rats for 28 days group; ZNS-50: 50 mg/kg zonisamide-administered rats for 28 days group; ZNS-100: 100 mg/kg zonisamide-administered rats for 28 days group.
a All data were expressed as mean ± standard error.
b Different from control group (p < 0.05).
c Different from ZNS-25 group (p < 0.05).
It was determined that the sperm concentrations were significantly lower in the ZNS-50 and ZNS-100 groups compared to the control group (p < 0.05).
When the groups were compared in terms of sperm motility, even though it was observed that the motility decreased in the ZNS-administered groups compared to the control group, these decreases were only statistically significant in the ZNS-100 group (p < 0.05).
The percentage of sperm with normal morphology was statistically decreased in the ZNS-50 and ZNS-100 groups compared to the control group (p < 0.05). Also, in the 50 mg/kg ZNS-treated group, the percentage of sperm with normal morphology was statistically decreased compared to the ZNS-25 group (p < 0.05). These abnormalities were found to occur more often in the tail, represented by bent tail, broken tail, and tailless (Figure 1) in the 50 and 100 mg/kg ZNS-administered groups at 15.48% and 15.69%, respectively. Additionally, the percentage of sperm head abnormalities, including banana-shaped head, bent neck, two-headed, and headless sperms (Figure 1), was determined as 13.98% and 9.06% in the 50 and 100 mg/kg ZNS-administered groups, respectively.
The results of comet assay, which were expressed as tail moment (extent tail moment: tail length × tail%DNA/100), for sperm exposed to different doses of ZNS and for control group are shown in Figure 2. When the groups were compared in terms of tail moment and DNA damage, no significant differences were observed.
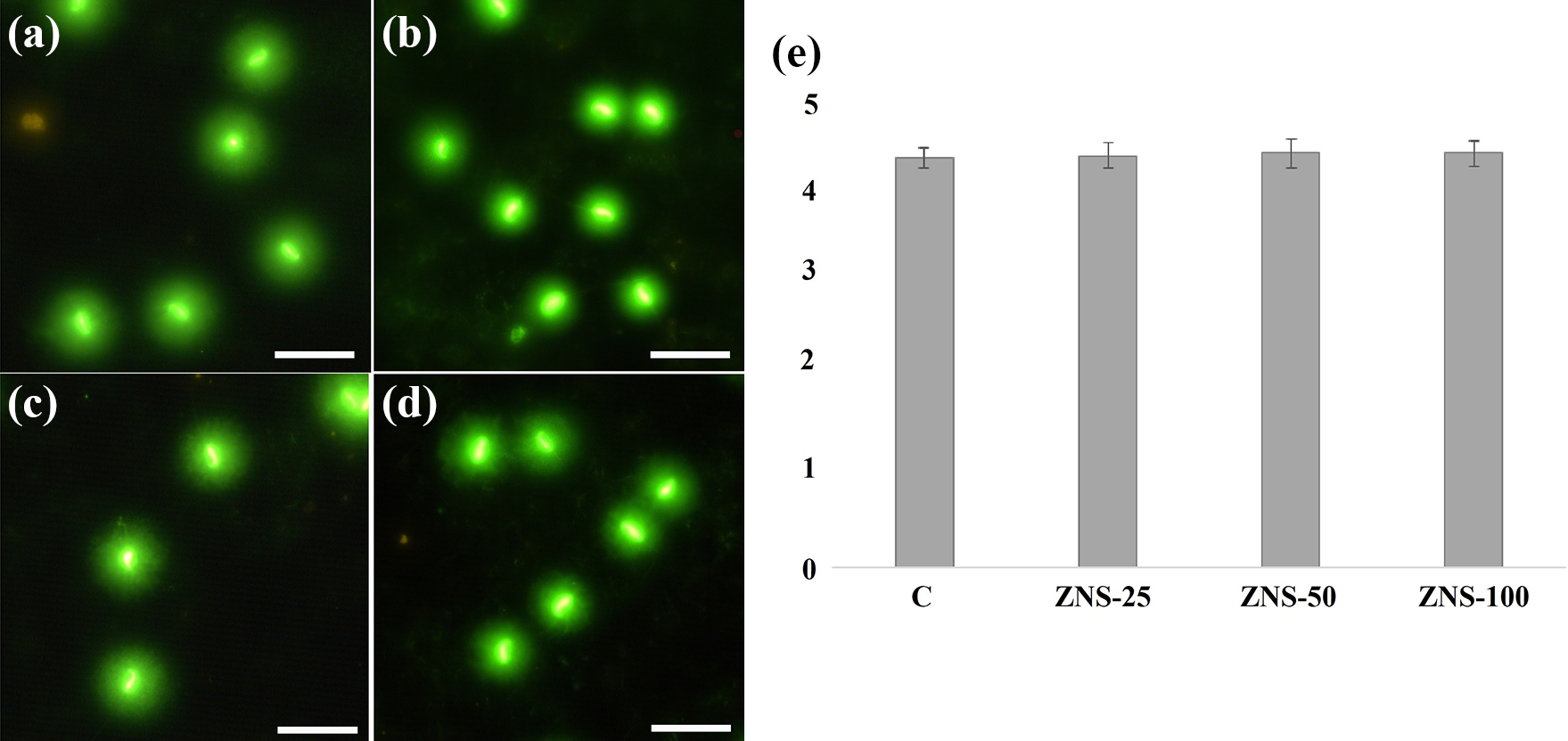
Effect of zonisamide on sperm DNA: (a) sperm comet assay photo of control group, (b) sperm comet assay photo of ZNS-25, (c) sperm comet assay photo ZNS-50, (d) sperm comet assay photo ZNS-100, and (e) tail moment graph. ZNS-25: 25 mg/kg zonisamide-administered rats for 28 days group; ZNS-50: 50 mg/kg zonisamide-administered rats for 28 days group; and ZNS-100: 100 mg/kg zonisamide-administered rats for 28 days group. All data were expressed as mean ± standard error.
Histological examination of testes of rats
Seminiferous tubules and interstitial cells of the testis tissue in control group animals were always normal. Spermatogenic series and Sertoli cells were in their usual structural organization. Numerous spermatozoa were observed in the lumen of the tubules. It was observed that Leydig cells in interstitial connective tissue are showed with normal histological appearance (Figures 3(a) and 4(a)).

(a) to (d) Low magnification of seminiferous tubules cross section. (a) Control: normal appearance of the seminiferous tubules (ST) and Leydig cells (L). (b) ZNS-25: seminiferous tubules with irregular shape (arrowhead). (c) ZNS-50: distortion of seminiferous tubules (arrowhead). (d) ZNS-100: enlarged intertubular spaces (*). Scale bar: 200 µm. ZNS-25: 25 mg/kg zonisamide-administered rats for 28 days group; ZNS-50: 50 mg/kg zonisamide-administered rats for 28 days group; and ZNS-100: 100 mg/kg zonisamide-administered rats for 28 days group.

(a) to (d) High magnification of seminiferous tubules cross section. (a) Control: normal aspect of the seminiferous tubules containing cells of the spermatogenic series (SS). (b) ZNS-25: mild intercellular vacuolation of spermatogenic series (v) and small vacuoles in Sertoli cells (arrowhead). (c) ZNS-50: thickening of basement membrane (arrowhead), mild swelling of cells in spermatogenic series (SS), and mild intercellular vacuolation (v). (d) ZNS-100: large vacuoles in sertoli cells (arrowhead), rarely observed atypical cells with dark nuclei (arrow), and detachment of basement membrane in some regions (*). Scale bar: 20 µm. ZNS-25: 25 mg/kg zonisamide-administered rats for 28 days group; ZNS-50: 50 mg/kg zonisamide-administered rats for 28 days group; and ZNS-100: 100 mg/kg zonisamide-administered rats for 28 days group.
Irregularity of the basement membrane and deformation were observed in seminiferous tubules of ZNS-25 group of animals. Sertoli cells and other cells of the spermatogenic series showed mild intercellular vacuolation (Figures 3(b) and 4(b)).
Histological analysis of testicular tissue sections belonging to ZNS-50 group of animals, thickening of the basement membrane was accompanying distortion and deformation of seminiferous tubules. Mild swelling and intercellular vacuolation were observed in spermatogenic series (Figures 3(c) and 4(c)).
Detachment of the basement membrane, deformation in seminiferous tubules and enlarged intertubular spaces were observed in ZNS-100 group of animals. There were large vacuoles inside Sertoli cells (Figures 3(d) and 4(d)). Testicular injury and spermatogenesis were evaluated using Johnsen's mean testicular biopsy score criteria and represented in Table 3.
Johnsen’s scores and semiquantitative comparison of pathology at the cellular level.a

ZNS-25: 25 mg/kg zonisamide-administered rats for 28 days group; ZNS-50: 50 mg/kg zonisamide-administered rats for 28 days group; ZNS-100: 100 mg/kg zonisamide-administered rats for 28 days group.
a All data were expressed as mean ± standard error.
b Different from control group (p < 0.05).
Serum hormone levels of rats
The serum FSH, LH, and testosterone levels of the groups are presented in Table 4.
Serum hormone levels of rats.a

ZNS-25: 25 mg/kg zonisamide-administered rats for 28 days group; ZNS-50: 50 mg/kg zonisamide-administered rats for 28 days group; ZNS-100: 100 mg/kg zonisamide-administered rats for 28 days group.
a All data were expressed as mean ± standard error.
b Different from control group (p < 0.05).
c Different from ZNS-25 group (p < 0.05).
When serum FSH levels of the groups were compared, no significant differences were observed among the groups.
According to the serum levels of LH, serum LH level of ZNS-50 and ZNS-100 groups decreased significantly compared to the control group (p < 0.05). Also, a significant decrease was observed in the ZNS-100 group compared to the ZNS-25 group (p < 0.05).
When serum testosterone levels of the groups were compared, testosterone level of the ZNS-100 group decreased compared with the control group (p < 0.05). No significant differences were observed between the ZNS groups.
Levels of oxidative stress biomarkers in testes of rats
The testis SOD, CAT, GSH, and MDA levels of the groups are presented in Table 5.
The levels of oxidative stress biomarkers in rat’s testes.a

ZNS-25: 25 mg/kg zonisamide-administered rats for 28 days group; ZNS-50: 50 mg/kg zonisamide-administered rats for 28 days group; ZNS-100: 100 mg/kg zonisamide-administered rats for 28 days group.
a All data were expressed as mean ± standard error.
b Different from control group (p < 0.05).
c Different from ZNS-25 group (p < 0.05).
SOD activities of testis tissues did not differ between the groups and no significant differences were observed between the ZNS groups when compared with the controls.
CAT activities were reduced in the ZNS-50 and ZNS-100 groups compared to the control group (p < 0.05). Also, CAT activities of the ZNS-100 group showed a significant decrease compared to the ZNS-25 group (p < 0.05).
GSH levels of testis tissues did not differ between the groups and no significant differences were observed between the ZNS groups when compared with the controls.
MDA levels were increased in the ZNS-50 and ZNS-100 groups compared to the control group (p < 0.05). Also, MDA levels of the ZNS-100 group showed a significant increase compared to the ZNS-25 group (p < 0.05).
Discussion
In our study, we evaluated the reproductive toxic effects of ZNS with repeated pharmacological dosing in male rats, independently of other risk factors related to reproductive toxicity. The results showed that ZNS administration decreased sperm concentration, motility, and normal sperm morphology, induced histopathological changes in testicular tissue in a dose-dependent manner. Oxidative stress observed in testicular tissues and the alterations occurred in serum hormones levels could be mechanisms in charge of reproductive toxic effects induced by ZNS administration.
Determination of organ weights is considered as an indicator of reproductive toxic effects. Additionally, the determination of relative organ weights is a more accepted standard than the absolute weights. Testis, epididymis, pituitary gland, seminal vesicle, and prostate weights are used in the evaluation of reproductive toxic effects. 36 Also, there are studies in which any change of the testis and epididymis weights were not observed, while the reproductive toxic effects were shown to be induced. 37 –40 According to our results, relative testis and epididymis weights did not change with ZNS administration. It can be stated that the reproductive toxic effect findings obtained with ZNS administration were not accompanied by the changes of the relative organ weights.
World Health Organization emphasizes that determination of sperm concentration, motility, and morphology in semen provides the basis for the evaluation of reproductive functions in men. Oligozoospermia, asthenospermia, teratozoospermia, or combination of these pathologies which can be induced by xenobiotics are interpreted as the indicators of reproductive toxicity. 41,42 It was determined that the sperm concentrations were significantly decreased with ZNS administration in our study. Although it is stated that ZNS has no effect on the neuronal responses to γ-aminobutyric acid (GABA), studies have shown that GABAergic transmission was evoked with ZNS treatment. 43 –45 At this point, it was emphasized that the increase of GABAergic transmission suppressed gonadotropin hormone-releasing hormone (GnRH) release from the hypothalamus. 46,47 Consequently, the decrease of GnRH release causes the decline of LH release from the anterior pituitary, therefore diminishing testosterone production in Leydig cells, resulting in both sexual dysfunction and inhibition of spermatogenesis. 11 It is possible that decreased sperm concentration might be associated with the suppression of the hypothalamic–pituitary–gonadal axis with ZNS administration in our study. Additionally, our results were in agreement with the results of Malaki et al.’s study which showed decreased sperm concentrations with ZNS administration dose dependently. 24 Similarly, sperm motility is also regulated by the hypothalamic–pituitary–gonadal axis. 48,49 In our study, sperm motility was also reduced due to the suppression of GnRH release based on the increase of GABAergic transmission by ZNS administration in the high-dose group. Furthermore, intracellular sodium and calcium levels are critically important for sperm motility, viability, and acrosome reaction. 50 –54 It is known that ZNS’s mechanisms of action were considered to be its inhibitory effects on voltage-gated sodium channel and voltage-sensitive calcium channels. 55,56 Therefore, it could be concluded that the decrease of sperm motility with ZNS administration may be associated with the mechanism of action of this drug including blocking of sodium channels and reducing calcium channel currents. Already it is expressed that antiepileptic drugs reduce sperm motility via stabilizing cellular membranes. 57,58 Also, it has been shown that xenobiotic-induced oxidative stress reduces sperm motility, 59,60 so at this point, it can be emphasized that ZNS administration-induced oxidative stress in testicular tissue dose dependently in our study. On the other hand, it is generally known that the decrease of sperm motility is accompanied by the increase of sperm morphological anomalies. 61 The fact that the percentage of sperms with abnormal morphology were increased in ZNS administration in our study dose dependently also supports the previous information. It could also be expressed that sperm morphological anomalies can also occur as a result of oxidative stress in testicular tissue similar to disturbances of sperm motility. 52
As a marker of reproductive function, the DNA’s structural integrity needs to be investigated to evaluate sperm function and its structural changes. 62 –64 Comet assay which is a simple and sensitive method for determining DNA strand breaks is used to measure sperm DNA damage in our study. Different parameters such as tail moment, tail DNA, and tail length for the quantification of DNA damage were calculated. 65,66 According to our results, it could be concluded that ZNS administration did not cause sperm DNA damage at the doses administered. Sperm DNA is susceptible to damage because of chromatin condensation, limited DNA repair mechanisms, and production of excess reactive oxygen species during spermatogenesis. 67 But, sperm DNA is protected against oxidative damage via its antioxidant defense mechanisms. Additionally, the highly organized and condensed structure of sperm contributes to the protection of the genome from damage. 68 At this point, it can be emphasized that sperm DNA damage occurs as a late result of oxidative stress. 69 On the other hand, in our study, the exposure period of ZNS did not reflect the intensive period of division and differentiation at early stages of spermatogenesis when DNA is more susceptible to damage. 70 Perhaps, DNA damage could occur in the later stages of longer ZNS administration periods.
Histological examination of reproductive system tissues is considered as an important biomarker for determining reproductive toxic effects induced by xenobiotics. Testis, epididymis, prostate gland, seminal vesicle, and pituitary gland are histologically evaluated in reproductive toxicity studies. It is thought that degenerative findings which are detected in tissues are associated with toxicity. 36 In testis tissues, major pathological findings were markedly observed in the high-dose group. It is known that the ongoing oxidative stress may cause morphological alterations in the testicular tissue. 71 Therefore, it is possible to associate the slight structural changes in testicular tissue with oxidative stress induced by ZNS administration in testicular tissue.
As is well-known, spermatogenesis is regulated by hypothalamic–pituitary–gonadal axis and serum hormone levels are also measured for determining reproductive function. 72 Especially the underlying pathophysiology is clarified by the determination of serum FSH, LH, and testosterone levels in patients having low sperm concentration. 49 In our study, decreased serum LH and testosterone levels were observed in the high-dose groups. It is known that GnRH neurons are responsible for the neuroendocrine control of reproductive function. 47 At this point, it is reemphasized that the release of GnRH from the hypothalamus is suppressed by GABA. 47,73 In addition, studies have shown that hypogonadotropic hypogonadism occurs with induction of GABAergic transmission. 74,75 Although it is stated that ZNS has no effect on neuronal responses to GABA, studies have shown that GABAergic transmission was evoked with ZNS treatment, 43 –45 consequently, this condition can result in the suppression of GnRH release from the hypothalamus. Additionally, a decrease of serum LH levels can directly decrease the testosterone production from Leydig cells. 76 –78 On the other hand, it is emphasized that calcium channel inhibition decreases gene expression of proteins controlling steroid hormone synthesis. 79 Also, calcium channel inhibition induced by ZNS administration may contribute to the decrease of testosterone levels. Similar to our results, the study performed by Khalil and Abdu demonstrated that serum testosterone and LH levels decreased in rats followed by ZNS administration. 23
Oxidative stress can be defined as an imbalance between oxidants and antioxidants in favor of the oxidants. 80 It is known that oxidative stress can be induced with exogenous sources such as drugs, cigarette, radiation, and endogenous conditions such as ion homeostasis disorders and ischemia. 81 Both enzymatic antioxidants (i.e. SOD, CAT, and glutathione peroxidase (GPx)) and non-enzymatic antioxidants (i.e. GSH) constitute the cellular defense mechanism against oxidative stress that damages cellular biomolecules such as lipid, protein, DNA, and RNA. 80 The measurement of the levels of antioxidants in biological samples along with MDA levels, the end product of lipid peroxidation, reflects oxidative status. 82 In our study, the decrease of CAT activity and the increase of MDA levels in the high-dose groups were interpreted as the evidence for oxidative stress in testicular tissue. The increase of oxidative stress in the testicular structure is known to cause sperm pathologies characterized by structural and functional deficiencies and inhibit spermatogenesis. 83 According to our results, it is possible that the degenerative findings in the testicular structure, reduced sperm concentration, motility, and normal morphology are associated with the oxidative stress induced in the high-dose groups. Additionally, it is also emphasized that the activities of antioxidants decreased in testicular tissue due to the decrease of testosterone levels, consequently, oxidative stress might be induced. 84,85 So, the decrease of serum testosterone levels with ZNS administration might contribute to the induction of the oxidative stress in testicular tissue.
Consequently, ZNS administration at repeated pharmacological doses induced reproductive adverse effects in our study. Additionally, the possible mechanism of the adverse effects on sperm parameters and testicular histology caused by ZNS may be testicular oxidative stress and hormonal disturbances, but the further study needs to be conducted to investigate the underlying molecular mechanism. On the other hand, further studies may also be conducted to investigate the effects of ZNS on fertility in animals. Especially ZNS therapy may be more critical in patients with anomalies of reproductive organ/functions and/or who want to have children. It is emphasized that investigating reproductive function and fertility levels in patients using ZNS through clinical studies is also necessary.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Anadolu University Scientific Research Projects Commission (Grant No. 1505S412).
