Abstract
Objective:
In this study, we aimed to investigate oxidative stress and antioxidant parameter levels in patients with carbon monoxide (CO) poisoning.
Methods:
The study was conducted prospectively between March 1, 2015 and April 30, 2016 in the pediatric emergency department. Eligible patients included children aged 0–18 years old with a diagnosis of CO poisoning. To determination of oxidative stress and antioxidant parameter levels, venous blood with heparinized and urine samples were drawn during the admission and after normobaric oxygen (NBO) and hyperbaric oxygen (HBO) treatment.
Results:
Forty-seven children with CO poisoning for study group and 29 patients as control group were included to the study. Sixteen patients treated with HBO. Basal plasma malondialdehyde levels were found to be significantly higher in the CO poisoning group when compared with the control group (p = 0.019). There is no significant difference in oxidative stress and antioxidant parameter levels except erythrocyte catalase enzyme levels in patients treated with NBO when comparing before and after NBO treatment (p > 0.05). Decreasing of basal erythrocyte catalase enzyme levels were found statistically significant after NBO treatment (p = 0.04). There was no significant difference in oxidative stress and antioxidant parameter levels in patients treated with HBO before and after therapy (p > 0.05).
Conclusions:
CO poisoning is associated with increased lipid peroxidation in children immediately after the poisoning. However, both treatment modalities including NBO or HBO do not have a significant effect on oxidative stress or antioxidant parameter levels.
Introduction
Carbon monoxide (CO) poisoning is an important cause of morbidity and mortality around the world. CO leads to hypoxia by forming carboxyhemoglobin (COHb) and shifting the oxyhemoglobin dissociation curve to the left. As less oxygen is available to the tissues, the cardiac function neuropsychological function and tissue perfusion are negatively effected. CO also causes inflammation. Hypoxia and inflammation result in oxidative stress, the release of nitric oxide and peroxynitrite, impairing mitochondrial function, the production of reactive oxygen species (ROS), lipid peroxidation, and apoptosis. 1 –3
Responses to CO exposures are variable depending on age. Children are more susceptible and may have more severe symptoms. Children exposed to CO become symptomatic earlier and recover faster than similarly exposed adults. Because children have lesser blood volume and increased minute ventilation per unit body mass as compared with adults. On the other hand, there are fewer studies on CO poisoning in children than in adults. In these studies, there is no clear explanation about pathophysiology of CO poisoning and its short- and long-term effects. However, several mechanisms especially for delayed onset of neuropsychiatric syndrome have been proposed from adult studies, including postischemic reperfusion injury, the effects of CO on the vascular endothelium and oxygen-radical-mediated brain lipid peroxygenation, and nitric oxide-related central nervous system damage. 2,4
The aim of the treatment in patients with CO poisoning is to reduce the amount of CO in the blood and restore oxygen delivery to be normal. Therefore, oxygen therapy is the key treatment for decreasing of COHb level. Hyperbaric oxygen (HBO) is used to treat some patients with CO poisoning, even though the uncertainty in identifying patients who will benefit from HBO therapy. HBO treatment elevates arterial and tissue oxygen tensions, decreases half-life of COHb level, increases adenosine triphosphate production, and reduces oxidative stress and inflammation. 2,3,5 On the other hand, there are few studies that HBO treatment may lead to an increase in the amount of dissolved oxygen and ROS, which can cause cellular damage with lipid, protein, and DNA oxidation. 6 –8 But these effects remain unclear.
Oxidative stress occurs when the production of free radicals overwhelms the antioxidant defense systems and results oxidative damage in cells. Previous studies showed that CO produces oxidative stress, causes lipid peroxidation, increases the production of ROS, and leads to cellular damage and neurotoxicity. However, effects of CO poisoning and HBO therapy on oxidative and antioxidant systems are not clear especially in children. 8 –12 In this study, we aimed to investigate oxidative stress and antioxidant parameters in children with CO poisoning treated with or without HBO therapy.
Method
CO poisoning (study) group
The study was conducted prospectively between March 1, 2015 and April 30, 2016 in the Pediatric Emergency Department (PED). Eligible patients included children aged 0–18 years old with a diagnosis of CO poisoning. The study was approved by our institution Ethics Committee (THD-2016-8971). Informed consent was taken from all patients with CO poisoning and healthy subjects.
All patients with suspected CO exposure included in the study. CO poisoning was diagnosed by using history, clinical findings, and COHb levels (more than 8%) of the patients. Patients with CO poisoning did not admit immediately after the poisoning, patients who admitted to another hospital and treated HBO therapy before coming to hospital and patients with chronic diseases excluded.
A standardized data collection sheet was prepared for this study. Demographic characteristics, causes of CO exposure, presenting complaints, physical examination findings, vital signs at triage, Glasgow Coma Scale (GCS) scores on admission, COHb level, other laboratory test results, clinical management (normobaric oxygen (NBO) or HBO therapy), length of stay in the PED and hospital ward, admission in PICU, and outcomes were all recorded on this form.
All patients were initially treated with high flow supplemental oxygen via non-rebreather mask (NBO therapy) at the emergency department. HBO is 100% oxygen at 5 atm pressure that is given to patients in a chamber for 90 min. HBO was given to the patients with abnormal neurological findings at the presentation such as transient loss of consciousness, altered mental status (GKS ≤ 14), seizure or history of loss of consciousness; COHb level equal or greater than 25% or evidence of myocardial injury such as elevation of troponin-t, ischemic echocardiograpy (ECG) changes, hypotension, or abnormal echocardiographic findings. According to our local protocol in patients with CO poisoning, cardiac biomarkers and ECG were ordered in all patients with suspected CO poisoning. If the patients had high level of troponin-t, ECG was obtained.
Sampling time in the CO poisoning and control group
Whole blood count, blood gas analysis, COHb level, troponin-t level, ECG were ordered for all patients with CO poisoning at the initial evaluation. At sixth hours after the admission and NBO therapy, only blood gas analysis and COHb level were repeated.
To determination of oxidative stress and antioxidant parameter levels, venous blood with heparinized (4 ml) and urine samples (4 ml) were drawn as shown below: For all patients: Blood and urine samples were obtained at the admission time (T1—initial time) and at sixth hours (T2). If the patient was not treated with HBO therapy in the first 6 h after the poisoning, he/she included in NBO treatment group. For patients treated with HBO therapy: Blood and urine samples were obtained at the admission time (T1—initial time), at sixth hours (T2), before HBO (T3), and after HBO (T4) therapies. (Samples were drawn as soon as possible, maximum in an hour after HBO therapy.) If the patient was not treated with HBO in the first 6 h, sampling at sixth hours (T2) was accepted as a sampling before HBO therapy (T3).
To compare of initial (T1) oxidative stress and antioxidant parameter levels in all patients with CO poisoning, venous blood with heparinized (4 ml) and urine samples (4 ml) were taken from the patients admitted to the general pediatric outpatient clinic during the healthy child visit (control group).
Preparing of samples
Heparinized blood samples were collected from patients and healthy controls. After centrifugation (2500 r/min, 10 min.), plasma samples were separated and portioned into small tubes. After removing the plasma, red blood cells were washed in an equal volume of phosphate buffer (100 mM, pH 7.4) twice. Following the second washing, supernatants were removed and erythrocytes were obtained. All samples were stored at −80°C until analysis. Additionally, the lower layer also transferred into a tube. All samples were stored at 80°C until the analyses. Similarly, all sterile urine samples were stored at 80°C until the analyses.
Analysis of oxidative stress and antioxidant parameter levels
All samples were analyzed at laboratory in the Department of Pharmaceutical Toxicology. For oxidative stress, urine 8-hydroxy deoxyguanosine (8-OHdG), plasma protein carbonyl (PK), and plasma malondialdehyde (MDA) levels; for antioxidant parameters, plasma total glutathion (GSH) levels and superoxide dismutase (SOD), catalase (CAT), and glutathion peroxidase (GPx) enzyme activities were determined. All parameters except for SOD and CAT were measured by using a commercial double-antibody sandwich enzyme-linked immunosorbent assay to assay kit following the manufacturer’s procedures. The kits were obtained from Eastbiopharm (Hangzhou Eastbıopharm Co., Ltd).
Erythrocyte CAT activity was measured as described by Aebi. 13
Erythrocyte SOD activity was measured spectrophotometrically as described by Marklund and Marklund. 14
Statistics
Statistical analysis was performed using the Statistical Package for the Social Sciences ver. 21.0 (SPSS Inc., Chicago, Illinois, USA). Numerical measurements were presented with mean ± standard deviation, median, and range; qualitative data with numbers and percentages. χ2 test was used for categorical variables. Depending on whether the distribution was normal or not, paired samples t-test, Mann–Whitney, Wilcoxon signed rank test, and Friedman test were performed to investigate the differences before and after the treatment of CO poisoning and in the control group. The p < 0.05 was considered statistically significant (IBM-SPSS 21.0 for Windows).
Results
General characteristics of the patients
Fifty-four children were diagnosed with CO poisoning during the study period. However, five patients were excluded from the study, because samples were not drawn at suitable time and two patients admitted to the emergency department after HBO treatment. Sixteen patients were treated with HBO. There is no patient treated with HBO in the first 6 h after the poisoning.
The mean age was 106.9 ± 56.6 months (13–214 months), 48.9% (23) of patients was male. Mean COHb level was 18.8 ± 7.2% (8.4–36.4) during the admission in patients with CO poisoning. There were no patients who treated with HBO in the first 6 h after the poisoning. The mean COHb level was 24.7 ± 7.1% (9.7–36.4%) in patients with CO poisoning treated with HBO. In all patients, CO exposure was dependent on defective heating systems. Thirty-four patients (72.3%) had complaints and the most common complaints were altered mental status (21.3%; 10 patients), vomiting (21.3%; 10 patients), fatigue (19.1%; 9 patients), dizziness (19.1%; 9 patients), and headache (17%; 8 patients).
Comparing basal oxidative system and antioxidant parameter levels, 29 patients were included as control group.
Comparison of CO poisoning and control groups
There were 23 male (48.9%) in the CO poisoning group and 13 male (44.8%) in the control group (p = 0.72). The mean age was 106.9 ± 56.6 months old (13–214 months) in the CO poisoning group and mean age was 78 ± 49.9 months old (6 months - 16 years) in the control group (p = 0.13) (Table 1).
Basal (T1) oxidant and antioxidant parameter levels in all patients with carbon monoxide poisoning and control groups.a

8-OHdG: 8-hydroxy deoxyguanosine; PC: protein carbonyl; MDA: malondialdehyde; SOD: superoxide dismutase; GPx: glutathion peroxidase; GSH: glutathion.
a Oxidative stress and antioxidant parameter levels were given as median (minimum–maximum).
b Mean ± standard deviation (minimum–maximum).
There are no significant differences in basal (T1) oxidant and antioxidant parameter levels when comparing CO poisoning and control groups except urine 8-OHdG and plasma MDA levels (p > 0.05). Basal urine 8-OHdG level was found lower (p = 0.002) and MDA level was found higher in the CO poisoning group (p = 0.019) (Table 1). In the CO poisoning group, when comparing basal (T1) oxidant and antioxidant status of patients with and without symptoms, there was no statistical significance (p > 0.05) (Table 2).
Basal (T1) oxidant and antioxidant parameter levels in patients with and without symptoms.a
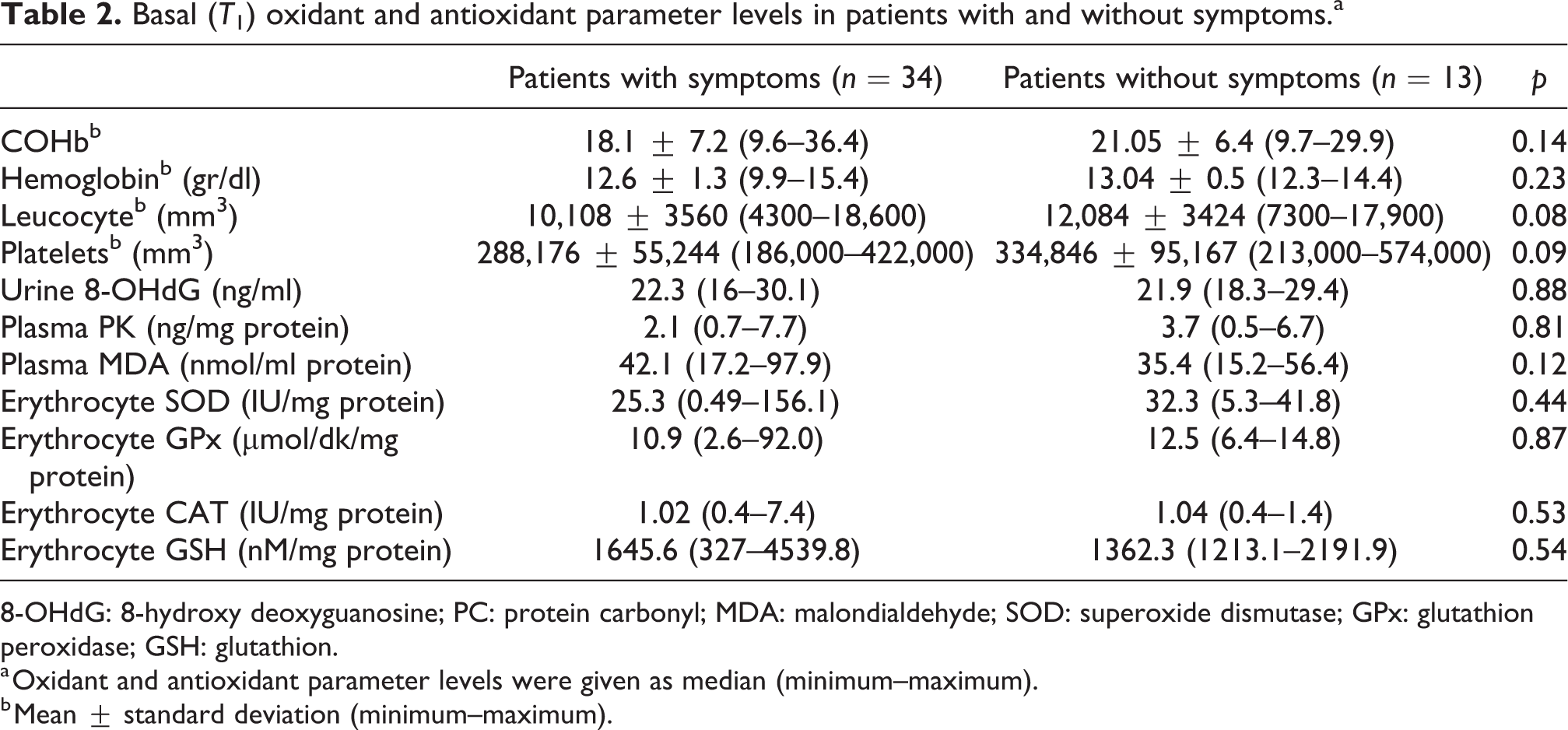
8-OHdG: 8-hydroxy deoxyguanosine; PC: protein carbonyl; MDA: malondialdehyde; SOD: superoxide dismutase; GPx: glutathion peroxidase; GSH: glutathion.
a Oxidant and antioxidant parameter levels were given as median (minimum–maximum).
b Mean ± standard deviation (minimum–maximum).
Comparison of oxidative stress and antioxidant parameter levels before (T1) and after (T2) NBO therapy
There are no significant differences in oxidative stress and antioxidant parameter levels except erythrocytes CAT enzyme levels in patients treated with NBO when comparing oxidant and antioxidant status of patients before (T1) and after treatment (T2—at sixth hours) (p > 0.05). Decreasing of basal erythrocytes CAT enzyme levels were found statistically significant after NBO treatment (p = 0.04) (Figure 1).

Oxidative stress and antioxidant parameter levels before (T1) and after (T2) normobaric oxygen therapy and comparison with control group. NBO: normobaric oxygen; 8-OHdG: 8-hydroxy deoxyguanosine; PC: protein carbonyl; MDA: malondialdehyde; SOD: superoxide dismutase; GPx: glutathion peroxidase; GSH: glutathion.
Evaluation of oxidative stress and antioxidant parameters in patients treated with HBO therapy
Sixteen patients were treated with HBO therapy. Three patients treated with HBO therapy were excluded from the study. Because samples were not drawn before and after the treatment. Eleven patients were treated with HBO at sixth hours and two patients were treated with HBO after first 6 h. The interval of sampling blood and urine before and after HBO therapy was maximum of 6 h.
There were no significant differences in oxidative stress and antioxidant parameter levels in patients treated with HBO before and after therapy (p > 0.05) (Table 3).
Oxidative stress and antioxidant parameter levels before and after hyperbaric oxygen treatment.a
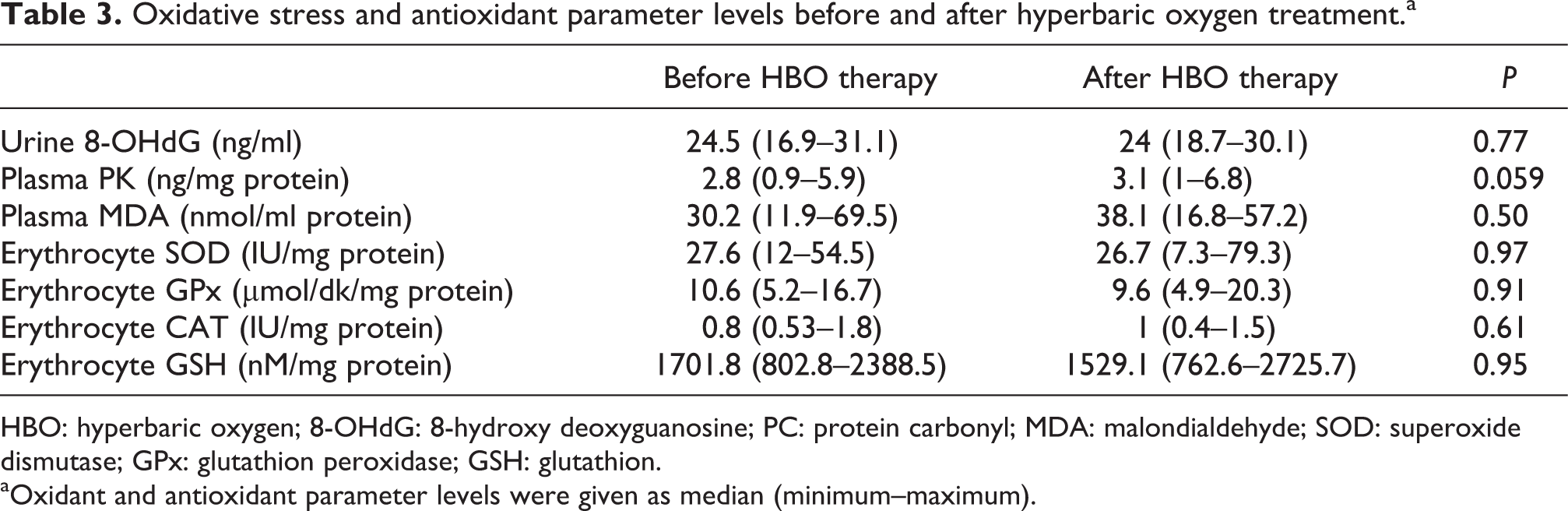
HBO: hyperbaric oxygen; 8-OHdG: 8-hydroxy deoxyguanosine; PC: protein carbonyl; MDA: malondialdehyde; SOD: superoxide dismutase; GPx: glutathion peroxidase; GSH: glutathion. aOxidant and antioxidant parameter levels were given as median (minimum–maximum).
There were also no significant differences in basal (T1) oxidative stress and antioxidant parameter levels when comparing patients treated with NBO (n = 31) and HBO (n = 16) (p > 0.05) (Table 4).
Basal (T1) oxidative stress and antioxidant parameter levels in patients treated with normobaric oxygen and hyperbaric oxygen treatment.a
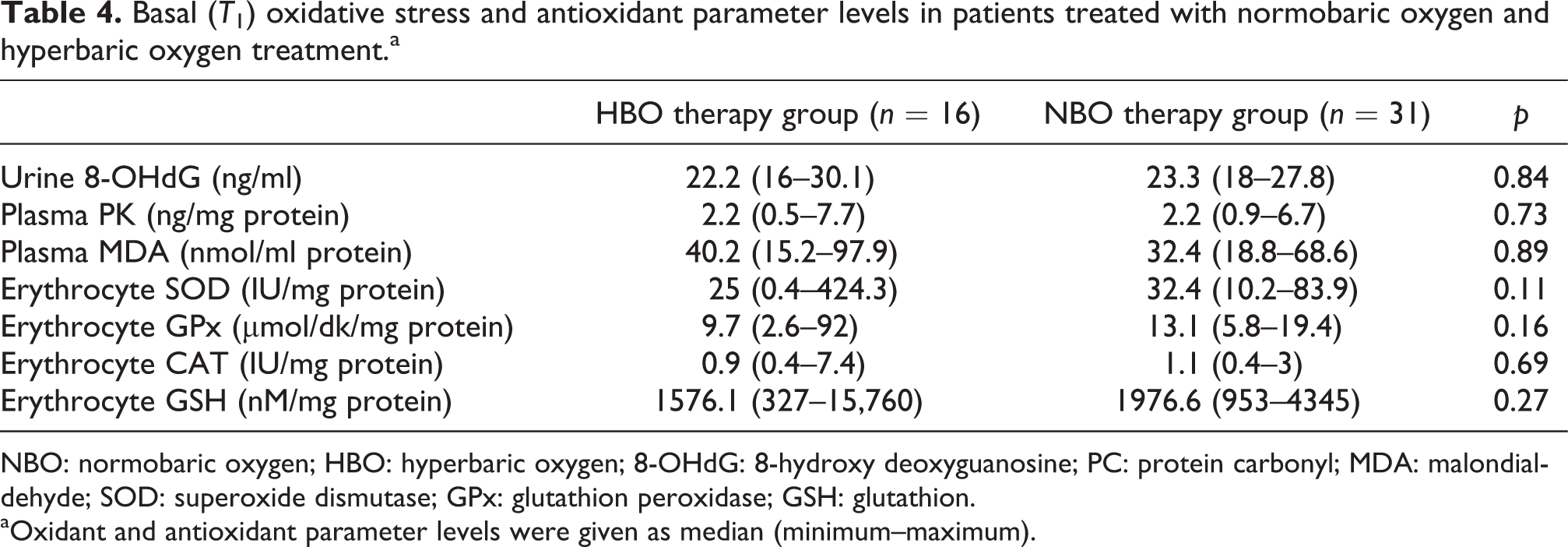
NBO: normobaric oxygen; HBO: hyperbaric oxygen; 8-OHdG: 8-hydroxy deoxyguanosine; PC: protein carbonyl; MDA: malondialdehyde; SOD: superoxide dismutase; GPx: glutathion peroxidase; GSH: glutathion. aOxidant and antioxidant parameter levels were given as median (minimum–maximum).
Discussion
In the present study, we showed that CO poisoning is associated with increased lipid peroxidation in children immediately after the poisoning. On the other hand, both treatment modalities including NBO or HBO do not have a significant effect on oxidative stress or antioxidant parameter levels except for CAT activity. As a cellular antioxidant, only CAT activity decreased at sixth hours after NBO therapy. To the best of our knowledge, this study evaluates oxidative stress and antioxidant parameter levels in children with CO poisoning is the first in the literature.
Oxidative stress defined as a disturbance in the balance between the production of ROS (free radicals) and antioxidant defenses. The shift in the balance between oxidants and antioxidants in favor of oxidants is termed as “oxidative stress.” Regulation of reducing and oxidizing state is critical for cell viability, activation, proliferation, and organ function. In recent years, there has been more evidence that pathophysiology of CO poisoning may be the result of increased free radical-mediated or ROS mediated neuronal and/or cellular injury, as shown in both experimental animal and clinical studies. 3,9,15 –17
In our study, plasma MDA level as a lipid peroxidation marker was found increased during the initial period of poisoning. But, there was no significant difference at sixth hours after the poisoning. Leucocyte count was also found higher in the poisoning group than in the control group during the admission. These results showed that lipid peroxidation and leucocyte count increased immediately after the poisoning. Previous studies also showed that CO causes lipid peroxidation through neutrophil activation to depend on tissue hypoxia and the production of ROS. 8 –12,15 –17 Yavuz et al. 17 showed that lipid peroxidation was found increased at sixth hours in rats after the CO poisoning. In an another animal study, Wang et al. 9 showed that MDA levels in serum, cerebral cortex, and hippocampus significantly increased on day 1, 3, and 7 when comparing with the level of day 0 after the experimental CO poisoning. On the contrary, the level of antioxidant enzymes including GSH-Px, GR, GSH, and anti-ROS decreased time dependent. As a result, they concluded that increased lipid peroxidation and decreased antioxidant enzymes can be responsible for CO-mediated delayed neuron damage. 9 According to these results, CO absolutely increases lipid peroxidation in children. However, it is still questionable whether the lipid peroxidation increased immediately or time-dependent after the CO poisoning. In addition, the results of these studies are not comparable of each other and our results. Because most of them are done in animals or evaluated in adults. Sampling time, methods, and measured markers were also different in these studies. Therefore, further studies must be done including more patients and evaluating the relation between delayed neurological outcome, oxidative stress, and antioxidant parameter levels.
In our study, when we compared antioxidant parameter levels during the first presentation and at sixth hours, only erythrocyte CAT activity after NBO therapy decreased. As a cellular antioxidant, catalase converts H2O2 into oxygen and water. 16 However, it was surprising that we did not observe any significant difference in erythrocyte CAT activity after HBO therapy. This result may show that after CO poisoning, H2O2 increases immediately and CAT level decreased after NBO therapy for cleaning of H2O2 in tissues, although there are no significant differences in basal CAT levels between study and control groups.
The predictors of severe CO poisoning and HBO therapy indications are not well known especially in children. In our previous study, we showed that GCS score < or = 14 and hypotension were found associated with myocardial injury. 18 In our another study, we evaluated predictors of severe clinical course in CO poisoning. In that study, severe clinical course defined as patients requiring treatment with inotropic drugs, mechanical ventilation, or having multi-organ failure. We found that low GCS score, high leucocyte count, and high troponin-t levels at the presentation were determined as risk factors associated with severe CO poisoning. 19 In the present study, we also evaluated whether oxidative stress or antioxidant parameters can be used to show the severity of CO poisoning. Therefore, we investigated the levels of oxidative stress and antioxidant parameters in patients with or without symptoms. However, there was no significant difference between patients with symptoms and without symptoms. Similarly, any significant difference was also not found between patients treated only with HBO or NBO. If patients treated with HBO therapy or patients with symptoms were taken into consideration as more severe poisoning, oxidative stress were expected significantly higher than asymptomatic patients or patients treated only NBO. Similarly, the levels of antioxidant parameters were also not different in patients treated with HBO or NBO therapy. Therefore, we should not speculate that oxidative stress and antioxidant parameter levels are enough for the decision of severe CO poisoning and therapy of choice. On the other hand, plasma MDA level might be a promising marker for the severe CO poisoning and HBO therapy decision.
Kavakli et al. 8 investigated that the effects of CO poisoning on oxidative stress parameters as total oxidant status (TOS) and total antioxidant status (TAS) in adult patients with CO poisoning. TOS level and oxidative stress index (OSI—the ratio of TOS to TAS levels) were found to increase during the first evaluation when compared to control group. After the treatment, TOS level and OSI in patients with CO poisoning were significantly decreased. However, TAS levels in the study and control groups were not changed before and after the treatment. They concluded that endogenous TAS levels do not increase in patients with CO poisoning, normal oxygen, or HBO therapies do not change TAS levels. They recommended that OSI may use for the decision of HBO therapy in patients with CO poisoning. In addition, oxidative stress parameters may be useful for long-term outcomes as an early biochemical marker in CO poisoning. However, only 12.5% of patients were received HBO therapy and the relation between treatment of choice, clinical findings and TOS, TAS levels did not investigate in their study. Therefore, the relation between oxidative stress and CO poisoning, the most useful biomarker which is shown severe poisoning and the factors affecting HBO therapy need to support by further studies. 8
HBO therapy elevates arterial and tissue oxygen tensions, promotes CO elimination and reduces oxidative stress and inflammation. HBO leads to increase of dissolved oxygen in the blood and it has been successfully used for the treatment of a variety of clinical conditions related to hypoxia. There are few studies that evaluate the effects of HBO in humans. In these studies, HBO therapy leads to increased oxidative stress that can cause cellular damage. Dennog et al. 12 showed that single HBO exposure in healthy adults has no significant differences in antioxidant vitamins A, C, E levels, and antioxidant enzymes including SOD, CAT, and GPx before and 24 h after HBO treatment. However, the heat shock protein (HSP70) which has known to play an important role in cellular protection against oxidative stress, was found significantly induced in lymphocytes after a single HBO treatment. In addition, they showed that supplementation of antioxidants including vitamin E and N-acetylcysteine before the HBO treatment did not influence the induction of DNA damage by HBO. In their study, they suggested that SOD, CAT, and GPx are not mainly responsible for the increased antioxidant defenses in the blood of humans after HBO and blood is not suitable for the detection of changes in the activity of these enzymes. 12
There are so many questions waiting to be solved about CO poisoning and treatment. One of them is the optimal number of HBO session in CO poisoning. However, there is not enough evidence about the number of HBO therapy, therapy of duration, and its pressure in patients with CO poisoning. 10 –12,15 –20 In our study, we evaluated how HBO therapy effects on oxidative stress and antioxidant parameters. However, neither oxidative stress parameters nor antioxidant parameters changed before and after HBO therapy. If patients treated with HBO therapy were considered as severe poisoning, basal oxidative stress, and antioxidant parameter levels expected to higher than patients treated only NBO therapy. However, these levels did not change before and after HBO therapy. According to these results, we can say only one HBO session does not have any significant effect. Benedetti et al. 10 evaluated the condition of oxidative stress in 12 patients undergoing prolonged exposure to HBO. The study showed that repeated exposures to HBO (15 HBO treatments) for different clinical conditions including diabetic feet, refractory chronic osteomyelitis, and aseptic osteonecrosis led to a significant accumulation of plasmatic reactive oxygen metabolites and MDA. At the end of 15 HBO sessions, reduced GSH, a-tocopherol, and retinol plasma levels were not changed. However, significantly decreased activity of erythrocytes SOD and CAT after 15th HBO treatment was observed when compared to the first HBO exposure; GPx activity remained unchanged. In this study, they concluded that prolonged HBO treatment leads to oxidative stress by affecting the response of the enzymatic antioxidant defense system. 10
Our study presents some limitations. First of all, small patients number may affect our results. If similar studies with larger sample size are done, different or significant results may yield. In addition, there was not enough study to investigate oxidative system and antioxidant status in patients with CO poisoning, especially in children. Moreover, measuring methods of different oxidant and antioxidant parameter levels, markers and sampling time are very various among in studies. Therefore, comparing of the studies is very difficult. Although we found that increased MDA level after the poisoning, we did not show any differences in all oxidative and antioxidant parameter levels, even in patients with or without symptoms due to CO poisoning. These findings may be related to sampling time, sampling type, methods, and different markers. For example, all our patients did not treated with HBO in the first 6 h after poisoning, because of given HBO therapy in another institution. Therefore, we may not have observed any significant difference in patients treated with HBO before and after treatment. Moreover, we did not show any differences in all oxidative and antioxidant parameter levels in patients treated with HBO and NBO. Finally, the most useful sampling is also unknown. It could be better the measurement of oxidative stress and antioxidant parameter levels in brain tissue instead of blood. Because these parameters may be normalized after the CO poisoning and then they increase in the brain tissue. Therefore, these questions that should be addressed by further studies.
Conclusion
This study showed that CO poisoning is associated with increased lipid peroxidation in children immediately after the poisoning. In addition, neither NBO treatment nor HBO treatment have significant effect on oxidative stress or antioxidant parameter levels except CAT activity. After NBO therapy, only CAT activity decreased at sixth hours. Therefore, it seems that only one HBO session may be safe in patients with CO poisoning. However, in our study, it is not clear that which patients should receive HBO therapy, optimal number of HBO sessions, the effects of repeated treatment on oxidative stress and antioxidant status, and long term effects of CO poisoning in children. Further studies should be done to investigate the possible relationship between oxygen treatment (HBO or NBO), oxidative stress and antioxidant parameter levels, and the long term effects of lipid peroxidation in children with CO poisoning.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Hacettepe University Scientific Research Project System.
