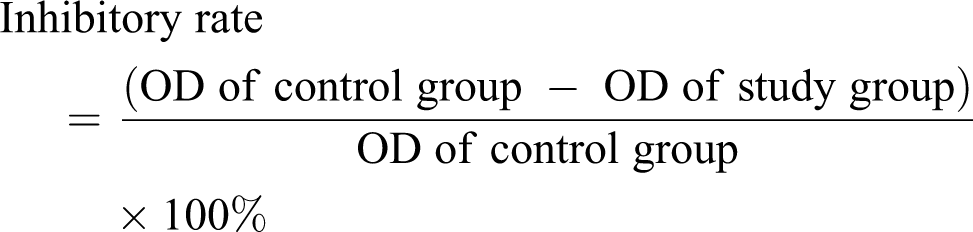Abstract
We examined the role of DNA methyltransferase 1 (DNMT1) in N-methyl-N′-nitro-N-nitrosoguanidine (MNNG)–induced malignant transformation of Kazakh esophageal epithelial (EE) cells to better understand the pathogenesis of esophageal cancer (EC). The 3-(4,5)-dimethylthiahiazo (-z-y1)-3,5-di- phenytetrazoliumromide method and colony formation assays were performed to determine the MNNG dose for malignant transformation. Colony formation assays showed the effects of different frequencies of MNNG exposure and different cell passages on malignant transformation. A nude mouse tumor experiment indicated the malignancy of Kazakh EE cells expressing high DNMT1 levels and of transformed cells. The result shows that when the dose, frequency, and time of MNNG exposure increased, cell morphology became irregular, cell-contact suppression disappeared, and cell tolerance and growth rate increased. Colony formation occurred in the Kazakh-DNMT1 group after 14 transfections and 27 passages. Significant differences in DNMT1 mRNA and protein levels were observed in different types of cells and tumor tissues (F = 140.644, p < 0.001; F = 105.545, p < 0.001). Our study demonstrated that DNMT1 could promote MNNG to induce malignant transformation of EE cells, and this study will help understand EC better in order to develop appropriate treatment strategies.
Introduction
Esophageal carcinoma (EC) is one of the most common malignant tumors worldwide. 1 About 50% of the 400,000 newly diagnosed esophageal cancer cases around the world have occurred in China, 2 making it the third leading malignant tumor of the digestive system in China after liver and gastric cancer. The occurrence of EC in China has significant regional and ethnic differences. Studies 3 have demonstrated that Linxian County in Henan Province, Yanting in Sichuan Province, and Kazakh, Xinyuan County of Yili, and Tori County of Tacheng in Xinjiang Uygur Autonomous Region are the main high-incidence areas of EC in China. The occurrence of EC is the result of multiple risk factors, such as dietary, genetic, and environmental, including nitrosamines. 4 Further, the cell malignant transformation assay is an important method to study carcinogenesis in vitro. 5 Although there have been some studies on the effect of exogenous compounds on malignant epithelial cell transformation, 6 methodologies vary and the duration of transformation is relatively long, making it difficult to gain insights into the pathogenesis of EC.
In the present study, we used N-methyl-N′-nitro-N-nitrosoguanidine (MNNG), a synthetic nitrosamine that is often used to induce tumor formation, as a carcinogen to induce high expression of DNA methyltransferase 1 (DNMT1) in vitro in Kazakh esophageal epithelial cells (EECs) and observed morphological changes occurring in passaged cell colonies, such as alterations in cell growth, composition, and malignant transformation, in an attempt to gain insight into cell malignant transformation and the development and progression of EC through clonal expansion. Through this study, we also attempt to construct a cell malignant transformation model to explore the effect of DNMT1 on malignant transformation of MNNG-exposed Kazakh EECs. We hope to provide a theoretical basis and research tool for further studies on the mechanism of the occurrence and development of esophageal cancer.
Methods
Reagents and instruments
Previously established Kazakh EECs expressing high levels of DNMT1 (6) were used in this study. Other reagents used in this study included MNNG (TCI Chemicals, Tokyo, Japan), epithelial cell medium-2 (EpiCM-2) base (ScienCell Research Laboratories, California, USA), Roswell Park Memorial Institute 1640 culture media, and fetal bovine serum (GIBCO, California, USA). We also used 0.25% trypsin, dimethyl sulfoxide (DMSO, Sigma, Saint Louis, USA), methyl thiazolyl tetrazolium (MTT, Sigma), and low melting point (LMP) agarose (HydraGene, New Jersey, USA).
Instruments used in this study included a fluorescence inverted microscope (Olympus, Japan), a carbon dioxide (CO2) constant-temperature incubator (Sanyo, Japan), a low-temperature centrifuge (Sony, Japan), and 6- and 96-well plates (Corning, South Corning, New York, USA).
Animals
BALB/c-nu nonpregnant and well-developed female nude mice aged 4–6 weeks and weighing 13–16 g (Beijing Weitong Lihua Experimental Animal Technology Co., Ltd., License No. SCXK, Beijing, 2012-0001, SPF grade) were raised in the barrier-maintained mouse facility of the First Affiliated Hospital of Xinjiang Medical University. Food and water in the feeding environment were disinfected by high temperature and high pressure. All persons who entered the environment were required to follow a strict microbial control protocol. This research has been approved by the ethics committee of the First Affiliated Hospital (permit code: IACUC20160303-08).
Cell culture
Cryogenic vials containing Kazakh EECs highly expressing DNMT1 were quickly moved to an enamel pot containing 37°C water, thawed by constant shaking, and then centrifuged at 1500 r/min for 5 min to remove the supernatant. The cell suspension in 1-mL complete medium (1 × EpicM-21 × EpicM-2; ScienCell, USA) was seeded into a 25-m2 tissue culture flask. Approximately, 80–90% confluent cells were grown in an aseptic environment, digested, and cultured for 1–2 passages.
MNNG preparation
DMSO (34 mL) was added to a brown glass bottle containing 5 g of MNNG, dissolved thoroughly, prepared into a 1-mol/L stock solution, and stored at 4°C until use. When used, 5 μL of the stock solution was aliquoted to 5 mL of medium to prepare a 1 × 10−3 mol/L storage solution.
Cell growth curve mapping
Cell growth curves were mapped by the 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide (MTT) method. Kazakh EECs expressing high levels of DNMT1 were routinely digested and prepared into a cell suspension, which was then seeded to a 96-well plate at a density of 1 × 104 cells/well with 100 μL in each well, and the edge was filled with 100-μL phosphate-buffered saline (PBS). After cells became completely confluent, 20 μL of freshly prepared MTT was added to all the holes except the edge holes and cultured at 37°C in 5% CO2 incubator away from light for 4 h. After removing the old medium, 50 μL of DMSO was added to each well, the plate was gently vortexed and incubated at 37°C for 10 min, and then, the optical density (OD) of each well was measured at 490 nm on a microplate reader for 7 consecutive days. Finally, cell growth curves were mapped according to the mean OD and time.
Determination of the MNNG concentration
Determination of the IC50 of MNNG
The 50% inhibitory concentration (IC50) of MNNG for Kazakh EECs expressing high levels of DNMT1 was determined by the MTT method. Cells were routine digested and prepared in a cell suspension, which was seeded in a 96-well plate at a density of 1 × 104 cells/well with 100 μL in each well, and the edge was filled with 100 μL of PBS. After cells grew to log phase on day 3, final concentrations of 0 mol/L, 0.500 × 10−5 mol/L, 1.000 × 10−5 mol/L, 2.000 × 10−5 mol/L, 4.000 × 10−5 mol/L, 8.000 × 10−5 mol/L, 16.000 × 10−5 mol/L, 32.000 × 10−5 mol/L, and 64.000 × 10−5 mol/L MNNG in complete medium were added. After a 24-h transfection, 20-μL MTT was added, vibrated gently, and incubated at 37°C for 10 min, and the OD of each well measured at 490 nm. The inhibitory rate of MNNG for cells was calculated using the following equation
Determination of the MNNG concentration for cell transformation
The MNNG concentration for cell transformation was determined by a colony formation assay. Cells were routinely digested and prepared into a cell suspension. About 200 Kazakh EECs expressing high levels of DNMT1 were seeded into a 6-well plate and cultured in a 37°C and 5% CO2 incubator for 24 h, followed by addition of MNNG medium at a final concentration of 0 mol/L, 0.75 × 10−5 mol/L, 1.5 × 10−5 mol/L, and 3 × 10−5 mol/L for transfection. After removing the MNNG containing medium, cells were washed three times and cultured for 10 days after the addition of fresh complete medium. The number of colonies containing more than 50 cells was counted under a microscope. The absolute colony formation efficiency (A-CFE) and relative colony formation efficiency (R-CFE) were calculated using the following equations
Malignant cell transformation
Kazakh EECs expressing high levels of DNMT1 were seeded into a 25-cm2 tissue culture flask. After cells grew to 70–80% confluency, MNNG was added at a final concentration (1.50 × 10−5 mol/L) of transformation for a 1-h transfection. After removing the MNNG-containing medium, cells were washed three times with PBS and digested with 0.25% pancreatin 1:2 passage, during which cells were cultured routinely. Passaged cells were treated once using the same method as described earlier. Transfection was performed 14 times altogether. Cell growth was observed daily under an inverted fluorescence microscope.
Soft agar colony formation assay
An agar solution (1.2%) was prepared by dissolving 1.2 g of LMP agar powder in 10 mL of ultrapure water and sterilized by high pressure for later use. At the same time, a 0.7% agar solution was prepared. Cells with different transfection frequencies were routinely digested and counted for later use. In addition, 5 mL of a 1.2% agar solution was mixed thoroughly with 5 mL of 2 × EpicM-2 medium. The mixture was seeded into a 6-well plate at 1.4 mL/well and kept at 37°C for about 1 h until solidification for use. Additionally, 4 mL of a 0.7% agar solution was thoroughly mixed with 4 mL of 2 × EpicM-2, and then with 0.8 mL of the cell suspension, from which a 1.1-mL agar suspension was spread over the bottom of the gel, kept at 37°C until solidified, incubated in a 37°C 5% CO2 incubator for 2 weeks, and finally observed under an inverted fluorescence microscope for cell colony formation.
Tumor formation in nude mice
Twenty-four mice were randomized equally into three groups: control group, Kazakh EECs expressing high levels of DNMT1 group (Kazakh EECs-DNMT1 group), and Kazakh EECs highly expressing transformed cells group (Kazakh EECs-zhz group). Log-phase cells were suspended, and the number was adjusted to 1 × 106 cells/mL using a cell counting plate. The prepared cell suspension was aseptically injected subcutaneously at a dose of 0.2 μL/body weight on the right side of the back of each mouse. Each mouse was weighed before injection, at the time of tumor appearance, and before sacrifice to observe the time of tumor formation and change in body weight after tumor appearance in the three groups. The animals were killed at 4 weeks after injection for pathological evaluation as follows. The tumor was dissected using sterile surgical scissors, photographed, fixed with paraformaldehyde overnight, wrapped with sterile filter paper the following day, placed in 95% ethanol, paraffin-embedded, and hematoxylin-eosin (HE)-stained to observe the structure of the tumor.
Detection of DNMT1 expression level
Detection of the mRNA expression level of DNMT1
Total RNA was extracted from cells, using the Trizol extraction kit (Invitrogen, Carlsbad, California, USA), and the purity and concentration of RNA were determined. According to the SYBR Premix Ex Taq II kit (Beijing Aide Lai, China), specifications for real-time fluorescent quantitative polymerase chain reaction (PCR) experiments were followed. The primers used for reverse transcription PCR (RT-PCR) were designed and synthesized by Premier 5 (Shanghai biotech company, Shanghai, China) (DNMT1-RT-F: AGGAGGCAGATGACGATGAG; DNMT1-RT-R: CGACCCAAGAGATGCGATTC), using the housekeeping gene (β-actin-RT-F: AGCGAGCATCCCCCAAAGTT; β-actin-RT-R: GGGCACGAAGGCTCATCATT) as a reference. The expression level of DNMT1 in different types of cells and tumor tissues was analyzed by the
Protein expression level of DNMT1 detected by Western blotting
Quantitative analysis of the extracted protein was performed by bicinchoninic acid protein assay. Next, 5× sodium dodecyl sulfate (SDS) sample buffer was added during the protein extraction process, mixed evenly and then samples were placed into a boiling water bath for 5 min to denature proteins, and Polyvinylidene Fluoride (PVDF) electrophoresis performed. The transfer membrane was then washed with Tris-base saline-Tween 20 three times and incubated for 2 h with 5% skim milk powder at 37°C.
Statistical analysis
Data were analyzed using the SPSS 17.0 statistical software. Measurement data are expressed as mean (
Results
Observation of normal cell morphology
Inverted fluorescence microscopy showed that normal Kazakh EECs were polygonal with no significant difference in size, and the nuclei were clear, bright, and organized with good refractivity. After 2–3 days of incubation, cells grew to 75–85% confluence in a flask in close arrangement with the background clearly visible; the rate of metabolite production was relatively slow (Figure 1(a)). Kazakh EECs expressing high levels of DNMT1 were significantly different in size; the cytomembrane had more ruffles, the amount of cytoplasm was small with small granules, and the nucleus was relatively large. After 1–2 days of culturing, cells grew to 75–85% confluence, the growth rate was accelerated, and cell metabolite production was vigorous (Figure 1(b)).

Morphological observation of cells. (a) Kazakh esophageal epithelial cells and (b) esophageal epithelial cells expressing high levels of DNMT1. DNMT1: DNA methyltransferase 1.
Determination of MNNG IC50
The OD values obtained from the different concentration groups are determined by the microplate reader. Using the modified Karber method, the IC50 of MNNG for Kazakh EECs highly expressing DNMT1 was calculated to be 23.897 × 10–5 mol/L.
Determination of the MNNG concentration for cell transformation
The number of clones from a single cell to a colony of more than 50 cells was calculated by a colony formation assay. The MNNG concentration that caused transformation of Kazakh EECs expressing high levels of DNMT1 was determined by calculating the A-CFE and R-CFE. Generally, an MNNG dose within the range of 70–80% R-CFE was chosen as the concentration for cell transformation (1.50 × 10–5 mol/L).
Cell growth curve mapping
Normal Kazakh EECs, Kazakh EECs expressing high levels of DNMT1, transformed Kazakh EECs expressing high levels of DNMT1, and ECa109 cells showed identical growth tendencies: Day 1–2 as the latent phase, day 3–5 as the proliferation phase, and day 6–7 as the degeneration phase (Figure 2). Of the four cell types, ECa109 cells grew the fastest, and transformed cells originating from Kazakh EECs expressing high levels of DNMT1 had a similar growth rate as ECa109 cells. In contrast, normal Kazakh EECs grew the slowest, and Kazakh EECs expressing high levels of DNMT1 showed moderate growth.

Growth curves of different cells. OD: optical density.
Soft agar colony formation assay
A soft agar colony formation assay was performed on normal Kazakh EECs transfected with MNNG 5, 11, and 14 times and was also performed on cells expressing high levels of DNMT1. After 2–3 weeks, no soft agar colony formation was observed in normal Kazakh EECs transfected with MNNG 5, 11, and 14 times, indicating that the result was negative. In contrast, cell colonies were observed in passage 27 of Kazakh EECs expressing high levels of DNMT1 after 14 transfections of MNNG, indicating that these cells had acquired malignant features. Nuclear condensation and death were observed in non-transfected cells. Microscopic observation after further expansion cultures of the “metastatic” cells showed multilayer growth of these cells with no contact suppression (Figure 3).

Soft agar colony formation experiment. (a) First day, (b) 14th day, (c) “transformed foci” cells, and (d) cells without transfection.
Tumor formation assay in nude mice
Weight change in nude mice
The nude mice grew well during the entire course of the experiment without showing adverse or inflammatory reactions. Weight change was observed before injection, in the mid-phase and late-phase of the experiment. There was no significant difference in body weights between the three groups before injection (F = 1.105, p = 0.350), whereas significant differences were observed in the mid-phase and late-phase of the experiment between the three groups (F = 6.536, p = 0.006). Compared with the control group and the group with high DNMT1 expression in Kazakh EECs, there were significant differences in the weight of the group with transformed cells from Kazakh EECs expressing high levels of DNMT1 (p = 0.011, p = 0.003). There was significant difference between the latter two groups (p = 0.001, p = 0.002). No tumor growth was observed in the normal control group, whereas topical tumors were observed in the other two groups, where body weight of the mice decreased initially and then increased. In addition, body weight in the group of transformed cells from Kazakh EECs expressing high levels of DNMT1 was always lower than that in the Kazakh EECs-zhz group (Table 1).
Comparison of body weight changes in three groups of nude mice with time (

DNMT1: DNA methyltransferase 1; EE: esophageal epithelial.
an = 24.
bp < 0.05: compared with the control group.
cp < 0.01: compared with the DNMT1 high-expression group.
dp < 0.01: compared with the control group.
Tumor formation and histopathological evaluation of nude mice
The animals were sacrificed within 4 weeks. Tumors were clearly observed in the EECs-DNMT1 group and the EECs-zhz group (Figure 4(a)).

Tumor formation and histopathological evaluation of nude mice. (a) Nude mice were compared between the three groups. Comparison of DNMT1 high-expression group and epithelial transformed cells-zhz group of nude mice, (b) DNMT1 high-expression cell group, and (c) esophageal epithelial cells transformed-zhz (the tumors of two nude mice were ruptured) in nude mice group. DNMT1: DNA methyltransferase 1; EE: esophageal epithelial.
Topical small vesicles were observed in the eight mice of the EECs-zhz group 8 days after cell seeding, and tumors were palpated on day 11. Within 3 weeks, the tumors began growing larger and became irregular and uneven. No significant change was observed in the eight mice belonging to the EECs-DNMT1 group after 8 days; however, small vesicles were observed on day 11, and topical subcutaneous nodules were noticed within 3 weeks. The tumors in both groups were nodular, with little adhesion to the skin, and showed clear tissue boundaries; they looked lobular or papillary, with abundant blood supply and smooth surfaces. The tumors in the EECs-DNMT1 group were generally larger than those in the EECs-zhz group (Figure 4(b) and (c)). At 4 weeks, the tumors were HE-stained and sectioned for pathological observation. It was found that cells in the EECs-DNMT1 group were structurally abnormal but without squamous cancer. In contrast, poorly differentiated squamous cancer cells were observed in the EECs-zhz group, and these cells were either nestle- or mass-shaped with different sizes and large nuclei, in which pathological mitosis was observed (Figure 5).

Histopathological examination of tumors in nude mice. (a) DNMT1 high-expression cell group (×20) and (b) esophageal epithelial cells-zhz group (×20). DNMT1: DNA methyltransferase 1.
DNMT1 gene expression level in different types of cells and tumor tissues
There were statistically significant differences in DNMT1 mRNA and protein expression levels in different types of cells and tumor tissues (F = 140.644, p < 0.001; F = 105.545, p < 0.001). Compared with that in the Kazakh EECs, the DNMT1 gene expression levels in Kazakh EECs with high levels of DNMT1 expression, EE transformation-zhz, and Ec109 cells showed statistically significant differences (p < 0.001). There was no statistically significant difference in DNMT1 mRNA and protein expression levels between EE transformation-zhz and tumor tissues (p > 0.05; Figure 6).

DNMT1 protein expression in different types of cells and tumor tissues. Note. 1: Kazakh esophageal epithelial cells; 2: Kazakh esophageal epithelial cells with high levels of DNMT1 expression; 3: EE transformation-zhz; 4: tumor tissues; and 5: Ec109. DNMT1: DNA methyltransferase 1; EE: esophageal epithelial.
Discussion
Research in recent years has demonstrated that both genetic and epigenetic mechanisms are involved in tumor formation. The former is mainly concerned with the relationship between change in DNA nucleotide sequences and tumor formation, whereas the latter mainly focuses on gene expression changes from the perspective of nongenetic sequence changes. 3 Among various epigenetic mechanisms, DNA methylation is the best studied and most important epigenetic modification. 7 DNA methylation involves covalent chemical modification, in which a hydrogen at the 5′ position on the cytosine ring of a CpG dinucleotide is replaced by an active methyl and transforms to 5-methyl cytosine, using S-adenosylmethionone as the methyl donor and under the catalysis of DNMTs. DNMTs are important enzyme tools, mainly including DNMT1, DNMT2, DNMT3a, and DNMT3b. Studies have shown that the expression of exogenous DNMT1 in murine and human fibroblasts can promote cell transformation through a series of carcinogenetic actions such as decreased contact suppression, increased methylation, increased oncogenesis, and abnormal spindles to increase the risk of carcinogenesis. 8 Some other studies have also found that DNMT1 shows abnormally high expression in multiple malignant tumors of the digestive system, such as in gastric cancer, 9 esophageal cancer, 10 and colon cancer, 11 due to activation of tumor cells. Thus, given the current understanding, we used the transcription activator-like effectors technique to establish Kazakh EECs expressing high levels of DNMT1, knowing that the growth rate of these cells is similar to that of ECa109 cells and that they would undergo changes in size and morphology. The establishment of this cell model provides a useful experimental tool for exploring the pathogenesis of EC.
Research on the pathogenesis of EC has revealed that nitrosamines are the most important contributing factor in EC. 12 However, the role of high levels of DNMT1 expression in MNNG carcinogenesis remains unclear. In the present study, we used Kazakh EECs expressing high levels DNMT1 as our research target and transfected passaged cells with MNNG. It was found that large numbers of cells died in early stages. Moreover, when the transfection frequency and number of passages increased, cell morphology became irregular, cell density increased, cell metabolite levels increased, contact suppression disappeared, and both cell tolerance and the growth rate gradually increased. A soft agar colony formation assay of Kazakh EECs expressing high levels of DNMT1 after 53 days of culturing with 14 transfections and 27 passages showed that cell colonies appeared on the soft agar after 2–3 weeks, and the number of such colonies increased gradually, indicating that these Kazakh EECs expressing high levels of DNMT1 exhibited cancer cell growth characteristics, which is different from results obtained by Sun et al., 13 who reported that MNNG intervention of normal untransformed cells did not have the ability to anchor independent growth after more than 50 consecutive passages of the culture in vitro.
The present study demonstrated that the body weights of the nude mice decreased at the beginning of tumor formation and increased to some extent at the time of killing. Using a human head and neck squamous cell carcinoma model formed by subcutaneous seeding of cells in nude mice, Zhang et al. 14 found that the body weight of the nude mice receiving such cells was significantly lower than that of normal mice. However, in their tumor-bearing model of human cervical carcinoma SiHa cells in nude mice, Hou et al. 15 found that the general condition of all tumor-bearing mice receiving SiHa cells was typical, with normal activity and alert reactions without significant changes in food and water intake or body mass index. A possible reason for these discrepancies in weight change may be due to the different tumor types involved, given that different tumor types have different mechanisms underlying their effects on nutritional absorption and metabolism. In the present study, the tumor formation rate in both the Kazakh EECs-zhz group and the EECs-DNMT1 group was 100%. In addition, both the degree of malignancy and the tumor growth rate in the former group were higher than those in the latter group. In their experiments with the primary gastric cancer cell lines HSC-44PE and HSC-58 as well as the passaged cell lines 44As3, 58Asl, and 58As9, Yanagihara et al. 16 found that the tumor growth rate in the passaged cells was significantly higher than that in the primary cells. In their in vitro and in vivo experiments with a highly invasive Taiwan EEC carcinoma subline that they screened out, Chen et al. 17 confirmed that the proliferative abilities and invasiveness of the passaged cells were higher than those of the primary cells. In addition, it was shown by RT-PCR and Western blotting that the mRNA and protein expression level of DNMT1 in Kazakh EECs, Kazakh EECs with high levels of DNMT1 expression, EE transformation-zhz, tumor tissues, and Ec109 cells showed statistically significant differences (F = 140.644, p < 0.001; F = 105.545, p < 0.001). Compared to those in the EE transformation-zhz group, the mRNA and protein expression levels of DNMT1 in tumor tissues did not show statistically significant differences (p > 0.05). However, the expression level was close to that of esophageal cancer cells (Ec109). On the basis of these findings, we conclude that the tumor formation model developed in the present study was successfully established, and the results suggest that the growth rate of Kazakh EECS expressing high levels of DNMT1 is significantly lower than that of EC cells. However, the high expression of DNMT1 and MNNG-mediated induction may promote tumor occurrence.
Conclusion
A model of malignant transformation of Kazakh EE cells expressing high levels of DNMT1 was successfully constructed, showing that DNMT1 could promote MNNG to induce malignant transformation of EE cells. This study will help understand EC better in order to develop appropriate treatment strategies.
Footnotes
Authors’ contribution
Yan Chen conceived and designed the study and provided administrative support; all authors helped in provision of study materials or patients, collection and assembly of data, data analysis and interpretation, manuscript writing, and final approval of the manuscript.
Declaration of Conflicting Interests
The author(s) declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a National Natural Science Foundation of China (NSFC) Grant (No. 81460502).