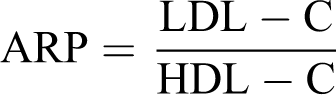Abstract
The current study investigated the role of guggulsterone (GS), a farnesoid X receptor antagonist, in the choline metabolism and its trimethylamine (TMA)/flavin monooxygenases/trimethylamine-N-oxide (TMAO) inhibiting potential in a series of in vitro and in vivo studies as determined by high-performance liquid chromatography (HPLC), mass spectroscopy (MS), and liquid chromatography (LC)-MS techniques. Atherosclerosis (AS) was successfully induced in a group of experimental animals fed with 2% choline diet for 6 weeks. Serum lipid profiles such as total cholesterol, triglycerides, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, and very low-density lipoprotein cholesterol were measured. Pro-inflammatory cytokines levels, markers for a hepatic injury, and oxidative stress markers were assessed. Interestingly, GS reduced the level of TMA/TMAO in both in vitro and in vivo studies as demonstrated by the peaks obtained from HPLC, MS, and LC–MS. Furthermore, GS exhibited cardioprotective and antihyperlipidemic effects as evidenced by the attenuation of levels of several serum lipid profiles and different atherogenic risk predictor indexes. GS also prevented hepatic injury by successfully restoring the levels of hepatic injury biomarkers to normal. Similarly, GS inhibited the production of pro-inflammatory cytokines levels, as well as GS, enhanced antioxidant capacity, and reduced lipid peroxidation. Histopathological study of aortic sections demonstrated that GS maintained the normal architecture in AS-induced rats. On the basis of results obtained from current investigation, we suggest that GS might have a great therapeutic potential for the treatment of AS.
Introduction
Cardiovascular diseases (CVDs) have been the dominant cause of death on the planet, seeing for 17.3 million deaths per year, which is expected to grow to >23.6 million by 2030. 1 Atherosclerosis (AS) is one of the major underlying causes of CVDs. 2 AS is a multifocal, smoldering, immunoinflammatory disease of medium-sized and large arteries fueled by lipids and endothelial cells, leukocytes, whereas intimal smooth muscle cells are the key players in the development of AS. 3 Meta organismal nutrient metabolism pathway linking CVD including AS has been documented earlier. 4 The trimethylamine-N-oxide (TMAO), a choline metabolite, is recently discerned as a new and independent risk factor for aiding AS 5 (Figure 1). Recent clinical studies have demonstrated that TMAO enhances platelet hyperreactivity and thrombosis risk. 6 It also has been recently reported that TMAO levels are elevated in chronic kidney disease patients which directly correlates to the acceleration of AS. 7,8 The gut microbiota (GM) forms a mutualistic relationship with the host and demonstrates an important role in host health. 9 GM has been documented to have a crucial role in the pathogenesis of TMAO-induced AS, 10 and decisive role of GM in the CVD has been reported earlier. 11

Schematic diagram illustrating involvement of TMA/TMAO pathway in AS. TMA: trimethylamine; TMAO: trimethylamine-N-oxide.
Human GM like Desulfovibrio desulfuricans, Anaerococcus hydrogenalis, Clostridium asparagiforme, Clostridium hathewayi, Clostridium sporogenes, Escherichia fergusonii, Proteus penneri, Providencia rettgeri, and Edwardsiella tarda are reported to exhibit trimethylamine (TMA) converting property from dietary choline. 12 Choline, a dietary nutrient, can be metabolized by GM to produce an intermediate compound TMA and then further oxidized by hepatic flavin monooxygenases (FMO3) to form TMAO. 13 The microbial enzyme produced by GM called choline-utilizing enzyme cutC (catalytic polypeptide) belonging to glycyl radical enzyme family and its activating protein cutD (regulatory polypeptide) holds the role of choline TMA lyase activity. These enzymes use choline as the substrate and selectively generate TMA. 14 The released TMA then reaches the liver rapidly via the portal circulation, where a large array of hepatic FMO3 enzymes systematically oxidizes TMA into TMAO. 15 FMO3 expression is induced by a mechanism that involves the farnesoid X receptor (FXR). 16 TMAO was reported to be associated with cardiovascular risks, and TMAO inhibits reverse cholesterol transport and also reduces bile acid (BA) synthesis, both of which are expected to aggravate AS. 17 TMAO promotes the assembling of cholesterol in macrophages, gathering of foam cells in artery walls, and eventually fostering to AS, which are connected with an increased risk of cardiometabolic diseases like heart attack, stroke, and so on. 5
Long-term exposure to mainstream statins therapy against AS might demonstrate side effects such as rhabdomyolysis, myopathy, liver injury, muscle toxicity, and acute renal failure. 18 Moreover, consumption of antibiotic could attenuate TMAO-induced AS by inhibiting TMAO synthesis. However, it comes along with the catastrophic antibiotics resistance which omits the regular use of antibiotics for languishing TMAO levels in plasma. Therefore, there is a pressing need of identification and development of novel compounds that could successfully attenuate TMAO level and terminates AS progression.
Guggulsterone (GS) isolated from guggul tree, Commiphora mukul, was identified as the main bioactive constituent responsible for guggul’s therapeutic effects. The FXR antagonism by GS has been proposed as a mechanism for its hypolipidemic effects. In addition, anti-inflammatory activity of GS was demonstrated by inhibiting nuclear factor kappa B activation. 19 It was established that GS is an antagonist at FXR, a key transcriptional regulator for the maintenance of cholesterol and BA homeostasis. 20 FMO3 expression is induced by dietary BA by a mechanism that involves the FXR, a BA-activated nuclear receptor. 21 The lipid-lowering activity of GS was also investigated in rats with hyperlipidemia induced by triton or cholesterol-feeding. 22
In current study, we reported the TMAO inhibiting the potential of GS which might demonstrate positive outcomes in the treatment of AS. Furthermore, plausible interventions to block the TMA/FMO3/TMAO pathway by GS are focused on current studies. We herein demonstrated TMAO inhibiting activity of GS in both in vitro and in vivo studies for the very first time. The TMAO inhibiting activity was evaluated with choline fed animal model of AS during in vivo studies. In addition, the effects of GS on serum lipids profile, atherogenic risk predictor (ARP) index, and hepatic and renal injury biomarkers were also reported. Similarly, the effect of GS on the production of pro-inflammatory cytokines and anti-oxidative capacity and lipid peroxidation was documented.
Material and methods
AS model
Wistar albino rats of weight 200–230 g were procured from animal house of Jamia Hamdard University. The rats were maintained under a standard 12-h light/12-h dark cycle and were housed in a controlled temperature (24 ± 2°C) and humidity (50 ± 5%) environment. They were housed in polypropylene cages (47 × 34 × 20 cm3) and fed with standard rat laboratory chow and water ad libitum. AS was induced by feeding the rats with 2% choline for 6 weeks. 4 Choline diet was prepared from normal chow diet by mixing 2-g choline chloride in powdered normal pellets and dough was made and dried. Normal pellet diet and saline were injected as the vehicle in control groups. All the experimental protocols were carried out with the approval from Institutional Animal Ethics Committee (IAEC) and Committee for the Purpose of Control and Supervision on Experiments on Animals (CPCSEA), Government of India, and application no. 1204/2016. The current study was conducted in 2016, lasting from December to June.
Drugs and chemicals
GS was obtained from Sigma-Aldrich chemicals Co., St Louis, Missouri, USA. Ciprofloxacin (CF) was obtained from Zydus Cadila, Ahmedabad, India.
Experimental design
After acclimatization, rats were randomly selected and divided into six groups, each containing six animals as follows:
Normal control (NC) received normal pellet diet with normal saline (0.1 ml/10 g, i.p.).
Pathogenic control (PC) received 2% choline diet for 6 weeks.
Pathogenic rats treated with GS received GS 20 mg/kg/day orally for 6 weeks (PC + GS 20).
Pathogenic rats treated with GS received GS 30 mg/kg/day orally for 6 weeks (PC + GS 30).
Per se group treated with GS received GS 30 mg/kg/day orally for 6 weeks (per se GS 30).
Pathogenic rats treated with CF received CF 50 mg/kg/day orally for 6 weeks (PC + CF 50).
Body weight was recorded weekly, whereas food intake was recorded daily.
In vitro studies
In vitro analysis of microbial inhibition by GS via cup plate method
In vitro analysis of microbial inhibition was performed based on earlier reported studies. 23,24 All the laboratory utensils to be used were kept in an autoclave at 15 psi at a temperature of 121°C for 15 min before initiation of the experiment. All the experiments were performed in aseptic condition. After preparation of agar, the inoculum containing gut microbes (E. fergusonii and C. sporogenes) was spread out in an agar plate. Two bores of same diameter and depth were made on the agar plate, and drugs (test group) of different concentrations 200 μg/ml (GS-L) and 500 μg/ml (GS-H) were poured on that bore. For the standard reference, CF disc (20 μg) was used. Then, the agar plate was kept at 4°C for 4 h to ensure the diffusion of the drug. The plate was transferred to incubator at 37°C for 24 h. The minimum concentration at which the drug shows the zone of inhibition was calculated as minimum inhibitory concentration (MIC) value.
In vitro analysis of TMA formed from choline by E. fergusonii in the presence and in the absence of GS
The qualitative extent of formation of TMA from choline was determined by estimating the amount of choline in the sample by high-performance liquid chromatography (HPLC) analysis. The standard plot was prepared with different series of dilution of standard choline chloride. Two groups of samples were divided as the sample containing GS (50 µg/ml) + choline chloride + E. fergusonii was labeled as a test group and sample without the test drug (GS), that is, choline chloride + E. fergusonii was labeled as control group; 1 ml of 100 µg/ml of choline chloride was added to the 50 ml of nutrient broth containing E. fergusonii with GS and without GS and was incubated for 2 h at 37°C. The incubated sample solutions were filtered with a 0.2-µm membrane filter. The extracted samples were kept in Eppendorf tubes (Recombigen Laboratories Private Limited, New Delhi) and were labeled accordingly.
The simple outline of the chromatographic system is as follows: Mode: LC. Detector: UV 208 nm. Column: 4.6-mm × 25-cm; packing L7. Column temperature: 30°. Flow rate: 1.0 ml/min. Injection size: 20 µl.
In vitro analysis of biosynthesized TMAO
Preparation of FMO microsomal homogenate from GS treated and untreated rats
A fresh liver was taken from experimental rats previously treated with choline fed diet and GS for 21 days. Liver for the control group was taken from the Wistar rat which was fed with normal diet and normal saline for 21 days. Microsomes were isolated from livers as per earlier described protocol. 24 Livers were homogenized in cold homogenization buffer (0.1-M potassium phosphate, 0.1-mM dithiothreitol, 2% sucrose, pH 9.0), and homogenates were centrifuged at 10,000 × g for 20 min at 4°C. The supernatant was then centrifuged at 100,000 × g for 60 min. The resulting pellet was washed and resuspended in 0.1-M potassium phosphate buffer (pH 7.4) containing 1-mM EDTA and stored at −80°C.
The standard TMA hydrochloride (HCl; pH adjusted to 8.5) and FMO homogenate was incubated as the standard group for 2 h. The FMO homogenate from treated group, test drug, and standard TMA HCl (pH = 8.5) were also incubated for 2 h. After incubation reaction was stopped, the samples were kept at −20°C for storage.
Sample extraction and preparation method for LC–mass spectroscopy analysis
Samples (20 µl) were aliquoted to a 1.5-ml Eppendorf tube and mixed in methanol. Protein in the samples was precipitated by vortexing for 1 min, and then, the supernatant was recovered following centrifugation at 20,000 × g at 4°C for 10 min.
LC condition: Mobile phase A: Water. Mobile phase B: Acetonitrile, Runtime: 2.5 min. Flow rate: 0.2 ml/min.
Mass spectroscopy condition: Capillary voltage: 3 kV. Temperature: 350°C. Detector: 1700. Nebulizing gas: 20 (Nitrogen). Desolvation gas: 500 l/h.
In vivo studies
Estimation of TMAO
The entire methodology was based on previously reported methods.
25,26
Preparation of sample: Samples (20 µl) were aliquoted to a 1.5-ml Eppendorf tube and mixed in methanol. Protein in the samples was precipitated by vortexing for 1 min and then the supernatant was recovered following centrifugation at 20,000 × g at 4°C for 10 min.
Biochemical estimation in serum
Immediately, after the study was terminated, blood sample (without anticoagulant) was collected by retro-orbital puncture. Experimental animals were killed under carbon dioxide (CO2) anesthesia by cervical dislocation. Organs (liver and heart) from experimental animals were removed, weighted before frozen in liquid nitrogen, and stored at –80°C until analysis. Serum was obtained by cold centrifugation at 3000 r/min for 10 min and was used for biochemical estimation.
Total cholesterol (TC), triglycerides (TG), low-density lipoprotein (LDL)-cholesterol and high-density lipoprotein (HDL)-cholesterol, and very low-density lipoprotein (VLDL)-cholesterol were determined in the serum samples that were collected under fasting conditions using commercially available kits (Sigma-Aldrich chemicals Co.).
Similarly, assessment of apolipoprotein B (ApoB) was done following earlier documented studies, 27 using the formula as follows
In addition, different ARP indexes were assessed using earlier reported method. 15
All the estimation was performed in the serum using commercially available kit according to instructions provided by manufacturers.
Quantification of pro-inflammatory cytokines level
The quantifications of tumor necrosis factor-α (TNF-α), interleukin (IL)-6, and IL-1β were performed in the serum according to the instructions provided by the rat ELISA kit manufacturer eBioscience (San Diego, California, USA).
Assessment of hepatic injury biomarkers
All the biochemical estimations in serum including aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were carried out using available commercial kit following the manufacturer’s protocol.
Biochemical estimations for oxidative stress markers
Cardiac tissue homogenates were prepared with 0.1-M Tris-HCl buffer (pH 7.4). The supernatant of homogenates was employed to estimate lipid peroxidation (malondialdehyde (MDA)), reduced glutathione (GSH), superoxide dismutase (SOD) activity, and catalase (CAT) activity. All these oxidative stress markers were determined as per the earlier reported study. 28
Histopathological evaluation
For histopathological examination, aorta (heart) of one animal from each group was isolated and fixed in 10% formalin solution. Immediately after the animals were killed, organs were isolated and washed with ice-cold normal saline. After fixing in 10% formalin solution, sections of 3–5 μm thickness were stained completely in hematoxylin and eosin (H&E) dye for histopathological examination.
Statistical analysis
Data were expressed as the mean ± standard error of the mean. For statistical analysis of the data, group means were compared by one-way analysis of variance with post hoc analysis. The Tukey–Kramer post hoc test was applied to identify significance among groups; p < 0.05 was considered to be statistically significant. Statistical analysis was carried out using Prism software package (version 5, Graph Pad, San Diego, California, USA).
Results
Effect of GS on body weight, relative heart weight, and food and water intake
Changes in body weight and relative heart weight after administration of choline and GS were presented in Table 1. Significant decrease (p < 0.001) in body weight and relative heart weight was observed in experimental animals after feeding animals with 2% choline for 6 weeks, that is, after induction of AS. Rats that received GS treatment had significantly elevated level (p < 0.001) of body weight and relative heart weight, as well as pathogenic rats treated with CF also showed significant (p < 0.001) elevation in the level of body and relative heart weight as compared to the PC rats. The elevation in the level of body weight and relative heart weight suggests that GS and CF treatment may successfully alleviate the organ injury caused by the induction of AS. The food intake and the water intake of the pathogenic rats were increased throughout the study period as compared with the NC group. However, after GS administration, the food intake and the water intake were markedly reduced as compared with the pathogenic untreated rats as demonstrated in Table 1. However, the effect of GS on both food intake and water intake was not dose-related.
Effect of GS on body weight, heart weight, water intake, and fluid intake in choline fed Wistar rats.a

NC: normal control group, normal saline (0.1 ml/10 g, i.p.); PC: pathogenic control group (2% choline diet for 6 weeks); GS: guggulsterone; CF: ciprofloxacin; PC + GS 20: PC and GS (20 mg/kg, p.o.) treated group; PC + GS 30: PC and GS (30 mg/kg, p.o.) treated group; per se GS 30: GS (30 mg/kg, p.o.) treated group; PC + CF 50: PC and CF (50 mg/kg, p.o.) treated group; SEM: standard error of the mean.
a Data are expressed as mean ± SEM (n = 6) and analyzed by one-way analysis of variance followed by Tukey’s multiple range test.
bp < 0.001 as compared to the normal control group.
cp < 0.01 as compared to PC group.
dp < 0.05 as compared to PC group.
ep < 0.001 as compared to PC group
In vitro analysis of the microbial inhibiting potential of GS
In vitro study of the microbial inhibiting potential of GS was done via analyzing zone of inhibition on the agar plate. The standard control group with CF disc has shown a significant zone of inhibition, that is, for E. fergusonii agar plate, 21 mm of zone of inhibition (see Figure 2(a)) and for C. sporogenes agar plate, 18 mm of zone of inhibition (see Figure 2(c)). On the other side, GS (test group) didn’t show any zone of inhibition, therefore couldn’t determine the MIC value. This led to the conclusion that GS did not demonstrate any antimicrobial activity against gut microbes, that is, E. fergusonii and C. sporogenes.

In vitro analysis of microbial inhibition by GS. Microbial inhibition potency of GS against several gut microbes based on zone of inhibition in agar plate as demonstrated on several plates. (a) Ciprofloxacin, (b) Escherichia fergusonii, (c) ciprofloxacin, and (d) Clostridium sporogenes. GS: guggulsterone.
In vitro analysis TMA formed from choline by E. fergusonii in the presence and absence of GS
The qualitative extent of formation of TMA from choline was determined by estimating the amount of choline in the sample by HPLC–UV analysis as shown in Figure 3(a). From the observed chromatogram, the retention time shown by choline chloride was at 3.925. As well as, from the standard plot of choline chloride as shown in Figure 3(b) different standard dilutions of choline chloride were prepared and their respective peak areas were obtained from the chromatogram as shown in Table 2. Strikingly, chromatograph didn’t show any peak on the respective retention time of 3.925 on either of the group. With this, we can conclude that choline chloride was completely digested by E. fergusonii. Similarly, we can also technically conclude that choline chloride present in nutrient broth was completely converted to TMA, and therefore, GS didn’t show any antimicrobial effects against E. fergusonii and displays no role in the inhibition of formation of TMA.
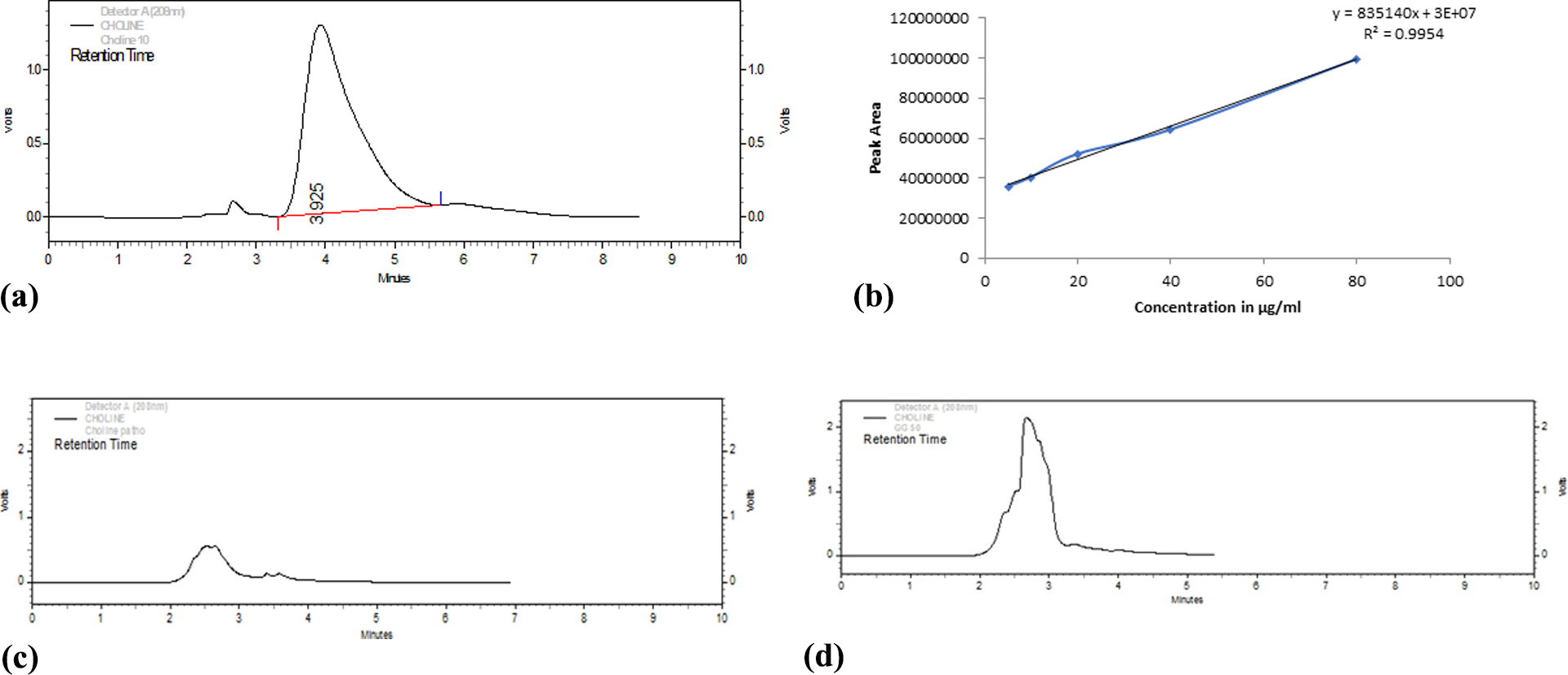
In vitro analysis of TMA. HPLC chromatogram for standard choline (a) and standard plot of choline chloride (peak area vs. concentration) (b). TMA: trimethylamine; HPLC: high-performance liquid chromatography (c) HPLC Chromatogram of choline chloride incubated with E. fergusonii in absence of drug (d) HPLC Chromatogram of choline chloride incubated with E. fergusonii in presence of drug.
Different standard dilutions of choline chloride and their respective peak areas.

In vitro analysis of biosynthesized TMAO
For in vitro analysis of biosynthesized TMAO, mass spectra of TMAO was obtained which demonstrated the peak at m/z value 76.0654, as shown in Figure 4(a). Table 3 shows different standard TMAO dilutions with their respective peaks as obtained from LC chromatogram. In addition, standard plot of TMAO with different dilutions is shown in Figure 3(b). The LC chromatogram of PC and test group is shown in Figure 4(c) and (d), respectively. From the standard plot, the concentration of the PC group and GS-treated group (test) was found to be 545.04 and 468.04 ng/ml, respectively. The present findings suggest that there was no significant decrease in TMAO levels as compared to the control group.

In vitro analysis of biosynthesized TMAO. Mass spectra of TMAO showing the peak at m/z value 76.0654 (a), standard plot of TMAO with different dilutions (b), LC chromatogram demonstrating the TMAO peak of sample without GS at the retention time of 1.26 min with the peak area 912.16 (c), and LC chromatogram demonstrating the TMAO peak of sample with GS at the retention time of 1.26 min with the peak area 821.86 (d). TMAO: trimethylamine-N-oxide; LC: liquid chromatography; GS: guggulsterone.
Different standard dilutions of TMAO and their respective peak areas.

TMAO: trimethylamine-N-oxide.
In vivo estimation of TMAO
For in vivo estimation, standard plot of TMAO with different dilutions was prepared as shown in Figure 5(a), which was obtained from Table 4. NC group did not show any mass peak at 76.0654 as it might be due to the absence of choline in the diet. Similarly, the pathogenic group treated with CF did not show any mass peak at 76.0654. This can be due to the fact that CF might have eradicated all the GM present in rats, thus inhibiting the conversion of choline to TMA.
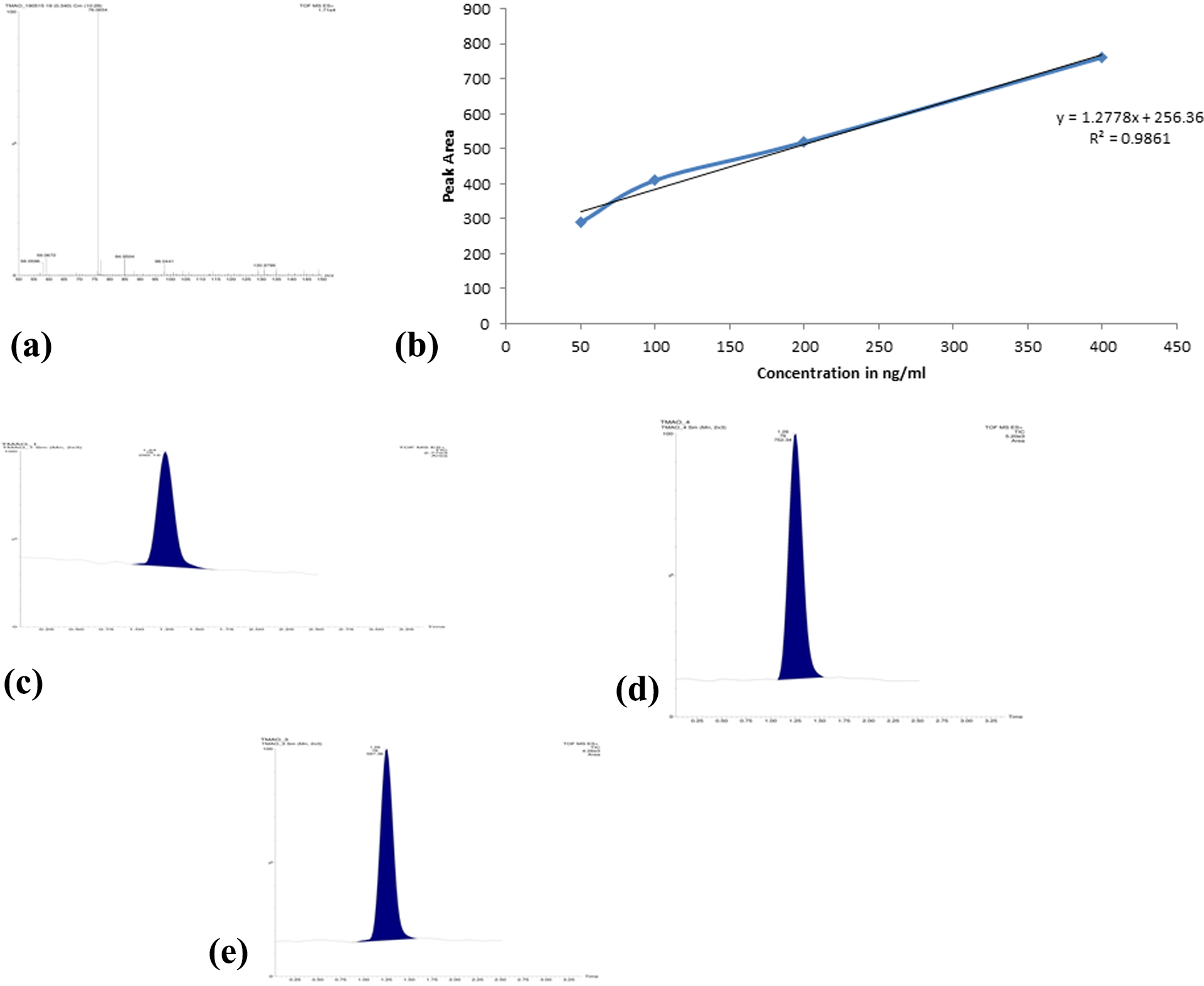
In vivo estimation of TMAO. Standard plot of TMAO with different dilutions (a), LC–MS chromatogram of pathogenic control group showing the peak area of 752.34 at retention time of 1.26 min (b), and LC–MS chromatogram of GS (30 mg/kg) given along with 2% choline diet (group IV) showing the peak area of 597.36 at retention time of 1.26 min (c). TMAO: trimethylamine-N-oxide; LC: liquid chromatography; MS: mass spectroscopy; GS: guggulsterone.
Different standard dilutions of TMAO and their respective peak areas.

TMAO: trimethylamine-N-oxide.
LC-mass spectroscopy chromatogram of PC group and test group (PC + GS 30 mg/kg) is shown in Figure 5(b) and (c), respectively. From the standard plot, the concentration of TMAO for the PC group and test group (PC + GS 30 mg/kg) was obtained as 388.15 and 266.87 ng/ml, respectively. By comparing the concentration of TMAO, we observed that GS has significantly reduced the level of TMAO during in vivo analysis.
GS improved serum lipid profile and ARP index and prevents hepatic injury
The results of serum lipid profile, ARP index, and hepatic injury biomarkers investigated in the present study are demonstrated in Table 5. ARP such as atherogenic index (AI) is one of the most important useful predictors of AS. The levels of TC, TG, LDL-C, VLDL-C, ApoB, AIP, CRI, ARP, ALT, and AST on PC rats were significantly (p < 0.001) increased as compared to the NC groups. On the contrary, the level of HDL-C was significantly (p < 0.001) reduced when compared to the normal rats. Pathogenic rats treated with GS significantly (p < 0.01) restored these altered levels to the normal value, suggesting its possibilities in treating lipid abnormalities and hepatic and renal injury. In addition, restoration of altered levels of serum lipid profile and hepatic injury markers has been significantly (p < 0.01) noted in the group treated with standard CF. On the other part, per se GS-treated groups did not show any significant effects as compared to the NC group.
Effect of GS on serum lipid profile, atherogenic risk predictor index, and hepatic and renal injury biomarkers on choline fed Wistar rats.a

TC: total cholesterol; TG: triglycerides; LDL-C: low-density lipoprotein cholesterol; and HDL-C: high-density lipoprotein cholesterol; VLDL-C: very low-density lipoprotein cholesterol; NC: normal control group, normal saline (0.1 ml/10 g, i.p.); PC: pathogenic control group (2% choline diet for 6 weeks); GS: guggulsterone; CF: ciprofloxacin; PC + GS 20: PC and GS (20 mg/kg, p.o.) treated group; PC + GS 30: PC and GS (30 mg/kg, p.o.) treated group; per se GS 30: GS (30 mg/kg, p.o.) treated group; PC + CF 50: PC + CF (50 mg/kg, p.o.) treated group; ALT: alanine aminotransferase; AST: aspartate aminotransferase; SEM: standard error of the mean.
a Data are expressed as mean ± SEM (n = 6) and analyzed by one-way analysis of variance followed by Tukey’s multiple range test.
bp < 0.001 as compared to the normal control group.
cp < 0.01 as compared to PC group.
dp < 0.05 as compared to PC group.
ep < 0.001 as compared to PC group.
##p < 0.001 as compared to normal control group.
GS suppressed the production of pro-inflammatory cytokines
The levels of TNF-α, IL-6, and IL-β were significantly (p < 0.001) elevated in the PC rats when compared to the normal rats as shown in Figure 6(a) to (c), respectively. Interestingly, PC rats administered with the test drug, GS, and standard drug CF showed a significant (p < 0.01) decline in the levels of these cytokines, which suggests the possibility of GS and CF in inhibiting the production of these pro-inflammatory cytokines in the choline fed Wistar rats. Furthermore, per se GS-treated groups did not demonstrate any significant effects as compared to the NC group.

Effect of GS on proinflammatory cytokines level on choline fed Wistar rats. Images showing inhibiting effect of GS on TNF-α (a), IL-6 (b), and IL-β (c). Data are expressed as mean ± SEM (n = 6) and analyzed by one-way analysis of variance followed by Tukey’s multiple range test. ###p < 0.001 as compared to the normal control group, ***p < 0.001 as compared to PC group, and **p < 0.01 as compared to PC group. NC: normal control group, normal saline (0.1 ml/10 g, intraperitoneal (i.p)); PC: pathogenic control group (2% choline diet for 6 weeks); GS: guggulsterone; CF: ciprofloxacin; PC + GS 20: PC and GS (20 mg/kg, per os (p.o.)) treated group; PC + GS 30: PC and GS (30 mg/kg, p.o.) treated group; per se GS 30: GS (30 mg/kg, p.o.) treated group; PC + CF 50: PC and CF (50 mg/kg, p.o.) treated group; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; IL-β: interleukin-β. SEM: standard error of the mean.
GS enhanced anti-oxidative capacity and reduced lipid peroxidation
TBARS is a marker of lipid peroxidation (MDA) and GSH and CAT are anti-oxidant enzymes. Rats fed with 2% choline for 6 weeks significantly (p < 0.001) reduced CAT, SOD, and GSH content and concomitantly increased MDA level in cardiac tissue of AS-induced rats, as shown in Table 6. However, oral administration of GS to the pathogenic rats improved the antioxidant status of cardiac tissues by increasing GSH level and the activities of SOD and CAT compared to the PC groups. In addition, it also significantly (p < 0.01) reduced the alleviated level of MDA in comparison with the PC rats. Results of elevation in the level of anti-oxidant enzymes such as GSH, SOD, and CAT as well as the decreased level of MDA have been demonstrated by pathogenic rats receiving CF. Similarly, no significant effects were documented on per se GS-treated group as compared to the NC group.
Effect of GS on cardiac oxidative stress markers on choline fed Wistar rats.a

NC: normal control group, normal saline (0.1 ml/10 g, i.p.); PC: pathogenic control group (2% choline diet for 6 weeks); GS: guggulsterone; CF: ciprofloxacin; PC + GS 20: PC and GS (20 mg/kg, p.o.) treated group; PC + GS 30: PC and GS (30 mg/kg, p.o.) treated group; per se GS 30: GS (30 mg/kg, p.o.) treated group; PC + CF 50: PC + CF (50 mg/kg, p.o.) treated group; MDA: malondialdehyde; GSH: glutathione; SOD: superoxide dismutase; CAT: catalase; SEM: standard error of the mean.
a Data are expressed as mean ± SEM (n = 6) and analyzed by one-way analysis of variance followed by Tukey’s multiple range test.
bp < 0.001 as compared to the normal control group.
cp < 0.01 as compared to PC group.
dp < 0.001 as compared to PC group.
Protective effect of GS on atherosclerotic pathology alteration in rat aorta
The findings obtained via biochemical assays were further confirmed by histopathological study. Representative aortic transverse sections obtained from each group are shown in Figure 7. Microscopic examination of H&E-stained aortic sections from rats (see Figure 7(a)) revealed that the aortic walls in the control rats were smooth and intact. However, by feeding 2% choline diet for 6 weeks in PC groups (see Figure 7(b)) demonstrated a thick layer of lipid deposition within the intima of an aorta, which is typical for AS. These results suggest that we successfully established an atherosclerotic model in rats. In comparison with the PC group (see Figure 7(b)), GS-treated groups (see Figure 7(c) and (d)), per se GS-treated group (see Figure 7(e)), and CF-treated group (see Figure 7(f)) showed a visible reduction in pathological changes; the vascular walls were slightly rougher and thicker, and fewer atherosclerotic plaques were found. Furthermore, the vascular structure of the thoracic aorta was typically normal in GS treated group suggesting its protective effects against AS which might be via reducing TMAO level.

Effect of GS on H&E staining of heart in atherosclerosis rats. Photomicrograph of sections of heart tissue of (a) NC, normal saline (0.1 ml/10 g, i.p.); (b) PC, (2% choline diet for 6 weeks); (c) PC + GS 20: PC and GS (20 mg/kg, p.o.) treated group; (d) PC + GS 30: PC and GS (30 mg/kg, p.o.) treated group; (e) per se GS 30: GS (30 mg/kg, p.o.) treated group; and (f) PC + CF 50: PC + CF (50 mg/kg, p.o.) treated group. GS: guggulsterone; H&E: hematoxylin and eosin; NC: normal control; PC: pathogenic control; CF: ciprofloxacin.
Discussion
There is an increasing understanding that hyperlipidemia, inflammation, and oxidative stress lead to the initiation and development of AS and which might even lead to endothelial dysfunction. 29 In the current investigation, an atherosclerotic rat model was developed via administering a choline diet for 6 weeks. 4 To overcome the limitations of currently available statin drugs, there is a continuous quest toward the evaluation of novel compounds from natural products which exhibit similar lipid-lowering, anti-inflammatory, and anti-oxidative activities as statins.
Recently, direct relations between dietary nutrients (choline, L-carnitine), GM, and cardiometabolic disease have been uncovered from the recent discovery of TMAO and its pathological links with AS and cardiometabolic diseases. 30,31 Intestinal microbiota causes the generation of TMAO via specific microbial choline 4 TMA lyases. Specifically, microbial conversion of dietary nutrients that possess a TMA moiety (such as choline, phosphatidylcholine, and L-carnitine) is converted to TMA by specific microbial enzymes (TMA lyase) through a wide variety of metabolic pathways. 31 TMA is then absorbed by the host and converted into TMAO by hepatic FMO3 and excreted by the kidneys. 31 Formally, it was reported that GM-driven TMO/FMO3/TMAO pathway is a key regulator of lipid metabolism and inflammation. 32 The studies described here along with the previous report linking TMAO to CVD risk in human 4 provide compelling shreds of evidence that the TMO/FMO3/TMAO pathway is a central regulatory pathway that deserves further exploration. However, the ability of TMAO to promote AS 4 may be mutually exclusive from the ability of FMO3 inhibitors to reorganize cholesterol balance and inflammation. 32
Microbial inhibiting property of GS was evaluated on agar plate during in vitro studies and observed that GS didn’t show any antimicrobial activity against E. fergusonii and C. sporogenes. However, the standard group with CF disc demonstrated significant zone of inhibition. The result obtained on the current investigation was in accordance with the earlier reported study. 33 Dietary choline is metabolized by the intestinal microbiota to TMA, which is further metabolized by FMO enzymes, in particular, FMO3, to produce TMAO in the liver. 21 Similarly, in vitro analysis TMA formed from choline by E. fergusonii in the presence and absence of GS is evaluated in the current study with the help of HPLC–UV. We interestingly, herein documented that choline chloride present in nutrient broth was completely digested by E. fergusonii and GS didn’t show any antimicrobial effects against E. fergusonii as evidenced by the absence of peak on the respective retention time of 3.925 as demonstrated in Figure 2. This led to the conclusion; GS has no antimicrobial properties against the GM and has no role in inhibiting TMA production during in vitro studies. Furthermore, in vitro analysis of biosynthesized TMAO was carried out. Mass spectra and LC chromatogram of TMAO were obtained in the presence and in the absence of GS. Mass spectra demonstrated the peak at m/z value 76.0654 as shown in Figure 3(a). Through analyzing with the standard plot obtained, the concentration of TMAO in control group without drug was found to be 545.04 ng/ml, and the test sample GS was found to be 468.04 ng/ml as shown in Figure 3(b). Findings from current investigation suggest that there was significant decrease in TMAO levels in test group as compared to the control group.
In vivo estimation of TMAO was carried out in experimental animals. Peaks obtained from the mass spectrum and LC chromatogram were used for the estimation of TMAO. Peak area obtained from different treatment groups was shifted on the linear equations and the respective value of TMAO was obtained. Interestingly, by comparing the concentration of TMAO with different treatment groups, we observed that GS has significantly reduced the level of TMAO during in vivo analysis. This result was in similar line with the earlier reported studies. 4 Decreased level of TMAO by GS might be due to its plausible antiatherogenic effects. Significant elevation in body weight and the decrease in relative heart weight were observed in the PC rats in the current study, which indicated that choline diet induced a significant obesity. However, GS treatment significantly attenuated the increased body weight. This finding corroborates with the earlier finding where body weight significantly increased on a high-fat diet fed animals. 34
The wide range of studies has demonstrated that the development of atherosclerotic lesions is associated with serum lipid levels; in particular, higher plasma levels of LDL-C are a definite risk factor for CVD. 35 In accordance with an earlier report, current studies demonstrated significant elevation in TC, TG, LDL-C, VLDL-C, and ARP index levels in PC rats and reversed by GS treatment. However, a higher content of HDL-C is very important in humans because it is correlated with a reduced risk of coronary heart disease. 27 Interestingly, Tsarouhas et al. reported that regular walking also can decrease cholesterol, TG, LDL, and other inflammatory biomarkers, which enhances the longevity with the patients of heart disease. 36 The AI is one of the most important useful predictors of AS. A lower index value indicates a lower extent of AS risk. AIP, CRI, and ApoB levels were increased in PC rats and these levels were attenuated with GS treatment and the findings were consistent with earlier studies. 34 Earlier finding reported that in the condition of chronic inflammatory burden with elevated oxidative stress, lipids are the first to be changed, followed by the altering of lipoprotein proteome, which coincides with our findings as there is altering of lipid profile with the elevation of inflammatory as well as oxidative stress biomarkers. 37 Several lines of evidence reported that hepatic injury 38 is associated with AS. Concomitantly with the earlier report, current investigation demonstrated increased levels of ALT and AST on choline fed PC rats. Interestingly, these elevated levels of ALT and AST were attenuated by the administration of GS. Herein, we suggest the plausible protective effects of GS in hepatic injury
It is evident that the expression of systemic inflammatory response factors such as TNF-α and IL-6 is high in metabolic syndrome and has prominent role in atherogenesis. 39 In agreement with the earlier study, the finding of current demonstrated revealed that the levels of several pro-inflammatory cytokines (TNF-α, IL-6, and IL-1β) increased in AS rats. 40 These increased levels were restored to the normal by GS supplementation. Similar result was clinically observed by Tsitsimpikou et al. where tomato juice significantly decreased the level of IL-6, TNF-α, and LDL along with slight increase in HDL level on the patients. 41 Henceforth, we suggest the protective role of GS against inflammation.
Disorders of lipid metabolism, which increase the overproduction of free radicals, disturb the antioxidant status, thereby causing damage to endothelial cells; this represents the initial step of atherogenesis. 42 Oxidative stress can also cause DNA damage which is also one of the pathogenesis for developing AS. 43 In consistency with earlier finding, the current study demonstrated altered SOD, GSH, CAT, and MDA levels in the cardiac tissues of AS rats. Decreased activities of SOD and CAT in PC rats clearly demonstrate that SOD and CAT are easily inactivated by reactive oxygen species and lipid peroxides. GS administration significantly restored the altered level of SOD, GSH, CAT, and MDA in GS-treated pathogenic rats. With this, we suggest that GS can reduce oxidative stress in choline fed AS rats. The H&E staining of aortic sections corroborates the above discussed molecular changes that GS almost maintained the normal architecture in AS-induced rats. Similar cardioprotective properties of resveratrol were observed by Hashemzaei et al. where resveratrol caused attenuation of histopathological lesions in rat’s heart tissue. 44
Conclusion
In summary, we found that 2% choline fed diet induces AS in the current study. The supplementation with GS acts on the TMA/TMAO/FMO3 pathway and subsequently reduces the level of TMAO through the inhibition of FMO3 expression in both in vitro and in vivo analysis. Moreover, GS exhibited an antihyperlipidemic property, reduced the lipid peroxidation process, and enhanced the antioxidant defense system. In addition, GS exhibits a strong cardioprotective effect as demonstrated by a significant reduction of several AIs. The results provide an insight into new avenues for research based on GM targeting TMA/TMAO/FMO3 pathway in designing therapy for AS. However, these results need to be substantiated by studies in primates and humans to ascertain the place of GS in AS clinical care pathways.
Footnotes
Acknowledgments
The authors would like to thank the Department of Pharmacology, Central Instrumentation Facility (CIF), and Central Animal House Facility, Jamia Hamdard, New Delhi, for providing the necessary facilities.
Authors’ contribution
AG performed the overall study, UB supervised the entire study, YNP drafted the manuscript, and SAZA contributed in the revised manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project receives no funding from any funding organization.