Abstract
Vanillin, a widely used flavoring agent, has antimutagenic and antioxidant properties. The current study was performed to evaluate its beneficial role against hepatotoxicity induced by maneb, a dithiocarbamate fungicide. Mice were divided into four groups of six each: group 1, serving as negative controls which received by intraperitoneal way only distilled water, a solvent of maneb; group 2, received daily, by intraperitoneal way, maneb (30 mg kg−1 body weight (BW)); group 3, received maneb at the same dose of group 2 and 50 mg kg−1 BW of vanillin by intraperitoneal way; and group 4, serving as positive controls, received daily only vanillin. After 10 days of treatment, mice of all groups were killed. Our results showed that vanillin significantly reduced the elevated hepatic levels of malondialdehyde, hydrogen peroxide, and advanced oxidation protein product and attenuated DNA fragmentation induced by maneb. In addition, vanillin modulated the alterations of antioxidant status: enzymatic (superoxide dismutase, catalase, and glutathione peroxidase) and nonenzymatic (reduced glutathione, nonprotein thiol, and vitamin C) antioxidants in the liver of maneb-treated mice. This natural compound was also able to ameliorate plasma biochemical parameters (aspartate aminotransferase, alanine aminotransferase, gamma glutamyl transpeptidase, alkaline phosphatase, total bilirubin, and total protein). The protective effect of vanillin was further evident through the histopathological changes produced by maneb in the liver tissue. Thus, we concluded that vanillin might be beneficial against maneb-induced hepatic damage in mice.
Introduction
Medicinal plants play an important role in pharmacology and medicine for many years. It is estimated that up to 80% of the world population use botanical preparations for their health care. 1 Vanillin, an o-methylcatechol (4-hydroxy-3-methoxybenzaldehyde) polyphenol, is present in the bean and pod of tropical vanilla orchid (Figure 1). It is widely used as a flavoring agent in various food and beverage products as well as in the perfumery and pharmaceutical industries. 2 Its annual worldwide human consumption is estimated to be more than 2000 tons. 3 In addition to its food and industrial applications, vanillin has been reported to possess anti-inflammatory, 4 antimutagenic, 2 antioxidant, and antitumor properties. 5,6 Besides, it has been demonstrated to control mitochondrial dysfunction, oxidative stress, and neurodegenerative disorders such as Parkinson’s and Huntington’s diseases. 7,6 Moreover, vanillin has been shown to reduce the expression of the pro-inflammatory cytokines interleukin-1ß (IL-1ß), IL-6, interferon-γ, and tumor necrosis factor in colonic tissues of mice with inflammatory bowel disease. 8 Recently, vanillin has been found to display a significant brain protective property through its antioxidant and anti-inflammatory activities. 9 Vanillin biological properties are mainly ascribed to its phenolic compounds, ether and aldehyde moieties. 6 It is used against oxidative damage induced by several xenobiotics like pesticides. 6,10 The latters have human serious chronic health effects, including diabetes, respiratory diseases, neurological effects, fetal diseases, genetic disorders, and cancer. 11 –14 Among pesticides, maneb is a manganese-containing ethylene-bis-dithiocarbamate fungicide, widely used to control numerous fungal diseases affecting vegetables, fruits, and field crops. 15 A variety of ailments has been reported in workers exposed to dithiocarbamates during foliar applications and seed treatments 16 and also through food chain. Despite its apparent low toxicity, numerous in vitro and in vivo studies have confirmed that maneb exposure causes lipid peroxidation, reactive oxygen species (ROS) generation, and alterations in antioxidant defense systems, as a result of complex III inhibition in mitochondrial electron transport chain. 17 –22 According to Ben Amara et al., 23 maneb, used at graded doses (1/8, 1/6, 1/4, and 1/2 of LD50), is able to generate free radicals in the liver of adult mice, leading to an impairment of liver defense system. Moreover, maneb increases the prevalence of Parkinson’s disease in humans 18 and in laboratory rodents. 24 Besides, it disturbs endocrine functions like thyroid and gonads, as reported previously by Mallem et al. 25 in rabbits.
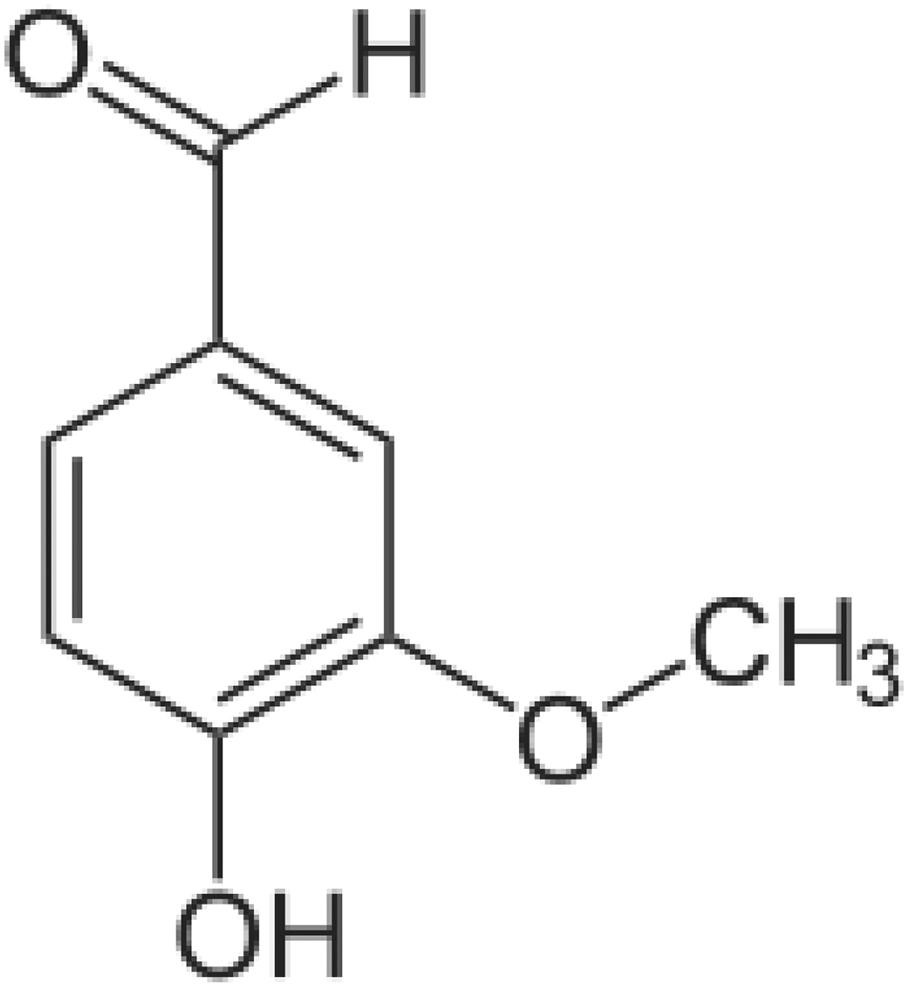
Chemical structure of vanillin.
To our knowledge, the effective ability of vanillin to attenuate maneb toxicity in the liver has not been previously investigated. Therefore, the present study focused for the first time on the hepatoprotective role of vanillin by analyzing the oxidative and anti-oxidative markers against maneb induced liver toxicity in adult mice.
Materials and methods
Chemicals
In the present experiment, we have used Ridanebe® fungicide (Dow AgroScience, Nice, France), a commercial formulation of maneb, which contained 80% of the active substance maneb and 20% of inert ingredients. Some compounds, such as vanillin, reduced glutathione (GSH), nicotinamide adenine dinucleotide phosphate reduced form, 5-5-dithio-bis-2-nitrobenzoic acid, and thiobarbituric acid, were purchased from Sigma-Aldrich (St Louis, Missouri, USA). Other compounds were purchased from other suppliers.
Animals
Adult male mice of the Swiss strain, weighing approximately 30 g at their reception, were purchased from the Central Pharmacy (SIPHAT, Tunisia). They were housed in plastic cages lined with husk and maintained under standard laboratory conditions (temperature 22 ± 2°C, relative humidity 40%, and 12-h light and 12-h dark cycle). A commercial standard pellet diet (SNA, Sfax, Tunisia) and drinking water were provided to the mice ad libitum. Animal care and all the experimental procedures were conducted with strict adherence to the Ethical Committee of Sciences Faculty of Sfax, with ethics approval number 1204, and were in accordance with the International Guidelines for Animal Care. 26
Experimental design
Mice were randomly divided into four groups of six each and treated as follows: Group 1 (control)—serving as negative controls, mice received daily by intraperitoneal way 0.5 ml of distilled water, a solvent of maneb. Group 2 (MB)—mice received daily, by intraperitoneal way, maneb at 30 mg kg−1 body weight (BW). Group 3 (MB+Van)—mice received maneb at the same dose of group 2 and 50 mg kg−1 BW of vanillin by intraperitoneal way. Group 4 (Van)—serving as positive controls, mice received daily by intraperitoneal way only vanillin at the same dose of group 3.
The maneb dose 30 mg kg−1 BW, used in the present study, was chosen based on the previous study of Yadav et al. 27 Concerning vanillin, we have used in our experiment the dose 50 mg kg−1 BW, according to Maurya et al. 28
At the end of the experimental period (10 days), all mice were euthanized by cervical decapitation to avoid stress. The trunk blood was collected into heparinized tubes and centrifuged at 2200 × g for 10 min. Plasma samples were drawn and stored at −80°C until analysis. Livers were dissected out, cleaned, and weighed. Some portions of the livers were rinsed, homogenized in phosphate buffer (0.1 M; pH = 7.4), and centrifuged. The resulting supernatants were maintained at −80°C for biochemical assays. Other portions were either fixed in 10% buffered formalin solution and embedded in paraffin for histological studies or taken for DNA assay.
Biochemical analysis
Protein quantification
Total protein content in liver homogenates was determined according to the method described by Lowry et al., 29 using bovine serum albumin as standard.
Liver malondialdehyde measurement
Malondialdehyde (MDA) content in liver, index of lipid peroxidation, was spectrophotometrically determined at 532 nm following the method described by Draper and Hadley, 30 and the values were expressed as nmoles MDA per milligram of protein.
Hydrogen peroxide measurement
Hydrogen peroxide (H2O2) measurement in liver was carried out by the ferrous ion oxidation xylenol orange method. 31 Values were expressed as micromoles per milligram of protein.
Liver advanced oxidation protein product levels
Liver advanced oxidation protein product (AOPP) levels were determined according to the method of Kayali et al. 32 For each sample, the AOPP concentration was calculated using the extinction coefficient 261 cm−1 mM−1, and the results were expressed as nanomoles per milligram of protein.
Liver nonenzymatic antioxidant levels
GSH levels in liver tissue were estimated at 412 nm by Ellman’s method 33 modified by Jollow et al., 34 and the values were expressed as nanomoles per milligram of protein.
Liver nonprotein thiol (NPSH) levels were determined using the method reported by Ellman. 33 The absorbance of the colorimetric reaction was measured at 412 nm. The total NPSH content was expressed as nanomoles per milligram of protein.
Liver ascorbic acid (vitamin C) content was determined using the dinitrophenyl hydrazine method described by Jacques-Silva et al. 35 The data were expressed as nanomoles per milligram of protein.
Liver enzymatic antioxidant activities
Catalase (CAT) activity in liver tissue was measured at 240 nm by Aebi’s method, 36 and the values were expressed as µmoles H2O2 consumed per minute per milligram of protein.
Superoxide dismutase (SOD) activity was determined at 560 nm by Beauchamp and Fridovich’s method, 37 and the values were expressed as Units per milligram of protein.
Glutathione peroxidase (GPx) activity was estimated according to the method described by Flohe and Günzler. 38 Enzyme activity was expressed as nmoles GSH oxidized per minute per milligram of protein.
Determination of biochemical parameters in plasma
The activities of plasma transaminases (aspartate aminotransferase (AST) and alanine aminotransferase (ALT)), gamma glutamyl transpeptidase (GGT), alkaline phosphatase (ALP), total protein and bilirubin levels, and lactate dehydrogenase (LDH) in plasma and in the liver were assayed spectrophotometrically according to standard procedures using commercially available diagnostic kits (Biomaghreb, Ariana, Tunisia, ref. 20042, 20046, 20021, 20015, 20161, 20103, 20012).
Qualitative DNA fragmentation assay by agarose gel electrophoresis
Genomic DNA was extracted from the hepatic tissue using a commercial kit and then electrophoresed on a 1% agarose gel stained with ethidium bromide (Pure Link Genomic DNA Invitrogen ref. K 182001, ThermoFischer, Carlsbad, CA, USA). The gel was then observed under ultraviolet light and photographed.
Histological studies
Some liver portions were fixed in 10% buffered formalin solution for 48 h. The specimens were washed and dehydrated through a series of graded ethanol solutions. Then, they were embedded in paraffin to form blocks, which were sectioned at a thickness of 5 µm and stained with hematoxylin and eosin. Liver sections were examined under light microscope. 39 Histopathological changes severity (granuloma inflammatory disorders, granuloma vascularization and inflammation) was scored using the blind study methodology, based on the following scale: 0 (no injury), 1 (injury to 25% of the field), 2 (injury to 50% of the field), 3 (injury to 75% of the field), and 4 (diffuse injury).
Statistical analysis
Data of the present work were analyzed using the statistical package program Stat view 5 Software for Windows (SAS Institute, Berkley, California, USA). Statistical analysis was performed using one-way analysis of variance followed by Fisher’s protected least significant difference test as a post hoc test for comparison between groups. When comparison between two groups was required, the student unpaired t-test was also used. All values were expressed as means ± SD. The differences were considered statistically significant at p < 0.05.
Results
Evaluation of BW, absolute and relative liver weights, and water and food intakes
Death was not observed during the experimental period. As shown in Table 1, food consumption by all treated mice was similar to that of controls. BWs were unaffected by maneb treatment; however, compared to that of the controls, absolute liver weight of MB and (MB+Van)-treated mice was significantly (p < 0.001) increased by 10 and 6%, respectively. In addition to that, compared to the controls, MB-treated mice showed increased relative liver weight (+8%). Furthermore, a significant (p < 0.001) rise in the water intake of maneb-treated mice was observed, compared to the controls. When vanillin was administered to maneb-treated mice, a recovery occurred in the water intake.
Final body weight, absolute and relative liver weights, and daily food and water consumption in control and treated mice during 10 days with maneb, maneb associated with vanillin, and vanillin alone.a

BW: body weight.
aValues are means ± SD for six mice in each group.
bp < 0.001: MB, (MB+Van), Van versus controls.
cp < 0.05: (Van+MB) versus MB.
dp < 0.05: MB, (MB+Van), Van versus controls.
ep < 0.001: (Van+MB) versus MB.
Liver biochemical markers evaluation
Oxidative stress biomarkers (MDA, H2O2, and AOPP)
The levels of lipid peroxidation, H2O2, and protein oxidation in the liver of control and experimental mice were illustrated in Table 2. Levels of MDA, H2O2, and AOPP significantly (p < 0.001) increased in maneb-intoxicated mice, when compared to control mice. Co-treatment with vanillin of maneb-intoxicated mice significantly decreased (p < 0.001) the levels of the parameters cited earlier. There were no differences between positive and negative control mice.
MDA, H2O2, AOPP, GSH, NPSH, and vitamin C levels in the liver of adult mice controls and mice treated during 10 days with maneb, maneb associated with vanillin, and vanillin alone.a

MDA: malondialdehyde; H2O2: hydrogen peroxide; AOPP: advanced oxidation protein product; GSH: reduced glutathione; NPSH: nonprotein thiol.
aValues are means ± SD for six mice in each group.
bp < 0.001: MB, (Van+MB), Van versus controls.
cp < 0.05: MB, (Van+MB), Van versus controls.
dp < 0.001: (Van+MB) versus MB.
ep < 0.05: (Van+MB) versus MB.
fp < 0.01: (Van+MB) versus MB.
Nonenzymatic antioxidant stress biomarkers (GSH, NPSH, and vitamin C)
Nonenzymatic antioxidant levels of control and experimental mice were shown in Table 2. In maneb-intoxicated mice, the levels of GSH, NPSH, and vitamin C were significantly (p < 0.001) decreased as compared to those of the control mice. Co-treatment with vanillin of maneb-treated mice significantly (p < 0.001) increased the levels of the above mentioned parameters in the liver tissue as compared to mice only treated with maneb.
Enzymatic antioxidant stress biomarkers (CAT, GPx, and SOD)
In the liver of MB-treated mice, CAT, SOD, and GPx activities decreased significantly (p < 0.001), when compared to those of controls. Vanillin administrated to maneb-treated mice restored partially CAT and SOD activities and totally GPx (Figure 2).
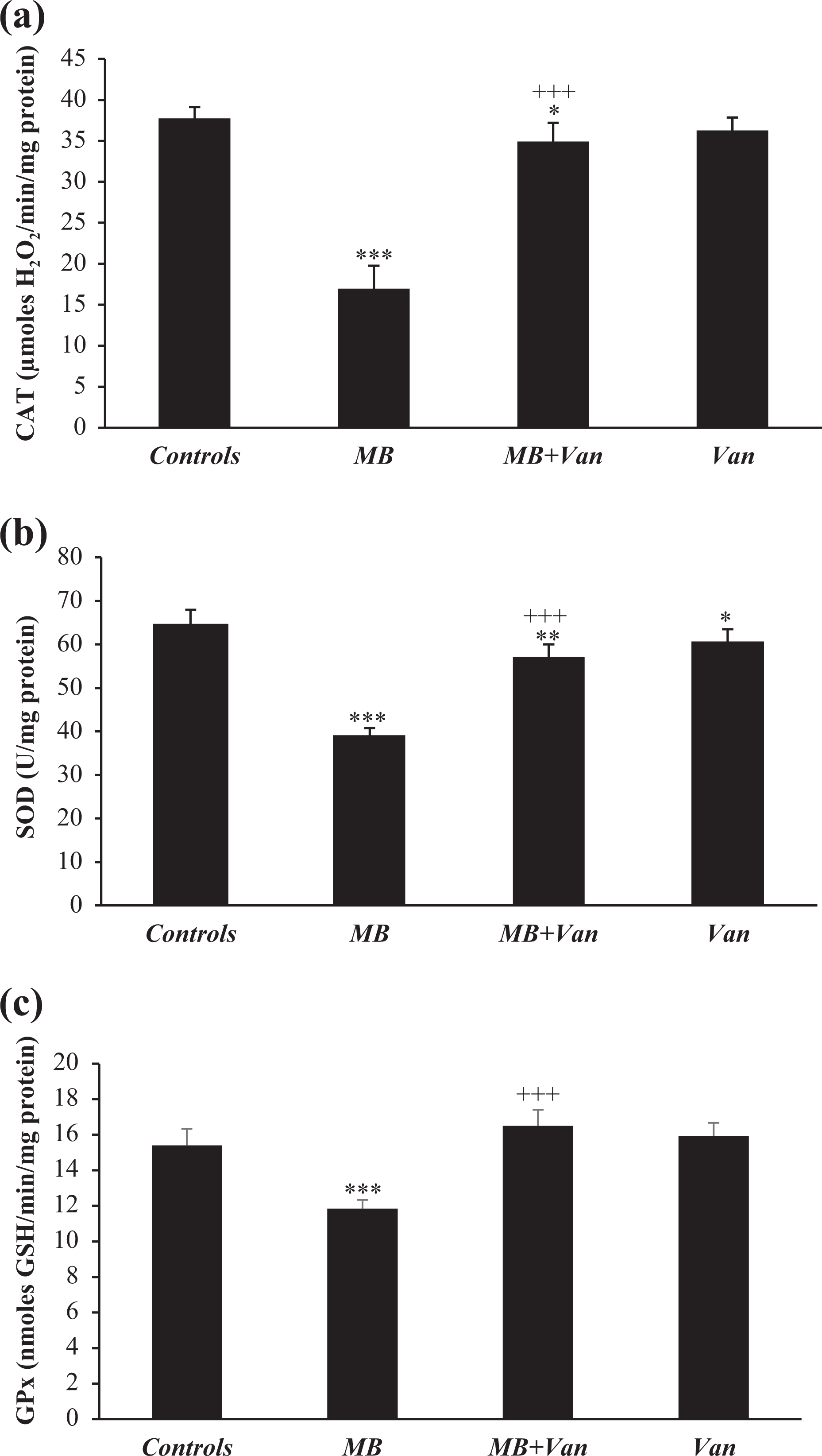
Liver enzymatic antioxidant activities (SOD, CAT, and GPx) in control and treated mice during 10 days with maneb, maneb associated with vanillin, and vanillin alone. Values are means ± SD for six mice in each group. MB, (MB+Van), and Van versus controls: *p < 0.05; **p < 0.01; ***p < 0.001. (MB+Van) versus MB: +++p < 0.001. SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase.
Plasma biochemical parameters
The AST, ALT, and ALP activities in the plasma of the maneb-treated mice increased, when compared to the controls, indicating hepatotoxicity induced by this fungicide. A significant increase in the total bilirubin levels and a decrease in total protein levels were also observed. Interestingly, the activity of GGT, a biomarker of liver dysfunction, was threefold increased in maneb-treated mice, as compared to controls (Table 3). LDH activity increased in plasma and decreased in the liver of maneb-treated mice (Figure 3). When vanillin was administered to the maneb-treated mice, AST, ALT, and GGT activities significantly decreased, as compared to maneb group. While the activity of ALP was not changed. The plasma total protein level showed improvement, without reaching the control values. Moreover, LDH activity significantly decreased in the plasma and increased in the liver of mice treated with maneb and vanillin, as compared to maneb group, without reaching control values.
Transaminase (AST, ALT), GGT, and ALP activities, total protein, and total bilirubin levels in plasma of adult mice controls and treated during 10 days with maneb, maneb associated with vanillin, and vanillin alone.a
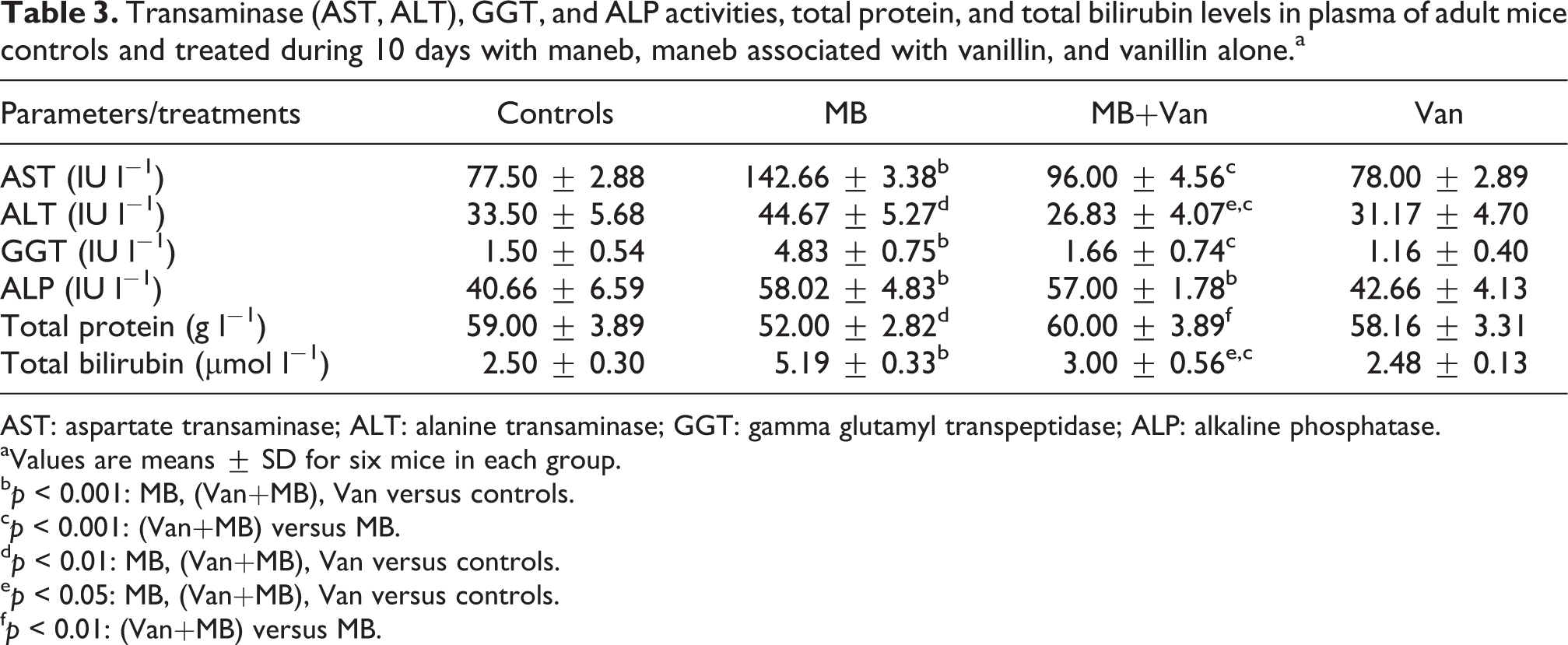
AST: aspartate transaminase; ALT: alanine transaminase; GGT: gamma glutamyl transpeptidase; ALP: alkaline phosphatase.
aValues are means ± SD for six mice in each group.
bp < 0.001: MB, (Van+MB), Van versus controls.
cp < 0.001: (Van+MB) versus MB.
dp < 0.01: MB, (Van+MB), Van versus controls.
ep < 0.05: MB, (Van+MB), Van versus controls.
fp < 0.01: (Van+MB) versus MB.

LDH activity in plasma (a) and liver (b) of control and treated mice during 10 days with maneb, maneb associated with vanillin, and vanillin alone. Values are means ± SD for six mice in each group. MB, (MB+Van), and Van versus controls: ***p < 0.001. (MB+Van) versus MB: +++p < 0.001. LDH: lactate dehydrogenase.
DNA fragmentation
As shown in Figure 4, a smear without ladder formation was observed in the liver of maneb-treated mice, indicating random DNA degradation. While mice treated with vanillin and maneb showed a decreased DNA smearing as compared to mice treated with maneb alone. No DNA damage was observed in vanillin-treated group.

Agarose gel electrophoresis of DNA in the liver of adult mice. M: marker; lane 1: MB+Van; lane 2: MB; and lane 3: Van.
Histopathological findings
Light microscopic examination of liver sections from the control and vanillin groups indicated a normal liver histo-architecture (Figure 5(a) and (d)). Exposure to maneb induced histopathological changes, with significant damage in hepatic cells like sinusoidal spaces as well as a central vein inflammation (Figure 5 (b) and (c)). Vascularized granuloma was also observed (Figure 5(b)). A normal histoarchitecture of the liver was also observed in mice treated with maneb and vanillin (Figure 5(d)).

Liver sections of control (a) and treated mice with maneb (b) and (c), maneb co-treated with vanillin (d), or treated only with vanillin (e). Coloration hematoxylin and eosin. Magnification: (×400). Arrows indicate inflammation of the central veins, granuloma vascularization, granuloma inflammatory disorders, and inflammatory cells among hepatocytes.
The severity of these pathological changes was scored and presented in Table 4. In fact, all the injury scores, including granuloma inflammatory disorders and granuloma vascularization and inflammation scores, were significantly increased in maneb group as compared to the control and vanillin groups. However, mice treated with maneb and vanillin showed lower injury scores when compared with maneb group.
Liver injury scores of adult mice controls and treated during 10 days with maneb, maneb associated with vanillin, and vanillin alone.a

aValues are means ± SD for six mice in each group.
b,c,dMeans, within each row, that do not share the same superscript letters, correspond to being significantly different from each other (p < 0.05).
Discussion
Due to its role in the metabolism and removal of environmental xenobiotics, the liver is at a great risk of injury leading to hepatotoxicity. To our knowledge, our article constitutes the first report exploring the relative cytoprotective efficiency of vanillin, a polyphenolic flavoring agent, against maneb-induced hepatotoxicity in mice.
In our experimental study, results showed no significant differences in BWs between maneb and control group. This fungicide didn’t probably affect the mouse appetite, since there were no differences between quantities of diet ingested by maneb and control mice. Our results were in agreement with the study of Rajeswary et al., 40 who have observed no changes in the BW of rats treated orally with carbendazim fungicide (25 mg kg−1 BW) for a long period (48 days). While a significant loss has been found by Ben Amara et al., 23 in BW of mice treated with graded high doses of maneb (1/8, 1/6, 1/4, and 1/2 of LD50) during 7 days, which may occur as a result of severe acute maneb toxicity. In our study, we have used a maneb dose of 30 mg kg−1 BW, representing (1/50 of LD50) which is the lower than the doses tested by Ben Amara et al. 23 On the other hand, the marked increase in liver weight, recorded in maneb group, could be a result of the hepatic inflammation caused by maneb, as confirmed by our histological findings. Our results were similar to those previously obtained by Razia and Siddiqui, 41 where a remarkable increase in liver weight of mice treated for 4 weeks with mancozeb fungicide (2.4 and 6.4 mg kg−1 BW) has been found.
In our present study, the morphological changes observed in liver of maneb-treated mice could have resulted from oxidative stress installation in this organ. Indeed, different mechanisms have been postulated to explain maneb-induced liver oxidative injury, such as interaction with membrane components and lipid peroxidation. 42 The last process represents one of the most frequent reactions resulting from free radical’s attack on biological structures. Hence, the increased level of lipid peroxidation in mice exposed to maneb, observed in our experiment, could not be only due to changes related to oxidative stress, but also due to the inhibitory effect of this fungicide on the activity of aldehyde dehydrogenases ALDH2 and ALDH5A, two mitochondrial enzymes implicated in the detoxification of lipid peroxidation. 43,21 This hypothesis was confirmed by the increase of liver H2O2 levels, suggesting the dysfunction of the mitochondrial respiratory chain. Our data were in accordance with the previous studies of Chaâbane et al., 44 who have indicated an increase of H2O2 levels in response to penconazole fungicide exposure.
In addition to lipids, proteins are also susceptible to reactive oxygen species attack, giving rise to carbonyl moieties, 45 AOPP, and many other undesired oxidation products. The occurrence of protein oxidative modification in the liver of experimental mice exposed to maneb was confirmed, in the present work, by a significant increase in their hepatic AOPP levels, indicating a putative mechanism of maneb-induced liver toxicity. Coadministration of vanillin, a polyphenolic flavoring agent, to maneb-treated mice restored MDA, H2O2, and AOPP levels, without reaching control values. Similar findings have been found by Ben Saad et al., 46 where vanillin attenuates the increase in lipid peroxidation, AOPP, and protein carbonyl formation in the liver of mice treated with potassium bromate. Also, Kadeche et al. 10 have reported that vanillin exhibits hepatoprotective effect against metribyzin pesticide toxicity in male Wistar rats. The improvement of MDA, H2O2, and AOPP levels after vanillin treatment could be explained by its important role in preventing lipid peroxidation and protecting the integrity of tissues and cells function. Indeed, vanillin presents different functional groups including the phenolic group. The antioxidant activity of phenols is attributed to their ability to scavenge free radicals. 47 Therefore, the vanillin-induced cytoprotection and hepatoprotective effects observed in our study might be ascribed to its direct antioxidant and free radical scavenging activity.
To advance our understanding of the mechanisms underlying maneb toxicity, the nonenzymatic antioxidant status in liver was assessed. GSH is the main thiol antioxidant which preserves and protects proteins against ROS, reactive nitrogen species (RNS), and electrophiles. 48,49 Its decrease, observed in our experiment, could be explained by its consumption to scavenge free radicals generated by maneb treatment. This is in agreement with the previous study of Grosicka-Maciag et al. 21 who have shown, in Chinese hamster V79 cells-exposed, that this fungicide decreases GSH and increases glutathione disulfide (GSSG) levels. Concerning NPSH, one of the essential primary defenses counteracting oxidative stress, its decrease demonstrated for the first time in our study after maneb treatment, is due to its interaction with free radicals. On the other hand, a significant decrease in vitamin C level was recorded in maneb-treated mice. This component is a very efficient scavenger of the H2O2, hypochlorite, hydroxyl, pyroxyl radicals, and singlet oxygen. 50 The antioxidant role of vitamin C is based on its reaction with the aqueous peroxyl radicals, and the formed product is the ascorbyl radical. By trapping peroxyl radicals in the aqueous phase, which they are ready to initiate peroxidation, vitamin C protects biomembranes and lipoproteins. 51,52 According to these authors, the vitamin C decrease could be the main reason lying behind directly increased production of lipid peroxidation products, including lipid hydroperoxides (LOOHs) and MDA. Besides, the improvement of GSH, NPSH, and vitamin C levels after vanillin treatment, in the current study, supported the beneficial role of this flavor molecule. In fact, the antioxidant activity of phenolic substances has been reported for several natural substances structurally similar to vanillin. The hydroxyl group of the aromatic ring contributes to the antioxidant activity through the hemolytic fragmentation of the O–H bond. 53,54 It has been reported that vanillin exhibits scavenging activities against hydroxyl radical 55 and of 2-2-azinobis (3-ethylbenzothiazoline-6-sulfonic acid (ABTS+) radical cation. 28 Its antioxidant property can be ascribed to the presence of hydroxyl group (OH–) linked to aromatic ring.
The effect of maneb treatment on the hepatic enzymatic antioxidant status was also investigated in the present work. Antioxidant enzymes activities such as SOD, CAT, and GPx were found to be decreased in the liver tissue, following maneb exposure. SOD is the primary step of the defense mechanism in the antioxidant system against oxidative stress by catalyzing the dismutation of two superoxide radicals (O2 −) into molecular oxygen (O2) and H2O2. The latter is neutralized, in all vertebrates, by the combined action of CAT and GPx. 56 These enzymes act in coordination; therefore, any change occurring in their levels may push the cell to an oxidative damage state. In the present study, the significant decrease of SOD activity observed in maneb group suggested, as reported by Fouchecourt and Riviere, 57 an increased superoxide radical production and other ROS thereby inducing oxidative damage. Moreover, the decreased CAT activity in maneb-treated mice indicated the presence of superoxide radical. The antioxidant enzyme CAT protected SOD against inactivation by H2O2. Reciprocally, SOD protected CAT against inhibition by superoxide anion that could be formed during maneb treatment. Our results were in agreement with the study of Ben Amara et al., 23 who have observed a significant decrease in the activities of CAT and SOD in the liver of adult mice exposed to graded doses of maneb. The significant recovery of hepatic antioxidant enzyme activities by vanillin suggested its chemopreventive effect against maneb-mediated hepatic injury.
Because the liver is regarded as the major metabolizing site in xenobiotic catabolism, 58 we assessed its integrity through the measurement of biochemical markers such as AST, ALT, and GGT. The significant increase observed in the activities of AST, ALT, ALP, GGT, and total bilirubin content and the significant decrease of total protein in the plasma of maneb-treated mice indicated a loss of hepatocyte membrane integrity. Similarly, LDH, another biomarker of liver toxicity, indicates also according to Gaskill et al. 59 cell membrane damage. Our results showed that LDH activity decreased in the liver tissue and increased in plasma of maneb-treated mice. This is in agreement with the findings of Sakr et al., 60 who have found that this enzyme increases in plasma of mancozeb-treated mice. This elevation could potentially be attributed to the hepatotoxicity, leading to an increase in permeability of liver membrane and a leakage of lysosomal enzymes in methomyl-treated mice. 61 Moreover, changes in aminotransferase activities (AST and ALT) could be expected to occur in association with a liver pathology involving necrosis. ALT, enzyme known to be present in large quantities in the liver, increases in plasma when cellular degeneration or destruction occurs in this organ. 62 In fact, our results showed that AST and ALT increased in plasma and decreased in the liver of maneb-treated mice escaping from the injured hepatic cells. Co-treatment with vanillin was effective in the prevention of oxidative damages induced by maneb, as objectified by the lower liver AST, ALT, and bilirubin concentrations than those of maneb-treated mice, indicating the important role of vanillin in protecting tissues and cells integrity and function.
In addition to the altered biochemical parameters, it is well established that DNA may be affected by free radicals’ accumulation, giving rise to mutations and/or cell death. 63 In the present study, DNA damage was evaluated by electrophoresis of DNA extracted from the liver. Maneb treatment induced DNA fragmentation resulting in DNA smear on agarose gel, a hallmark feature of necrosis, confirming maneb genotoxicity. DNA breakdown could be due, according to Domico et al., 64 either directly to the prooxidant effect of maneb and/or to one of its metabolites or to ROS generated by the fungicide in mesencephalic cells. Maneb principal metabolite, ethylene thiourea, has shown a genotoxic effect on Wistar rats. 65 In our experimental study, treatment with vanillin was effective in reducing DNA smearing effects induced by maneb. Our results were in line with previous studies showing that vanillin, an effective radical’s scavenger, reduces the oxidative DNA damage in the liver of rats exposed to γ-radiation. 28
DNA damage and biochemical changes might account for the occurrence of histopathological alterations in the liver of maneb-treated mice. In fact, maneb caused various categories of inflammations ranging from simple to complicated, depending on the location and intensity. In fact, a few inflammatory cells were observed in hepatocytes and around the central vein. Furthermore, the marked granuloma inflammatory disorders along with granuloma vascularization were also found in the hepatic tissue. Treatment with vanillin resulted in a marked improvement in liver histoarchitecture, highlighting its protective potential.
There is a little information regarding the molecular mechanism of maneb-induced cytotoxicity. According to the literature data, the main mechanisms by which fungicides cause damage to the cell macromolecules leading even to hepatic cell death could be ROS-mediated mitochondrial dysfunction and inflammatory pathways. 66 –68 As reported by Domico et al., 19 maneb is metabolized to the degradation product ethylene thiourea, which may have toxic properties by interfering with the mitochondrial respiratory chain. Mitochondrial toxicity evaluation is a useful approach in the comprehensive understanding of chemical-induced toxicity. In this context, it has been demonstrated, in human hepatoma HepG2 cells, that difenconazole fungicide induced mitochondrial toxicity through inhibition of cellular respiration and the Krebs cycle. 69 On the other hand, the inflammation observed in liver histological sections represents a naturally occurring reaction of the system to the damaged cells. 70 Consistently, in our mouse model, maneb administration induced simple to complicated categories of inflammations. Hepatic inflammatory response is mediated by the pro-inflammatory cytokines, especially tumor necrosis factor-α (TNF-α), which can further modulate the effects of other cytokines, as IL-1, IL-2, IL-4, IL-6, IL-10, IL-12, IL-18, and IL-23. 71 In normal liver, TNF-α is almost detectable. However, in many types of liver injury, TNF-α production represents one of the first events happening. Recently, it has been demonstrated that critical drug-induced toxicity pathways act in synergy with TNF-α. 72 TNF-α promotes the inflammatory response directly or indirectly by stimulating pro-inflammatory mediators such as IL-6 secreted by Kupffer cells. IL-6 is the predominant mediator of the hepatic acute-phase response. It modulates liver fibrosis by degrading extracellular matrix proteins and this is through the inhibition of proteases or the binding to other cytokines. 73,74 In an experimental model of liver injury in rats, Xiao et al. 75 found that Lycium barbarum polysaccharides prevent nonalcoholic steatohepatitis-induced liver injury by the hepatic pro-inflammatory mediator’s reduction and the attenuation of inflammation. Hence, to improve our understanding of the molecular mechanisms ruling maneb-induced hepatotoxicity, it would be interesting to examine, in the future studies, the target pathways activated by ROS in hepatic cells exposed to this fungicide and the protective effects of vanillin.
Conclusion
Based on our results, it could be concluded that vanillin possesses remarkable protection against maneb-induced hepatic injury and could reduce the damaging effect of this fungicide on liver by exerting antioxidant activity and reducing oxidative stress. Further studies are required to evaluate the protective and antioxidant effects of vanillin in human beings.
Footnotes
Acknowledgements
The authors are indebted to Mrs Raoudha Ben Amar Abdennadher and Mr Chedli Tmar for their skillful technical assistance.
Authors’ Contribution
Awatef Elwej and Mariem Chaâbane contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Higher Education and Scientific Research in Tunisia.
